Abstract
Background:
New-onset sarcoidosis has been previously described in three case reports in patients affected by rheumatoid arthritis treated with tocilizumab (TCZ). The existence of a cause–effect mechanism between the biological treatment and the onset of the illness is still being debated.
Patient concerns:
A 74-year-old woman was diagnosed with giant cell arteritis (GCA). The first-line treatment with glucocorticoids; and the second-line with methotrexate and low-dose glucocorticoids were stopped due to multiple pathological vertebral fractures and insufficient biological and clinical response. The cytotoxic agent, cyclophosphamide, was then introduced and in turn stopped, because of gastrointestinal side effects. Thereafter a treatment with TCZ was begun. The patient experienced good clinical response; however, 8 months later she developed painful hyper-pigmented reddish cutaneous micronodular lesions localized to the abdomen and thorax. A cutaneous biopsy was performed, and histological analysis showed noncaseating epithelioid granulomas in the hypodermis. The diagnosis of cutaneous sarcoidosis was made.
Interventions:
Topical corticosteroids were administered and, as requested by the patient, TCZ was discontinued with slow but complete resolution of the skin lesions. After TCZ discontinuation however, the GCA flared and the patient’s symptoms and biological abnormalities reappeared. Thus, after a 6-month suspension, TCZ was re-administered. At 2 months later the skin lesions compatible with cutaneous sarcoidosis reappeared. Topical corticosteroids were once again prescribed and as suggested by the patient the TCZ posology was reduced. The patient’s symptoms disappeared, and the cutaneous lesions resolved.
Lessons:
The time elapsed from TCZ treatment start and the onset of cutaneous sarcoidosis, as well as its recurrence after TCZ suspension and rechallenge supported the diagnosis of a drug-induced reaction. To the best of our knowledge, this case report represents the first instance of cutaneous sarcoidosis most likely induced by TCZ in patients affected by GCA. In addition, our case emphasizes that although TCZ in monotherapy confirms to be an effective treatment for GCA, further immunological disorders could be unmasked, and the discussed side effect of the drug could be dose-dependent.
Background
Tocilizumab: clinical implications
Interleukin-6 (IL-6) represents a pivotal biological proinflammatory cytokine with a wide range of properties including regulation of immune response and inflammation, oncogenicity and a variety of molecular and biological effects on B-cells, T-cells, blood vessels, bone cells, cardiac muscle cells and hepatocytes. 1 Furthermore, to date, IL-6 is considered as a fundamental cytokine in the pathogenesis of several autoimmune diseases. 1
Tocilizumab (TCZ), also known as myeloma receptor antibody or Actemra®, is a humanized monoclonal antibody which competes with IL-6 in binding to the IL-6 receptor subunit alpha (IL-6Rα), in two existing forms, transmembrane and soluble, preventing the transduction of the signal via the formation of the complex IL-6R/glycoprotein 130. 2 The inhibition of the intracellular cascade, contemplating in particular the Janus kinase 1 (JAK1) and the signal transducer and activator of transcription 3 (STAT3), results in a decrease in neutrophil count, myeloid dendritic cells, monocytes, lymphocyte T-helpers 17 and in neutrophil joint infiltration. 3
TCZ belongs to the class of drugs called biological disease-modifying antirheumatic drugs (bDMARDs) and represents the first humanized anti-IL6R monoclonal antibody approved for the treatment of patients with rheumatoid arthritis (RA); in which it slows down the radiological progression of joint lesions and induces the remission of arthritis activity. In recent years, several clinical trials and data from real-world clinical practice have confirmed the efficacy of TCZ in the treatment of different rheumatological diseases. Nowadays TCZ is approved by the United States Food and Drug Administration (US FDA) and by the European Medicines Agency as a treatment for: Castleman’s disease, polyarticular and systemic juvenile idiopathic arthritis, and GCA as well as for the treatment of severe chimeric antigen receptor T-cell-induced cytokine release syndrome. Its use has been recommended by the European League Against Rheumatism and the American College of Rheumatology as a first-line biological agent for treating active RA after unsuccessful treatment with conventional synthetic disease-modifying antirheumatic drugs (csDMARDs). 4
Many concerns have been raised in the last years about the long-term safety of TCZ treatment, for many side effects reported in the literature. Common adverse events, which deserve close laboratory controls at the beginning of the therapy, include: abnormalities of liver function (with in particular increase in alanine and aspartate aminotransferases), cytopenia (neutropenia and thrombocytopenia) and rise in low-density lipoprotein levels. Other significant adverse events include in particular: infections which, although rare, can be severe (i.e. pneumonia, cellulitis, herpes zoster, tuberculosis), gut perforation, anaphylaxis and increased blood pressure.
Sarcoidosis induced by Tocilizumab: previous reports
New-onset sarcoidosis during TCZ treatment was until now described in only three case reports (Table 1); all of them referring to patients with RA.5–7 In the three previous reports, what was supposed to be a TCZ-induced sarcoidosis, presented during treatment for RA, in females and at least 1 year after the beginning of the treatment. In two cases5,6 there was involvement of both skin and lung (bilateral hilar lymphadenopathy and pulmonary infiltration) and in one case an isolated involvement of the skin. 7 In all cases diagnosis was performed on the basis of histological evidence of noncaseating epithelioid granulomas. The sarcoidotic manifestations resolved after discontinuation of TCZ and introduction of a treatment with glucocorticoids.
Reported cases of sarcoidosis onset during tocilizumab treatment.

F, female; GCA, giant cell arteritis; M, male; RA, rheumatoid arthritis; TCZ, tocilizumab.
We report here the first case of TCZ-induced cutaneous sarcoidosis in a patient treated for GCA.
The evidence of the potential benefit as well as the safety of TCZ as second-line therapy for GCA is widely known and TCZ was first approved for the treatment of GCA in May 2017. Furthermore, results of multicenter randomized, double-blind, placebo-controlled trials, have demonstrated that early initiation of TCZ in patients with GCA is associated with a reduction in the total amount of glucocorticoids administrated, in the incidence of flare and in the rate of long-term complete remission.8,9 Nevertheless despite the proven effectiveness of TCZ in GCA, the safety profile is limited by the maximum 1 year follow up published in clinical trials today. A longer follow up on the benefit–safety profile is therefore necessary.
Case presentation
A 74-year-old White woman was admitted to our internal medicine service in June 2016. Her past medical history included: hypertension, dyslipidaemia, obesity, an ischaemic stroke affecting the pons and chronic hepatitis C with a benign clinical course and without alterations of alanine and aspartate aminotransferases. The patient was investigated for sudden onset of intermittent, increasing in severity, bi-temporal headaches concomitant to a recurrent temporo-mandibular pain syndrome. Biopsies of the temporal arteries showed a granulomatous vasculitis with uniform full thickness inflammation and multinucleated giant cells. The diagnosis of GCA was made. The clinical presentation of the GCA was severe and, from the beginning, glucocorticoids were prescribed. With 1 mg/kg/day of prednisone a prompt clinical and biological response was obtained, allowing a progressive dose tapering. A bone densitometry gave normal results.
After 3 months of therapy however, she was readmitted to the hospital with severe acute back pain localized to the region of the thoracolumbar spine junction. A computed tomography (CT) scan of the lumbar spine was performed, revealing a T12 compression fracture. The patient was treated with a vertebral cementoplasty with a rapid improvement in the pain intensity. The diagnosis of secondary, steroid-related, osteoporosis was postulated. In investigating the reason for the unfavourable bone outcome, a misunderstanding leading to the nonintroduction of a protective bisphosphonate therapy was pointed out. Thereafter, methotrexate (MTX) was proposed as a glucocorticoid-sparing drug at the dosage of 10 mg/week, and a second-line treatment with MTX and low-dose glucocorticoids (prednisone 7.5 mg/day) was started. At the same time intravenous bisphosphonates were administered.
Nevertheless, in October 2016 the patient was readmitted to hospital for the recurrence of acute low-back pain and the CT of the lumbar spine found new compression fractures of L1 and L3. The patient was again treated with vertebral cementoplasties. Considering the several osteoporotic vertebral fractures associated with a limited clinical and laboratory response, it was decided to stop the association MTX/glucocorticoids and to start a third-line therapy with per-oral cyclophosphamide (CYC), tapering the posology to 50 mg every 4 days due to lymphocytopenia. After 2 months of therapy however, at the end of January 2017, CYC was interrupted for gastrointestinal intolerance (nausea, loss of appetite, stomach pain, diarrhea), persistent lymphocytopenia and subjective hair loss.
In February 2017, considering the persistence of clinical and biological signs of GCA activity, a monotherapy with TCZ 8 mg/kg intravenously every 4 weeks, was started. TCZ was offered as an ‘off-label’ option, since the medication was not registered for GCA in Switzerland, considering the strength and the amount of findings present in the literature at the time. 10 The medication was given after discussing the risk–benefit ratio with the patient and after approval by the review board of the private health care insurance in charge of the patient. The monotherapy with TCZ, determined a rapid clinical improvement and after 4 weeks, knowing that the normalization of both, C-reactive protein (CRP) and erythrocyte sedimentation rate could have only been a direct consequence of IL-6 inhibition, we were relieved by the absence of signs and symptoms of active GCA.
In September 2017, the patient was once again seen in our clinic, this time for the appearance of hyper-pigmented reddish skin nodules localized to the abdomen and thorax. A cutaneous biopsy was performed. A Quantiferon test was negative, as well as microbiological testing for bacteria and fungi. Serum angiotensin-converting enzyme and calcium levels were within the normal range. A thorax CT scan excluded the presence of pathological lymphadenopathy or pulmonary abnormalities. Histological analysis of the skin lesions showed noncaseating epithelioid granulomas in the hypodermis (Figure 1). The diagnosis of cutaneous sarcoidosis was made. The new onset of the skin lesions, the absence of other sarcoidotic manifestations and the typical histological and clinical presentation of GCA at the beginning, make the hypothesis that the sarcoidosis was initially misdiagnosed for a GCA or that the disease was already active before unlikely.
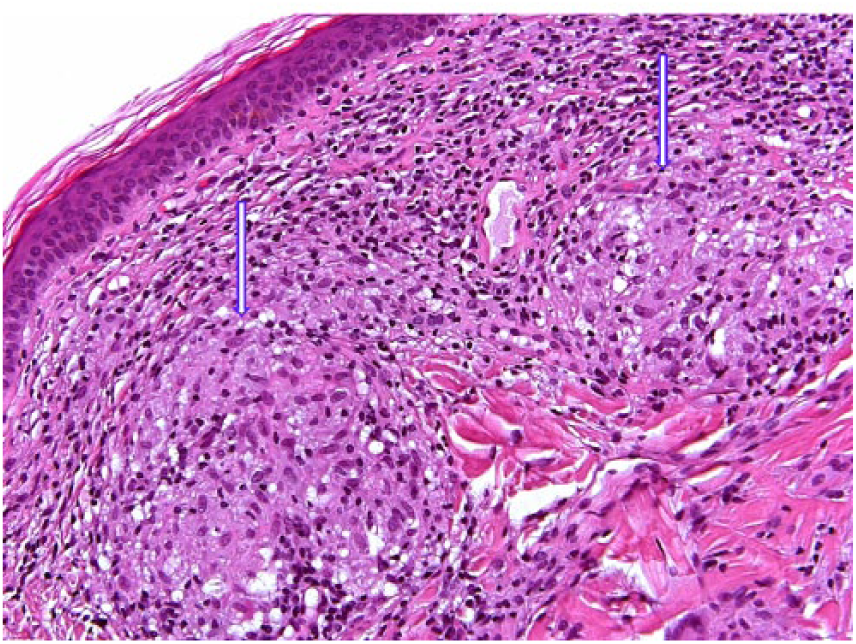
Photomicrograph of the cutaneous biopsy showing the pathological findings of noncaseating epithelioid granulomas (arrows) consisting of epithelioid cells with vacuolated cytoplasm and round or oval nuclei (hematoxylin-eosin stain, 40×).
To treat the skin lesions, a topical corticosteroid was administered and on patient request, TCZ was discontinued with progressive resolution of the cutaneous manifestations. In March 2018, however we were faced with a new clinical and biological flare of the GCA and after discussion of the pro and contra with the patient and considering the benign course of the cutaneous sarcoidosis, TCZ at the same dosage of 8 mg/kg every 4 weeks was re-administered. After two TCZ doses, the reddish skin nodules localized to the abdomen and thorax previously diagnosed as cutaneous sarcoidosis reappeared. Thanks to a reduction in TCZ posology to 4 mg/kg every 4 weeks and topical corticosteroids, the skin lesions disappeared without recurrence and the patient did not experience a new clinical flare of the GCA.
Discussion
GCA represents the most common vasculitis after the age of 50 years and is characterized, if untreated or active, by the risk of relevant clinical consequences, potentially life-threatening, that could, in particular, lead to vision loss, stroke and involvement of large vessels, with secondary development of aortic aneurysms, and more rarely, myocardial infarctions. 11 Glucocorticoids are usually efficient in inducing clinical and biohumoral remission, nevertheless they may lead to several long-term complications (e.g. skin atrophy, weight gain, diabetes and osteoporosis.). 12 During recent years an increasing number of clinical trials have been conducted to verify the efficacy of TCZ on GCA.
Thanks to their findings, and especially to the results of the GiACTA trial, in which a good profile in terms of glucocorticoid dose reduction, relapse-free remission and number of adverse events was shown, 9 in May 2017, TCZ was approved by the US FDA for treating GCA in adults. The ideal illness-specific posology, the optimal treatment duration and the TCZ long-term safety and efficacy remain however to be determined.
In wider terms, TCZ is recommended as a valid option with a tapering course of glucocorticoids or alone, in patients at risk or experiencing glucocorticoid toxicity or in the case of relapsing disease.
Sarcoidosis is a granulomatous multisystemic disease with a still debated etiology. Previous studies carried out on affected patients demonstrated elevated IL-6 levels in bronchoalveolar lavages and in induced sputum. These findings together with the experience obtained in other autoimmune disorders successfully treated with IL-6 inhibitors generated the rationale that IL-6 could also represent a potential therapeutic target in sarcoidosis. The hypothesis has however, until now, not been investigated in a consistent way.
Recently, a published case series, showed a good clinical response in sarcoidosis-associated uveitis after TCZ administration. 13
We thus face a paradox in which TCZ, on one hand has been used successfully to treat the peculiar clinical manifestation of sarcoidosis and on the other, has been considered the trigger for new onset of the disease.
To the best of our knowledge, this is the first report suggesting that TCZ could induce cutaneous sarcoidosis in patients affected by GCA. We hope that this case, adding some new elements to the complex picture depicting the relationship between IL-6 and sarcoidosis, will help in defining further aspects to be investigated. Unlike other case reports, in our case, some elements could reinforce the hypothesis of a causative relationship between TCZ treatment and sarcoidosis: (1) the temporal relationship with <1 year exposure, (2) the onset of sarcoidosis in the context of a long lasting clinical observation without previous cutaneous manifestations, (3) the recurrence of the cutaneous abnormalities after TCZ rechallenge and (4) the resolution after a 50% reduction in TCZ posology.
In addition, our case suggests that lower doses of TCZ could be enough, and better tolerated, to control the clinical manifestations of the disease. However, knowing that the normalization of the currently available biological markers of inflammation cannot be used to exclude, in asymptomatic patients, a residual GCA activity, 14 in cases of doubt a second agent that has been proven to be effective, such as MTX, could be added. Furthermore, facing the failure of all cited options, preliminary studies suggest the promising potential utility of other biological agents, such as abatacept and ustekinumab. 15
Although only a few clinical cases have been reported to date, we believe that, the possibility of new-onset sarcoidosis, including the isolated cutaneous form, should be considered in the watchful observation after the start of the biological drug TCZ.
Footnotes
Acknowledgements
Rosaria Del Giorno and Alfonso Iodice have joint first authorship. We would like to thank Franco Fulciniti, MD, who kindly performed the microscopic histopathological examination. This paper has been seen and approved by all authors; it is an original contribution not previously published and it is not under consideration for publication elsewhere.
Written informed consent was obtained from the patient for publication of the case details and accompanying images.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study is exempt from institutional review board approval by the Swiss Ethics Committee.
