Abstract
Glucocorticoid-induced osteoporosis (GIOP) is the most common form of secondary osteoporosis. To date, six large randomized controlled clinical trials on the efficacy of pharmaceutical treatment in GIOP have been conducted. All of these studies have focused predominately on bone mineral density outcomes, and none of them have been statistically powered to address fracture endpoints. The purpose of this review is to highlight differences in the design and results within these large randomized GIOP clinical trials, and how these differences might affect clinical decisions. Differences between studies in trial design, populations studied, and variable efficacy impact the comparability and generalizability of these findings, and ultimately should affect practitioners’ behavior. We review the clinical trials that provide the best quality evidence on comparative efficacy and safety of GIOP treatments. We also propose suggestions on the design of future GIOP clinical trials with attention to improved generalizability, and, ideally, study designs that might achieve fracture outcomes.
Introduction
Glucocorticoids are widely prescribed for the treatment of inflammatory, autoimmune, and respiratory diseases. 1 Osteoporosis is a common, at least partially preventable, adverse event related to glucocorticoid treatment. Glucocorticoid-induced osteoporosis (GIOP) is the most common form of secondary osteoporosis. Pathophysiologic mechanisms leading to GIOP work in concert to increase bone fragility via reduced bone formation and increased bone resorption. Increased bone resorption is seen mainly during the early phases of glucocorticoid treatment, while the effect on bone formation is prominent after several months of treatment. GIOP risk increases in both a dose- and time-dependent manner;2,3 however, the use of glucocorticoids is associated with an increased risk of fracture within the first 6 months of treatment. 4 To date, six large randomized clinical trials have been conducted,5–10 and seven therapeutic agents have been approved for the treatment of GIOP in the United States, all with demonstrated efficacy in improving bone mineral density (BMD).5–26 However, these clinical trials have been conducted with different study designs, leading to a limited comparability between studies testing different therapeutic options. Most of these studies also lack some generalizability to glucocorticoid users overall. We reviewed pivotal, multinational, randomized controlled trials on pharmacologic treatment in GIOP that have enrolled more than 200 patients each. Such large randomized trials, together with meta-analyses of randomized controlled trials, are considered the highest level of evidence, which, in the process of formulating guidelines, leads to the strongest level of recommendation.27,28 The aim of our review was to provide clinicians with a critical comparative interpretation of the results of GIOP clinical trials, and to transfer information from these clinical trials into clinical practice.
Overview of clinical trials in GIOP
Designs of large trials evaluating therapeutic options in GIOP have been diverse. Only two therapeutic drugs, alendronate and risedronate, were tested against placebo, while teriparatide, zoledronic acid, and denosumab were each tested against an active comparator.5–10 Teriparatide was compared with alendronate, while zoledronic acid and denosumab were compared against risedronate.8–10 Clinical trials in GIOP differed in many important baseline characteristics, such as participant’s age, sex, ethnicity, menopausal status, underlying conditions, comorbidities, cotherapies, and bone health at baseline (see Table 1). The first large randomized clinical trial in GIOP was published more than 20 years ago, 5 and, since that time, guidelines, preventive strategies, and cotherapies for the underlying conditions have changed substantially, resulting in different patient population makeup participating in such trials. Moreover, since the first study was conducted in this area, many more therapeutic options are now available to patients, somewhat limiting patients’ interest in clinical trials of newer drugs. In contrast, glucocorticoid use patterns have not changed all that much in the last decade, and, if anything, glucocorticoid use may be increasing. 29 Etidronate was not included in the present review since it is not approved for the treatment of GIOP in the US, and its use has been surpassed in many regions by newer bisphosphonates. Nevertheless, etidronate has been studied in GIOP and is used in GIOP in Europe. Data supports its efficacy in preventing and treating GIOP. 30
Characteristics of the treatment arm of GIOP clinical trials.

COPD, chronic obstructive pulmonary disease; GCA, giant cell arteritis; GIOP, glucocorticoid-induced osteoporosis; IBD, inflammatory bowel diseases; N/A, not applicable; N/S, not specified; IU, international units; PMR, polymyalgia rheumatica; SLE, systemic lupus erythematosus.
Prevention design refers to participants on glucocorticoids for up to 3 months; treatment designation refers to participants on glucocorticoids for ⩾3 months. In square brackets expected characteristics for the glucocorticoid users in the general population (epidemiological data by Overman and colleagues 1 and Walsh and colleagues 31 ).
Designation of GIOP clinical trials study type
One of the most noteworthy differences between and within clinical trials is the ‘treatment’ or ‘prevention’ designation. The terms ‘treat GIOP’ or ‘prevent GIOP’ refers to whether the study subjects are prevalent glucocorticoid users (typically defined as ⩾3 months of treatment) or incident users, having very recently started glucocorticoids (<3 months of treatment), respectively. Among the six clinical trials published, one had an exclusive prevention designation (risedronate), 6 two included only prevalent glucocorticoid users (risedronate and teriparatide), 7 and the remaining three included both prevalent and incident glucocorticoid users (alendronate, zoledronic acid and denosumab).5,8–10 Another key difference between studies, mentioned before, resides in the ‘placebo’ or ‘comparator’ designation of the study. All studies had been conducted in multiple countries, including the US. These multisite designs led to greater population genetic diversity. For example, there is a variability in susceptibility to glucocorticoids adverse events in patients with polymorphisms of 11β-hydroxysteroid dehydrogenase. 32 Nevertheless, when results were reported for each study site, there was no significant difference in BMD changes across geographic regions.5,8
Differences between clinical trials in GIOP
Gender, age, menopausal status, and ethnicity
Gender, age, menopausal status, and ethnicity may importantly affect bone health. For example, older white females have a greater risk of fracture compared with younger black males. 33 Moreover, bone turnover depends strictly on menopausal status. 34 Large epidemiological data showed 0.5–1% of the general population are on long-term glucocorticoids, with a nearly equal representation of women and men. 1 However, the proportion of men enrolled in GIOP clinical trials was considerably lower compared with the reported proportion receiving treatment with glucocorticoids in the general population, ranging from 19.6% to 38% of the overall population of glucocorticoid users. Although the mean age of the patients enrolled in the clinical trials (means range from 53 ± 14 to 66 ± 10 years) was comparable to the mean age of chronic glucocorticoid users in the general population (56 ± 4 years), 1 the proportion of premenopausal women enrolled in GIOP clinical trials varied considerably, from 0.4% to 22%. In the general population, the proportion of women younger than 50 years taking glucocorticoids is approximately 2%. 1 Ethnicity of subjects enrolled was reported in three GIOP trials (teriparatide, denosumab, and alendronate).5,8,10 White patients largely dominated these studies, representing 72% of subjects in the teriparatide trial, and 90% in the denosumab treatment trial. These proportions are comparable to the estimated proportion of white persons among glucocorticoid users in at least one of the general populations in which this has been reported (approximately 80%). 35 Nevertheless, some glucocorticoid requiring diseases affect predominantly black patients (e.g. systemic lupus erythematosus), limiting the validity of the results in such populations. In summary, there were important demographic differences between the glucocorticoid users in the general population and patients enrolled in the GIOP clinical trials that might raise concerns about the generalizability of trial results.
Prednisone equivalent dose
Doses lower than 5 mg/day (prednisone equivalent) are generally considered safer for bone health and much less likely to raise major concerns about GIOP prevention. A dose of 7.5 mg per day is normally considered the ‘physiologic threshold’ based on doses above this level leading to far greater effects on the hypothalamic-pituitary-adrenal axis. For this reason, most GIOP clinical trials enrolled patients treated with ⩾ 7.5 mg/day of prednisone equivalent. Nevertheless, mean and median glucocorticoid doses were substantially different between studies, ranging from a median dose of 7.5 mg/ day in the teriparatide clinical trial, 8 to a mean dose of 21 mg/day in the risedronate prevention clinical trial, 6 affecting comparisons between study populations.
Baseline bone health
Inclusion criteria in GIOP clinical trials required a BMD threshold or a history of past fragility fractures in the teriparatide and denosumab trials only.8,10 In both these trials, patients with a T-score, at any site, of ⩽ –2.0 or ⩽ –1.0 with a prevalent fragility fracture were considered eligible. These inclusion thresholds were largely based on prior considerations of these drugs for primarily higher risk patients, based on guidance from regulatory agencies at the time of GIOP study design. The mean baseline T-score measured at the lumbar spine ranged from ‒2.5 in the teriparatide trial, 8 to ‒0.7 in the risedronate prevention trial. 6 In addition, the proportion of patients with a prevalent vertebral fracture in all GIOP trials ranged from 14% (denosumab prevention trial) to 37% (risedronate treatment trial).
Calcium and vitamin D supplementation
Adequate calcium and vitamin D supplementation to a pharmaceutical therapy is essential for the prevention and treatment of bone loss in GIOP.36,37 Patients enrolled in most of the major randomized controlled trials for the treatment of GIOP received vitamin D, ranging from 0 (risedronate prevention trial) to 1200 IU/day (zoledronic acid trial) or calcium [ranging from 500 (risedronate prevention trial) to 1000 mg/day (all other five GIOP trials)]. In 2017, the American College of Rheumatology (ACR) recommended optimizing calcium (1000–1200 mg/day) and vitamin D (600–800 IU/day) intake over no treatment, or over any other pharmaceutical treatment alone. 38 However, this was a conditional recommendation based on low-quality evidence related to the impact of calcium and vitamin D on fractures, at least in monotherapy.39–43
Underlying conditions, comorbidities and cotherapies
Rheumatoid arthritis was the most commonly represented underlying disease in GIOP clinical trials, followed by other rheumatologic conditions, including polymyalgia rheumatica, giant cells arteritis, and lupus. Other, less represented, conditions included pulmonary diseases such as asthma and chronic obstructive pulmonary disease (COPD).5–7,9,10,20 However, although the proportion of the represented diseases were largely similar across studies, the disease distribution differed somewhat from the general GIOP population. After rheumatoid arthritis, emphysema, COPD, and asthma constitute the diseases more frequently associated with chronic glucocorticoid use.1,44 These differences likely represent exclusion criteria, the types of physicians involved in these studies as investigators, and less interest in clinical trials among patients with more severe or active disease. Notably, patients with inflammatory bowel diseases requiring glucocorticoids were not adequately represented in GIOP trials.
Outcomes and results
The primary outcome in all GIOP clinical trials was the change from baseline in vertebral BMD compared with placebo or the active comparator (see Table 2). All six randomized controlled trails proved the superiority or non-inferiority of the medication tested in increasing BMD compared with placebo or active-comparator. Secondary outcomes also included changes in hip BMD and the incidence of vertebral and nonvertebral fractures. The overall number of fractures in reviewed studies, which included younger adults, men and those with well-preserved baseline BMD, was small and the follow-up short. For these reasons, all GIOP clinical trials were underpowered to detect a reduction in fragility fracture. Only one study, the comparison of teriparatide to alendronate found a significant reduction in a small number of reported vertebral fractures as part of the secondary analysis. 8
Outcomes of GIOP clinical trials.
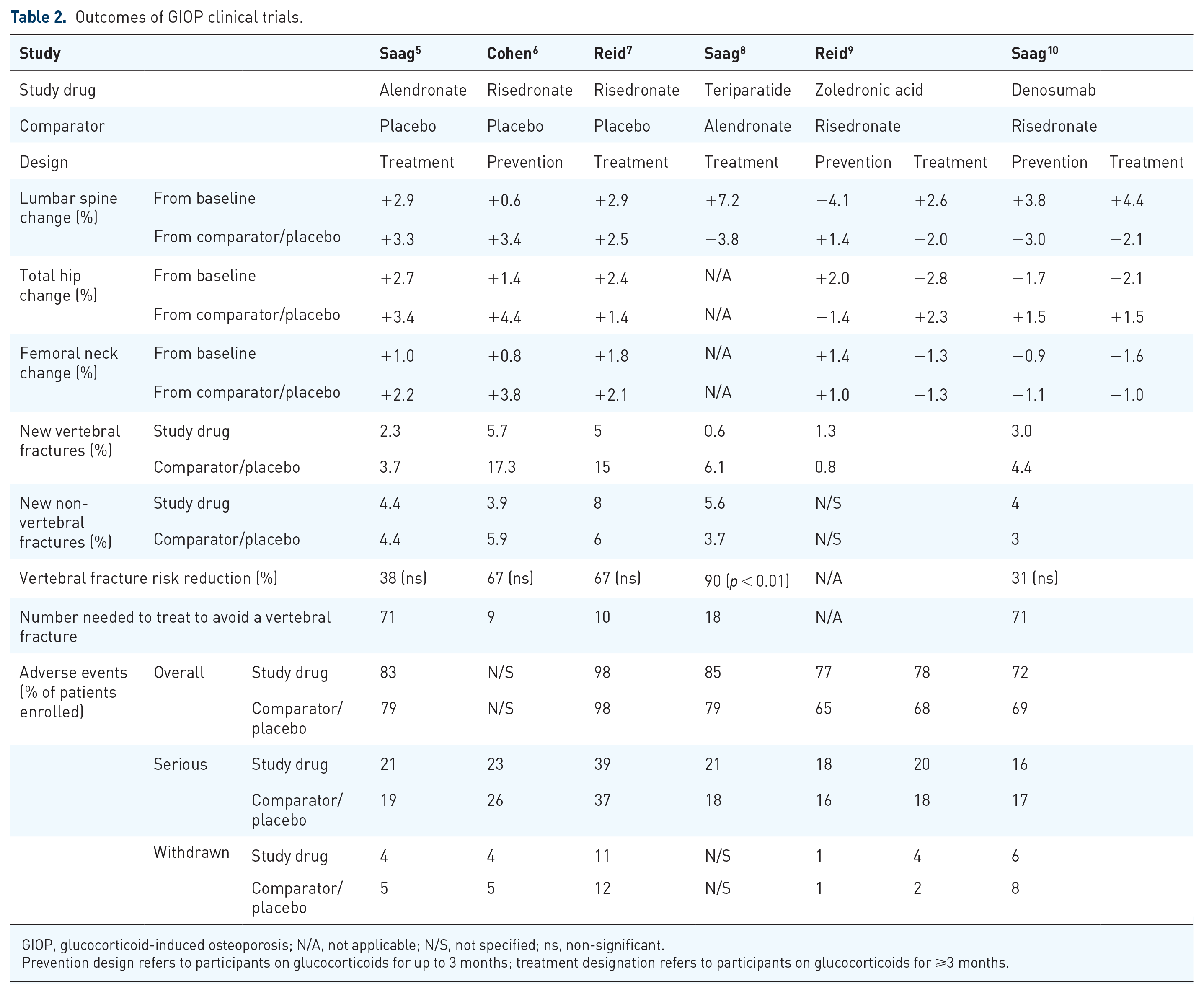
GIOP, glucocorticoid-induced osteoporosis; N/A, not applicable; N/S, not specified; ns, non-significant.
Prevention design refers to participants on glucocorticoids for up to 3 months; treatment designation refers to participants on glucocorticoids for ⩾3 months.
Safety
The safety of the medications, compared with placebo or the active comparator, was overall reassuring. However, adverse events and serious adverse events were reported in approximately 80–20% of patients, respectively, confirming that GIOP patients are a sicker patient population overall. Moreover, trials on GIOP were short; for this reason, conclusion on long-term safety of these medications cannot be drawn. However, extended clinical trials in postmenopausal osteoporosis showed a favorable risk/benefit profile of anti-osteoporotic medications.45,46 Unfortunately, long-term data on the safety of anti-osteoporotic drugs in GIOP are lacking and should be the topic of future, larger, more pragmatic designs.
Future perspectives on clinical trials in GIOP
When considering the design of future randomized clinical trials on GIOP, researchers need to consider several issues. First, is a consideration of inclusion and exclusion criteria. We recommend that the prednisone equivalent dose criteria be set at ⩾7.5 mg/day, and standardized definitions of prevention versus treatment designation be used consistently in trial designation (⩾3 months is the standard cut-point to define the treatment population). In addition, a BMD or prevalent fracture inclusion criteria (i.e. T-score, at any site, of ⩽–2.0 or ⩽–1.0 with a prevalent fragility fracture) should be considered for participants treated for GIOP, and, conversely, should be avoided for those enrolled in the prevention arm of such a study. Pregnant women and patients at risk for short-term adverse outcomes (e.g. patients with severe systemic conditions requiring glucocorticoids or transplant patients) should be excluded from future clinical trials, for safety considerations. Beyond these key exclusion criteria, we value a less exclusive group of patients, ideally including many patients seen in primary care rather than specialty settings.
Another important issue to consider is whether the use of a placebo is currently ethical or unethical in GIOP, a condition where effective therapies exist. In 2013, the Declaration of Helsinki was revised, and the paragraph regarding the placebo use in clinical trial was left open to various interpretation. Indeed, drug regulatory authorities allow the use of placebo only in some instances. 47 A 2002 FDA advisory committee report noted that randomized, placebo-controlled clinical trials are still appropriate in osteoporosis under certain circumstances 48 : for women with osteoporosis who cannot tolerate existing treatment, when all trial participants get calcium and vitamin D supplements, and as discontinuation is always an option for participants with new fractures or significant bone loss that occur post-trial initiation. This recommendation was specific to postmenopausal osteoporosis, but similar considerations might be applied to GIOP as well. The FDA also issued guidance for when randomized active-controlled clinical trials might be indicated. 49 They recommended that active-controlled trials should be designed for non-inferiority when comparing medications in the same class (e.g. anti-resorptives), while superiority designation should be reserved for comparison between bone anabolic and antiresorptive treatments. Regardless of whether a placebo control is ethical or not, it may be impractical to recruit patients to a placebo-controlled osteoporosis trial in 2019.
The six large historical GIOP trials were all designed based on a BMD endpoint. Although a large part of the reduction in the fracture risk with the use of anti-osteoporotic treatment in postmenopausal osteoporosis is attributable to the BMD increase, 50 BMD alone is not the optimal endpoint for comparative efficacy claims between two approved drugs in GIOP, where BMD values do not explain the full pathophysiology of the disease process. A major difficulty for future GIOP trials is that fracture outcome requires much larger number of patients than studies to date have included to demonstrate a change in such an outcome. This has clear cost considerations for future studies if they maintain the traditional study designs employed. For example, since the annual incidence of fragility fracture in the risedronate control arm of denosumab GIOP trials was 3.2%, and, assuming that denosumab might reduce the annual incidence of fragility fracture to 2.7%, one would need to randomize more than 15,000 individuals in a non-inferiority study of these two drugs to establish non-inferiority with a non-inferiority margin of 1%. Newer studies designs may offer promise in circumventing this dilemma, as we discuss below.
Another relevant consideration for randomized controlled trials in GIOP is the statistical approach to outcomes. Intention-to-treat (ITT) analysis provides an estimate of the treatment differences that is most familiar, and represents the standard approach in superiority studies, but, due to crossovers and noncompliance in non-inferiority active comparator trials, may bias clinical researchers towards falsely rejecting the null hypothesis of inferiority. To avoid this source of bias, per-protocol (PP) analysis should be reported together with ITT analysis in non-inferiority studies. Another possible solution for this statistical problem in such studies could be the approach of a hybrid ITT/PP analysis that may be able to contend more effectively with issue of missing data in non-inferiority trial designs. 51 This hybrid approach excludes noncompliant patients similar to the PP analysis, and properly addresses impact of nontrivial missing data in congruous fashion to a maximum likelihood estimation-based ITT analysis.
Although properly designed randomized clinical trials represent the highest level of evidence, historically, typical explanatory randomized clinical trials in GIOP, while necessary for drug approvals, fail to address certain limitations (e.g. large number of patients requiring rigorous and costly clinical follow up) and lack generalizability. Epidemiological studies are an important source of data on effectiveness of medications, 52 albeit potentially suffering from confounding by indication a type of channeling bias due to the lack of randomization. They do, however, provide a more real world look at drug use in routine clinical practice. For example, bone-forming agents are commonly used in patients with more severe osteoporosis, making the comparison with antiresorptives not feasible. A possible alternative to explanatory randomized clinical trials are pragmatic clinical trials.53,54 Pragmatic clinical trials (PCTs) in GIOP would be executed in more general clinical practice settings (and less so in research clinics), where individuals receive their real world care. Eligibility and recruitment criteria of PCTs are simpler; the intervention implementation and participants’ adherence to protocol are less rigid. 55 Moreover, pragmatic clinical trials employ natural endpoints (e.g. fractures) in preference to BMD, which necessitates measurement in clinics. Fracture endpoints might better inform physicians’ decisions and policymakers. The intention of pragmatic clinical trials is to measure real-world effectiveness (generalizability); conversely, the intent of explanatory randomized clinical trials is to measure efficacy, usually for regulatory approval. For example, in GIOP, the exclusion of patients with significant comorbidities reduces the generalizability of obtained results. Based on their simpler study design, pragmatic clinical trials can include larger sample sizes at a lower cost. Employing other innovations such as data linkages (for example connecting study patients passively to their administrative claims and billing data) allows capture of more important fragility fractures, especially hip fractures (of greatest clinical relevance) during the study.
In summary, to date, randomized clinical trials have provided high quality evidence that support the efficacy of several anti-osteoporotic medications in GIOP. Although these studies have been rigorously conducted and answer many key comparative efficacy and safety questions, they have limitations in their generalizability and have not addressed fracture endpoints adequately. Future innovations in GIOP studies such as pragmatic clinical trials can better address true drug effectiveness and provide imperative fracture outcomes allowing more rigorous comparison of these efficacious medications for the treatment of GIOP.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and publication of this article.
Conflict of interest statement
This review does not contain any original data collection or intervention with human participants or animals performed by any of the authors. For this type of review article a formal consent and ethical board approval is not required.
Giovanni Adami and Elizabeth J Rahn declare that they have no conflict of interest. Kenneth G Saag declares research grant from Amgen and Merck and consultant fee from Amgen, Lilly, Merck and Radius and Roche.
