Abstract
Polymyalgia rheumatica (PMR) is a common inflammatory condition that often affects people over the age of 50 years. Characteristic symptoms are shoulder and hip girdle pain and prolonged morning stiffness. Markers of inflammation are often elevated. Clinicians are often faced with the challenge of distinguishing PMR from other conditions, particularly rheumatoid arthritis and spondyloarthropathy that can mimic symptoms of PMR in older people. Additionally, there is an association between PMR and giant cell arteritis, a common large-vessel vasculitis which also affects people over the age of 50 years. Imaging of the large vessels in asymptomatic patients with PMR often reveals findings of subclinical vasculitis.
Presently, there are no tests that are specific for the diagnosis of PMR and clinicians rely on a combination of history, physical examination, laboratory tests and imaging studies to make a diagnosis. A recent undertaking by the European League Against Rheumatism/American College of Rheumatology has led to the publication of provisional classification criteria of PMR. Ultrasonography, which is being increasingly used by rheumatologists, can greatly aid in the diagnosis of PMR and often shows changes of synovitis and tenosynovitis.
Treatment consists of low doses of glucocorticoids which are associated with morbidity. Evaluation of newer biologic therapies targeting inflammatory cytokines is underway. Despite treatment, relapses are common.
Introduction
Polymyalgia rheumatica (PMR) is an inflammatory rheumatic condition that affects individuals over the age of 50 years. The etiology of PMR is currently unknown, although genetic and environmental factors are thought to contribute [Kermani and Warrington, 2013]. Patients with PMR often report pain and stiffness involving the neck, shoulders, low back, hips and thighs which are worse in the morning and improve with activity. The diagnosis of PMR poses many challenges since there are no diagnostic tests that are specific for this condition. Therefore, careful consideration and exclusion of several conditions that can mimic PMR is important. Collaborative efforts by the European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) have resulted in the publication of provisional classification criteria for PMR [Dasgupta et al. 2012a, 2012b]. Several studies have demonstrated the potential utility of ultrasonography as a diagnostic aid [Koski, 1992; Salvarani et al. 1997; Cantini et al. 2001; Frediani et al. 2002; Falsetti et al. 2011; Dasgupta et al. 2012a, 2012b]. Glucocorticoids (GCs) remain the mainstay of treatment for PMR but are associated with significant morbidity [Gabriel et al. 1997; Hernandez-Rodriguez et al. 2009; Dasgupta et al. 2010; Mazzantini et al. 2012]. Recent anecdotal reports suggest interleukin (IL)-6 may be a helpful therapeutic target in PMR and large-vessel vasculitis but additional studies are needed [Hagihara et al. 2010; Unizony et al. 2012; Alten and Maleitzke, 2013; Ashraf et al. 2013; Macchioni et al. 2013].
Epidemiology and pathogenesis
PMR is the second most common inflammatory autoimmune rheumatic condition in the United States. The estimated annual incidence of PMR is 58.7 per 100,000 people aged 50 years and over [Salvarani et al. 1995; Lawrence et al. 2008; Crowson et al. 2011]. The prevalence of PMR in the United States (based on 2005 estimates from the Census Bureau and the prevalence rates reported in Olmsted County) is calculated at 711,000 people [Lawrence et al. 2008]. It is typically only seen in individuals who are aged 50 years and over with a mean age at diagnosis of 73 years [Salvarani et al. 1995]. The incidence of PMR increases with age [Salvarani et al. 1995; Lawrence et al. 2008; Gonzalez-Gay et al. 2009]. In the United States, the lifetime risk of developing PMR is estimated to be 2.43% for women and 1.66% for men [Crowson et al. 2011].
PMR is more common in white people. Also, there is geographic variation in incidence, with the highest incidence rates being reported in populations in northern latitudes like Aust Agder, Norway while lower incidences have been reported in southern European countries like Italy and Spain [Salvarani et al. 1991; Gran and Myklebust, 1997; Gonzalez-Gay et al. 1999, 2009]. A higher prevalence of PMR was reported in rural areas of the province of Manitoba, Canada compared with urban areas [Bernatsky et al. 2009].
The etiology of PMR is unknown. Genetic polymorphisms in human leukocyte antigen (HLA) genes and other genes related to immune regulation have been associated with PMR [Mattey et al. 2000; Salvarani et al. 2000b; Alvarez-Rodriguez et al. 2009]. There is an association between HLA-DRB1 and PMR [Gonzalez-Gay et al. 2003]. To date no infectious etiology has been confirmed.
As with other chronic autoimmune conditions, there may be an imbalance between the immunosuppressive T-regulatory (Treg) lymphocytes and proinflammatory T-helper 17 (Th17) cells in PMR and giant cell arteritis (GCA) [Samson et al. 2012]. The frequency of circulating Treg cells appears to be reduced in patients with PMR compared with age-matched volunteers, while Th17 cells are increased [Samson et al. 2012]. Another intriguing hypothesis is that of relative adrenal insufficiency (inadequate cortisol secretion in response to the inflammatory status) [Straub et al. 2000; Cutolo et al. 2002, 2006; Demir et al. 2006; Narvaez et al. 2006].
Finally, there is a well recognized association between PMR and GCA and it has been suggested that PMR is a forme fruste of GCA [Weyand et al. 1994]. Temporal artery biopsies from patients with PMR without clinical features of GCA show evidence of subclinical inflammation based on expression of cytokines in the absence of overt vasculitis [Weyand et al. 1994]. Messenger RNA transcripts for IL-2, IL-1 and IL-6 but not interferon γ were detected in the temporal artery specimens from patients with PMR [Weyand et al. 1994; Weyand and Goronzy, 2003].
Clinical features
Patients with PMR present with pain and stiffness affecting the shoulder and hip girdle, low back and neck muscles. Stiffness lasting half an hour or longer, generally occurring in the morning or after periods of inactivity, is common. As opposed to the symptoms of osteoarthritis, the stiffness and pain tend to be bilateral or symmetric and improve with activity. Constitutional symptoms such as fatigue, malaise, anorexia, weight loss and fever are also common [Chuang et al. 1982]. There are many conditions that can mimic PMR, particularly rheumatoid arthritis (RA) and spondyloarthropathies (SpA) [Gonzalez-Gay et al. 2000; Caporali et al. 2001]. Pain and swelling of distal joints like wrists, hands and feet should raise concern for an inflammatory arthritis. A subset of patients with PMR may present with swelling and pitting edema of the hands and feet due to tenosynovitis, so called remitting seronegative symmetrical synovitis with pitting edema syndrome [Salvarani et al. 1999]. However, this variant can also be a presenting feature of other inflammatory forms of arthritis, including SpA and RA.
GCA and PMR share many similarities, including age at onset, female predominance and similar geographic distribution, and are often thought to represent different spectrums of the same disease entity. Clinically, 40–60% of patients with GCA have PMR symptoms at diagnosis while 16–21% of patients with PMR develop GCA [Salvarani et al. 2008b].
Given the association between PMR and GCA, it is important to elicit symptoms of GCA such as headaches, jaw claudication, scalp tenderness, visual symptoms, carotidynia or limb claudication. If present, evaluation for GCA should be pursued. The differential diagnosis and common mimics of PMR are listed in Table 1.
Differential diagnosis of patients presenting with symptoms of polymyalgia rheumatica.

Diagnosis
One of the challenges in the diagnosis of PMR is the lack of any diagnostic tests that are specific for this entity. Additionally, given the multitude of other conditions that can mimic this diagnosis or present with polymyalgic symptoms, the diagnosis requires exclusion of other conditions.
Diagnostic and classification criteria
Several diagnostic criteria for PMR have been proposed based on retrospective clinical series [Bird et al. 1979; Jones and Hazleman, 1981; Chuang et al. 1982; Healey, 1984]. Most of these include an age cutoff, presence of bilateral shoulder girdle and hip girdle pain, morning stiffness and elevated markers of inflammation. Given the absence of any specific tests for PMR, clinicians often use a prompt response to GCs as a confirmation of the diagnosis. However, other conditions that mimic PMR also respond to GC therapy [Matteson, 2010]. Additionally, several recent studies have called into question the validity of using GC responsiveness as a diagnostic tool by demonstrating that only a subset of patients with PMR respond to GCs even after 3–4 weeks of therapy [Hutchings et al. 2007; Dasgupta et al. 2012a, 2012b].
To address some of these issues, an international collaborative initiative between EULAR and ACR is underway. The provisional classification criteria for PMR have been published (Table 2) [Dasgupta et al. 2012a, 2012b]. In addition to clinical features, the use of ultrasonography of the shoulders and hips improved the criteria specificity for PMR (Table 2). It should be noted that these are classification criteria (criteria to separate patients with PMR from a group of patients with conditions that mimic PMR) and are not diagnostic criteria.
Proposed provisional classification criteria for polymyalgia rheumatica.

Required criteria: age ≥ 50 years, bilateral shoulder pain and abnormal erythrocyte sedimentation rate or C-reactive protein.
Using only clinical criteria (without US), a score of ≥ 4 out of a total possible score of 6 had a 68% sensitivity and 78% specificity for discriminating PMR from comparison subjects. When ultrasound criteria were included, a score of ≥ 5 out of a total possible score of 8 had a sensitivity of 66% and specificity of 81% for discriminating PMR from comparison subjects.
N/A, not applicable; PMR, polymyalgia rheumatica; US, ultrasound.
Physical examination
All patients suspected of having PMR should undergo a comprehensive physical examination with special attention to the vascular, neurologic and musculoskeletal components. Musculoskeletal examination often reveals a painful and limited range of motion of the shoulders and hips. Swelling of the wrists and knees has been reported in up to one-third of patients with PMR [Myklebust and Gran, 1996; Salvarani et al. 1998, 2000; Gran and Myklebust, 2000; Ceccato et al. 2006b]. While wrist synovitis and synovitis of metacarpophalangeal or proximal interphalangeal joints have been reported in patients with PMR, these findings would be more suggestive of RA [Pease et al. 2009; Dasgupta et al. 2012a, 2012b]. Patients may have give-way muscle weakness due to pain, but true muscle weakness is not a feature of PMR. Examination should also include evaluation of the temporal arteries, peripheral pulses and auscultation for bruits. If vascular abnormalities are present, evaluation for GCA should be undertaken.
Laboratory findings
Laboratory findings in PMR are nonspecific and may include anemia, leukocytosis and elevated markers of inflammation [Bird et al. 1979; Chuang et al. 1982]. Erythrocyte sedimentation rate (ESR) and C-reactive protein are often elevated, although low (≤30 mm/h) or normal ESR has been reported in 6–20% of patients with PMR [Ellis and Ralston, 1983; Helfgott and Kieval, 1996; Gonzalez-Gay et al. 1997; Olsson et al. 1997; Proven et al. 1999; Cantini et al. 2000; Kyle et al. 1989; Myklebust and Gran, 1996; Cantini et al. 2000].
Autoantibodies including rheumatoid factor and anti-cyclic citrullinated peptide are usually negative in PMR; while if positive, are more suggestive of late-onset RA [Chuang et al. 1982; Lopez-Hoyos et al. 2004; Pease et al. 2005; Ceccato et al. 2006a; Dasgupta et al. 2008]. Thyroid-stimulating hormone, muscle enzymes (creatine phosphokinase), calcium, electrolytes, renal function, serum protein electrophoresis and urinalysis are also recommended tests to exclude PMR mimics [Michet and Matteson, 2008; Dasgupta et al. 2010].
Imaging studies
Imaging studies are not required to establish the diagnosis of PMR, but ultrasonography appears to add to the specificity of the diagnosis and therefore should be considered [Dasgupta et al. 2012a, 2012b]. Radiographs of the affected joints in PMR are often normal and are only useful in excluding other conditions like degenerative joint disease or crystalline arthritis (chondrocalcinosis). The presence of bony erosions on radiographs is more consistent with RA [Pease et al. 2009].
Based on ultrasonography and magnetic resonance imaging (MRI) findings, PMR predominantly affects the periarticular structures. Bicipital tendonitis (Figure 1(a)), subacromial bursitis (Figure 1(b)), subdeltoid bursitis and trochanteric bursitis are commonly seen in patients with PMR [Salvarani et al. 1997; Cantini et al. 2001, 2005; McGonagle et al. 2001]. Glenohumeral and hip joint effusions and synovitis have also been reported [Koski, 1992; Cantini et al. 2001; Frediani et al. 2002; Cantini et al. 2005; Falsetti et al. 2011; Dasgupta et al. 2012; Dasgupta et al. 2012]. It should be noted that the abnormalities described above can also be seen in other inflammatory forms of arthritis like RA or SpA. In general, the presence of subacromial/subdeltoid bursitis or tenosynovitis of the long head of the biceps appears to be more prevalent in patients with PMR [Salvarani et al. 1997; Cantini et al. 2001; Frediani et al. 2002]. In the EULAR/ACR classification study, ultrasonography had high specificity in discriminating PMR from other shoulder conditions (89%) but not in discriminating PMR from RA (70%) [Dasgupta et al. 2012a, 2012b]. Despite the limitations, ultrasonography and MRI can be very helpful in distinguishing common causes of shoulder pain such as rotator cuff tendinopathy and degenerative joint changes from inflammatory conditions like PMR.
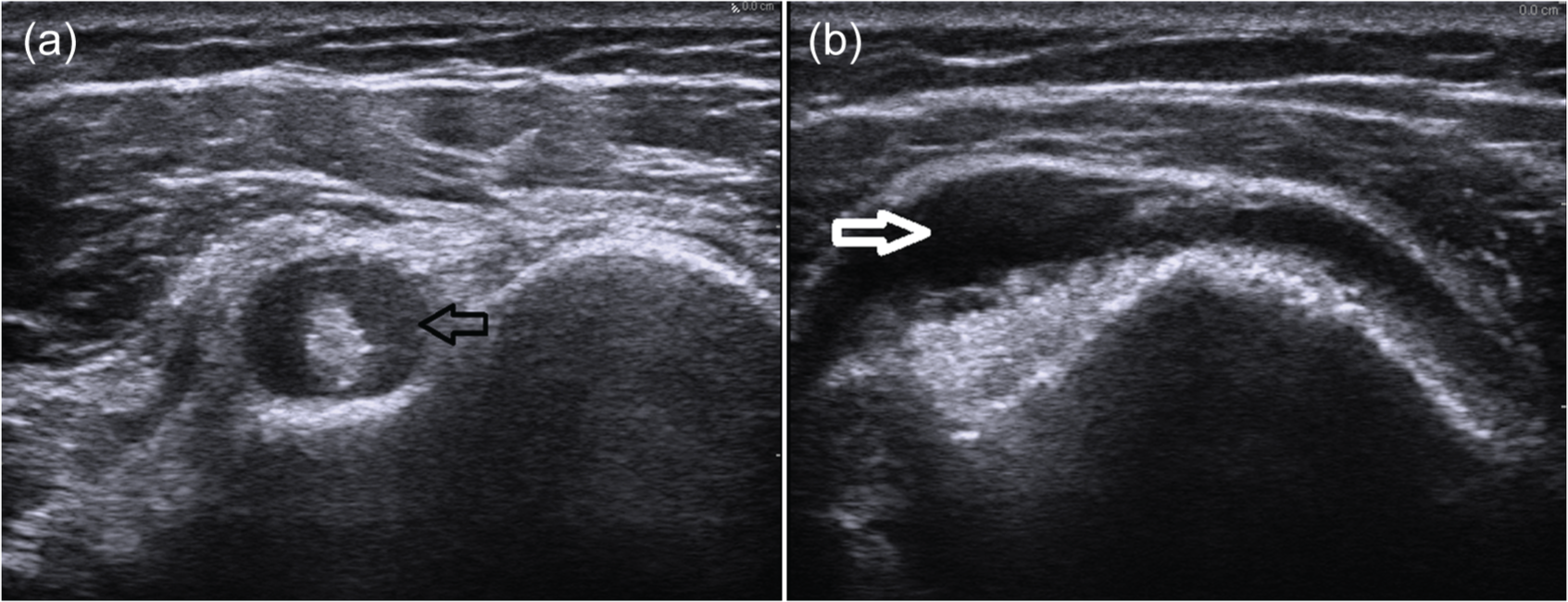
Ultrasonography of the shoulder in two patients with polymyalgia rheumatica showing bicipital tenosynovitis (short axis view) (a) and subdeltoid bursitis (b).
Patients with PMR also often report neck and back pain and imaging of these areas has also shown inflammatory changes. MRI of the cervical spine and lumbar spine in patients with newly diagnosed PMR showed evidence of cervical and lumbar interspinous bursitis [Salvarani et al. 2008a, 2013]. On positron emission tomography (PET) study, increased fluorodeoxyglucose (FDG) uptake in the shoulders, hips, and cervical and lumbar interspinous processes was observed in patients with PMR [Blockmans et al. 2007; Adams et al. 2012; Yamashita et al. 2012]. While it requires considerable expertise, ultrasound can also be useful in detecting typical spinous lesions in PMR.
Imaging of blood vessels in patients with PMR shows findings of vasculitis even in patients without signs and symptoms of GCA. In a prospective study of 102 patients with PMR and no clinical findings to suggest GCA, 8% had ultrasonographic findings on examination of the temporal arteries to suggest GCA [Schmidt and Gromnica-Ihle, 2002]. Temporal artery biopsy was positive in 50% of these patients with PMR in whom ultrasonography was abnormal [Schmidt and Gromnica-Ihle, 2002].
Patients with PMR may also have vascular uptake in other large vessels if imaged with sensitive modalities like PET scans, suggesting subclinical vessel inflammation (Figure 2) [Moosig et al. 2004; Blockmans et al. 2007; Kermani and Warrington, 2010]. In one study, approximately one-third of patients with isolated PMR were noted to have vascular FDG uptake (predominantly in the subclavian arteries) [Blockmans et al. 2007]. The significance of these incidentally noted abnormalities is presently unknown. PET is a costly modality and is rarely necessary in the routine evaluation of patients with PMR. Ultrasonography of the temporal arteries and large vessels may add to the diagnostic evaluation of patients with PMR.

Positron emission tomography in a patient with polymyalgia rheumatica showing fluorodeoxyglucose uptake in both shoulders but also in the large vessels, indicating concurrent, subclinical giant cell arteritis.
Biopsy
Tissue biopsies are not necessary in patients with PMR. Muscle biopsy would only be indicated in those suspected of having an inflammatory myositis based on clinical or laboratory evaluation. Temporal artery biopsy should be pursued in patients with PMR who have cranial symptoms or vascular abnormalities that may suggest GCA [Salvarani et al. 2008b].
Treatment
Glucocorticoids
Nonsteroidal anti-inflammatory medications are generally not recommended for treatment of PMR [Hernandez-Rodriguez et al. 2009]. GCs are the mainstay of therapy for patients with PMR. Low doses of GCs (prednisone or equivalent of 10–20 mg daily) are usually effective [Hernandez-Rodriguez et al. 2009; Dasgupta et al. 2010]. Higher doses should be initiated only in patients suspected of having GCA.
Clinicians often use the rapid resolution of symptoms to low doses of prednisone (≤20 mg) as a confirmation of the diagnosis. However, this approach has limitations. In a prospective study of 129 subjects with newly diagnosed PMR, 26% of patients still reported proximal myalgias 3 weeks after standard therapy and 29% still had over half an hour of morning stiffness after 3 weeks of treatment [Hutchings et al. 2007]. In the recent ACR/EULAR prospective study of 125 patients with PMR, 71% of patients met the definition for a complete response to GC therapy at 4 weeks [Dasgupta et al. 2012a, 2012b]. When there is no or only partial clinical response to GCs, or when markers of inflammation remain persistently elevated, other diagnoses should be considered.
There are no standardized protocols for tapering prednisone in PMR. The initial dose is usually maintained for 2–4 weeks. Subsequently, it has been recommended that prednisone be decreased by 2.5 mg every 2–4 weeks until the patient is at 10 mg daily following which the daily dose of prednisone is tapered by 1 mg a month [Hernandez-Rodriguez et al. 2009; Dasgupta et al. 2010].
In patients with multiple comorbidities, intramuscular methylprednisolone at an initial dose of 120 mg every 3–4 weeks followed by a reduction by 20 mg every 2–3 months can be used [Dasgupta et al. 2010]. In a double-blind, controlled trial intramuscular methylprednisolone was associated with similar remission rates but a lower mean cumulative GC dose and fewer GC-related adverse events than oral prednisone [Dasgupta et al. 1998].
More recent approaches with GC have been looking into targeted therapy. Chronotherapy, the coordination of the release of GC based on biological rhythms of cytokine concentrations, has been evaluated in RA. Two clinical trials evaluating delayed release (DR) or modified release prednisone have found these formulations to be effective in alleviating symptoms in patients with RA [Buttgereit et al. 2008, 2013]. In Europe the DR formulation is approved only for the treatment of RA, while in the United States it has also been approved by the Food and Drug Administration for other inflammatory conditions, including PMR [Buttgereit, 2012]. However, this medication is expensive. Additionally, whether there is any advantage of this formulation over short-acting GCs in terms of adverse event profile is unclear. Other approaches to optimize GCs through development of selective GC receptor agonists or more targeted delivery of GCs are some of the areas being actively investigated and may hopefully lead to safer more effective options [Buttgereit, 2012].
Other immunosuppressive medications
Relapses in PMR are common and most patients require prolonged GC therapy. The typical duration of treatment is 1–2 years and often longer [Gabriel et al. 1997; Maradit Kremers et al. 2007; Barraclough et al. 2008]. Prolonged GC exposure is associated with significant morbidity [Gabriel et al. 1997; Mazzantini et al. 2012]. Other alternatives to GC have been evaluated for treatment of PMR but results have been disappointing to date. Few randomized therapeutic clinical trials have been conducted in patients with PMR [De Silva and Hazleman, 1986; Ferraccioli et al. 1996; van der Veen et al. 1996; Caporali et al. 2004; Salvarani et al. 2007]. The efficacy of methotrexate for the initial treatment of PMR has been studied in three randomized clinical trials with mixed results [Ferraccioli et al. 1996; Caporali et al. 2004; van der Veen et al. 1996]. In clinical practice, methotrexate may be considered for the subset of patients with frequent relapses resulting in the inability to taper prednisone, or in patients with GC toxicity [Dasgupta et al. 2007; Hernandez-Rodriguez et al. 2009; Spies et al. 2010]. Azathioprine has also been evaluated in a small, randomized, double-blind, placebo-controlled trial and was found to have a steroid-sparing effect but the study had a high dropout rate and the results need to be interpreted with caution [De Silva and Hazleman, 1986]. In a multicenter, randomized, placebo-controlled trial in 40 patients with newly diagnosed PMR treated with infliximab and prednisone or placebo and prednisone, similar rates of relapses and recurrences were observed [Salvarani et al. 2007]. Other medications that have been reported to be useful in anecdotal cases and case series are leflunomide, anti-tumor necrosis factor medications, and most recently, an anti-IL-6 agent tocilizumab [Adizie et al. 2012; Aikawa et al. 2012; Unizony et al. 2012; Macchioni et al. 2013]. Therapies targeting cytokines IL-1, IL-6 and IL-17 are being actively evaluated for treatment of PMR (www.clinicaltrials.gov).
Prognosis
Despite therapy, relapses occur in approximately 50% of patients [Kremers et al. 2005a; Salvarani et al. 2005]. Several prospective studies have re-evaulated the final diagnosis in patients with PMR with between 2–30% of patients being diagnosed at a later date as having RA [Dasgupta et al. 1998; Caporali et al. 2001; Pease et al. 2005; Hutchings et al. 2007; Falsetti et al. 2011]. GC-related adverse events are common in patients with PMR [Gabriel et al. 1997; Hutchings et al. 2007; Mazzantini et al. 2012].
The risk of vascular disease (peripheral arterial disease, cardiovascular and cerebrovascular events) was increased in a systematic review of patients with PMR perhaps secondary to chronic inflammation or subclinical vasculitis [Hancock et al. 2012].
There are many reports of PMR in association with neoplasms, suggesting a paraneoplastic phenomenon. Several studies have evaluated this question. In a population-based study from Norway, malignancy risk was not increased in patients with PMR (also included GCA) compared with age- and gender-matched controls [Myklebust et al. 2002]. However, two database studies have suggested increased risk of cancer particularly in the first year of diagnosis. A database study from Sweden included 35,928 patients with GCA and PMR but this utilized the Swedish Hospital Discharge Register [Ji et al. 2010]. There was a marginal increase in the incidence of cancer in patients with GCA compared with the general population (standardized incidence ratio 1.19; 95% confidence interval 1.06–1.23) [Ji et al. 2010]. However, when evaluating malignancy risk in the first year after diagnosis of PMR or GCA, the standardized incidence ratio was 2.26 (95% confidence interval 2.10–2.42) [Ji et al. 2010]. Another study utilized the UK General Practice Research Database and evaluated cancer risk in 2877 patients with PMR compared with age- and sex-matched controls [Muller et al. 2013]. Over a median follow up of 7.8 years, 23.2% of patients with PMR developed cancer compared with 19.5% of controls [Muller et al. 2013]. The risk of malignancy was increased in patients with PMR during the first 6 months after diagnosis (hazard ratio 1.69; 95% confidence interval 1.18–2.42) [Muller et al. 2013]. Misdiagnosis may account for these findings and this highlights the importance of careful evaluation of patients presenting with polymyalgic symptoms to exclude other conditions. Another possibility is surveillance bias. Increased healthcare utilization has been reported in patients during the first year after diagnosis compared with the general population of age- and gender-matched controls [Kremers et al. 2005b]. Immune dysregulation which has been associated with increased risk of malignancy in other rheumatologic disease may also be at play. Caution should be used interpreting database studies since the diagnosis of PMR is not confirmed by chart review and the possibility of misclassification is a concern. One study included subjects with GCA in addition to PMR and used a hospital registry [Ji et al. 2010]. Overall survival in PMR is similar to or slightly better than the general population [Salvarani et al. 1995; Gran et al. 2001].
Conclusion
PMR is a common inflammatory condition affecting individuals aged 50 years and over. There is a close association between PMR and GCA. Given the absence of any diagnostic tests specific to PMR, careful consideration should be given to excluding other mimics, particularly SpA and RA. Ultrasonography of affected joints may complement the clinical evaluation for the diagnosis of PMR. Treatment consists of prolonged GC therapy, but relapses are common. Moreover, GCs are associated with significant morbidity. Evaluation of therapies targeting inflammatory cytokines as alternatives to GCs in PMR is underway.
Footnotes
Acknowledgements
We thank Dr Mihaela Taylor, Associate Professor of Medicine, Division of Rheumatology, University of California, Los Angeles for kindly providing us with the ultrasound images used in this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
We declare we have no conflict of interest.
