Abstract
Bone marrow (BM) contains stem cells for both hematopoietic and nonhematopoietic lineages. Hematopoietic stem cells enable hematopoiesis to occur in a controlled manner in order to accurately compensate for the loss of short- as well as long-lived mature blood cells. The physiological role of nonhematopoietic BM stem cells, often referred to as multipotential stromal cells or skeletal stem cells (SSCs), is less understood. According to an authoritative current opinion, the main function of SSCs is to give rise to cartilage, bone, marrow fat and hematopoiesis-supportive stroma, in a specific sequence during embryonic and postnatal development. This review outlines recent advances in the understanding of origins and homeostatic functions of SSCs in vivo and highlights current and future SSC-based treatments for skeletal and joint disorders.
Keywords
Introduction: bone marrow determined osteogenic progenitors
The discovery of multipotential stromal cells (MSCs) (‘determined osteogenic progenitors’) in the bone marrow (BM) emanates from seminal experiments by Friedenstein and colleagues, who demonstrated the presence in the BM of clonogenic and multipotential cells, able to generate mesenchymal tissues in vivo [Friedenstein et al. 1974]. Whereas an innate ability to form bony ‘ossicles’ in vivo has also been shown for MSCs resident in other tissues, such as periosteum [De Bari et al. 2006; Arnsdorf et al. 2009; Roberts et al. 2015] only BM-resident MSCs possessed an additional strong ability to support ectopic hematopoiesis [Miura et al. 2006; Sacchetti et al. 2007; Yamaza et al. 2009].
Whereas an ability to support hematopoiesis cannot be considered as an indispensable feature of all MSCs, its relevance to skeletal development and physiological live-long bone remodeling should not be underestimated. A well-known age-related phenomenon of red-to-yellow marrow conversion in human long bones clearly illustrates that BM MSC differentiation towards fat goes hand in hand with reduced hematopoiesis in the same bone [Bianco and Robey, 2015]. On the other hand, recent advances in the hematopoietic stem cell (HSC) field indicate that HSC stromal ‘niche’ cells, that is the same BM MSCs, may be involved in the progression of hematopoietic malignancies [Garcia-Garcia et al. 2015]. These unique features rightly justify positioning BM MSCs [or according to alternative terminology, skeletal stem cells (SSCs)] ‘above and beyond’ other tissue-resident MSCs, as they are critically important in the whole organism physiology, rather than purely in their host tissue maintenance [Oreffo et al. 2005; Kassem and Bianco, 2015]. This review article will summarize rationale, progress to date and clinical application of BM MSCs or SSCs in the field of bone regeneration and repair. While not attempting to describe the whole spectrum of tissue-resident MSCs, we will nevertheless give some pertinent examples of other MSC types (adult and fetal) that have been successfully used in preclinical and clinical investigations aimed at bone repair.
SSCs: one cell or many?
The fact that BM MSCs or SSCs should be able to produce four distinct lineages, cartilage, bone, fat and hematopoiesis-supportive stroma, has over the years posed a question of their potential heterogeneity and, if it existed, whether it was predetermined (i.e. developmentally regulated) or environmentally induced. Early studies have initially focused on tripotential features of BM MSCs [Muraglia et al. 2000] and attempted to assign certain specific markers to subpopulations of committed osteogenic and nonosteogenic progenitors [Satomura et al. 1998]. It was also recognized that standard cultivation leads to significant phenotypic changes in MSCs [Digirolamo et al. 1999] and that the presence of markers on certain MSC subsets in vitro does not warrant their presence in vivo [Buhring et al. 2007]. Cell-sorting studies using a number of surface marker combinations such as CD45/CD271 [Jones et al. 2002; Buhring et al. 2007; Battula et al. 2009; Churchman et al. 2012; Li et al. 2014] in humans, and platelet-derived growth factor (PDGFR)-α/Sca-1 in mice [Morikawa et al. 2009; Houlihan et al. 2012] have succeeded in isolating the whole fraction of BM MSCs, rather than its specific functional subsets. Interestingly, Tormin and colleagues have shown that two different phenotypes of BM MSCs were linked to their in vivo topography, rather than differentiation predisposition [Tormin et al. 2011]. The lin–/CD271+/CD45–/CD146–/low) subset characterized bone-lining MSCs located near the bone surfaces whereas the lin–/CD271+/CD45–/CD146+ subset characterized reticular cells located perivascularly, however up to now it remained unclear whether they had the same or different embryonic origins.
The neural crest (NC) is a transient embryonic tissue. During development, NC cells migrate to various locations and persist as undifferentiated cells at least until late gestation [Nagoshi et al. 2009]. According to earlier reports, NC stem cells were found to migrate through the bloodstream and reach the BM coinciding with the arrival of HSCs [Dzierzak and Speck, 2008; Nagoshi et al. 2008], where they persisted into adulthood and played a role in HSC maintenance. In more recent reports, NC stem cells were traced as travelling along developing nerves rather than via the bloodstream [Isern et al. 2014]. Significantly, only these NC stem cells (nestin+ MSCs) were located in neonatal BM and endowed with HSC support function while a separate population of mesoderm-derived cells (nestin– MSCs, located in the appendicular skeleton) formed a pool of progenitors for endochondrogenesis [Isern et al. 2014]. Other recent articles provided additional evidence for developmental heterogeneity of mouse SSCs. For example, Wortley and colleagues recently demonstrated the existence of osteochondroreticular stem cells generating articular and growth plate cartilage during development, which were in late adulthood complemented by traditional ‘perisinusoidal’ adipogenesis-competent MSCs [Worthley et al. 2015]. Different phenotypic profiles of early postnatal and adult mouse SSCs were also reported in the studies of Ono and colleagues [Ono et al. 2014], Zhou and colleagues [Zhou et al. 2014] and Chan and colleagues [Chan et al. 2015]. Therefore, according to the most recent findings, SSCs do not in fact represent a ‘singularity’ [Bianco and Robey, 2015] but are rather a mixture of stem cells of different embryological origins [Komada et al. 2012; Isern et al. 2014], this being dependent on an anatomical location as well as a ante- and postnatal developmental stage [Kassem and Bianco, 2015].
Although therapeutic exploitation of the hematopoiesis-supporting subpopulation of SSCs is outside the scope of this article, it is worth noting that manipulating HSC-niche cells has broad implications for the treatment of leukemias and other BM diseases (reviewed by Garcia-Garcia and colleagues) [Garcia-Garcia et al. 2015]. Furthermore, recent evidence suggests that BM MSCs have a major role in ‘emergency hemopoiesis’, that is ‘on demand’ myeloid-lineage cell production in response to infection [Boettcher et al. 2012; Welner and Kincade, 2014; Ziegler et al. 2015]. This is because MSCs possess toll-like receptors [Liotta et al. 2008] and are able to respond to pathogens by producing soluble mediators that support the survival of innate immune cells [Cassatella et al. 2011] or skew HSC differentiation towards innate immune cell lineages [Welner and Kincade, 2014]. Furthermore, perisinusoidal MSCs are likely controlling the egress of hematopoietic cells into systemic circulation [Cariappa et al. 2005; Allende et al. 2009]. In some way, the ability of BM MSCs to orchestrate emergency hematopoiesis can be seen as one example of their native or in vivo immunoregulatory function, which is easily reconciled with their HSC niche support function.
The use of SSCs in orthopedics
Early reports have shown that standard culture expansion of BM MSCs or SSCs is permissive for the maintenance of osteo- and chondrogenic progenitors [Muraglia et al. 2000]. Early clinical studies utilized culture-expanded BM MSCs seeded on ceramic scaffolds to enable repair of large (segmental) bone defects [Quarto et al. 2001]. Several important clinical studies employing autologous cultured BM MSCs for large bone defect repair including avascular necrosis of the femoral head are outlined in Table 1. Avascular necrosis of the femoral head is a serious orthopedic condition often occurring in young individuals (Figure 1). Although limited in their level of evidence, a combination of these clinical case reports and retrospective studies suggests good clinical safety of cultured BM MSCs for a follow-up period of up to 7 years. Although local application of MSCs resulted in complete fusion/healing in cases of fractures and nonunions, and decreased necrotic lesions in cases of osteonecrosis (Table 1), it needs to be noted that larger-cohort, prospective, randomized studies are needed to confirm the observed beneficial effects.
Clinical studies using cultured autologous BM MSCs/SSCs for the treatment of orthopedic defects.

According to ISCT minimal criteria, MSCs must express CD73, CD90, CD105 and lack expression of CD11β or CD14, CD19 or CD79α, CD34, CD45, and HLA-DR surface molecules [Dominici et al. 2006].
BM, bone marrow; MSC, multipotential stromal cell; NR, not reported; SSC, skeletal stem cell ; ISCT, international society of cellular therapies; HLA-DR: human leukocyte antigen-D related.

Anteroposterior radiograph of a 19-year-old man with post-traumatic late-stage avascular necrosis (a). Note the large cartilage defect discovered intraoperatively (b).
With the use of cultured MSCs, that remain undifferentiated, the main challenge remains how to insure their maximal osteogenic differentiation with minimal untoward commitment to other lineages. Recently proposed approaches for this include preselection of stage-specific osteogenic progenitor subsets [Twine et al. 2014; Loebel et al. 2015] or the use of ‘smart’ new-generation osteoconductive scaffolds with optimal pore sizes, calcium release and resorption profiles [Roberts et al. 2015] or precise nanosurface geometries or cell-material interfaces [Dalby et al. 2007; Crowder et al. 2016].
More recently, uncultured BM MSCs in the form of BM ‘concentrates’ or ‘viable allografts’ have emerged as a new way of bone defect treatment (Table 2). Bone marrow concentrates are normally prepared by minimal manipulation of a BM aspirate such as density centrifugation and volume reduction and their MSC content is normally assessed based on colony-forming unit-fibroblast (CFU-F) assay [Woodell-May et al. 2015]. Viable allografts are prepared from cadaveric cancellous bone after the removal of the immune cell component from the graft while preserving the multipotential nonimmune cells [Baboolal et al. 2014]. In their pioneering clinical studies, Hernigou and colleagues (studies described in Table 2) used BM concentrates for a wide range of orthopedic indications, such as avascular necrosis, fracture nonunion and rotator cuff repair. Overall, these and other studies showed the safety of local or systemic transplantation of uncultured BM MSCs with a follow up of up to 22 years. Furthermore, positive outcomes were found to be correlated with the number of transplanted MSCs (Table 2). It is noteworthy that although these treatments represented a mixture of cells, rather than a pure population of MSCs, some ‘contaminant’ cell fractions, for example monocytes, could contribute to bone repair, by virtue of being precursors of osteoclasts. Furthermore, concentrated injected platelets may also be highly beneficial as producers of high local levels of vascular endothelial growth factor, PDGFs and fibroblast growth factors [Dhillon et al. 2012], the molecules very important in bone repair [Giannoudis et al. 2007]. Although positive clinical outcomes were reported for both cultured and noncultured MSC preparations, the above evidence suggests that their mechanisms of action are likely to be very different and requiring further investigations in preclinical studies.
Clinical studies using uncultured BMAC or viable allografts for the treatment of orthopedic defects.
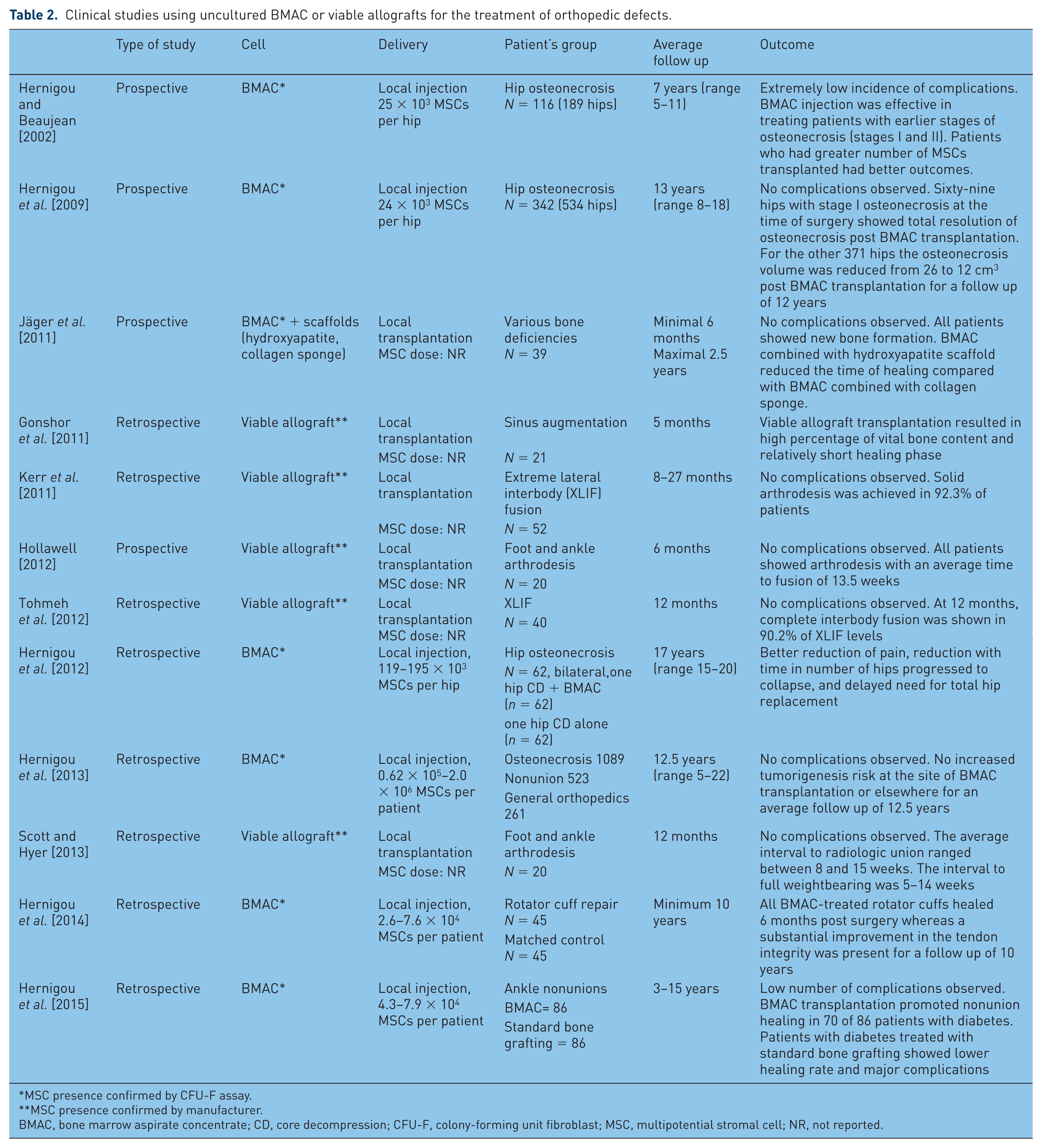
MSC presence confirmed by CFU-F assay.
MSC presence confirmed by manufacturer.
BMAC, bone marrow aspirate concentrate; CD, core decompression; CFU-F, colony-forming unit fibroblast; MSC, multipotential stromal cell; NR, not reported.
Despite encouraging clinical results, the dosages of implanted MSCs (either cultured or uncultured) remain in most cases empirical, and the fate of implanted cells remains to be elucidated. For the quantification of uncultured MSCs in BM aspirates, Cuthbert and colleagues developed a rapid (40 min) flow cytometry based assay that can be used to determine MSC dose intraoperatively [Cuthbert et al. 2012]. Another study from our laboratory [Baboolal et al. 2014] has established an average MSC dose in a viable allograft product, Osteocel (Nuvasive, Inc, San Diego, USA) (0.5 million MSCs/g of graft). In terms of in vivo tracking of implanted or injected MSCs, the most widely used noninvasive approach in animal models has been the green fluorescent protein (GFP) labeling [Guo et al. 2012] whereas magnetic resonance imaging (MRI) was applied to detect iron oxide nanoparticle or fluorine-19 agent labeled MSCs in other studies [Mathiasen et al. 2013; Gaudet et al. 2015].
Regarding the mechanisms of MSC action for their use in orthopedics, apart from their ‘classical’ roles as osteo- and chondroprogenitors (driving intramembranous and osteochondral repair processes, respectively) [Thompson et al. 2015], MSCs may also regulate later bone repair phases, including the remodeling of the transitional tissue and osteointegration via their immunomodulatory [Le Blanc and Davies, 2015], prosurvival and other ‘trophic’ functions [Caplan and Correa, 2011]. To retain both regenerative and immunomodulatory MSC subsets in therapeutic MSC preparations, and in order to capitalize on their combined functions, a better knowledge of their differential phenotypes [James et al. 2015], and methods for their prospective selection would be required. Finally, to achieve a broader understanding of the effectiveness of MSC delivery in orthopedics, particularly in terms of their survival and engraftment in hypoxic environments such as avascular necrosis lesions, more clinical studies with MRI-detectable labeled MSCs are definitely needed.
Exploiting SSCs for the conditions of systemic bone loss
Apart from bone damage due to injury, other conditions of bone loss are represented by systemic bone diseases such as osteoporosis (OP) and osteogenesis imperfecta (OI). In a pioneering study, Pereira and coworkers were the first to apply allogeneic BM MSC therapy in an OI mouse model, showing significant increase in collagen and mineral content 2.5 months after local transplantation [Pereira et al. 1998]. In other studies, systemic infusion of allogeneic BM MSCs in OI mouse models showed MSC engraftment in recipient BM, trabecular bone and cortical bones where they directly differentiated into osteoblasts and synthesized type I collagen [Wang et al. 2006a; Li et al. 2007].
The first human clinical trial to treat OI has been performed by Horwitz and colleagues [Horwitz et al. 2002] who systemically infused allogeneic BM MSCs to treat six children with severe OI. Although four out of five children showed increased bone mineral density and accelerated linear growth at 18–36 months of follow up, only 1% of infused BM MSCs could be detected in bone, skin and other tissues. Low engraftment of systemically infused MSCs in humans could be explained by their entrapment in capillaries of various tissues, including lungs. Because of the observed low osteopoietic cell engraftment, another study from the same group explored an ‘indirect’, collagen mutation-independent mechanism of MSC action in OI where they demonstrated that infused MSCs induced ‘the production of a soluble mediator by a second tissue that activates the proliferation of chondrocytes in the growth plate’ [Otsuru et al. 2012]. These two complementary mechanisms of MSC action in OI (direct and indirect) were also described in another study, although the researchers used fetal chorionic stem cells, not BM MSCs, in their preclinical investigation [Guillot et al. 2008].
Allogeneic fetal MSCs (fMSCs) rather than BM MSCs, have been also transplanted in two separate patients with OI (one with OI type III and one with OI type IV) at 32 and 31 weeks of gestation [Le Blanc et al. 2005]. Neither patient showed alloreactivity towards donor fMSCs or manifested any evidence of toxicity. Postnatal boosting with fMSCs resulted in low-level engraftment in bone but improved linear growth, mobility, and fracture incidence, possibly as a result of an indirect mechanism of MSC action [Gotherstrom et al. 2014].
Regarding OP treatment, local transplantation of autologous BM MSCs has been first assessed in an ovariectomized OP rabbit model. Four to eight weeks after transplantation, newly formed osteoids and enhanced trabecular thickness were evident [Wang et al. 2006b]. In allogeneic settings, when BM MSCs were transduced with receptor activator of nuclear factor κB Fc or bone morphogenetic protein 2 and locally transplanted in ovariectomized OP models, increased bone mineral density and mature bone formation were observed [Kim et al. 2006; Tang et al. 2008]. These preclinical studies indicated the potential of autologous or allogeneic BM MSCs to engraft in recipients’ bones contributing to improved bone mineral density and biomechanical stiffness.
Most recent preclinical studies for OP and OI cell therapies are outlined in Table 3. Some of these therapies use MSCs as vehicles to target miRNAs (noncoding single-stranded RNAs) involved in post-transcriptional expression regulation of bone-related genes. For example, Li and colleagues generated baculovirus-engineered rat BM MSCs expressing microRNA sponges to antagonize microRNA-140 and microRNA-214. Local transplantation of engineered MSCs in osteoporotic mice not only resulted in healing of critical sized bone defects but also in amelioration of bone quality [Li et al. 2016].
Recent preclinical studies using allogeneic BM MSCs/SSCs for the treatment of systemic bone diseases.

BM, bone marrow; MSC, multipotential stromal cell; NR, not reported; RANK-Fc, receptor activator of nuclear factor κB Fc; SSC, skeletal stem cell.
In parallel to the development of cell-based therapies for the treatment of OP and OI, new-generation treatments are aimed at targeting native MSCs or their progeny in these diseases. As mentioned already, the enhancement of MSC anabolic (i.e. bone forming) ability can be achieved via in vivo delivery of miRNAs driving MSC differentiation to osteoblasts or by using miRNA ‘sponges’ to sequester and neutralize miRNAs controlling MSC differentiation to other lineages [Taipaleenmaki et al. 2011]. Other pharmacological interventions for OP treatment include targeting DLK1/Pref-1, a protein expressed by BM MSCs and involved in bone-fat homeostasis, or targeting other mediators such as Wnt pathway molecules in OP MSCs [Janeczek et al. 2015]. Monoclonal antibodies to molecular targets, such as sclerostin, primarily produced by osteocytes (terminally differentiated bone cells derived from MSCs) have also been investigated for the treatment of postmenopausal OP. In preclinical studies, a sclerostin neutralizing antibody increased bone formation, bone mineral density and bone strength [Li et al. 2009; Ominsky et al. 2010, 2011; Suen et al. 2015]. Two humanized antisclerostin monoclonal antibodies, romosozumab and blosozumab, have been shown to possess osteoanabolic properties with the potential to improve clinical outcomes in patients with OP in phase II clinical trials [McClung et al. 2014; Recknor et al. 2015]. With increased knowledge on the in vivo abnormalities in OP MSCs, new pharmacological agents (for example, Wnt pathway modifiers) or biological agents, similar to antisclerostin antibodies, to ‘correct’ their altered function in OP could be developed.
Targeting SSCs to reverse sclerotic bone formation and restore cartilage in osteoarthritis
Some musculoskeletal conditions are characterized by deranged bone formation or resorption processes, one example of which is osteoarthritis (OA). OA involves complex pathologies, which depend in many ways on the type of joint involved (hip, knee, etc.). Nevertheless, the loss of cartilage and subchondral bone abnormalities including bone marrow lesions (BMLs), represent the most common features of all types of OA. Bone marrow lesions represent mesenchymal tissue abnormalities within OA bone and are associated with OA structural progression and pain [Xu et al. 2012; Wenham and Conaghan, 2013]. Although the OA primary lesion may be indeed located in cartilage, abnormal joint loading and shock absorbance are most certainly transduced to the underlying bone, leading to excessive remodeling within the bone tissue and the subsequent formation of BMLs and osteophytes.
The initial appreciation of these processes in OA bone has firstly led to the proposed use of antiresorptive drugs such as bisphosphonates for OA treatment, with a potential to combine those with bone anabolic compounds such as calcitonin, strontium ranelate, parathyroid hormone, estrogens, or selective estrogen receptor modifiers [Kwan Tat et al. 2010; Karsdal et al. 2014]. Being the precursors of both bone and cartilage, MSCs have also attracted a considerable interest as cell and gene therapy modalities for OA (reviewed by Coleman and colleagues) [Coleman et al. 2010]. More recently, it has been recognized that OA MSCs may in fact contribute to abnormal remodeling processes in OA bone [Zhen et al. 2013]. Zhen and colleagues clearly demonstrated that the numbers of MSCs were dramatically increased in OA subchondral bone. Furthermore, the authors proposed that the altered biomechanical pressures on the OA joint have led to increased numbers of osteoclasts and transforming growth factor (TGF)-β1 release from the bone matrix, leading to the TGF-β1-induced MSC recruitment to the subchondral bone contributing to aberrant bone formation and disease progression [Zhen et al. 2013]. In agreement, we have recently found increased numbers and altered transcriptional profiles of CD271+ MSCs in BML areas of hip OA bone and showed their accumulation at the sites of damage [Campbell et al. 2016]. Based on these studies, new OA disease-modifying treatments to target abnormally activated signaling pathways in OA MSCs have been proposed [Zhen and Cao, 2014]. The timing and the mode of delivery of such future therapies remain to be established.
Conclusion
MSCs have been isolated from various tissues of the musculoskeletal system, including periosteum [De Bari et al. 2008], synovium [De Bari et al. 2001], tendon [Lui, 2013], joint fat pad [Wickham et al. 2003; English et al. 2007] and others. Although periosteal MSCs seeded on osteoconductive scaffolds have been shown to contain BM elements following implantation in vivo, only BM-resident MSCs possess homeostatic HSC-supporting niche properties without additional stimulation and in steady-state physiological conditions. This unique feature sets them apart from other tissue-resident MSCs making the use of a specific term, SSCs, justifiable. Not only have their stem cell nature and self-renewal ability been proven in vivo [Sacchetti et al. 2007], their native phenotypes and developmental origins [Bianco, 2011; Isern et al. 2014; Worthley et al. 2015] are now mapped much better compared with other types of MSCs.
There have been many reports on the potential use of adipose-derived MSCs for bone repair applications [Dragoo et al. 2003; Khan et al. 2009; Mesimaki et al. 2009]. However, it does appear more ‘natural’ to capitalize on native abilities of BM MSCs/SSCs to regulate bone formation and remodeling when considering the therapeutic use of MSCs for bone repair. Autologous bone grafting with uncultured SSCs surrounded by their native extracellular matrix (ECM) and other supportive cell populations remains the gold standard for repairing bone in orthopedics. Ex vivo expansion and implantation of cultured SSCs is safe and efficacious, however it may now be surpassed by newer, less laborious and less expensive treatments based on uncultured SSCs such as BM concentrates. Although encouraging clinical results have been obtained by transplanting either uncultured SSCs or cultured SSCs, the exact dosage and route of application remain to be optimized and the fate of transplanted cells and their mechanisms of action need to be better monitored in larger clinical trials. For the treatment of systemic bone diseases, the implantation of cultured or noncultured SSCs appears efficacious and newer approaches could involve targeting of SSCs in situ using pharmacological compounds and biologic treatments. Better understanding of the pivotal functional roles of SSCs in health, ageing and disease will provide novel and advanced therapeutic strategies for skeletal and joint disorders.
Footnotes
Acknowledgements
EJ and PVG are partly supported by NIHR-Leeds Musculoskeletal Biomedical Research Unit (LMBRU).
Funding
This review article received no grant from any funding body in the public, commercial, or non-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
