Abstract
Objective:
The objective of this study was to analyze health outcomes, resource utilization, and costs in osteoarthritis patients with chronic nociceptive pain who began treatment with an opioid in real-world practice in Spain.
Methods:
We designed a non-interventional, retrospective, longitudinal study with 36 months of follow-up using electronic medical records (EMRs) from primary care centers, of patients aged 18+ years who began a new treatment with an opioid drug in usual practice for chronic pain due to osteoarthritis. Health/non-health resource utilization and costs, treatment adherence, pain change, cognitive functioning, and dependence for basic activities of daily living (BADL) were assessed.
Results:
A total of 38,539 EMRs [mean age (SD); 70.8 (14.3) years, 72.3% female; 53.3% hip/knee, 25.0% spine, and 21.7% other sites] were recruited. A total of 19.1% of patients remained on initial opioid at 36 months, without significant differences by osteoarthritis site (p = 0.125). Mean total adjusted cost was €17,915, with 27.7% corresponding to healthcare resources and 72.3% to lost productivity. Hospital admissions for osteoarthritis-related surgical interventions accounted for 15.8% of total healthcare cost. A slight mean pain reduction was observed: –1.3 points, –16.9%, p < 0.001, with increases in cognitive deficit (+3.3%, p < 0.001) and moderate to total dependence for BADL (+15.6%, p < 0.001) in a median duration of opioid use of 203 days (IQR: 89–696).
Conclusions:
In real-world practice in Spain, opioid use in osteoarthritis was high, but with low adherence. There were meaningful increases in resource use and costs for the National Health System. Pain reduction was modest, whereas cognitive impairment and dependence for BADL increased significantly.
Keywords
Lay Summary
Osteoarthritis is a degenerative joint disease characterized by cartilage deterioration, pain and impairment in functionality. It is highly prevalent all over the world and one of the most frequent causes of disability. Therefore, osteoarthritis is a substantial public health problem, associated with frequent utilization of health resources and related cost.
The therapeutic approach includes non-pharmacological measures, drugs, and joint replacement surgery in the most disabling cases. Opioids are a pharmacological group widely used to treat chronic pain non-responsive to other treatments, although they are not recommended for osteoarthritis by many scientific societies.
In this study, we analyzed the health outcomes (pain change, cognitive functioning, and dependence for basic activities of daily living), resource utilization, and cost consequences of using opioids in the treatment of adult patients with chronic pain associated to osteoarthritis under usual care practice (real world) in Spain. A total of 38,539 patients were analyzed.
We concluded that in the real world in Spain, opioid use in osteoarthritis was high, but with low adherence and does not meet patient needs for adequate pain management. There were meaningful increases in resource use and costs for the National Health System, whereas pain reduction was modest and cognitive impairment and dependence for basic activities of daily living increased significantly.
Introduction
Osteoarthritis is a degenerative joint disease characterized by cartilage deterioration, with proliferative reaction of the subchondral bone and inflammation of the synovial membrane.1,2 Chronic nociceptive joint pain (>3 months duration) is the most common symptom of osteoarthritis, and one of the most frequent causes of disability, and therefore osteoarthritis is a substantial public health problem. 3 Studies show chronic pain affects 10–30% of European adults. 4 In Spain, the estimated prevalence of any-type chronic pain is 16.6%, and more than 50% of patients have limitations on the basic activities of daily living (BADL), 30% felt sad and/or anxious, and 47.2% indicated the pain was affecting their family life. 5 There are wide variations in the prevalence of osteoarthritis according to the types of study and the criteria used to define the disease. 6 Around 10% (range: 6–24%) of adults have moderate or severe osteoarthritis, and the incidence increases with age. 7 According to EPISER data, 8 the prevalence of knee osteoarthritis is 13.9%, osteoarthritis of the hands 7.9%, and osteoarthritis of the spine, the most frequent site, 10.1–15.5%. Pain and disability associated with osteoarthritis have a negative effect on the perceived quality of life and are one of the main causes of work absenteeism, causing substantial health and non-health costs (temporary or permanent disability) for the Spanish National Health System (NHS) and society.1,4,8
The therapeutic approach includes non-pharmacological measures, pharmacological treatments, and joint replacement surgery in the most disabling cases.2,9 Opioids are a pharmacological group characterized by a selective affinity for central and peripheral opioid receptors. 10 They are widely used to treat acute pain and moderate–severe chronic pain non-responsive to other treatments,11,12 although they are not recommended for osteoarthritis at any location by the most recent Osteoarthritis Research Society International (OARSI) non-surgical treatment guidelines. 13 The utility of strong opioids is proven in the relief of cancer pain but is controversial in the relief of chronic non-cancer pain, with doubts about their long-term efficacy and functional results in the relief of pain in osteoarthritis compared with other analgesics.13,14 Opioids have adverse effects that may lead to drug withdrawal and dependence in a percentage of cases. Therefore, treatment must be individualized according to the patient and type of pain, assessing the benefits and risks.10,15 Opioid prescriptions have increased in the last decade in both Europe and in Spain, although their use remains lower than in the United States. 16 In Spain, according to the Spanish Medicinal Agency, opioid consumption has increased from 10.3 defined daily doses (DDD) per 1000 inhabitants in 2010 to 18.9 DDD in 2018 (close to 87% in 9 years). 17 The most frequently prescribed opioids were tramadol, tramadol in combination and tapentadol, representing 65.7% of total opioid consumption in the same year. The most widely used weak and strong opioids were tramadol in combination (1.8–5.3 DDD) and fentanyl (1.4–2.3 DDD), respectively. 17 For strong opioids, the transdermal route is by far the most frequently used.18,19 The weak opioids included codeine, dihydrocodeine and tramadol, alone or in combination. The strong opioids included morphine, oxycodone, hydromorphone, pethidine, fentanyl, buprenorphine, methadone, and tapentadol.17,20
In Spain, opioid use and that of other resources associated with the treatment of osteoarthritis patients with chronic nociceptive pain have increased in recent decades, justifying assessing the clinical and economic consequences in usual clinical practice (real world). The objective of this study was to analyze the use of health and non-health resources and associated costs in osteoarthritis patients with chronic nociceptive pain who began opioid treatment from the NHS perspective. In addition, drug persistence, the medication possession ratio (MPR), and the clinical effectiveness of opioid treatment on pain, BADL, and cognitive functioning were evaluated during a 3-year follow-up period.
Method
Design and study population
A non-interventional, multicenter, longitudinal retrospective study was designed, with a new user design, based on the review of electronic medical records (EMRs). The study population was obtained from the unified health records of health providers contained in the BIG-PAC® anonymized database (http://www.encepp.eu/encepp/search.htm). The data comes from EMRs and complementary databases of the financing/provision of public services of seven Spanish Autonomous Communities including 1.9 million patients. The patient data included in the database are anonymized as specified in Spanish Law 15/1999, of 13 December, on Personal Data Protection. EMRs of patients who started a new treatment (index date) with any weak or strong opioid drug in monotherapy or in combination with other analgesics between 1 January 2010 and 31 December 2015 (recruitment period) were included. Patients were followed from the index date for a maximum of 3 years and/or until discontinuation of opioid treatment (follow-up period), change to another analgesic, loss to follow-up, and/or until any-cause death.
EMRs fulfilling the following inclusion criteria were recruited: (a) patients aged ⩾18 years starting treatment with a new opioid for the treatment of osteoarthritis for the first time with chronic nociceptive pain of more than 3 months of evolution from the date of diagnosis; (b) refractory to previous analgesia with at least one usual analgesic, such as non-steroidal anti-inflammatory drugs (NSAIDs), paracetamol, metamizole, etc.; (c) active patients (two or more health records in the database in the 12 months before study inclusion; (d) inclusion in the chronic medication prescription program (with a record of the daily dose, the time interval and the duration of each treatment administered (⩾ 2 prescriptions during the follow-up period); and (e) regular monitoring (⩾ 2 health records in the computer system). Exclusion criteria were: (a) subjects transferred out to other centers, displaced, or out of area; (b) permanently institutionalized patients; (c) terminal disease and/or dialysis (ICD-10: N18); (d) associated neuropathic/radiculopathy pain (ICD-10: G50-65) or oncological pain (ICD-10: G89.6). The records of osteoarthritis patients were obtained using the International Classification of Diseases (10th edition) Clinical Modification (ICD-10-CM): (a) hip and knee (M16, M17), (b) spine (M54.5), and (c) other types (M15, M18, M19, M40, M41). The criteria followed were always at the discretion of the attending physician. The diagnoses were related to the prescription made by the type of chronic osteoarthritis pain. Chronic pain was defined as pain ⩾3 months and refractory pain as no adequate analgesic control at doses within the therapeutic range recommended by product data sheets [pain severity ⩾5 in an 11-points numeric rating scale (NRS)]. 21 Three study groups were differentiated according to the osteoarthritis site: (a) hip and knee, (b) spine, and (c) other sites. Since a patient may suffer more than one type of chronic osteoarthritis pain, the inclusion of a patient in a specific group was made according to the indication of the opioid medication recorded in the database. The follow-up period from the patient’s index date was a maximum of 3 years (index date to 12, 24, and 36 months, respectively, in accumulated periods).
Demographic and comorbidity variables
The demographic and comorbidity variables collected were: age (continuous and by ranges), time from diagnosis, sex, and history (ICD-10-CM) of arterial hypertension, diabetes, dyslipidemia, obesity, smoking, ischemic heart disease, cerebrovascular accident, heart failure, kidney failure, asthma, chronic obstructive pulmonary disease (COPD), dementia, depression, and malignant neoplasia (all types). As a summary variable of general comorbidity: (a) the Charlson comorbidity index was used as an approximation to severity, 22 and (b) the number of chronic comorbidities were collected. These data were obtained on the index date.
Treatment, incidence rate, and adherence with opioids
The medications (active ingredients) indicated for chronic osteoarthritis pain were obtained according to the Anatomical Therapeutic Chemical Classification System (ATC) classification N02AA01 to N02AX06. The information was obtained from pharmacological prescription records. The choice of medication in a specific patient was at the discretion of the physician. The medical specialty that initiated the prescription was determined. Prescriptions included: (a) non-opioid analgesics (NSAIDs, paracetamol, metamizole), (b) weak opioids (codeine, dihydrocodeine, tramadol, dextropropoxyphene), and (c) strong opioids (buprenorphine, fentanyl, hydromorphone, morphine, naloxone, oxycodone, pethidine, tapentadol). The study records were obtained during the 12 months prior to the index date, and for the following 36 months. The index date was the start of a new opioid treatment (weak or strong), from the date of diagnosis of chronic nociceptive pain due to osteoarthritis. The incidence rate of opioid treatment, expressed as the number of patients with new treatments with any weak/strong opioid per 1000 inhabitant-years, was determined for each year of recruitment.
Adherence was defined as the percentage of patients that at 12, 24, and 36 months of index date, continued in the study with possession of the initial opioid drug that led to be included in the study. 23 The MPR was measured as the ratio between the number of days with medication dispensed to the patient and the days of follow-up (time in treatment) in the study, as expressed as a percentage. Persistence was defined as the days of follow-up in the study and was calculated as the difference between the start date of the medication (day of reception of the opioid motivating study inclusion) and the date of completion of the study for the patient. 23 The end date was the first to occur during the 3-year follow-up: (a) discontinuation due to poor tolerability (> 30 days without renewing the initial medication dispensed in community pharmacy without refills during study follow-up), (b) discontinuation due to poor effectiveness (pain on a 11-point NRS > 5 points in the last available measurement, and >1 prescription), (c) change to another analgesic treatment with a drug other than an opioid or change to an strong opioid if treated with a weak opioid, (d) loss to follow-up, and/or (e) any-cause death.
Resource use and cost analysis
The societal and Spanish NHS perspectives were considered to calculate healthcare and indirect costs. Direct healthcare costs were those relating to healthcare activity (medical visits, days of hospitalization, emergency visits, diagnostic and therapeutic requests, etc.) made by healthcare professionals. Non-healthcare costs (indirect costs) were those relating to lost productivity (days of sick leave due to temporary or permanent disability). Costs were expressed as the mean cost per patient (mean per unit) throughout study. Cost of analgesic drugs, non-opioids and opioids, were also computed daily while on therapy. Supplemental Table 1 shows the unit costs of healthcare resources and days of productivity lost applied in the economic evaluations (in 2018 euros). Prices were based on hospital accounting, except for medication and days of productivity lost (days of sick leave). All analgesic medications included opioids, were quantified by the retail price per pack at the time of dispensing from the community pharmacy (according to the Drug Catalogue of the General Council of Associations of Official Pharmacists of Spain, available from: https://botplusweb.portalfarma.com/botplus.aspx). 24 Days of productivity lost were quantified according to the mean inter-professional wage [source: Spanish Statistical Office (Instituto Nacional de Estadística, INE)]. 25 The analysis did not consider non-healthcare direct costs, that is, out-of-pocket costs or costs paid by the patient/family, as these are not recorded in the database and the study had no direct access to patients. Total resource use was differentiated (health and lost productivity) during the 12 months before the index date and at 12 and 36 months of follow-up.
Clinical effectiveness
Clinical effectiveness was measured by: (a) the variation in pain intensity (11-point NRS), 21 and, especially in subjects aged ⩾65 years, (b) the functional variation in the basic activities of daily living (Barthel index), 26 and (c) cognitive variation [Mini-Mental State Examination (MMSE)] 27 between the closest date before the start date (index date) and the end date (discontinuation) of the study. The Barthel scale is an ordinal scale used to measure performance in BADL. Each activity item is scored on this scale with a given number of points assigned to each level of activity or performance. 26 It uses 10 different basic activities and mobility. A lower number is associated with a greater likelihood of not being able to live at home with independence, therefore, needing help from a caregiver for one or more basic BADL. For all scales, the absolute variation in natural units and the relative percentage change over the baseline value were calculated. To interpret the changes, patients were classified into three groups according to pain severity: 21 <5, no pain/mild pain; 5–7, moderate pain; >7, severe pain. For the BADL, the criteria for interpreting the Barthel scale considered dependence as a moderate to total limitation on functionality (limitation <90). 26 For cognitive functioning, a MMSE score <20 was considered as a mild to severe cognitive deficit. 27
Statistical analysis
The data was validated in the BIG-PAC® database through specific computer scripts and underwent exploratory analysis, with observation of frequency distributions, searching for possible registration or coding errors. External representativity of this database is also guaranteed. 28 A descriptive-univariate statistical analysis was made. Qualitative data were described using absolute and relative frequencies and quantitative data using means, standard deviation, medians, and the 25th and 75th percentiles of the distribution [interquartile range (IQR)]. The 95% confidence intervals (CIs) for the estimate of parameters were based on the total number of subjects without missing values. The normality of distribution was checked using the Kolmogorov–Smirnov test. A survival analysis was made by estimating the Kaplan–Meier curves (log rank test) to analyze the persistence of opioid treatment. The bivariate analysis used the corresponding tests for paired groups for each subgroup (before and after). The before–after differences are presented with the 95% CIs of the difference calculated by non-parametric resampling (1000 bootstrap iterations). An analysis of covariance (ANCOVA; estimation of marginal means; Bonferroni adjustment) was used to correct healthcare costs when independent groups were compared. Covariates included were sex, age, general comorbidity (number and Charlson comorbidity index), and time from diagnosis. The incidence rates of new weak/strong opioid treatments were calculated annually in each of the six study years as the number of new treatments in the year per 1000 inhabitant-year. The statistical interpretation of the evolution of the incidence rate was made using trend analysis, accepting trends with an R2 value ⩾ 0.7 as relevant. Likewise, the Mantel–Haenszel χ2 test was applied to compare incidence rates between weak and strong opioids and, in each group of opioids, each year with the previous year. The analysis was made using SPSS (version 23.0).
Confidentiality of information/ethical aspects and reporting guidelines
The confidentiality of the records (anonymous and dissociated) was respected according to the Law on Protection of Personal Data. The study was classified by the Spanish Agency for Medicines and Health Products as EPA-OD and subsequently approved by the Research Ethics Committee of the Hospital de Terrassa, Barcelona, Spain. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement guidelines for reporting observational studies were followed. 29
Results
Of an initial selection of 1,280,684 subjects aged ⩾18 years who sought medical care during the 6-year recruitment period, 124,798 were diagnosed with osteoarthritis and, of these, 34% started a new opioid treatment (n = 42,429). A total of 3891 records (9.2%) were excluded due to non-compliance with selection criteria, loss to follow-up, or data missing/inconsistencies. Missing/inconsistent data were observed in 3.2% of cases only, and due to this, imputation was not done. Therefore, 38,539 EMRs that met the study selection criteria were extracted (Figure 1). The prevalence of osteoarthritis was 9.7% (95% CI: 8.6–10.8%). The distribution of patients by osteoarthritis type was: 53.3% hip/knee, 25% spine, and 21.7% other sites. The incidence rate (× 1000 inhabitant-years) of opioid use is shown in Figure 2. In the study period, a significant increase in weak and strong opioid use was observed (p < 0.001). In both cases, there was a highly significant linear growth: R2 = 0.963 for weak opioids and R2 = 0.991 for strong opioids.

Study flow diagram.

Incidence rate (× 1000 inhabitant-years) of opioid use in the years of study recruitment.
Table 1 shows the sociodemographic characteristics and general comorbidity of study participants. The mean age was 70.8 (SD: 14.3) years and 72.3% were female. Treatment adherence, persistence, MPR, and reasons for discontinuation by study groups are detailed in Table 2. The median duration was 203 days (IQR: 89–696 days). At 36 months of follow-up, overall treatment adherence to initial opioid was 19.1% (95% CI: 18.7–9.5%): 18.8% hip/knee, 19.2% spine, and 19.8% other locations (p = 0.125). The MPR was 74.0% (by groups: 74.8%, 73.8%, and 71.4%, respectively; p < 0.001). Supplemental Figures S1 and S2 show the Kaplan–Meier curves for treatment persistence and the median treatment duration according to the reason for discontinuation and at 36 months follow-up, respectively. Patients with hip/knee osteoarthritis had a longer persistence time, although the difference was small. The change to strong opioid was significantly associated with a longer persistence time on weak opioid, both with respect to any-cause abandonment and changes for other reasons (p < 0.001 in both cases).
Baseline characteristics (demographic and morbidity) of patients by study groups.

Values expressed as percentage or mean (SD, standard deviation).
COPD, chronic obstructive pulmonary disease.
Treatment persistence and adherence, medication possession ratio and deaths by study groups (during the follow-up period).
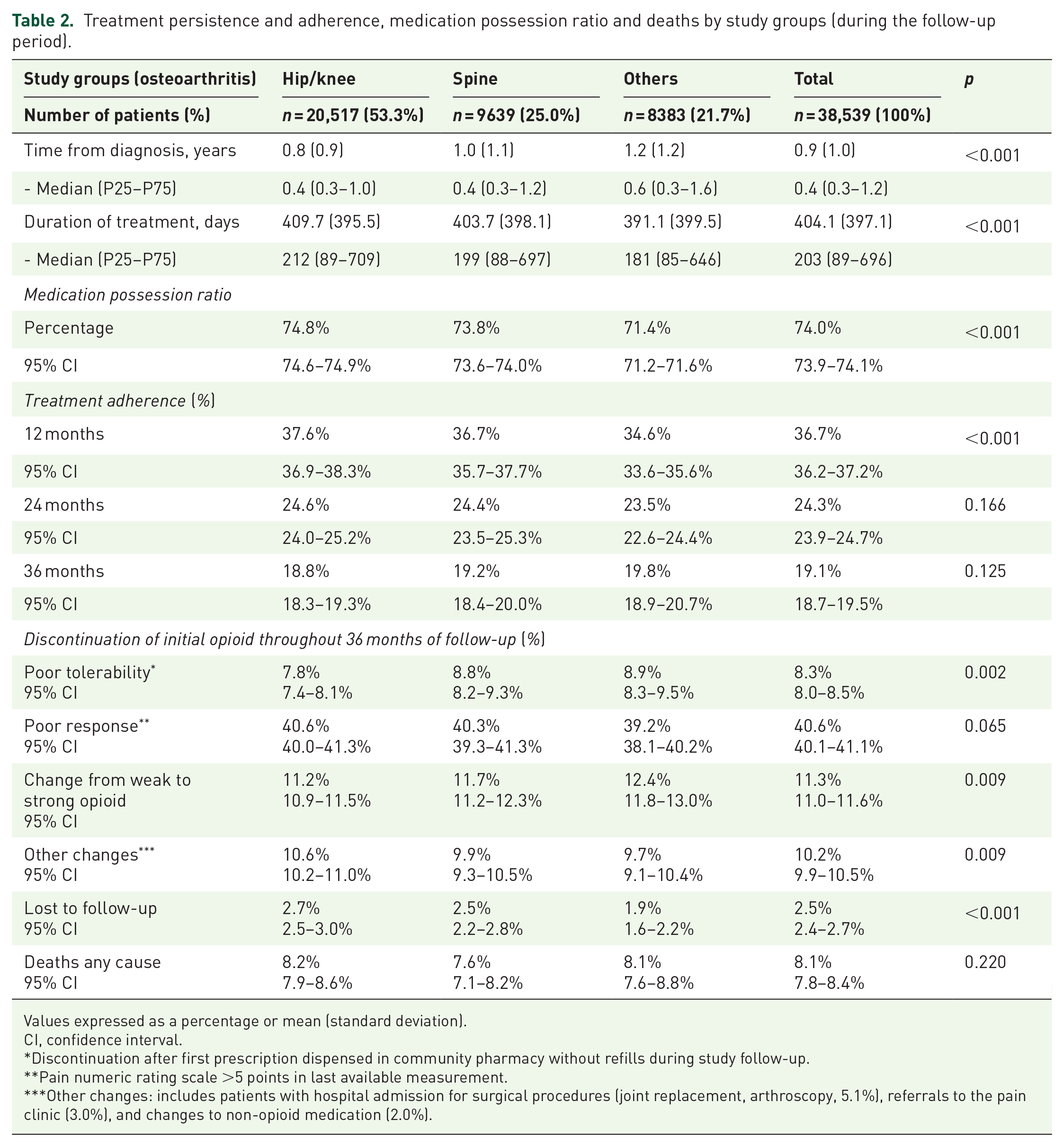
Values expressed as a percentage or mean (standard deviation).
CI, confidence interval.
Discontinuation after first prescription dispensed in community pharmacy without refills during study follow-up.
Pain numeric rating scale >5 points in last available measurement.
Other changes: includes patients with hospital admission for surgical procedures (joint replacement, arthroscopy, 5.1%), referrals to the pain clinic (3.0%), and changes to non-opioid medication (2.0%).
Table 3 shows the concomitant medication prescribed by groups and follow-up periods. In the 12 months prior to opioid medication, the mean concomitant medication was 1.4 analgesics per patient compared with 1.1 at 36 months. The reduction was mainly due to lower paracetamol use (82.4% versus 42.4%, p < 0.001), and was observed in all study groups, whereas the proportion of patients who continued to take concomitant NSAIDs did not vary significantly, except for a discrete reduction in all groups at 12 months (Table 3): 28.6% of all patients who started treatment with a strong opioid were previously prescribed a weak one.
Concomitant medication by study groups and follow-up periods.

Values expressed as a percentage or mean (SD, standard deviation).
NSAIDs, Non-steroidal anti-inflammatory drugs.
Percentage of patients using weak opioids before starting treatment with strong opioid, in which the use of strong opioid is the reason for study inclusion.
Resource use and gross costs by the different study groups (disaggregated by each component) are shown in Supplemental Tables S2 and S3. At 36 months, the total cost was €690.4 million, of which 27.7% corresponded to direct healthcare costs and 72.3% to days off-work. The costs of hospital admissions accounted for 42.9% of healthcare costs, primary healthcare visits 15.5%, and opioid medication 14.2%. Hospital admissions for osteoarthritis-related surgical interventions accounted for 36.8% of hospital costs, which doubled the annual hospital cost from €411 in the first year of follow-up to €856 during the second and third years (calculated from Supplemental Table S3), which contrasts with the cost of €491 before the start of opioid treatment. Table 4 shows the health and non-health costs corrected by covariates, and the costs of non-narcotic analgesia and opioids. The mean/unit of the total cost during the 12 months before opioid medication was €5994 (Table 4), while during the subsequent 12 months it was increased by a 3.3% to €6193: a difference of €199 (196–202, p < 0.001, effect size; 0.02) and up to 36 months of €17,915 (equivalent to €5972/year). Healthcare cost grew on average by a 21.5% from an initial annual mean cost of €1276 (1252–1300) to €1550 (1521–1579) after 12 months of starting opioid analgesia: a difference of €274 (238–310, p < 0.001; effect size 0.12). The significant increase in the total cost at 12 months was generated by the cost of introducing opioids in the patient’s therapy and, in the case of hip/knee and spinal osteoarthritis, because a significant increase in the costs of other healthcare resources (Table 4). The opioid medication administered and the medical specialty that initiated the prescription during the follow-up period are described in Table 5. Weak opioids were prescribed to 61.2% of patients and strong opioids to 38.8% (p < 0.001). Almost all weak opioids prescribed were tramadol or tramadol + paracetamol (97.2%).
Health and non-health costs corrected by covariates (in euros) according to study groups and total.
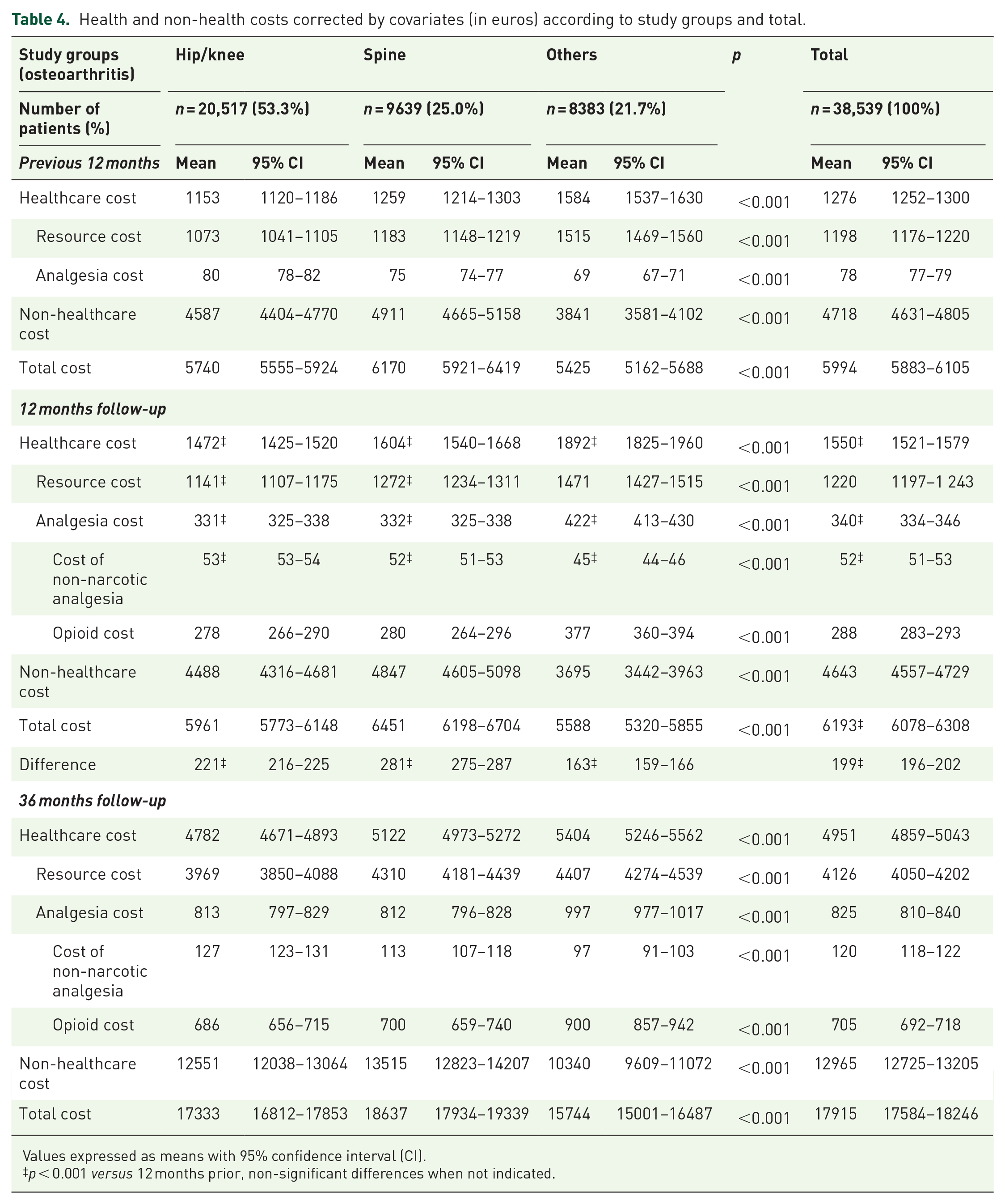
Values expressed as means with 95% confidence interval (CI).
p < 0.001 versus 12 months prior, non-significant differences when not indicated.
Opioid administered and prescribing medical specialty during the follow-up.

Values expressed in absolute numbers (n) and percentage (%).
AAS, acetylsalicylic acid; ATC, Anatomical Therapeutic Chemical Classification System; NSAID, non-steroidal anti-inflammatory drug.
Tramadol was the most frequent weak opioid (40.5%) and fentanyl the most frequent strong opioid prescribed (14.8%) with small between-group differences. The most frequent prescribing specialties were family medicine (74.2%) and traumatology (9.7%). Strong opioids were proportionately more frequently prescribed by reference specialists (33.4% versus 21.1%, p < 0.001). Supplemental Table S4 shows the mean cost per day of treatment (analgesics and opioids) by study groups. The mean cost/day of treatment was €2.65/day per patient distributed as follows: (a) cost of non-opioid analgesic medication €0.15/day, (b) cost of opioid medication €2.50/day, (c) weak opioid cost €0.50/day, and (d) strong opioid cost €5.68/day. During the follow-up, the cost/day of non-narcotic analgesics decreased, and the cost of opioids increased significantly (Supplemental Table S4, p < 0.001).
Table 6 shows the evolution of clinical effectiveness. Pain intensity decreased significantly, but discretely by –1.3 points (–16.9%, p < 0.001), and only 6.4% of patients reducing their grade of pain to mild/no pain from initial moderate or severe pain after opioid abandonment. However, the degree of moderate to total dependence for BADL increased significantly by 15.6% with opioid treatment (from 80.4% to 96.4% of users) and was slightly higher (16.5%) in patients with hip/knee osteoarthritis, whereas the proportion of people with cognitive deficits according to the MMSE increased significantly by 3.3% (p < 0.001). These results were similar for all osteoarthritis sites but were slightly higher in patients with spinal osteoarthritis (4.7%).
Clinical effectiveness of opioid use expressed as effect on pain intensity, cognitive functioning, and disability for BADL, in the global sample and by osteoarthritis site.

Values expressed as a percentage or mean (SD).
p < 0.001, †p < 0.01, *p < 0.05 in the final baseline adjusted differences. Not significant when not indicated.
Pain intensity was measured with a 11-point NRS [scored 0 (no pain) to 10 (worst imaginable pain)]. Cognitive function was assessed with the MMSE test, establishing a cognitive deficit for scores ⩽20. BADL were evaluated using the Barthel test, interpreting a moderate to total disability (dependence) for scores ⩽90 points. MMSE and Barthel results were obtained in the subsample of patients ⩾65 years old; NRS = 38,426, Barthel = 23,356, MMSE = 22,874, with response rates of 99.7%, 91.0%, and 89.1%, respectively.
BADL, basic activities of daily living; MMSE, Mini-Mental State Examination; NRS, numerical rating scale.
Discussion
The findings of this study show a high current utilization of strong and weak opioids for the treatment of chronic pain associated with osteoarthritis, despite not being recommended by the OARSI experts, with a trend to a continuing increase.13,17 We observed a low treatment adherence and early medication abandonment of opioids, causing considerable use of health and non-health resources and high costs for the NHS and society, despite that in terms of clinical effectiveness, opioid use was related to a modest reduction in pain and an increase in cognitive impairment and the degree of dependence for BADL in people aged ⩾65 years, in a relatively short period of time receiving the treatment. The lack of real-life observational studies makes it difficult to compare the results but supports the need for this study. In addition, the large number of patients included, together with the long follow-up period, may support the degree of external validity of the results in Spain and can be considered a strength of the study. The prevalence of chronic pain osteoarthritis found was 9.7%, consistent with the recent findings of the EPISER epidemiological study carried out by the Spanish Society of Rheumatology. 8 The literature reviewed showed that osteoarthritis is a highly prevalent chronic disease from 50 years of age onwards, with important socioeconomic implications, as it results in disability (especially due to knee and hip involvement), with a high economic cost related to medical and surgical treatment in advanced stages.2,4,6 The use of long-term opioids in these patients is controversial and there is little scientific evidence.11,16 The study highlights an increase of its utilization over time, in line with current trends worldwide.16,17,30–33 We cannot say whether physicians made risk–benefit assessments before starting treatment, but opioid use was high and is increasing. Ackerman quantified the current use of opioids for osteoarthritis pain in Australia and concluded it will continue to increase substantially, 34 and that their projections were a conservative estimate of the total financial burden, given the costs associated with the adverse effects of opioids, especially in older people. Therefore, the implications of our results for health decision makers and society in general are worth noting, because an increase in the analgesic cost per patient contributes to increasing healthcare costs. In this study, most of the increment in healthcare cost was supported by the cost of opioid therapy, and although this increase showed an small effect size, due to the high prevalence of osteoarthritis, its extrapolation to the whole nation generates an important economic effect on the NHS: approximately the expenditure in opioid drugs would account for the 3.1% of the NHS pharmaceutical budget in 2018. This is of importance, considering that the improvement in health outcomes was modest or even poor. Except for methodological differences, our results are in line with the literature reviewed.16,30
At 36 months of follow-up, adherence rate to opioid treatment was 19.1% (first year: 36.7%). Kostev et al. evaluated the persistence of opioids in a large cohort of patients (n = 32,158) who were treated for chronic pain. 35 As of the following year, 69.0% had discontinued medication. The authors concluded that adherence to opioids was associated with chronic comorbidity and depression, whereas younger subjects with chronic noncancerous pain (back osteoarthritis) increased the likelihood of opioid discontinuation. The study of Shcherbakova et al., 36 in a retrospective cohort of 302 patients who initiated treatment with buprenorphine/naloxone for chronic pain, describes an adherence of 40.4%, per year of follow-up. The authors of the study emphasize that: (a) healthcare providers should educate patients about the risks of opioid use and establish alternative methods of pain management, before prescribing an opioid, and (b) there appears to be an inverse association between the adherence to treatment and hospital admissions. Although neither study was conducted specifically in patients with osteoarthrosis, our results of adherence to treatment (the year of follow-up, and saving methodological comparisons/limitations), appear to be in line with these reported data (adherence of 31.0% and 40.4%, respectively).
The economic impact of osteoarthritis was high. Overall, 27.7% of costs were health costs and 72.3% non-health costs (lost productivity). In addition, 36.8% of the cost in patients requiring hospital admission corresponded to osteoarthritis-related surgical interventions (5.1% of patients abandoned the study due to any surgical or invasive procedure related with osteoarthritis requiring hospitalization), which was a considerable source of increasing costs, mainly during the last two years of follow-up. The percentage of surgical procedures reported here is aligned with that showed by Postler et al., 37 who found 5.3% of patients receiving a total joint replacement in 1 year in Germany in subjects with hip or knee osteoarthritis above 60 years old, if we consider that our patients were younger. In Postler et al.’s study, 37 63.4% of patients were receiving analgesic drugs including opioids prior to surgery. In addition, Inacio and coworkers 38 reported a considerable percentage of patients using opioids (54% and 44%, respectively) in the year prior to a hip or knee joint replacement. The economic burden of osteoarthritis has been shown in numerous studies.2,8,10,16,30 A review by Xie et al. found that direct costs (mean/year) in the US ranged from $1442 to $21,335 and indirect costs ranged from $238 to $29,935 and concluded that the costs of osteoarthritis are considerable, and the patient’s quality of life remains poor. 39 In addition, they showed that standardization of the definition of costs and measurement of lost productivity would facilitate the comparison of results. Salmon et al. also highlighted the heterogeneity between studies and the lack of methodological consensus on how to obtain comparable estimates of the disease cost and highlighted the high health costs (€500–10,900) and non-health costs (€200–12,300) of osteoarthritis. 40 Zhao et al. pointed out that patients on opioids had greater resource use and total costs (healthcare: $13,595; non-healthcare: $2331). 41 These results suggest the need to evaluate alternative strategies for pain management. Our study provides similar results to those described previously, although non-health costs may be are proportionately higher, as patients on opioids may have greater disease severity.
In terms of clinical effectiveness, there was a slight reduction in the proportion of patients with moderate to severe pain (6.4%), while the percentage of patients with cognitive impairment (3.3%) and moderate to total dependence for BADL increased (15.6% in total and 16.5% in patients with hip/knee osteoarthritis). NSAIDs and opioids may offer discrete reductions in pain and improve functionality in osteoarthritis patients, but potential harm must be considered, and therefore they are not recommended or recommended with limitations by OARSI. 13 Smith et al. found that NSAIDs and opioids offer similar pain relief in osteoarthritis patients, 42 which could encourage physicians and patients to evaluate the possible benefits of alternative painkillers. Wei provided evidence of the economic impact of opioid use and the poor improvements in pain intensity. 43 Fuggle et al. concluded there were considerable safety and tolerability problems in opioid use for the treatment of osteoarthritis and defended international recommendations for short periods of opioid use after another analgesic treatment. 44 Some authors have found that opioid treatment for osteoarthritis patients produces an acceptable degree of analgesia without affecting cognitive functioning or BADL, although for short periods. 45 Our study seems not to be consistent with these contributions. Opioid use (and increased costs) did not correlate with an improvement in health, there was a high short- to medium-term dropout rate with increased cognitive deterioration and physical dependence for BADL, which may assume greater importance in this especially vulnerable population in accordance with OARSI recommendation and other authors;13,15,46 67% of patients who start opioid treatment are aged ⩾65 years and 43% ⩾75 years, and this may have a major impact on the NHS.
The possible limitations of the study are those inherent to retrospective studies, such as disease under recording or possible professional and patient variability owing to the observational design, the measurements used for the main variables, and possible classification bias. Likewise, possible inaccuracies in the diagnostic coding of osteoarthritis or the lack of variables that could have influenced the results (socioeconomic level, evolution of the prescribed pharmacological dose, etc.), may also be considered as limitations. Patients with missing/inconsistent data were excluded in the analysis, so they could cause potential bias in the study; however, because they were a low number of subjects, we do not believe it could interfere with the results of the study. Other possible limitations include the causes of opioid withdrawal (adverse reactions, etc.) could not be quantified, and the possible combinations of analgesic medication with non-pharmacological therapies were not considered. However, in our opinion, the main limitation was selection bias by attending physicians when initiating opioid treatments, as this was not randomized, as is usual in real-life situations. Further studies are required in usual clinical practice to reinforce our results. That is, opioid treatment can increase health and non-health costs (especially analgesia costs), without significantly improving pain control and worsening cognitive functioning and the degree of dependence. It may be necessary to restrict opioid use to highly selected patients and assess whether they are used in accordance with available recommendations. 13 New therapeutic approaches to the treatment of chronic pain in osteoarthritis patients are also necessary, given its high prevalence, especially in older patients.
In conclusion, opioid use for the treatment of pain in osteoarthritis patients was high, with a tendency to increase. These patients had low treatment adherence, and high resource use and health and non-health costs for the Spanish NHS and society. In addition, in terms of clinical effectiveness, opioid use was related to a modest reduction in pain and an increase in cognitive impairment and the degree of dependence for basic activities of daily living observed in a relative short duration of treatment.
Supplemental Material
Opioids_OAC_supplementary_materials_v2 – Supplemental material for Health outcomes and costs in patients with osteoarthritis and chronic pain treated with opioids in Spain: the OPIOIDS real-world study
Supplemental material, Opioids_OAC_supplementary_materials_v2 for Health outcomes and costs in patients with osteoarthritis and chronic pain treated with opioids in Spain: the OPIOIDS real-world study by Antoni Sicras-Mainar, Carlos Tornero-Tornero, Francisco Vargas-Negrín, Isabel Lizarraga and Javier Rejas-Gutierrez in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors state that the manuscript is based in an investigation classified by the Spanish Agency for Medicines and Medical Devices (AEMPS) as a Post-Authorization Study - Other Design (EPA-OD), and subsequently was approved by the Institutional Research Board of the Hospital de Terrassa in Barcelona (Code: PFI-OP-2018-01) on 11 March 2019). Patient consent was not obtained as Spanish legislation excludes existing data that are aggregated for analysis and personal data are stripped of identifying details as specified in Spanish Law 15/1999, of 13 December, on Personal Data Protection.
Author contributions
The conception of the study was made by ASM, JRG, and IL. ASM, CTT, FVN, JRG, and IL participated and contributed to the design of the study. Data collection and statistical analysis was performed by ASM. Interpretation of data was made by all authors. All authors drafted or revised critically and approved the final version of submitted manuscript.
Availability of data and materials
All data generated or analyzed during this investigation are included in this published article. Any additional information is available from the corresponding author on reasonable request.
Conflict of interest statement
Antoni Sicras-Mainar is an employee of Real-Life Data who were paid consultants to Pfizer in connection with the development of this manuscript. Javier Rejas-Gutiérrez and Isabel Lizarraga are employees of Pfizer, SLU. Carlos Tornero-Tornero and Francisco Vargas-Negrín declare that they have no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The OPIOIDS study was funded by Pfizer, SLU. JRG and IL are full employees of Pfizer, SLU and participated in the conception and design of the study, interpretation of data and revising critically the final version of manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
