Abstract
Background:
Long-term corticosteroid use after kidney transplantation is associated with a decrease in bone mineral density (BMD) and a high fracture risk. We hypothesized that patients with early steroid withdrawal (ESW) would display a gain in BMD in the year following kidney transplantation, when compared with patients on long-term corticosteroid therapy.
Methods:
In a cohort of kidney transplant recipients, 356 patients were included between 2012 and 2019. Dual-energy X-ray absorptiometry was performed 1 and 12 months after transplantation. The data were analyzed using linear regression with inverse probability-of-treatment weighting (based on a propensity score).
Results:
At 1 year after transplantation, the gain in BMD was significantly greater in recipients with ESW than in recipients on long-term corticosteroid therapy for the lumbar spine (+0.036 g/cm2, p < 0.001) and the femoral neck (+0.020 g/cm2, p = 0.035). Among patients with ESW, (i) none had osteoporosis, (ii) the percentage with normal BMD increased from 33.3% at month 1 to 54.4% at month 12, and (iii) the percentage with osteopenia fell from 56.2% to 45.6%. In patients undergoing long-term corticosteroid therapy, the fracture incidence was 13.5 per 1000 person-years. None of the patients in the ESW group experienced a fracture.
Conclusion:
ESW has a positive effect on bone in kidney transplant recipients.
Introduction
Immunosuppressive regimens for kidney transplantation have progressed since the 1990s, especially with regard to early steroid withdrawal (ESW). During the decade following the introduction of cyclosporine, several retrospective studies documented the benefits of ESW in general, and, in particular, the reduction in cardiovascular complications (lower blood pressure, lower cholesterol levels, and less diabetes mellitus).1–3 However, some reports have suggested that steroid withdrawal is associated with a significant risk of acute allograft rejection and graft loss.4,5
Nevertheless, the development of additional immunosuppressants (such as tacrolimus and mycophenolate mofetil) and the use of induction treatments (such as basiliximab and antithymocyte globulin) have stimulated renewed interest in steroid-free immunosuppressive treatments, and similar benefits have already been demonstrated.6–10 Furthermore, two randomized multicenter trials have similar long-term renal allograft survival rates and functions for recipients with ESW and those continuing to take corticosteroids – even though the rate of acute allograft rejection was higher in the ESW group.8,10 Moreover, Barbour et al.’s recent study of a large cohort of kidney transplant patients suffering from recurrent glomerulonephritis showed that ESW was not associated with a major risk of death or all-cause graft loss. 11
Most of the studies of ESW have focused on cardiovascular and metabolic complications, and there are only few reports of the consequences of early withdrawal on bone. 12 Prior to the introduction of steroid-free immunosuppressive regimens, several studies evidenced a decrease in bone mineral density (BMD) in corticosteroid-treated recipients (particularly 6–12 months after transplant)13–16 and a higher fracture risk. 17 Thus, it has been suggested that corticosteroid treatment is a risk factor.18,19 Indeed, corticosteroid-induced osteoporosis is the leading cause of secondary osteoporosis. 20
Bone fractures are major complications of osteoporosis, and constitute a major public health issue and a socio-economic burden. Indeed, osteoporotic hip fractures are responsible for excess mortality, with many deaths occurring within 6 months of the fracture. 21 The excess mortality rate varies from one study or population to another but ranges from 8% to 36% within the first year following hip fracture.22,23 Unsurprisingly, BMD is a major determinant of osteoporotic disease and is associated strongly with the fracture risk.24,25
Hence, in addition to its cardiovascular and metabolic benefits, ESW in kidney transplant recipients might limit bone damage by increasing BMD and reducing the fracture risk – even though bone quality is another major determinant of the fracture risk, 26 particularly in patients suffering from chronic kidney disease-mineral and bone disorder (CKD-MBD). Indeed, CKD-MBD is characterized by one or more of the following manifestations: (i) renal osteodystrophy; (ii) vascular and soft tissue calcification; (iii) abnormal metabolism of calcium, phosphorus, parathyroid hormone (PTH); and 25(OH) vitamin D. 27 Renal osteodystrophy includes all the changes in bone histology seen in uremic patients: abnormalities in bone turnover, mineralization, volume linear growth, or strength. 28 Mild hyperparathyroid-related bone disease (reflected histologically by high-turnover bone, leading to osteitis fibrosa in advanced cases) is more frequently encountered in early-stage kidney disease. 29 In end-stage kidney disease, skeletal resistance to the action of PTH action leads to adynamic bone disease (ABD). 30 The latter corresponds to low-turnover bone with normal mineralization and a normal or low bone volume. 31 Osteomalacia and mixed uremic osteodystrophy can also be encountered in CKD patients. 28 Regardless of the origin, a low BMD in CKD patients is a marker of bone fragility. 32
In steroid-treated patients with normal renal function, treatment withdrawal is associated with a rapid increase in bone mineral density and a decrease in the fracture risk.33–35 One can reasonably expect the same to be true of kidney transplant patients but these changes would need to be confirmed – especially since the bone microarchitecture is impaired in this population (as described above).
In view of different characteristics in patients on long-term corticosteroid therapy and those with ESW, comparisons based on corticosteroid therapy duration may seem difficult. However, a number of innovative methods are now available to minimize the indication bias inherent in pharmacoepidemiological studies, such as high-dimensional propensity scores. 36 We hypothesized that patients with ESW could display a gain in BMD during the year following kidney transplantation, when compared with patients on long-term corticosteroid therapy. Therefore, we assessed BMD in both types of patient during the 12 months after kidney transplantation using inverse probability-of-treatment weighting (IPTW) to reduce indication bias.
Methods
Study design and participants
We performed a longitudinal study of a cohort of adult patients (aged 18 and over) having undergone kidney transplantation at Amiens University Medical Center (Amiens, France) between 1 January 2012, and 13 February 2019, and with available data on BMD measured at several different anatomic sites (the lumbar spine, the hip, and femoral neck) 1 month (M1) and 12 months (M12) after transplantation. BMD was determined using dual-energy X-ray absorptiometry (DXA; Hologic Discovery System, Hologic Inc., Waltham, MA, USA). Osteoporosis was defined as a T-score ⩽ −2.5 at one or more sites, and osteopenia was defined as a T-score between −1 and −2.5.
Collected data
Data at study baseline
The variables recorded at baseline included sociodemographic characteristics, osteoporosis risk factors, the use of drugs potentially influencing BMD at the time of transplantation (steroids, calcium, cholecalciferol, alfacalcidiol, calcifediol, and bisphosphonates), any history of chronic kidney disease, the characteristics of the donor, and the characteristics of the transplantation.
Serum levels of calcium, phosphate, 25(OH) vitamin D, PTH, bone alkaline phosphatase, osteocalcin, and creatinine at baseline and glomerular filtration rate were also recorded.
Follow-up data
Data on prevalent osteoporotic fractures and intakes of calcium, cholecalciferol, alfacalcidiol, calcifediol, and bisphosphonate during the study period were collected, together with post-transplantation complications: acute graft rejection (defined according to the Banff classification after an pathologic assessment of a graft biopsy at M3), the appearance of donor-specific antibodies (DSAs), bacterial and viral infections, and cardiovascular risk factors and cardiovascular events. 37
Data on serum levels of calcium, phosphate, 25(OH) vitamin D, PTH, and creatinine at M12 and on the glomerular filtration rate were also collected.
Assays of serum PTH, 25(OH) vitamin D, bone alkaline phosphatases, and osteocalcin
Serum PTH was assayed using a chemiluminescent immunoassay (ADVIA Centaur PTH from Siemens Healthcare Diagnostics SAS; intra-assay coefficient of variation <2.05%, inter-assay coefficient of variation <4.04%; detection limit of the assay: 4.6 pg/ml), according to the manufacturer’s instructions. Serum 25(OH) vitamin D was assayed using an ELISA (assay kit total Vitamin D -ADVIA Centaur from Siemens Healthcare Diagnostics SAS). The detection threshold for serum 25(OH) vitamin D was 4.20 ng/ml (intra-assay coefficient of variation <9.79%, inter-assay coefficient of variation <7.32%). Bone alkaline phosphatase and osteocalcin were assayed using Liaison-XL from DiaSorin SA (Antony, France). The detection thresholds for serum bone alkaline phosphatase and osteocalcin were 3.0 µg/l and 1.5 ng/ml, respectively. The intra-assay coefficients of variation were <2.16% and <4.99%, respectively, and the inter-assay coefficients of variation were <4.19% and <5.33%, respectively.
Immunosuppressive regimens
Since March 2016, kidney transplant recipients with a low immunological risk (defined as a peak panel-reactive antibody <20%) being treated in the Department of Nephrology at Amiens University Medical Center have discontinued their corticosteroid treatment on day 7 post-transplantation. The treatment consisted of a bolus of methylprednisolone (500 mg) at the time of transplantation, followed by oral prednisolone (20 mg/day for 4 days and then 10 mg/day for 3 days). Patients who had not resumed corticosteroid treatment by M12 formed the ESW group. The patients had received induction therapy with basiliximab and maintenance therapy with mycophenolate mofetil plus tacrolimus or mycophenolate mofetil plus cyclosporine, depending on the risk of developing type 2 diabetes mellitus or exacerbating preexisting diabetes.
Before March 2016, all patients (regardless of the immunological risk) received the same long-term corticosteroid treatment, consisting of a bolus of methylprednisolone (500 mg) at the time of transplantation and then oral prednisolone (20 mg/day, with a gradual dose reduction to between 5 and 10 mg/day by M3). Patients treated before March 2016 and patients with a low immunological risk rechallenged with steroids before M12 since March 2016 formed the “other steroid regimens” (OSR) group.
Identification of patients with ABD
In order to take account of the heterogeneity of skeletal involvement, patients with PTH <150 pg/ml (positive predictive value for ABD: 97%) and bone alkaline phosphatases <10 ng/ml (which can further bolster the diagnosis of ABD, as it is 100% sensitive and 93.7% specific) were classified as having ABD. 38
Statistical methods
In our descriptive analysis, categorical variables were expressed as the number (percentage), and continuous variables were expressed as the mean ± standard deviation (SD) or the median (range), depending on the data distribution. The Shapiro-Wilk test was used to determine whether or not data were normally distributed.
In bivariate analyses of the ESW and OSR groups, continuous variables were compared using Student’s t test or a Wilcoxon’s rank sum test (depending on the data distribution), and categorical variables were compared using a chi-square test or Fisher’s exact test.
Bivariate analyses were performed for the change over time (between M1 and M12) in BMD and in all other variables, in order to determine factors influencing BMD in the study population. For continuous variables, Pearson’s correlation coefficients or Spearman’s rank correlation coefficients were calculated (depending on the data distributions). For binary variables, Student’s t test or Wilcoxon’s rank sum test was applied. For categorical variables (>2 categories), an analysis of variance was performed (Supplemental Tables S1–S3).
IPTW was used in the primary analysis. The rationale for using IPTW was that the distribution of the probability of receiving one treatment or another is similar among the weighted population; this enables observational studies to be designed on the same lines as a randomized controlled trials. 36 A propensity score (PS) was estimated using a logistic regression model. Treatment assignment (ESW versus OSR) was regressed only on variables with p < 0.2 in the bivariate analysis for the prediction of changes in BMD (Supplemental Tables S1–S3). Indeed, it is better to include either covariates with prognostic value (those related to outcomes, i.e. BMD changes) or confounding covariates (those related to treatment, i.e. ESW and outcomes, i.e. BMD changes) in the PS model than variables that affect the treatment selection process.39,40 Thus, three PSs were produced – one for each BMD measurement site (the lumbar spine, the femoral neck, and the total hip). Next, simple IPTW linear regressions were performed for each site: patients in the ESW group were weighted by the reciprocal of their PS, and patients in the OSR group were weighted by one minus the reciprocal of their PS. We checked that the conditions for a valid linear regression were met.
The PSs were balanced by excluding counterfactuals, when required. 41 Next, the PS distributions were evaluated using a kernel density plot (Supplemental Figure S1). Lastly, the balance of each PS was assessed by examining the standardized mean differences (Supplemental Figures S2–S4). For regression adjustment to be trustworthy, the absolute standardized differences of means should be less than 0.25. 42
In sensitivity analyses, (i) simple IPTW linear regressions (one for each BMD measurement site) were performed after all the covariables had been included in a PS, and (ii) the previous analyses were performed in patients with initial ESW [with or without resumption of steroids, ESW (M0)] and those with initial long-term steroid therapy [OSR (M0)].
The main analyses were stratified by sex, age group (with a cut-off at 60, to limit the loss of statistical power) and bone involvement (i.e. patients with versus without ABD).
All analyses were performed using R software (version 3.6.0, R Foundation for Statistical Computing, Vienna, Austria).
Ethics approval and informed consent statements
In line with the French legislation on retrospective analyses of routine clinical practice, patients were not required to give their informed consent. On admission to hospital, however, patients could refuse the use of their medical data for research purposes. This protocol was approved by an institutional committee (with competency for studies not requiring approval by an independent ethics committee) and was registered with the French National Data Protection Commission (Commission Nationale de l’Informatique et des Libertés, Paris, France; reference: PI2019_843_0055).
Results
Study population
Of the 477 patients having received a kidney transplant between 1 January 2012, and 13 February 2019, 356 (including 226 men, 64.5%) were included in the present study. Of the 85 recipients having discontinued steroid at M0, 25 had resumed steroid therapy during the study period (Figure 1).

Patient inclusion flow chart.
The reasons for the resumption of corticosteroid therapy were acute graft rejection (based on an M3 biopsy; n = 7), the appearance of DSAs (n = 3); delayed recovery of renal function (n = 5), glomerular lesions on the graft biopsy without signs of acute rejection (n = 5), and dosage reduction or the discontinuation of other immunosuppressive drugs following an adverse effect (n = 5: three patients with mycophenolate-mofetil-related leukopenia, one patient with tacrolimus-related hyperkaliemia, and one patient with tacrolimus-related BK virus viremia).
The mean ± SD age of the study population at the time of transplantation was 50.9 ± 13.2 years. The median time to the first BMD measurement was 32 days, and the median time to the second was 12 months (Table 1).
Characteristics of the study population.

BMD, bone mineral density; BMI, body mass index; BP, bisphosphonate; ESA, erythropoiesis-stimulating agent; ESW, early steroid withdrawal; FUEF, fracture of the upper end of the femur; GFR, glomerular filtration rate; HPT, hyperparathyroidism; IVIg, intravenous immunoglobulins; MMF, mycophenolate mofetil; OSR, other steroid regimens; PRA, panel-reactive antibody; PTH, parathyroid hormone.
Number of days between kidney transplantation and serum creatinine <30 mg/day.
The most common cause of kidney disease necessitating kidney transplantation was recurrent glomerulonephritis (29.8%) (Table 1).
The proportion of patients with prior steroid intake was higher in the OSR group than in the ESW group (19.3% versus 5.0%, respectively; p = 0.012) (Table 1). There were no significant intergroup differences in the intakes of native vitamin D or its metabolites before or after transplantation. A given patient could have received cholecalciferol and one of its metabolites but not simultaneously. After transplantation, the mean daily dose of alfacaldiol in the total population was 1.2 ± 0.3 µg/day, that of calcifediol was 28.2 ± 12.7 µg/day, and that of cholecalciferol was 1,00,000 IU/month [regardless of 25 (OH) vitamin D3 levels].
Influence of osteoporosis risk factors and other variables on BMD
Factors influencing BMD (p < 0.2) at each measurement site are described in Supplemental Tables S1–S3. The factors that influenced the three sites were recipient age, the serum PTH level, and the serum osteocalcin level at M0 and deceased donor. For each measurement site, variables included in the propensity score and influencing BMD are listed in Supplemental Figures S2–S4. Absolute mean differences between groups were well balanced after adjustment using IPTW (Supplemental Figures S2–S4).
Changes over time in BMD, and the incidence of fractures
When comparing OSR and ESW groups, in a bivariate analysis of the data at M1, there were no intergroup differences in BMD at the three measurement sites, despite the greater proportion of patients with prior steroid intake in the OSR group - suggesting that steroids received before transplantation had no impact on the overall trend. At M12, the lumbar spine BMD was significantly higher in the ESW group than in the OSR group - particularly in men (Table 2).
Bone mineral densities and T-scores at M1 and at M12.
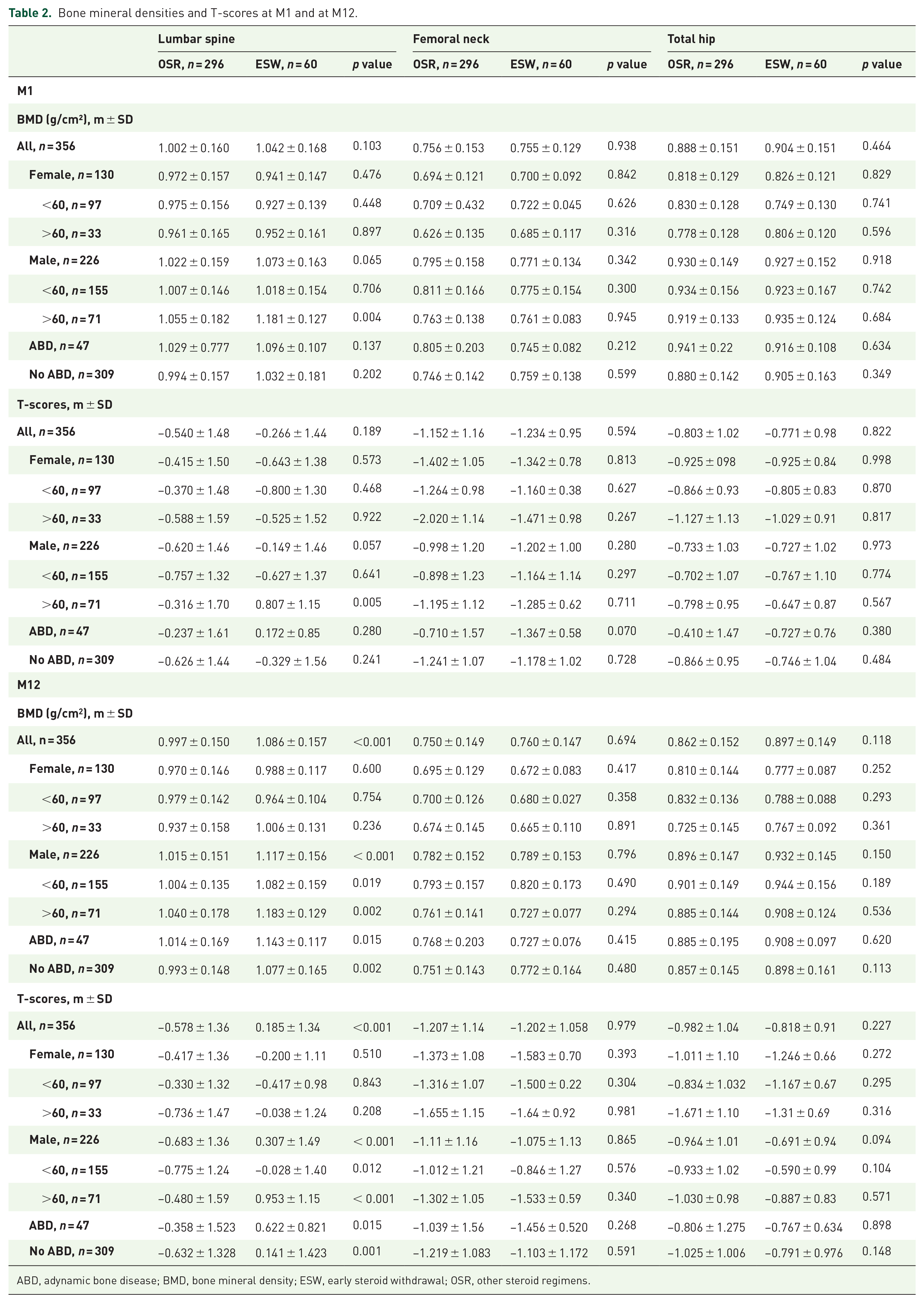
ABD, adynamic bone disease; BMD, bone mineral density; ESW, early steroid withdrawal; OSR, other steroid regimens.
In the ESW group, the lumbar spine BMD increased significantly by 0.038 ± 0.07 g/cm2 (+4.2 ± 6.8, p < 0.01) between M1 and M12. This increase in BMD was particularly evident in women over 60 (+0.054 ± 0.06 g/cm2, p = 0.028), men under 60 (+0.053 ± 0.06 g/cm2, p < 0.001), and patients without ABD (+0.048 ± 0.07 g/cm2, p < 0.001). In contrast, the lumbar spine BMD in the OSR group decreased by 0.005 ± 0.06 g/cm2 (−0.2 ± 5.9%, p = 0.141) over the same period. The intergroup difference in the change in lumbar spine BMD was statistically significant (p < 0.001), and was most evident in women over 60 (p = 0.008), men under 60 (p < 0.001), and patients without ABD (p < 0.001) (Table 3).
Changes in BMD between M1 and M12.

ABD, adynamic bone disease; ESW: early steroid withdrawal; OSR: other steroid regimen.
p*: intra-group p-value; p**: inter-group p-value.
In the OSR group, the proportions of patients with normal BMD, osteopenia, or osteoporosis did not change greatly between M1 and M12. In the ESW group (in which the patients did not receive bisphosphonate), the proportion of osteoporotic patients fell from 10.5% upon transplantation to 0% at M12. Furthermore, the percentage of patients in the ESW group with normal BMD increased from 33.3% to 54.4%, and the percentage with osteopenia decreased from 56.2% to 45.6% (Figure 2). There were no incident fractures in the ESW group (Table 4).

Prevalence of normal BMD, osteopenia, and osteoporosis at M1 and at M12 in recipients in the OSR group (a) and the ESW group (b).
Complications after kidney transplantation.
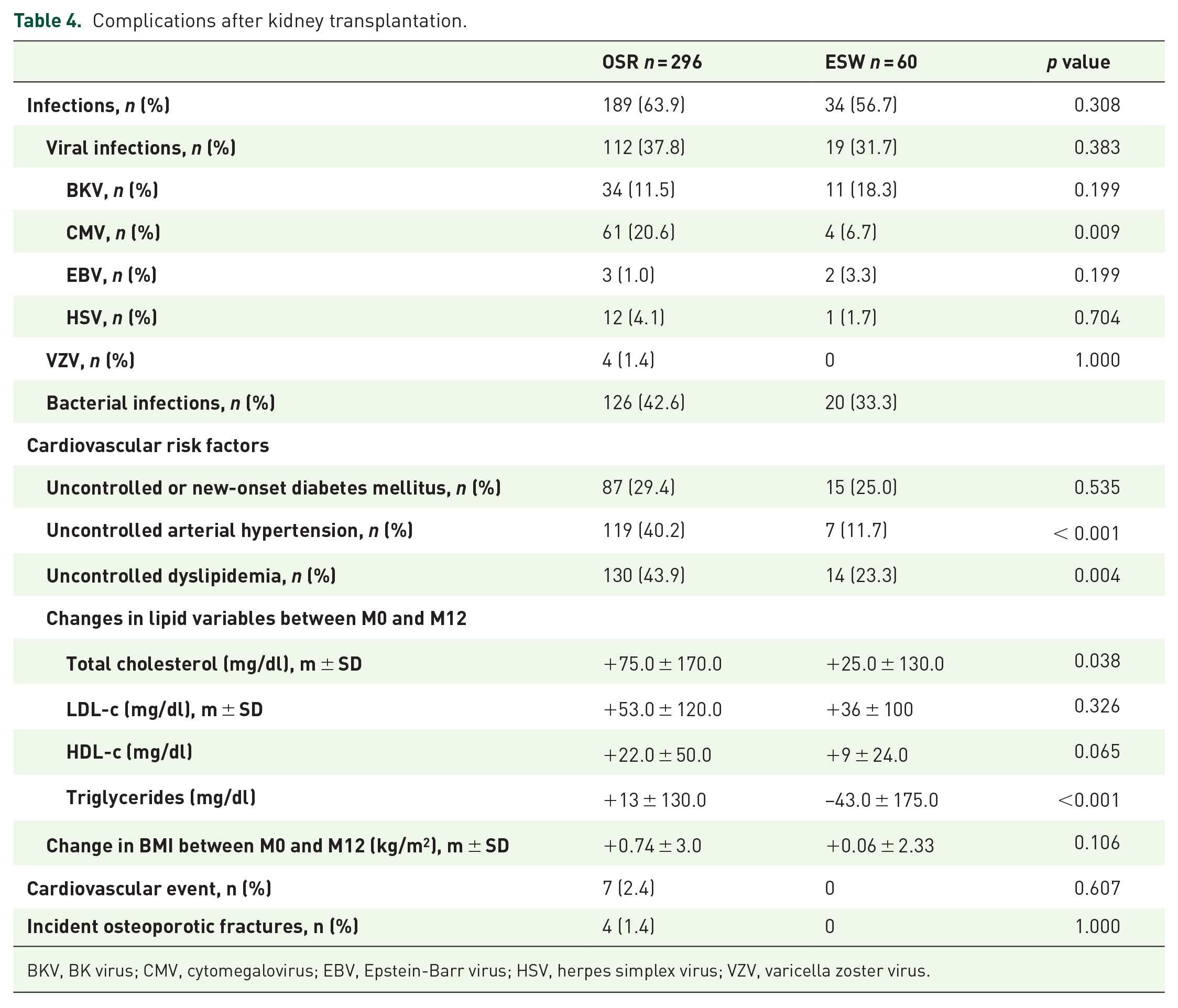
BKV, BK virus; CMV, cytomegalovirus; EBV, Epstein-Barr virus; HSV, herpes simplex virus; VZV, varicella zoster virus.
During the 12-month follow-up period, the incident fracture rate in the OSR group was 13.5 per 1000 person-years.
Comparison of BMD values in the ESW and OSR groups
In the primary analysis at M12, patients in the ESW had a significantly greater increase in BMD at (i) the lumbar spine (0.036 g/cm2 (p < 0.001), corresponding to a T-score gain of 0.299 (p < 0.001), particularly in women, men under 60, and patients without ABD), and (ii) the femoral neck (0.020 g/cm2 (p = 0.035), corresponding to a T-score gain of 0.121 (p = 0.013), particularly in men under 60 and patients without ABD), relative to patients in the OSR group (Table 5).
BMD and T-scores variations (IPTW, main analysis).

ABD, adynamic bone disease; BMI, body mass index; ESW, early steroid withdrawal; OSR, other steroid protocols; PS, propensity score; PTH, parathyroid hormone.
Variables included in the PS (influencing changes in the lumbar spine BMD; exclusion of counterfactuals was not necessary): age, BMI at M0, change in BMI, a history of autoimmune disease or primary, immunosuppressive maintenance therapy, blood calcium at M0, osteocalcin at M0, PTH at M0 and M12, 25 (OH) vitamin D3 at M0 and M12, blood creatinine at M12, prior calcium, erythropoiesis-stimulating agent or cinacalcet intake, calcium or bisphosphonate intake during the study period, donor age and sex, deceased donor, uncontrolled arterial hypertension, uncontrolled dyslipidemia, bacterial infection, cross-match results, warm ischemia time.
Variables included in the propensity score (influencing changes in the femoral neck BMD; exclusion of counterfactuals was not necessary): age, BMI, a history of primary or secondary hyperparathyroidism, smoking, etiology of the chronic kidney disease, PTH at M0 and at M12, osteocalcin at M0, PAO at M0, cross-match results, bacterial infection, uncontrolled or new-onset diabetes mellitus during the study period, uncontrolled dyslipidemia during the study period, donor age, erythropoiesis-stimulating agent intake, deceased donor.
Variables included in the propensity score (influencing changes in the total hip BMD, exclusion of counterfactuals was not necessary): age, sex, race, change in BMI, induction therapy (basiliximab and thymoglobulin), PTH at M0, osteocalcin at M0, PAO at M0, blood calcium at M0, prior vitamin D intakes, vitamin D intake during the study period, donor age, deceased donor, acute graft rejection, time on hemodialysis before transplantation, uncontrolled arterial hypertension and dyslipidemia during the study period.
The results of the sensitivity analyses were consistent with the results of the primary analysis (Supplemental Tables S4–S7).
Comparison of complications in the ESW and OSR groups
There was no intergroup difference in the incidence of viral or bacterial infections during the study period. However, 1 year after transplantation, cardiovascular risk factors were more prominent in the OSR group. Relative to patients in the ESW group, patients in the OSR group (i) were more likely to have high blood pressure (11.7 versus 40.2%, respectively; p < 0.001), and (ii) displayed a greater increase in serum triglyceride levels (+9.7 ± 118.6 mg/dl versus −37.2 ± 177.9 mg/dl, respectively, p = 0.004). Likewise, the proportion of patients with cardiovascular events (arterial thrombosis of the lower limbs and myocardial infarction) was higher in the OSR group (2.5%) than in the ESR group (0%), although the intergroup difference was not significant (Table 4). These results were consistent when comparing patients with initial ESW with or without steroids resumption and those with initial long-term steroid therapy (Table S8).
At M0, the ESW (with or without steroids resumption) and OSR groups did not differ significantly with regard to the acute graft rejection rate (8.2% versus 5.5% respectively, p = 0.437). Likewise, the two groups did not differ significantly with regard to the appearance of DSAs during the study period (5.9% in the ESW group versus 2.6% in the OSR group; p = 0.167) or the change in kidney graft function (measured as the serum creatinine level) (Table S8).
Discussion
The primary objective of the present study was to evaluate the putative beneficial effects of ESW on the gain in BMD during the year following kidney transplantation. Whereas most published studies of ESW have focused on cardiovascular complications, the present study is the first to have evaluated the change in BMD during the 12 months following kidney transplantation in patients with ESW versus patients with other steroid regimens. Our results evidenced a significant increase (by about 0.04 g/cm2, +4.2%) in BMD at the lumbar spine and the femoral neck; this corresponds to a T-score gain of about 0.3 in the ESW group (particularly in men under 60 and patients without ABD), relative to the OSR group. Moreover, the percentage of patients in the ESW group with normal BMD had increased by 21.1 percentage points 1 year after kidney transplantation and the proportion of patients with osteopenia decreased by 10.6 percentage points. None of the patients in the ESW group had osteoporosis at M12, despite the absence of specific anti-osteoporotic treatments (whereas 10.5% of patients in this group had densitometrically confirmed osteoporosis at M1).
In addition to treating any osteoporosis-inducing diseases present (such as hypogonadism), or withdrawing any medications that induce bone loss (such as steroids), only specific anti-osteoporotic drugs can produce an increase in BMD. Indeed, in a recent meta-analysis including 18 randomized controlled trials with a total of 1200 kidney recipients with long-term steroid therapy (median follow up of 12 months), bisphosphonate plus calcium and/or vitamin D revealed a significant gain in percent BMD change than calcium and/or vitamin D alone at the femoral neck (mean difference, 5.83, 95% credible interval, 1.61–9.27) and at the lumbar spine (mean difference, 5.48, 95% credible interval, 2.57–8.42). 43 Thus, in our population, ESW had much the same positive effect on BMD as specific anti-osteoporotic treatments do in kidney recipients with long-term steroid therapy. The spontaneous BMD gain observed in our ESW group might be related to the restoration of normal calcium, phosphate and calcitriol levels and the absence of the steroids’ harmful effects on bone. Indeed, low BMD in patients with end-stage renal disease and renal osteodystrophy (i.e. osteitis fibrosa as a result of hyperparathyroidism, osteomalacia or ABD) is due to perturbed calcium, phosphate and calcitriol levels; 44 however, these variables normalize within 3–12 months of transplantation.45,46 Nevertheless, two of the patients had a PTH value at M12 below the assay’s limit of detection (4.6 pg/ml); there were few changes in these variables during this time frame.
In the present study, the patients with OSR displayed a decrease in BMD at all three measurement sites 1 year after transplantation; this was probably linked to the ongoing steroid therapy. In the literature, the post-transplantation decrease in BMD in patients receiving long-term steroid therapy is mainly attributed to the cumulative dose of corticosteroids. In an Indian study, there was a significant post-transplantation decrease in lumbar spine BMD at 3 months (11.8%, p < 0.0001) and 6 months (16%, p < 0.0001). 47 In a study in the United States, Julian et al. reported bone losses of 6.8 ± 5.6% and 8.8 ± 7.0% at the lumbar spine respectively 6 and 18 months after transplantation. 16 Similar losses have been observed at the hip 5 months after kidney transplantation. 15 Other risk factors have been identified, including the time on dialysis prior to transplantation, age at transplantation, vitamin D deficiency, and a low body mass index (<23 kg/m2).18,19
Calcitriol [1,25(OH)2D3] regulates calcium homeostasis and bone cell differentiation, 48 and its deficiency leads to osteoporosis through an increase in PTH levels. 49 Glucocorticoids also lead to osteoporosis by decreasing intestinal calcium absorption, increasing the urinary excretion of calcium, enhancing bone resorption, and decreasing bone formation. 50 Furthermore, the use of corticosteroids contributes to low serum levels of 25(OH) vitamin D. 51 Hence, vitamin D prophylaxis might be more effective in patients on long-term steroid therapy; however, the risk of associated hypercalcemia and hypercalciuria through excessive calcium and vitamin D supplementations should also be considered. 52
It has been firmly established that the fragility fracture risk is correlated with BMD.53,54 Indeed, in postmenopausal women, there is a continuum between the BMD decrease and the fracture risk, with no threshold value: the fracture risk is multiplied by a factor of two for a one SD decrease in the BMD T-score. 24 Our present results evidenced significant BMD T-score gains for the lumbar spine and the femoral neck (~0.3 and ~0.2, respectively), which suggest that the fracture risk is lower in the patients with ESW. In fact, none of the patients in the ESW group suffered a fragility fracture. In contrast, four patients in the OSR groups suffered a fragility fracture (L3 vertebrae, the hip, a rib, and the scapula glenoid), corresponding to an incidence of 13.5 fractures per 1000 person-years. This intergroup difference in fracture incidence was not statistically significant – probably due to the small number of events. Longer-term follow up would be required before firm conclusions can be drawn in this respect. In a recent, prospective, observational study of 518 kidney transplant recipients with an average follow-up period of 5.2 years, Evenepoel et al. found a fracture incidence of 14.2 per 1000 person-years – much the same value as in the present study. 55 Moreover, Evenepoel et al. found an association between incident fractures on one hand and BMD at the lumbar spine and at the femoral neck on the other. 55
To the best of our knowledge, only one study [a single-center study of 259 patients, including 75 (29%) with ESW] has evaluated the impact of ESW on BMD. Segaud et al. evidenced a significant increase in BMD at the lumbar spine (+3.1 ± 5.6%, p < 0.001), the femoral neck (+3.2 ± 7.3%, p < 0.0001) and the total hip (+4.1 ± 6.9%, p < 0.0001) in patients with ESW between 9 and 24 months after kidney transplantation, with greater gains in patients treated with bisphosphonates. 56 One of the main limitations of Segaud et al.’s study was the absence of a DXA assessment before or following kidney transplantation. This lack of data prevented the researchers from estimating the BMD variation in the year following transplantation – a period during which the fracture risk appears to be higher. 57 Furthermore, Nikkel et al.’s study of a large American cohort of kidney recipients found that corticosteroid withdrawal was associated with a 31% reduction in the fracture risk [hazard ratio: 0.69; 95% confidence interval (CI): 0.59–0.81], which also suggests that ESW has a beneficial positive effect on bone. 58 However, the latter study had a number of limitations because it was based on a registry and claims data: the documentation of osteoporosis risk factors was not exhaustive, and it was not possible to distinguish between traumatic fractures and fragility fractures. Moreover, Nikkel et al.’s study population was younger than our population, and contained more women and more black recipients. Lastly, the study period was different: the patients had undergone kidney transplantation between 2000 and 2006. 58
Along with positive effects on bone, ESW was associated with a reduction in several cardiovascular risk factors in our study population. Indeed, patients in the ESW group were less likely to have uncontrolled arterial hypertension at M12, and had a significantly lower serum triglyceride level and significantly greater weight gain. Multicenter studies with long follow-up periods (3 and 5 years) have shown that steroid withdrawal is associated with a reduction in cardiovascular risk factors (hyperlipidemia, hypertension, and diabetes mellitus) and provides similar long-term outcomes for renal allograft survival and function.6,8 In the present study, ESW group experienced more acute graft rejections (6.7% in the ESW group versus 4.7% in the OSR, p = 0.766) and more appearance of DSAs during the study period (6.7% in the ESW group versus 1.9% in the OSR group; p = 0.099) even if the two groups did not differ significantly.
One of the main strengths of our study was its exhaustive documentation of osteoporosis risk factors, which thus limited confounding bias. Indeed, age, sex, body mass index, previous fragility fractures, glucocorticoid use, secondary osteoporosis (i.e. a history of diabetes mellitus, osteogenesis imperfecta, hyperparathyroidism, or hypogonadism), rheumatoid arthritis, a history of hip fracture in the parents, current cigarette smoking, alcohol intake, and low femoral neck BMD are the risk factors used in a fracture risk assessment tool for predicting the 10-year major osteoporotic fracture rate (vertebrae, the hip, the forearm, and the proximal humerus). 59 Moreover, other risk factors associated with low BMD or a high fracture risk in kidney transplant recipients (i.e., duration of dialysis before transplantation and vitamin D deficiency) were exhaustively documented.18,19 Another strength of our study relates to the use of IPTW; this approach enables observational studies to be designed in much the same way as randomized controlled trials, and thus reduces residual confounding bias and misclassification bias. 36
The main limitations of our study are related to the small number of incident fractures, which probably prevented us from observing intergroup differences. Despite the small number of patients in the ESW group, our data highlighted a significant BMD gain at the lumbar spine and the femoral neck in patients with ESW, and thus suggest that ESW has a positive effect on BMD in kidney transplant recipients, especially since patients in the ESW group were exposed to a higher risk of osteoporosis. Furthermore, our follow-up period may have been too short for an adequate investigation of the fracture risk in this population. Pre-transplant DXA data could not be exploited because (i) they were not collected systematically during the dialysis period, and (ii) even when they were available, the time interval between the DXA assessment and transplantation was very variable. Nevertheless, we consider that the BMD at M1 post-transplantation was probably similar to the value at M0 at the time of transplantation because (i) it is known that PTH, phosphate, calcium, calcitriol, and FGF-23 levels stabilize within 3 to 12 months of transplantation45,46 and (ii) corticosteroid-induced bone loss appears to peak 3–6 months after the initiation of treatment.60,61 Alternatively, and considering the broad age range in our population (77 of the patients were under the age of 40), a Z-score could have been used but these data were frequently missing. Given that the DXA data were collected mainly from consultation reports, the Z-score was (unlike the T-score) rarely reported – probably because it is not intended for the diagnosis of osteoporosis in adults. 62 Furthermore, several other risk factors for osteoporosis (such as the lack of physical activity or late puberty) could not be taken into account because of the study’s retrospective design. Moreover, ESW and OSR patients had the same steroid induction therapy, that could explain a similar deleterious effects of this on BMD at M1 (BMD at M1 was similar between groups). Lastly, this was a single-center study, with all the inherent limitations; however, bone density was always evaluated with the same device, and thereby made our intergroup comparisons more robust.
Conclusion
In a study of a cohort of kidney transplant recipients, we found that ESW was associated with a spontaneous increase in BMD at 12 months post-transplantation (relative to patients on long-term steroid therapy). The BMD gain appeared to be linked to a lower fracture risk, although firm conclusions on this matter would require a longer follow-up period.
Supplemental Material
Supplementary_information – Supplemental material for Early steroid withdrawal has a positive effect on bone in kidney transplant recipients: a propensity score study with inverse probability-of-treatment weighting
Supplemental material, Supplementary_information for Early steroid withdrawal has a positive effect on bone in kidney transplant recipients: a propensity score study with inverse probability-of-treatment weighting by Benjamin Batteux, Valérie Gras-Champel, Mathilde Lando, François Brazier, Romuald Mentaverri, Isabelle Desailly-Henry, Amayelle Rey, Youssef Bennis, Kamel Masmoudi, Gabriel Choukroun and Sophie Liabeuf in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Author contributions
Benjamin Batteux contributed to the conception/design of the work, acquisition, analysis, and interpretation of data for the work, and drafting the manuscript. He approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Valérie Gras-Champel contributed to interpretation of data for the work, revised it critically for important intellectual content, approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Mathilde Lando revised the work critically for important intellectual content, approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
François Brazier revised the work critically for important intellectual content, approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Romuald Mentaverri revised the work critically for important intellectual content, approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Isabelle Desailly-Henry revised the work critically for important intellectual content, approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Amayelle Rey contributed to analysis of data for the work, revised it critically for important intellectual content, approved the version to be approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Youssef Bennis revised the work critically for important intellectual content, approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Kamel Masmoudi revised the work critically for important intellectual content, approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Gabriel Choukroun contributed to the conception/design of the work, revised the work critically for important intellectual content, approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Sophie Liabeuf contributed to the conception/design of the work, the interpretation of data for the work, and the drafting of the manuscript, approved the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
