Abstract
Anti-interleukin (IL) therapies have emerged as a major treatment for patients with moderate-to-severe psoriasis. This article reviews the up-to-date results of pivotal clinical trials targeting the interleukins used for the treatment of psoriasis, including IL-1, IL-2, IL-6, IL-8, IL-10, IL-12, IL-17, IL-20, IL-22, IL-23, IL-36 and bispecific biologics IL-17A/tumor necrosis factor alpha (TNF-α). Cytokines involved in the circuits of psoriasis inflammation without ongoing clinical trials are also mentioned (IL-9, IL-13, IL-15, IL-16, IL-18, IL-19, IL-21, IL-24, IL-27, IL-33, IL-35, IL-37, and IL-38).
Introduction
Psoriasis is a chronic immune-mediated inflammatory disease with far-reaching systemic implication beyond the skin, such as cardiovascular comorbidities. 1 It also affects quality of life profoundly 2 and imposes high economic burden. 3 Currently, interleukin (IL)-17/IL-23 pathways are identified as playing the key roles in the immunopathogenesis of psoriasis, 4 but numerous other cytokines are also involved in the pathway. Herein, we review the up-to-date results of pivotal clinical trials targeting the interleukins involved in the pathogenesis of plaque-type psoriasis.
Part I: Interleukin-12, 17, 23 (Tables 1-2)
Anti-interleukin agents with promising results for psoriasis treatment.

R, receptor; IL, interleukin; MACEs, major adverse cardiovascular events.
Structures and properties of anti-interleukin agents for psoriasis treatment.
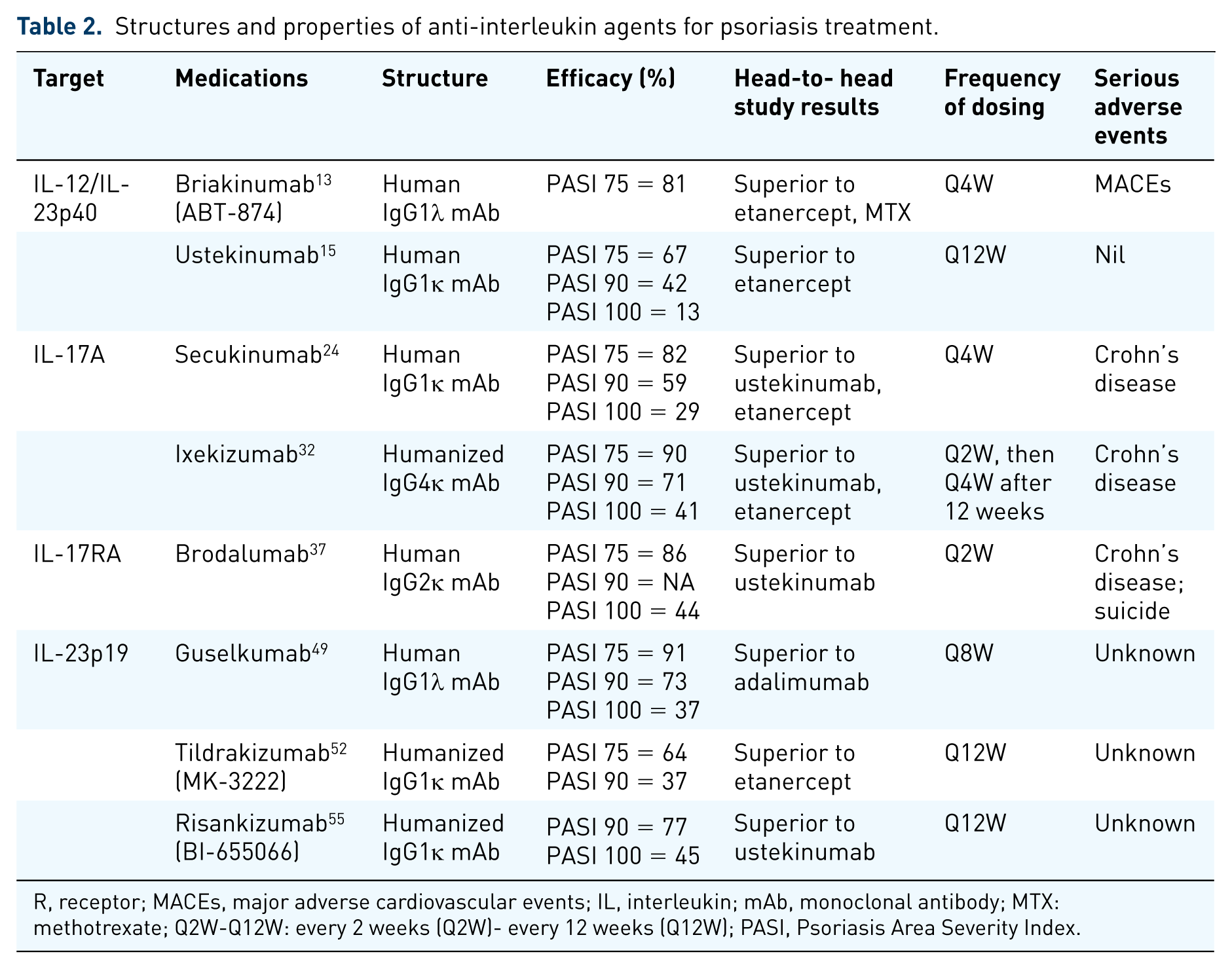
R, receptor; MACEs, major adverse cardiovascular events; IL, interleukin; mAb, monoclonal antibody; MTX: methotrexate; Q2W-Q12W: every 2 weeks (Q2W)- every 12 weeks (Q12W); PASI, Psoriasis Area Severity Index.
Interleukin-12/interleukin-23 p40 antagonists
IL-12 is a key cytokine for T-helper 1 cells’ polarization. In psoriasis, it is mainly produced by macrophages. 5 It is also produced by peripheral nerve cells, forming the basis of neurogenic inflammation in psoriasis.6,7 The initial success in using the anti-IL-12 approach to treat psoriasis was found to act through the anti-IL-23 pathway by binding to the shared p40 protein of IL-12 and IL-23. In experiments, targeting p35 of IL-12 did not show adequate treatment response. 8
Briakinumab (ABT-874)
Briakinumab targets the shared p40 subunit of IL-12 and IL-23. One phase II
9
and four phase III,10–13 randomized, placebo-controlled, double-blind trials verified its efficacy in moderate-to-severe psoriasis. Among the phase III trials, the Psoriasis Area Severity Index (PASI) 75 response at week 12 showed similar results in briakinumab-treated patients (80.6–81.9%), and was superior to etanercept (39.6–56.0%) and placebo (4.5–7.4%) (
Ustekinumab
Ustekinumab acts similarly to briakinumab. On 25 September 2009, the US Food and Drug Administration (FDA) approved ustekinumab to treat moderate-to-severe psoriasis. It is given subcutaneously at weeks 0 and 4, and then every 12 weeks and the dose is adjusted by body weight. For body weight equal or less than 100 kg, 45 mg is recommended; for body weight more than 100 kg, 90 mg is suggested. Phase III trials revealed better efficacy in ustekinumab than in etanercept and placebo.15–19
Efficacy
The Phoenix I and II trials recruited 766 and 1230 patients. At week 12, PASI 75 responses were 67.1%, 66.4%, and 3.1% in the 45 mg, 90 mg, and placebo groups of Phoenix I.
15
Similar PASI 75 results were noted in Phoenix II with 66.7%, 75.7%, and 3.7% in three respective arms. In Phoenix II, partial responders (PASI improvement ⩾ 50 and <75) at week 28 were reallocated to two groups with different frequency of drug administration. The results showed that at week 52, ustekinumab 90 mg every 8 weeks was superior to every 12 weeks (PASI 75 = 68.8%
CADMUS, a phase III study (
ACCEPT (
Safety
Nasopharyngitis (7–8%), upper respiratory tract infection (4–5%) and headache (5%) accounted for the three most common adverse reactions in both 45 mg and 90 mg doses. 20 A 5-year follow-up of clinical trial data 21 and registry data with more than 12,000 patients 22 revealed no increase of serious infections, MACEs, malignancies and mortality rates.
Ustekinumab, an anti-IL-12/IL-23 agent, is the first biologic which is approved first in psoriasis and then in other indications. Due to the high efficacy and relative absence of reactivation of tuberculosis risk, it has emerged as the most widely used biologic agent for psoriasis in many countries. However, ustekinumab will remain the only biologic agent for this mechanism, knowing the unnecessity of inhibiting IL-12 for psoriasis treatment. Also, the dubious increased risk of MACEs from briakinumab put an end to future development of novel IL-12/IL-23 blockers. The use of ustekinumab is expected to fall sharply after the launch of pure IL-23 blockers.
Interleukin-17 antagonists
IL-17, as well as IL-23, have emerged as the most important cytokines in psoriasis. Patients with psoriatic lesions had 30 times more IL-17 level than those with normal skin. 23 Three agents directed against either IL-17 or IL-17 receptors have been approved for psoriasis.
Secukinumab
Secukinumab is an IgG1κ monoclonal antibody that targets IL-17A. Standard dosage is given by 300 mg subcutaneously at weeks 0, 1, 2, 3 and 4, and 300 mg every 4 weeks thereafter. For patients with body weight less than 60 kg, 150 mg is also acceptable. Several phase III studies showed the efficacy of secukinumab 300 mg to be better than 150 mg; both 300 mg and 150 mg were superior to placebo, etanercept and ustekinumab.24–26
Efficacy
In the ERASURE study (
In the FIXTURE study, 1306 patients were randomly allocated to secukinumab 300 mg, secukinumab 150 mg, etanercept 50 mg twice-weekly (BIW) for 12 weeks followed by 50 mg once-weekly, or placebo groups. PASI 75 response at week 12 was 77.1%, 66.7%, 44%, and 4.9%, respectively. At week 52, 78.6% in the 300 mg group, 65.7% in the 150 mg group, and 55.4% in the etanercept group achieved PASI 75 response. According to these two double-blind, randomized controlled trials, the efficacy of both secukinumab 300 mg and 150 mg was superior to placebo and etanercept, with peak response at week 16 maintaining to week 52. 24
In the head-to-head CLEAR study (
SCULPTURE, another phase III, double-blind, randomized parallel-group trial (
The partial responders (definition: PASI 50–74 at week 12) in SCULPTURE entered the STATURE study. Participants were reallocated to the secukinumab 10 mg/kg intravenous group or the 300 mg subcutaneous group. At week 8, PASI 75 response rates were higher in the intravenous group than in the subcutaneous group (91%
Subjects who administered prefilled syringes by themselves also demonstrated good efficacy in the FEATURE study (
Safety
The most common adverse reactions of secukinumab were nasopharyngitis (11.4–12.3%), diarrhea (2.6–4.1%), and upper respiratory tract infection (2.5–3.2%). Neutropenia was observed in clinical trials, but most conditions were transient and reversible without serious accompanying infections. It is worth noting that exacerbation of inflammatory bowel disease was reported in some cases and one randomized controlled, double-blind trial proved that secukinumab gave no benefit to Crohn’s disease and induced more adverse effects.30,31 Mucocutaneous candidiasis rate was higher in the secukinumab group than in the control group (1.2%
Ixekizumab
Ixekizumab specifically binds to IL-17A to inhibit inflammation. The recommended dosage is 160 mg at week 0, then 80 mg Q2W until week 12 followed by 80 mg Q4W. Three phase III studies corroborated the efficacy of ixekizumab.
Efficacy
In the UNCOVER-2 (
The UNCOVER studies were extended to 60 weeks. In UNCOVER-1 (
In IXORA-S, another phase III trial, ixekizumab 160 mg loading dose/80 mg Q2W was superior to ustekinumab dosed by body weight. At week 12, PASI 90 responders were 75% in the ixekizumab group and 42% in the ustekinumab group; sPGA 0/1 was 43% in the ixekizumab group and 18% in the ustekinumab groups. After 12 weeks, ixekizumab 80 mg Q4W was given. Superiority of ixekizumab to ustekinumab was maintained through week 24 (PASI 90 = 83.1%
Safety
Pooled data from three UNCOVER trials revealed the rates of candida infection (3.4%), inflammatory bowel disease (0.3%), and grade 3/4 neutropenia (0.3%) through weeks 0–60 of all ixekizumab exposure patients. During the placebo-controlled period (weeks 0–12), the rate of grade 3/4 neutropenia was 0.2% in the ixekizumab arm and 0.1% in the placebo arm. The proportion of neutropenia of ixekizumab-treated patients at weeks 13–60 was less than that at weeks 0–12.33,35
Interleukin-17 receptor-A antagonist
Brodalumab
Brodalumab, an IgG2κ monoclonal antibody, binds to IL-17 receptor A (IL-17RA) and inhibits IL-17A, IL-17F, IL-17A/F, IL-17C and IL-25. The recommended dosage is 210 mg given at weeks 0, 1, 2, and then every 2 weeks. The black boxed warning emphasized that the Risk Evaluation and Mitigation Strategy (REMS) program should be performed before prescribing due to the risk of suicidal ideation and behavior. 36
Efficacy
Phase III trials (AMAGINE-2 and 3) demonstrated that brodalumab 210 mg was statistically superior to ustekinumab standard dose and placebo. At week 12, PASI 75 was 86% in the brodalumab group and 8% in the placebo group; PASI 100 was 44% in the brodalumab group and 22% in the ustekinumab group (AMAGINE-2). 37
Safety
Arthralgia, headache and fatigue were the three most common adverse reactions of brodalumab. Crohn’s disease is contraindicated because brodalumab may aggravate this disease. In clinical trials, four completed suicides occurred in the brodalumab-treated group. 36
Interleukin-17A and interleukin-17F
ALX-0761/M1095. ALX-0761/M1095, a bispecific IL-17A/IL-17F nanobody, showed promising results in a phase I study for moderate-to-severe plaque psoriasis. All patients in the highest-dose group (240 mg, subcutaneously) achieved PASI 90, and 56% of subjects achieved PASI 100. By day 85, 91% among the treated arm and 0% among the placebo arm achieved sPGA 0/1. 38
Bimekizumab
Bimekizumab is an IgG1 monoclonal antibody neutralizing both IL-17A and IL-17F.
A phase I study (
IL-17 is proved to be the key cytokine which drives psoriasis directly, and the controlled clinical trials have demonstrated all the three approved IL-17 or IL-17R antagonists have superiority over ustekinumab. In addition,
Interleukin-23 p19 antagonists
As described above, IL-23 shares with IL-12 the same p40 protein as heterodimers, that is, p40p35 for IL-12 and p40p19 for IL-23. IL-23 is produced by keratinocytes and dendritic cells. Targeting the p19 protein has the potential advantage of bypassing the Th1 pathway, which has physiologic roles in antiviral host defense. IL-23 helps proliferation, maturation and maintenance of Th17 cells, and promotes the production of IL-17 cytokine.43–48
Guselkumab: efficacy
Three randomized–controlled, double-blind, phase III trials revealed the efficacy and safety of guselkumab, and the FDA is reviewing it as a therapy for plaque psoriasis. In VOYAGE 1 and 2, guselkumab 100 mg was superior to placebo and adalimumab in IGA 0/1 and PASI 90. At week 16, IGA 0/1 was 85.1%, 65.9%, and 6.9%; PASI 90 was 73.3%, 49.7%, 2.9% in the guselkumab, adalimumab, and placebo groups (VOYAGE 1), respectively. 49 Similar results were shown in VOYAGE 2. Moreover, 66.1% of patients who had poor response to adalimumab achieved PASI 90 after shifting to guselkumab at week 48. 50
The NAVIGATE study compared the efficacy of guselkumab and ustekinumab among ustekinumab users who failed to reach IGA 0/1 at week 16. From weeks 28 to 40, guselkumab-treated patients had a significantly higher mean number of visits: IGA 0/1 and ⩾2-grade improvement was twice as often as ustekinumab-treated patients (1.5
Tildrakizumab (MK-3222)
Two phase III trials (reSURFACE 1 and 2) confirmed the efficacy of tildrakizumab: at week 12, PASI 90 was 37% in the 100 mg arm, 36% in the 200 mg arm; PGA 0/1 was 57% and 59%, respectively. At week 28, the percentage of patients achieving PASI 90 were 54% in 100 mg, 59% in 200 mg, and PGA 0/1 was 66% and 69%, respectively. The results of the reSURFACE 2 trial also showed that poor responders to etanercept (PASI < 75) who were shifted to tildrakizumab 200 mg achieved PASI 75, 90, 100, and a PGA 0/1 of 81%, 42%, 16%, and 69%, respectively, at week 52 (over 28 weeks of tildrakizumab treatment).52,53
Risankizumab (BI-655066)
The results of a phase II head-to-head study showed superior efficacy of risankizumab to ustekinumab. At week 12, the proportion of subjects who achieved PASI 90 was 78.6% in the 180 mg risankizumab group and 40% in the ustekinumab group. At week 48, 37% more patients maintained PASI 90 between the two groups (47.6%
Mirikizumab (LY3074828)
Mirikizumab, a humanized IgG4κ monoclonal antibody, antagonizes IL-23. A phase II, randomized controlled trial is ongoing to evaluate the efficacy in participants with plaque psoriasis [ClinicalTrials.gov identifier: NCT02899988]. 56
The FDA approval of guselkumab for the treatment of psoriasis has opened a new era in psoriasis treatment. However, the efficacy of anti-IL-23 agents seems to be similar to that of the currently approved anti-IL-17/IL-17R agents. However, a different safety profile exists in the two classes of drugs and, in theory: anti-IL-23 targets the upstream of psoriasis inflammation pathway and anti-IL17 targets the downstream. Thus, studies to compare their long-term drug survival and duration of remission in addition to safety will be necessary. A clinical study is already ongoing to answer this question (ECLIPSE) [ClinicalTrials.gov identifier: NCT03090100]. 57
Part II: interleukin-1, 2, 6, 8, 10, 20, 22, 36, and dual targets (Table 3)
Anti-interleukin and interleukin agents with clinical data for psoriasis treatment.

R, receptor; IL, interleukin; GPP, generalized pustular psoriasis; PPPP, palmoplantar pustular psoriasis; TNF-α, tumor necrosis factor alpha.
Interleukin-1 antagonist
IL-1, especially IL-1 alpha (α), appears to be important in the formation of T-cell antigen-presenting cell (APC) dermal clusters, providing an extra-lymphoid environment for the contact between dendritic cells, T cells and macrophages. IL-1 also exhibits rapid and profound effects on keratinocytes. 58
Gevokizumab
Gevokizumab, an IL-1 beta inhibitor, improved two cases of generalized pustular psoriasis (GPP) with 79% and 65% decrease of GPP area and severity index (GPPASI). 59 Further randomized controlled trials are needed to evaluate its safety and efficacy in treating plaque-type psoriasis.
Rilonacept, canakinumab, and anakinra
Two IL-1 blockers, rilonacept and canakinumab, and one IL-1 receptor antagonist (IL-1Ra), anakinra, have been approved for the treatment of rheumatoid arthritis and various autoinflammatory disorders, such as cryopyrin-associated periodic syndromes (CAPS), and neonatal-onset multisystem inflammatory disease. They have also been reported as effective in many cutaneous diseases, including Schnitzler syndrome 60 and pustular psoriasis, in case reports.61–64
Among the three drugs, only anakinra has been tested in patients with plaque-type psoriasis. 65 Although effective in some patients with pustular psoriasis and psoriatic arthritis, anakinra fails in psoriasis treatment possibly due to abundance of IL-1Ra in psoriatic lesions. 66 In addition, new-onset psoriasis has been reported following treatment with anakinra. 67
As an early phase inflammatory mediator, current evidence does not support a role of anti-IL-1 therapy in the treatment of chronic plaque psoriasis. However, for pustular psoriasis, due to the lack of controlled trials or large case series to support the use of any biologic drugs, anti-IL-1 agents remains a reasonable treatment choice.
Interleukin-2 antagonists
In psoriatic lesions, IL-2 and interferon-γ predominance had been demonstrated and psoriasis was once thought to be a type I cytokine inflammation. 68 Besides, it was noted that the level of the IL-2 receptor was proportional to the disease activity. 69 Moreover, IL-2 immunotherapy could induce erythrodermic psoriasis. 70
DAB389 interleukin-2
DAB389IL-2, a fusion protein of IL-2 and diphtheria toxin, has been approved for the treatment of refractory cutaneous T-cell lymphoma. It also showed benefits in patients with psoriasis during a double-blind, phase II trial (
Basiliximab
Basiliximab is a chimeric monoclonal antibody of IL-2 indicated for prophylaxis of acute graft rejection in renal transplantations. It specifically targets the α-chain of IL-2 receptor. Case reports in 2000 had demonstrated its efficacy in severe recalcitrant psoriasis vulgaris and GPP by intravenous infusion.72–74
Daclizumab
Daclizumab is a humanized IgG1 monoclonal antibody which blocks IL-2 receptor by binding to CD25 (an α-subunit of the IL-2 receptor) on T lymphocytes. One clinical trial enrolled 19 patients to receive 2 mg/kg of daclizumab initially, and then 1 mg/kg at weeks 2, 4, 8, and 12. The results showed reduced PASI score by 30% at week 8 for subjects with initial PASI less than 36 (
Anti-IL-2 agents are important in the developmental milestone of biologic therapy for psoriasis. However, due to relatively high toxicity and only intermediate treatment response compared with TNF blockers present at the same time, anti-IL-2 treatment is no longer used for psoriasis treatment.
Interleukin-6 antagonists
IL-6 and transforming growth factor beta are important for the IL-23-induced differentiation of Th17 cells, and are therefore important in the initiation of psoriasis. FDA-approved IL-6-receptor antagonists included tocilizumab and sarilumab, which were approved for rheumatoid arthritis and giant cell arteritis (tocilizumab only). Siltuximab, an anti-IL-6 monoclonal antibody, was approved for the treatment of human immunodeficiency virus-negative and human herpes-virus-8-negative patients with multicentric Castleman’s disease. Other anti-IL-6 antibodies such as olokizumab and sirukumab are undergoing clinical phase II/III clinical trials for rheumatoid arthritis. However, more deaths in patients taking sirukumab were reported in patients with rheumatoid arthritis.
Tocilizumab
Several case reports existed to show the successful treatment with tocilizumab for palmoplantar pustular psoriasis (PPPP) triggered by TNF-α inhibitors and rituximab in patients treated for arthritis.76–78 Psoriatic arthritis was also been reported to improve after tocilizumab use. 79 However, there were reports of psoriasis onset after tocilizumab administration.80,81
Although IL-6 plays a pivotal role in the maturation of Th17 cells, search of literature and clinical trial registration websites did not reveal psoriasis studies with anti-IL-6/IL-6 receptors.
Interleukin-8 antagonists
IL-8 is a pro-inflammatory and growth-promoting factor. IL-8 induces keratinocyte overgrowth in psoriasis. In addition, overexpression of IL-8 in skin is associated with accumulation of large amounts of neutrophils, and neutrophil infiltration relates to inflammatory and treatment-resistant psoriasis. IL-8 receptors are highly expressed on the surface of both keratinocytes and neutrophils. Moreover, angiogenesis in the psoriatic lesions are accelerated by IL-8 cytokine, which further promotes keratinocyte proliferation.82–84
Abcream (Enboke)
A phase II/III, double-blind, placebo-controlled trial enrolled 412 psoriasis patients to test the efficacy of Abcream, a topical IL-8 inhibitor. After 6 weeks, the efficacy was higher in the active treatment group than in the control group (49%
ABX-interleukin8
ABX-IL8 is a fully human monoclonal antibody that targets IL-8. Although it showed efficacy in a phase IIa study, the result could not be replicated in a phase IIb trial. In the phase IIb study, PASI 75 improvement was 6% in the 300 mg group, 2% in the 200 mg group and 3% in the placebo group. There were no further trials of ABX-IL8 conducted in treating psoriasis.87,88
Although anti-IL-8 is approved as a topical treatment for psoriasis in China, it has not been accepted as an effective treatment for psoriasis elsewhere. The use of anti-IL-8 biologics is still under development for oncologic indication, but no further development was in progress for psoriasis.
Interleukin-10 agonist
IL-10 is a type 2 cytokine with numerous immunosuppressive and anti-inflammatory capacities. IL-10 inhibited antigen-presenting cells including dendritic cells, monocytes and macrophages.89–91
Recombinant human IL-10
One pilot
94
and two phase II studies92,95 revealed promising results of IL-10 in improving psoriasis. Then, in a double-blind, randomized controlled trial, subcutaneous recombinant human IL-10 20 μg/kg or placebo was given three times a week for 12 weeks to 28 moderate-to-severe psoriasis patients. Although type I and pro-inflammatory cytokines decreased gradually during the treatment period, there were no significant differences between the two groups in the improvement of PASI scores. At week 12, PASI decrease from baseline was 17% in the IL-10 group and 13% in the placebo group (
The therapeutic use of recombinant IL-10 as an anti-inflammatory cytokine had been highly anticipated for more than 20 years. However, the unexpected failure in a phase II study had put an end to its development in psoriasis, but trials continue in other indications which are all still pending approval. 97
Interleukin-20 antagonist
In psoriatic-lesional skins, IL-20 mRNA, IL-20 proteins and IL-20 receptors were increased in keratinocytes. IL-20 upregulates proliferation and prevents differentiation of keratinocytes.98–101
NNC0109-0012
NNC0109-0012 is a fully recombinant human monoclonal antibody which neutralizes IL-20 cytokine. There were 48 patients enrolled in a phase 1, placebo-controlled, double-blind trial. Drugs were tolerable and no dose-related toxicity under the dose of 0.05–3.0 mg/kg. However, no apparent improvement of PASI scores was demonstrated and the study was terminated early [ClinicalTrials.gov identifier: NCT01261767]. 102
Interleukin-22 antagonists
IL-22, a cytokine secreted from Th17 and Th22 cells, regulates keratinocyte differentiation and seems to be essential for Th17-dependent inflammatory diseases. Overexpression of IL-22 induced psoriasis-like lesions (acanthosis and hypogranulosis) in mice and the mechanism was associated with upregulating signal transducer and activator of transcription 3. Another mouse model pathology corresponded to this finding: blocking IL-22 reduced acanthosis and production of Th17 cytokines.97,98,103,104 In psoriasis, limited production of IL-22 binding protein, a natural IL-22 neutralizer, has been demonstrated leading to the intensification of skin inflammation.99,105
Fezakinumab (ILV-094)
There was a phase I study [ClinicalTrials.gov identifier: NCT00563524] performed to evaluate the efficacy and safety of fezakinumab, an anti-IL-22 monoclonal antibody, in treating psoriasis. The result was unpublished and ILV-094 had been listed as a discontinued project in the Pfizer Pipeline, published in August, 2011.100,106
ILV-095
ILV-095 is another anti-IL-22 antagonist. A phase I study [ClinicalTrials.gov identifier: NCT01010542] of ILV-095 300 mg was terminated in March 2011 because the study was unable to meet the primary efficacy endpoint.101,107
IL-22 is the key cytokine responsible for skin thickening (acanthosis) seen in both psoriasis and atopic dermatitis. More recently, it is also found to induce new bone formation in psoriatic arthritis. 108 Despite the failure of current anti-IL-22 agents in the treatment of psoriasis, therapeutic opportunities targeting IL-22/IL-22R still exist, at least for other indications. 109
Interleukin-36 antagonists
IL-36 belongs to the same family as IL-1. In humans, IL-36 receptors are widely expressed by epithelia and antigen-presenting cells, but not human T cells and neutrophils. GPP is an orphan inflammatory disease without any FDA-approved medication. It has been known that the mutations of IL-36 receptor antagonists (IL-36Ra) are present in a high proportion of patients with GPP and acrodermatitis continua. 110
ANB019
ANB019 is an antibody which blocks IL-36-receptor (IL-36R). A Phase I trial was planned to commence in 2017 for GPP and PPPP. Patients with PPPP also have high serum IL-36 levels and were recruited in this trial, too. 111
BI 655130
BI 655130 is another compound of undisclosed mechanism also tested for GPP and PPPP treatment.
A phase I, single-arm, open-label trial [ClinicalTrials.gov identifier: NCT02978690] has been initiated to evaluate the efficacy, safety, pharmacokinetics and pharmacogenomics of BI 655130 in adult subjects with a flare of GPP covering more than 10% body surface area. A single intravenous dose of BI 655130 will be given. 112
Anti-IL-36 is still in the early development for the treatment of pustular psoriasis. Its role in plaque psoriasis also remains to be determined. Pustular psoriasis is a rare disease, and drugs indicated only for this indication will be exceedingly expensive. In fact, several existing biologics, such as secukinumab, infliximab, ixekizumab and brodalumab are approved for the treatment of pustular psoriasis in Japan based on small open-label studies. Potential high cost of IL-36 blockers will only be justified if improved efficacy and safety can be demonstrated for this new class of drugs.
Dual targets
Dual targets of biologics are thought to offer two benefits. One of them is to exert a more efficient suppression of main cytokines; the other is to prevent forming another circuit that drives inflammation. 113
Interleukin-17A and tumor necrosis factor alpha
COVA 322. COVA 322 is an IL-17A and TNF-α fusion protein. A phase Ib/IIa trial [ClinicalTrials.gov identifier: NCT02243787] for moderate-to-severe plaque psoriasis was terminated due to observed safety profile. 114
ABT-122, another bispecific agent blocking IL-17A and TNF-α simultaneously, demonstrated no significant difference of efficacy compared with adalimumab in rheumatoid arthritis and psoriatic arthritis phase II trials. Currently, no clinical trials are registered for psoriasis treatment (Table 4). 115
Anti-interleukin agents pending results for psoriasis treatment.

IL, interleukin; R, receptor; GPP, generalized pustular psoriasis; PPPP, palmoplantar pustular psoriasis.
Part III: interleukin-9, 13, 15, 16, 18, 19, 21, 24, 27, 33, 35, 37, 38
Interleukin-9
IL-9R levels were significantly higher in psoriatic skin compared with healthy control. Intradermal injection of IL-9 induced Th17-associated inflammation, and IL-9 promoted IL-17A production significantly. Therefore, it was suggested that IL-9 might have a role in the psoriasis formation through the Th17 pathway.116,117
Interleukin-13
IL-13 has been found to be a risk allele for psoriasis. It is also upregulated in psoriatic lesions. 118 However, its functional role in psoriasis inflammation remains unclear, and no clinical trials have been directed against IL-13.
Interleukin-15
IL-15 acts early in the inflammation reaction and promotes the production of cytokines including IFN-γ, TNF-α, and IL-17. In a mouse model, an IL-15 receptor antagonist, 146B7, inhibited TNF-α and T-cell proliferation. 119 In a PPPP study with 20 patients and 29 healthy individuals, serum IL-15 and IL-22 levels significantly increased in the disease group than in the control group. 120 These two reports suggested the role of IL-15 in psoriasis.
Interleukin-16
IL-16 is a cytokine that recruit CD4+ cells. One study revealed significantly higher serum IL-16 levels in plaque type psoriasis (
Interleukin-18
IL-18 is one of the cytokines which belong to the IL-1 family. IL-18 expression is increased in keratinocytes from lesional skin, and significantly decreased after treatment. In psoriatic skin, IL-18 levels are significantly correlated with disease severity. 122 IL-18 might cooperate with IL-23 to induce inflammation and epidermal hyperplasia. 123 It was also noted that serum concentrations of IL-18 had positive correlation with GPP disease severity scores.85,124
Interleukin-19
IL-19, signaling through the IL-20 receptor, belongs to the IL-10 family (including cytokines IL-10, IL-19, 20, 22, 24, 26). Elevated serum and lesional levels of IL-19 were significantly associated with psoriatic disease severity. It potentiates IL-17A effects on keratinocytes. 125
Interleukin-21
Serum IL-21 levels significantly elevated in psoriasis patients compared with healthy individuals, and IL-21 concentrations were positively correlated with PASI severity. 126 This suggested IL-21 plays an important role in the pathogenesis of psoriasis. It has been demonstrated that IL-21 promoted CD4+ T cells to differentiate into Th17 cells.127,128 It also induced human keratinocyte proliferation and epidermal hyperplasia. Blockade of IL-21 in mouse model demonstrated the resolution of inflammation and the inhibition of keratinocyte proliferation. 129 Furthermore, the baseline level of IL-21 might be a potential marker for prediction of ustekinumab response. 130
Interleukin-24
IL-24 belongs to the IL-20 subfamily (IL-19, 20, 24) and it acts through binding to heterodimer receptors: IL-20R1/IL-20R2 or IL-22R1/IL-20R2. Signal transduction by activating STAT-1 and STAT-3 is related to cell survival and proliferation. In mice, TNF-α stimulated IL-24 expression in keratinocytes and resulted in the development of psoriasis-like inflammation. 131 Etanercept, an anti-TNF-α blocker, suppressed gene expression of the IL-20 subfamily within 1 week of administration and inhibited Th17 elements later, at week 3–4. 132
Interleukin-27
IL-27 is a member of the IL-12 family and mainly secreted by antigen-presenting cells. Serum IL-27 levels were higher in psoriatic patients than in normal controls and were related to the psoriasis severity. 133 It has been reported that IL-17 and IL-27 presented an opposite reaction to the production of TNF-α-induced chemokines. This suggested that the balance of IL-17 and IL-27 in psoriatic lesional skin influenced the recruitment of neutrophils, dendritic cells, and Th17 cells, which affected inflammation. 134
Interleukin-33
IL-33 belongs to the IL-1 superfamily. Originally, IL-33 was regarded as a Th2 cytokine inducer, but later, it was found to be associated with Th1/Th17-mediated inflammatory diseases including rheumatoid arthritis and inflammatory bowel disease. In psoriatic patients, increased expression of IL-33 was found in lesional skin. 135
Compared with healthy controls, serum levels of IL-33 were significantly elevated in psoriasis, psoriatic arthritis, and pustular psoriasis patients. Serum IL-33 correlated with inflammation of psoriasis, and IL-33 levels were reduced following anti-TNF-α treatment. 136
Interleukin-35
IL-35, a member of the IL-12 family, is secreted by regulatory T cells. IL-12 and 23 promote inflammation, whereas IL-35 suppresses immune reaction. IL-35 is a heterodimer composed of p35 (encoded by IL-12 genes) and Ebi3 subunits. 137 IL-35 inhibited the activity of Th17 cells in autoimmune uveitis and collagen-induced arthritis. 138 In psoriasis models, IL-35 reduced pro-inflammatory cytokines and decreased the numbers of macrophages. 139
Interleukin-37
IL37 is decreased in psoriatic lesions. Transfection of IL-37b to human macrophage cell lines resulted in a decrease of pro-inflammatory cytokines such as IL-1, IL-6, TNF-α and macrophage inflammatory protein 2. 140
Interleukin-38
IL-38 is a member of IL-1 cytokine family and IL-36 subfamily. IL-38 binds to IL-36R and shows antagonistic effects. It was suggested that IL-38 limited the inflammation of psoriasis. 141 In a human study (n=40), serum levels of IL-38 was higher in patients with pustular psoriasis than in patients with psoriasis vulgaris or healthy controls. 142
Conclusion
With the clarification of the pathogenesis of psoriasis, its treatment has changed significantly. Anti-interleukin and interleukin therapies have become the mainstay method for patients with moderate-to-severe psoriasis. Both the quality of life and treatment efficacy have met with increasingly high satisfaction without compromised safety consideration. Although scientists and clinical specialists have established IL-23/Th17/IL-17 pathway as the major axis, there is still a small group of patients who cannot be alleviated by the existing agents, both in Western and Asian subjects. 143 Thus, clinical trials targeting other cytokines or interleukins, either alone or in combination, might still bring new insights into our understanding and armamentarium of psoriasis treatment in the future. In addition, the need for long-term maintenance treatment still brings about huge pharmaco-economic burden. Modalities to induce long-term remission and disease modification are still awaited.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Dr Tsen-Fang Tsai has conducted clinical trials or received honoraria for serving as a consultant for AbbVie, Boehringer Ingelheim, Celgene, EliLilly, Galderma, GSK-Stiefel, Janssen-Cilag, Leo-Pharma, Merck, Novartis, Pfizer, and Serono International SA (now Merck Serono Intenational). Dr. Ya-Chu Tsai has received speaking fees from Pfizer and Janssen-Cilag Pharmaceuticals.
