Abstract
Pannexin 1 (Panx1) is an ubiquitously expressed protein that forms plasma membrane channels permeable to anions and moderate-sized signaling molecules (e.g., ATP, glutamate). In the nervous system, activation of Panx1 channels has been extensively shown to contribute to distinct neurological disorders (epilepsy, chronic pain, migraine, neuroAIDS, etc.), but knowledge of the extent to which these channels have a physiological role remains restricted to three studies supporting their involvement in hippocampus dependent learning. Given that Panx1 channels may provide an important mechanism for activity-dependent neuron-glia interaction, we used Panx1 transgenic mice with global and cell-type specific deletions of Panx1 to interrogate their participation in working and reference memory. Using the eight-arm radial maze, we show that long-term spatial reference memory, but not spatial working memory, is deficient in Panx1-null mice and that both astrocyte and neuronal Panx1 contribute to the consolidation of long-term spatial memory. Field potential recordings in hippocampal slices of Panx1-null mice revealed an attenuation of both long-term potentiation (LTP) of synaptic strength and long-term depression (LTD) at Schaffer collateral-CA1 synapses without alterations of basal synaptic transmission or pre-synaptic paired-pulse facilitation. Our results implicate both neuronal and astrocyte Panx1 channels as critical players for the development and maintenance of long-term spatial reference memory in mice.
Introduction
Pannexins (Panxs) are a group of proteins that belong to the gap junction family but do not form gap junction channels. Of the three pannexins (Panx1, Panx2, and Panx3), Panx1 is the best characterized, with studies indicating that this protein forms plasma membrane channels that are ubiquitously expressed in vertebrate tissues (Bruzzone et al., 2003; Hanstein et al., 2013). In the central nervous system (CNS), Panx1 is present in glia and neurons (Ray et al., 2005; Rigato et al., 2012; Santiago et al., 2011; Seo et al., 2020; Vogt et al., 2005). Panx1 channels are permeable to anions and larger negatively charged molecules such as glutamate and ATP (Bao et al., 2004; Ma et al., 2009) and are activated by voltage, caspases (Sandilos et al., 2012), and following membrane receptor activation (purinergic P2 receptors, glutamate receptors, alpha-adrenergic receptors, thromboxane receptors (Billaud et al., 2011; da Silva-Souza et al., 2014; Locovei et al., 2006; Locovei et al., 2007; Pelegrin & Surprenant, 2006; Weilinger et al., 2012). Despite the strong evidence in support of the participation of Panx1 channels in neurological disorders (Burma et al., 2017; Cisneros-Mejorado et al., 2015; Dossi et al., 2018; Karatas et al., 2013; Lutz et al., 2013; Santiago et al., 2011; Thompson et al., 2006; Velasquez et al., 2016; Weaver et al., 2017), little is known about their physiological role in the CNS. Few studies have indicated that these channels have a physiological role during neurodevelopment (Wicki-Stordeur et al., 2012; Wicki-Stordeur et al., 2016) or in the regulation of synaptic plasticity involved in learning and memory (Ardiles et al., 2014; Gajardo et al., 2018; Prochnow et al., 2012).
Evidence that Panx1 contributes to learning and memory comes from studies performed on transgenic mice showing that global deletion of Panx1 impaired spatial learning, novel object recognition, and spatial reversal learning (Gajardo et al., 2018; Prochnow et al., 2012). These memory-related behaviors, which are dependent on hippocampal synaptic plasticity (both long-term potentiation and depression: LTP and LTD), are altered in mice lacking Panx1 (Ardiles et al., 2014; Gajardo et al., 2018; Prochnow et al., 2012). These two major forms of synaptic plasticity require, at least in part, glutamate binding to the N-methyl-D-aspartate receptor (NMDAR) (Malenka & Bear, 2004). LTP is induced by strong stimuli leading to large influx of calcium through NMDAR and activation of CaMKII, leading to persistent enhancement of synaptic strength, while weaker stimuli are believed to lead to LTD by driving smaller flux of calcium through NMDAR, resulting in preferential activation of protein phosphatases leading to long-term reductions in synaptic strength (Lisman, 1989; Malenka, 1994). NMDARs have been linked to Panx1 activation via ionotropic and metabotropic NMDAR signaling (Bialecki et al., 2020; Patil et al., 2022). The activation of Panx1 via ionotropic calcium influx-dependent NMDAR signaling relies on the interaction of the Ca2+-sensor stromal interaction molecules (STIM1/2) with the N-terminus of Panx1 (Patil et al., 2022), while Panx1 activation via NMDAR metabotropic signaling relies on Src kinase recruitment and phosphorylation of Panx1 C-terminal domain (Weilinger et al., 2012; Weilinger et al., 2016). Based on these reports and on studies showing that the calcium sensor STIM1/2 is important for LTP (Garcia-Alvarez et al., 2015), and those showing that the metabotropic NMDAR signaling contributes to LTD (Nabavi et al., 2013; Stein et al., 2015; Stein et al., 2020), it is likely that Panx1 contributes to both forms of synaptic plasticity, via NMDAR-STIM1/2 and NMDAR-Src, respectively.
Given that Panx1 in the CNS is expressed in astrocytes and neurons (Santiago et al., 2011; Zoidl et al., 2007 ) and that astrocytes participate in the induction of long-term synaptic plasticity through the release of gliotransmitters, including glutamate, ATP/adenosine, and D-serine (reviewed in Durkee et al., 2021; Perea et al., 2009), we investigated the extent to which memory deficits reported in mice with global deletion of Panx1 can be attributed to either astrocyte or neuronal Panx1. Using behavioral tests and electrophysiological recordings from Panx1 transgenic mice, we found that Panx1 does not participate in locomotor activity or anxiety-like behavior, but that it does contribute to long-term reference memory formation. Electrophysiological recordings of hippocampal slices confirmed that impairments in long-term synaptic plasticity in Panx1-null mice reduced the magnitude of both LTP and LTD, compared to wild-type control mice. Because deletion of neuronal or astrocytic Panx1 induced similar impairments of long-term reference memory as seen in the global Panx1 knockout, we concluded that both astrocyte and neuronal Panx1 are necessary for the acquisition/consolidation of long-term spatial memory.
Materials and Methods
Animals
Panx1f/f mice were generated in our facility from crosses between Panx1tm1a(KOMP)Wtsi (Panx1-null; obtained from KOMP; RRID:IMSR_KOMP:CSD66379-1a-Wtsi) and a flippase deleter mouse (B6.ACTFLPe/J; RRID:MGI:5014383) in the C57BL/6 background. For astrocyte and neuronal deletion of Panx1, mGFAP-Cre (B6.Cg-Tg(Gfap-cre)73.12 Mvs/J; RRID:MGI:4829613) in the C57BL/6 background and mNFH-Cre (Tg(Nefh-cre)12 Kul/J; RRID:MGI:3043822) mice in a mixed (129/Sv * 129S7/SvEvBrd * C57BL/6 * FVB) background purchased from the Jackson Laboratory were crossed with Panx1f/f to generate mGFAP-Cre:Panx1f/f and mNFH-Cre:Panx1f/f mice and maintained in our animal facility. These transgenic mice were previously described and characterized, as follows: For the global Panx1 KO, we showed Panx1 deficiency by immunostaining and qPCR (Hanstein et al., 2013: Figures 2 and 3; Santiago et al., 2011: Figure 1; Velasquez et al., 2016: Figure 6), by western blots (Cone et al., 2013: Figure 3; Negoro et al., 2013: Figure 2), and by functional assays (Lutz et al., 2013: Figure 4; Santiago et al., 2011: Figures 2 and 4). For the NFH-Cre:Panx1f/f and GFAP-Cre:Panx1f/f, we showed the lack of Panx1 expression in these mice by immunostaining (Hanstein et al., 2013: Figure 6; Hanstein et al., 2016: Supplemental Figure 5S) and by functional assays (Scemes et al., 2019: Figure 3 and Supplemental Figure S1). Additional evidence for changes in Panx1 expression levels in brains of mice with cell-type specific deletion of Panx1 is here provided using Western blot analysis (Supplemental Figure 1S). To avoid ectopic expression of Cre in the germlines of Panx1f/f derived from GFAP-Cre or NFH-Cre crosses (Hanstein et al., 2013), we maintain Panx1f/f mice and Cre-expressing mice as separated colonies and did not use Panx1f/f littermates derived from crosses of Cre expressing mice in our experiments. Animals were housed in individually ventilated cages containing three to four males or females. Mice were located in a single room and maintained under specific pathogen-free conditions in the Animal Resource Facilities of New York Medical College and all experiments were pre-approved by the Animal Care and Use Committee.
Behavioral Studies
For behavioral experiments, 2-month-old male and female mice were maintained in a light/dark cycle (light on: 7:00 AM, light off: 7:00 PM) and experiments were performed during the light cycle at a time particular to each test as described below. Animals had free access to water and chow, except for 2 days prior to the radial maze testing when mice received a restricted diet (see below). Sex as a biological variable was assessed only in Panx1f/f and Panx1 KO. As two-way ANOVA showed that neither the sex nor interactions of sex with the experimental groups had any statistically significant effect (Supplemental Figures 2S and 3S), data from male and female mice were combined for behavioral analysis.
Open Field
To determine general locomotor activity levels and thigmotaxis (tendency to remain close to the walls, and a proxy of anxiety-like behavior), we used the open field and behavioral testing performed during 9:00 AM to 12:00 PM. Animals were placed for 10 min in an arena (43.5 cm × 43.5 cm) with infra-red sensors attached to a computer. Mouse activity (total distance traveled and time spent in inner and outer zones) was recorded using Activities® software. The number of fecal boluses was determined after each mouse completed the open field test.
Eight Arm Radial Maze
To investigate the spatial orientation and memory performance in mice, we used an automated eight arm radial maze (Ugo Basile) with central circular area (16 cm) and eight arms (36 cm × 5 cm × 15 cm) that could be accessed by automated sliding doors. The maze was operated via ANY-maze video tracking software interface. The radial maze was placed on a platform (90 cm across and 38 cm on each side) which was placed directly on the floor of the testing room. External visual cues included marks on the walls near the maze, and furniture. All cues remained in the same position for all trials. Behavioral testing occurred during 1:00–5:00 PM. Two days before the start of the experiment, mice were put on a restricted diet and their weight were maintained at 80–85% of original weight throughout the experiment. The behavioral test consisted of three sessions: (1) one-day habituation session followed by (2) 13 daily training sessions, followed by (3) a single test session 24 h later. During habituation, mice were placed in the middle of the maze with access to all eight baited arms for 15 min or until the mice ate all the baits (chocolate cheerios). The baits were placed at the far end of each arm. For the training and test sessions, different sets of four baited arms were designated for each mouse and remained constant for any single mouse over the 2 weeks of training. Daily training sessions consisted of two phases, a forced and a choice phase, and lasted for 13 consecutive days. The forced phase of the training session consisted of placing the mouse on the central area for 5 s before the four doors of the baited arms were simultaneously opened. After the mouse visited a baited arm, the door was closed; the forced training ended when the mouse ate all four baits which occurred within the 15 min forced training. The choice phase, performed 40 s after the forced phase, consisted of placing the mouse on the central area and allowing it to enter any of the eight (four baited and four non-baited) arms. The choice phase lasted for 10 min. Twenty-four hours after the last training day, a single test session was performed. The test session was similar to the training session, but without the forced phase. The number of working and reference memory errors (see below) and the number of correct choices (number of first entries into baited arms) were recorded during the training and test sessions. Reference memory errors (RME) consisted in the first entry into a non-baited arm, while working memory errors (WME) consisted of repeated entries into previously visited arms. WME was further divided into correct (WM-C: re-entries into baited arms that have been previously visited) and incorrect (WM-I: re-entries into un-baited arms) errors, as described by others (Jarrard et al., 1984; Pirchl et al., 2010; Yan et al., 2004). For analysis, the number of WM-C, WM-I, RME and of correct choices obtained during the training and test sessions obtained for each mouse genotype were normalized to their respective mean value obtained at the first training day (day 1).
Hippocampal Slice Electrophysiology
To determine the extent to which synaptic neurotransmission is modified by the genetic deletion of Panx1, electrophysiological field potential recordings from Schaffer collateral-CA1 synapses in hippocampal slices were performed, as previously described (Sullivan et al., 2018). Two-month-old Panx1f/f and Panx1-null male and female-naïve mice (not subjected to behavioral tests) were deeply anesthetized with isoflurane, and their brains were quickly removed and submerged in ice-cold (2–4 °C) sucrose-containing artificial cerebrospinal fluid (ACSF) (in mM): NaCl 87, NaHCO3 25, NaH2PO4 1.25, KCl 2.5, CaCl2 0.5, MgCl2 7, D-glucose 25 and sucrose 75, saturated with 95% O2/5% CO2. Coronal slices (350 μm) were obtained using a vibratome (Leica VT1000 S), and then placed in a holding chamber containing the same sucrose-containing ACSF for at least 30 min before being transferred to another holding chamber containing recording ACSF (in mM: NaCl 126, NaHCO3 26, NaH2PO4 1.25, KCl 3, CaCl2 2.5, MgCl2 2, and D-glucose 10) for another 30 min. Once transferred to an interphase-type recording chamber, slices were continuously perfused with recording ACSF maintained at 32 °C. A borosilicate-glass recording electrode filled with recording ACSF (1–2 MΩ; Model P-97, Sutter Instruments, Novato, CA, USA) was placed in the stratum radiatum area to record field excitatory postsynaptic potentials (fEPSPs) from distal dendritic arbors of CA1 pyramidal neurons. Evoked fEPSPs were generated by stimulating the Schaffer collaterals with pulses delivered using a bipolar stainless-steel stimulating electrode placed approximately 200-300 µm from the recording electrode, in the Schaffer collaterals/commissural fibers of stratum radiatum. Electrical stimulation was delivered by an ISO-Flex isolator controlled by a Master 8 pulse generator (AMPI, Jerusalem, Israel) and triggered and recorded with a MultiClamp 700B amplifier (Molecular Devices, San Jose, CA, USA). Prior to running experimental protocols, fEPSP slopes were recorded for a 15-min baseline. Slices exhibiting drifts larger than ±10% during baseline recording were considered not suitable and excluded from experimentation. One to two hippocampal slices per animal were used.
Basal Synaptic Transmission
To evaluate for changes in basic synaptic transmission caused by deletion of Panx1, we assessed neuronal excitability and synaptic transmission by measuring the input-output (I/O) relationship and pair-pulse facilitation (PPF), respectively. I/O relationship was determined from field excitatory post-synaptic potentials (fEPSPs) generated at the CA1 stratum radiatum evoked by gradually increasing the stimulus intensity (20 µA to 200 µA; 100 µs duration; 20 s intervals) applied at the Schaffer collaterals; the input was considered as being the stimulus intensity and the output was the initial slope of fEPSPs. PPF was measured over five inter-pulse intervals of 10, 20, 50, 100, and 200 ms with intensity (50–100 µA) adjusted to evoke ∼50% of the maximal fEPSP. The ratio of the amplitude of the second to the first fEPSP, averaged over five sweeps per interval, was used to characterize PPF ratio profiles.
Long-term potentiation (LTP) and long-term depression (LTD): A 15-min baseline was recorded prior to experimental induction of LTP or LTD, with the stimulus intensity of the current pulses (50 µA to 100 µA; 100 µs duration; 20 s intervals) adjusted to evoke ∼50% of the maximal fEPSP. The high-frequency theta burst stimulus (TBS) paradigm for induction of LTP consisted of three theta burst trains separated by 20 s. Each train consisted of 10 bursts, with five pulses per burst, at a burst frequency of 100 Hz and inter-burst interval of 200 ms. LTD was induced using low frequency stimulation (LFS: 2 Hz/10 min). The slopes of fEPSPs were measured by linear interpolation from 20% to 80% of maximum negative deflection (SciWorks Software, DataWave Technologies, Parsippany, NJ, USA). The magnitude of LTP and LTD was calculated as the average of the fEPSP responses recorded 50–60 min after conditioning stimulation compared to the pre-conditioning 15-min baseline.
Statistical Analyses
Data were analyzed and graphed using GraphPad Prism (9.4.1). The Shapiro-Wilk test was used to assess whether non-categorical data came from a normally distributed population prior to further analyses. Non-Gaussian distributed data were analyzed using the non-parametric Kruskal-Wallis's test followed by Dunn's multi-comparison test, and the normally distributed data analyzed using parametric test (ANOVA) when homoscedasticity was verified by visual inspection of plots of predicted Y values versus the absolute value of residuals. In the latter case, one-way and two-way ANOVA without/with repeated measures (RM) followed by Šidák's multiple comparison tests were used for data analyses when appropriate. For RM ANOVA, we used the Geisser-Greenhouse correction ε to adjust for potential violation of sphericity assumption (equal variance of independent variables). For analysis of LTP and LTD data, unpaired Student t-test was used.
Results
Panx1 Does Not Influence General Locomotor Activity or Thigmotaxis
To evaluate the overall locomotor activity and thigmotaxis (as a proxy for anxiety-like behavior) of mice with global and cell-type-specific deletion of Panx1, the total distance traveled, and the percent time spent in outer and inner zones of the open field arena were measured. Given that no significant sex differences in terms of thigmotaxis were detected in Panx1f/f or Panx1-null mice (Supplemental Figure 2S), subsequent behavioral analysis was performed by grouping male and female mice. There was no significant difference in total distance traveled during the 10 min test between the four mouse genotypes (Figure 1A; Kruskal-Wallis test, p = 0.8313). Figure 1B shows the percent time that the animals spent in the outer zone; mice of all genotypes spent 92 to 96% time in the outer zone (Kruskal-Wallis test, p = 0.4739). These results show that Panx1 does not affect overall locomotor activity and does not contribute to anxiety-like behavior as measured by thigmotaxis (time spent in the outer zone).
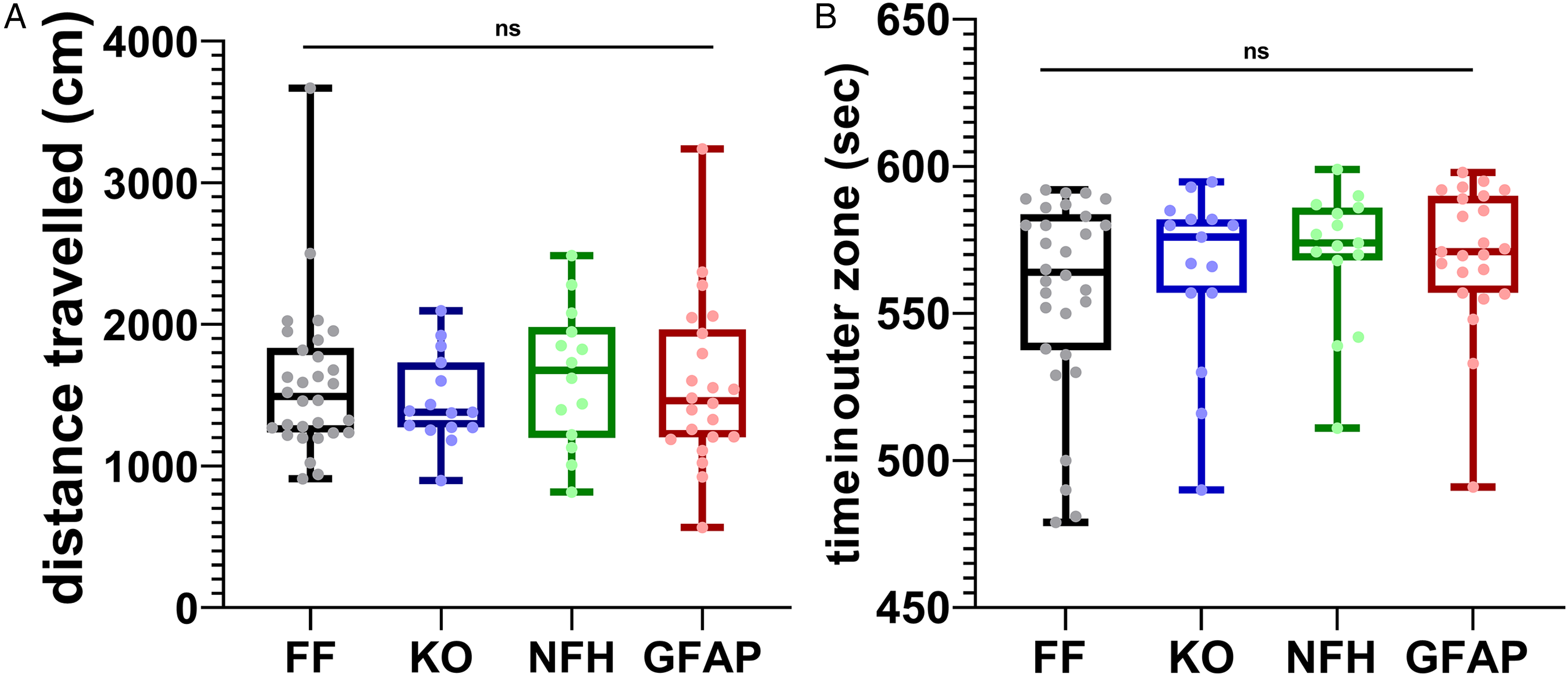
Locomotor activity and thigmotaxis are independent of Panx1. Median ± min/max values of the (A) total distance traveled and (B) time spent in the outer zone of the arena recorded from Panx1f/f (FF, black bars; n = 30), Panx1-null (KO, blue bars; n = 15), NFH-Cre:Panx1f/f (NFH, green bars; n = 14), and GFAP-Cre:Panx1f/f (GFAP, red bars; n = 23) mice exposed for 10 min to the open field.
Long-Term Spatial Reference Memory is Impaired in Mice Deficient in Panx1
The contribution of Panx1 to spatial memory was assessed using the eight-arm radial maze that allows for the simultaneous evaluation of working and reference memories. Spatial working memory is a short-term storage of task-relevant information to guide the ongoing and upcoming behavior, that is, a temporally based representation of a spatial location that is used within a single trial of an experimental session to guide behavior. Spatial reference memory is a form of long-term memory representing the spatial, contextual, and factual aspects of a task that remains constant between trials, thus it is the representation of an association between spatial locations that remains consistent across several trials of an experimental session and is used to guide behavior.
Working and reference memory errors in the eight-arm radial maze were measured daily over 13 days during the choice phase of the training sessions (see Methods). As with thigmotaxis (Supplemental Figure 2S), no sex differences were detected in terms of working memory errors (WME) and reference memory errors (RME) in Panx1f/f or Panx1-null mice (Supplemental Figure 3S). In terms of working memory, no significant differences were detected among the genotypes during the training and test sessions (Figure 2; Supplemental Figure 4S). Figure 2A and C shows that during the 13 days’ training sessions, the fraction of working memory correct (WM-C) and working memory incorrect (WM-I) errors decreased significantly over time for each mouse genotype (repeated measure ANOVA; Ftime (12, 660) = 4.396, p < 0.0001, and Ftime (12, 660) = 4.537, p < 0.0001, respectively), indicating that mice learned the task by reducing the number of errors. In addition, during the training sessions, no significant differences in terms of WM-C and WM-I were detected between genotypes (Figure 2A and C; Fgenotype (3, 55) = 0.9708, p = 0.4132, and Fgenotype (3, 55) = 2.290, p = 0.0885, respectively). In terms of absolute number of WM errors, the number of WM-I measured for each mouse genotype was higher than the number of WM-C (Supplemental Figure 5S). At day 14 (test day), WM-C and WM-I did not differ significantly among the genotypes (Figure 2B and D; Kruskal-Wallis test, p = 0.2940, and p = 0.2598, respectively). Thus, these results show that independently of the genotype, mice improved their working memory performance over time and that Panx1 does not play a role, at least in this form of learning.
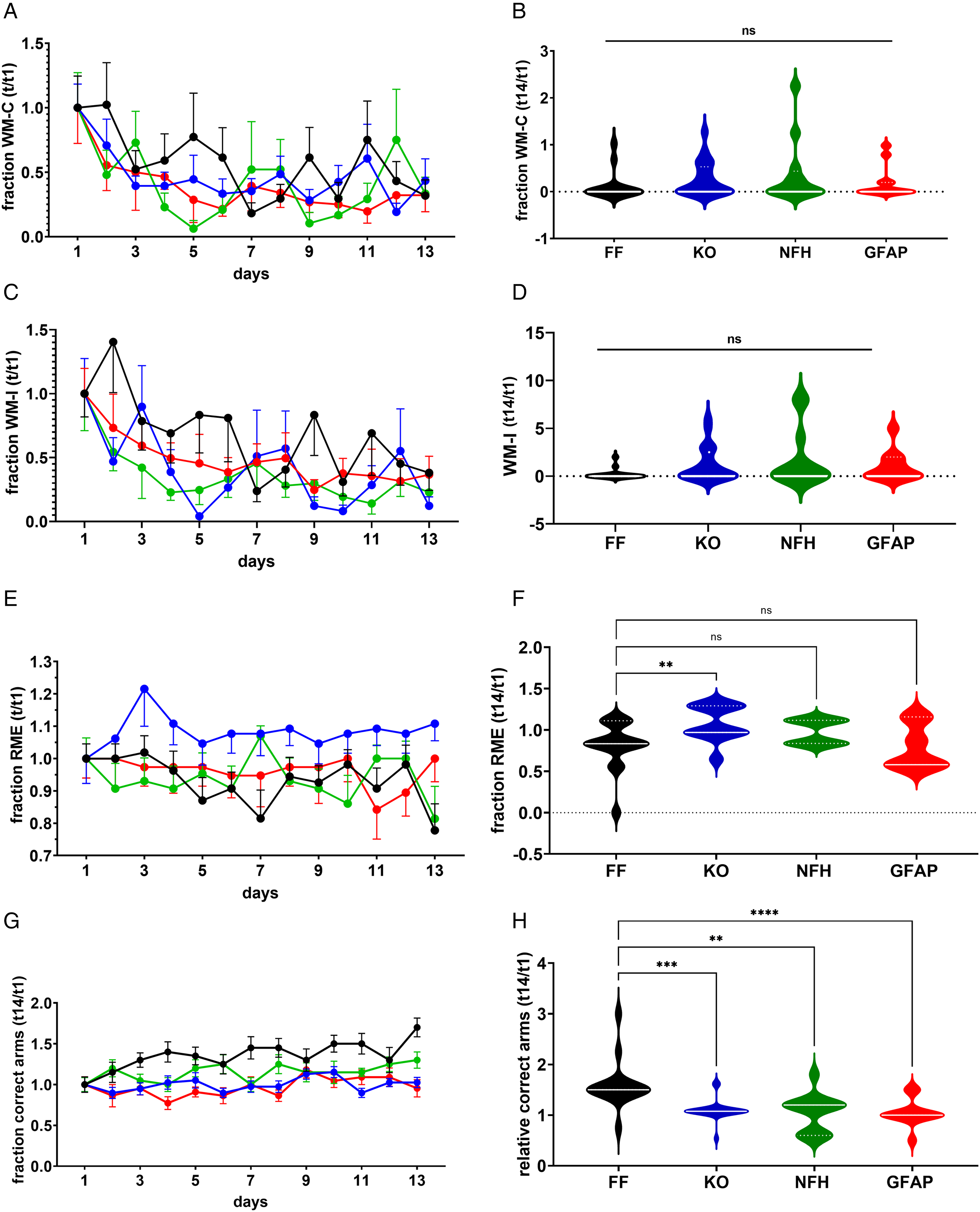
Impaired long-term reference memory in mice lacking Panx1. (A, C, E, G) Mean ± sem of the (A) fraction of working memory correct errors (WM-C), (C) working memory incorrect errors (WM-I), (E) reference memory errors (RME), and (G) correct choices (correct arms) obtained during the training sessions. (B, D, F, H) Violin plots showing the median values of WM-C (B), WM-I (D), RME (F), correct arms (H) obtained during the test phase for Panx1f/f (FF, black symbols; n = 15), Panx1-null (KO, blue symbols; n = 21), NFH-Cre:Panx1f/f (NFH, green symbols; n = 12), and GFAP-Cre:Panx1f/f (GFAP, red symbols; n = 11) mice.
Differently from WME, the fraction of RME recorded during the training sessions did not vary overtime within each genotype (two-way ANOVA Ftime (12, 660) = 0.5277, p = 0.8975), indicating that mice did not significantly improve their performance during the training phase (the number of errors did not change; see Supplemental Figure 4S). However, there was a significant difference in RME among the genotypes (Figure 2E; repeated measures ANOVA: Fgenotype (3, 55) = 11.27, p < 0.0001), particularly between Panx1f/f and Panx1-null mice (Šidák's multi-comparison test: p < 0.0001) that was maintained during the test session (Figure 2F; Panx1f/f: 0.8 folds, Panx1-null: 1.1 folds; Kruskal-Wallis test followed by Dunn's multiple comparison test: p = 0.0009).
In terms of fraction of correct (baited) arms first visited, significant differences were recorded during the 13 days of training sessions (Figure 2G; repeated measures ANOVA: Ftime (12, 660) = 2.253, p = 0.0085) and among the genotypes (Fgenotype (3, 55) = 56.44, p < 0.0001). At day 14 (test session), a significant deficit in long-term reference memory, in terms of correct entries into baited arms, was detected in the three genotypes that were Panx1 deficient (Figure 2H). The fold changes (mean ± sem) in the number of correct entries relative to day 1 recorded for Panx1-null (1.10 ± 0.05 fold, n = 20), NFH-Cre:Panx1f/f (1.05 ± 0.11 fold, n = 12), and GFAP-Cre:Panx1f/f (1.05 ± 0.08 fold, n = 11) mice were significantly lower than those recorded in Panx1f/f mice (1.60 ± 0.12 fold, n = 15) (Figure 2H; Kruskal-Wallis test followed by Dunn's test; p < 0.01). No significant difference in terms of fraction of correct entries was detected between Panx1-null, NFH-Cre:Panx1f/f and GFAP-Cre:Panx1f/f mice during the test session (Dunn's test, p > 0.55).
Thus, these results show that Panx1 contributes to long-term reference memory and that deletion of Panx1 from either astrocytes or neurons results in similar memory deficits, at least in terms of correct choices, as seen in the global Panx1-null mice.
Panx1 Does not Contribute to Basal Synaptic Transmission
To assess potential changes in excitatory synaptic transmission and cell excitability that could underlie the altered behavior in Panx1-deficient mice, we recorded fEPSPs in response to different stimulation intensities in the apical dendritic region of stratum radiatum of field CA1 in hippocampal slices from young adult (2-month-old) male and female Panx1-null and Panx1f/f mice. Given that no sex differences were detected when performing behavioral studies (see above), electrophysiological data derived from slices of female and male mice were pooled together. As shown in Figure 3A, no significant differences in fEPSP slopes as a function of stimulus intensity (20–200 µA) were detected between the two genotypes (p > 0.93; two-way RM ANOVA with Geisser-Greenhouse correction, followed by Šídák's multiple comparisons test), indicating no differences in excitatory synaptic transmission.
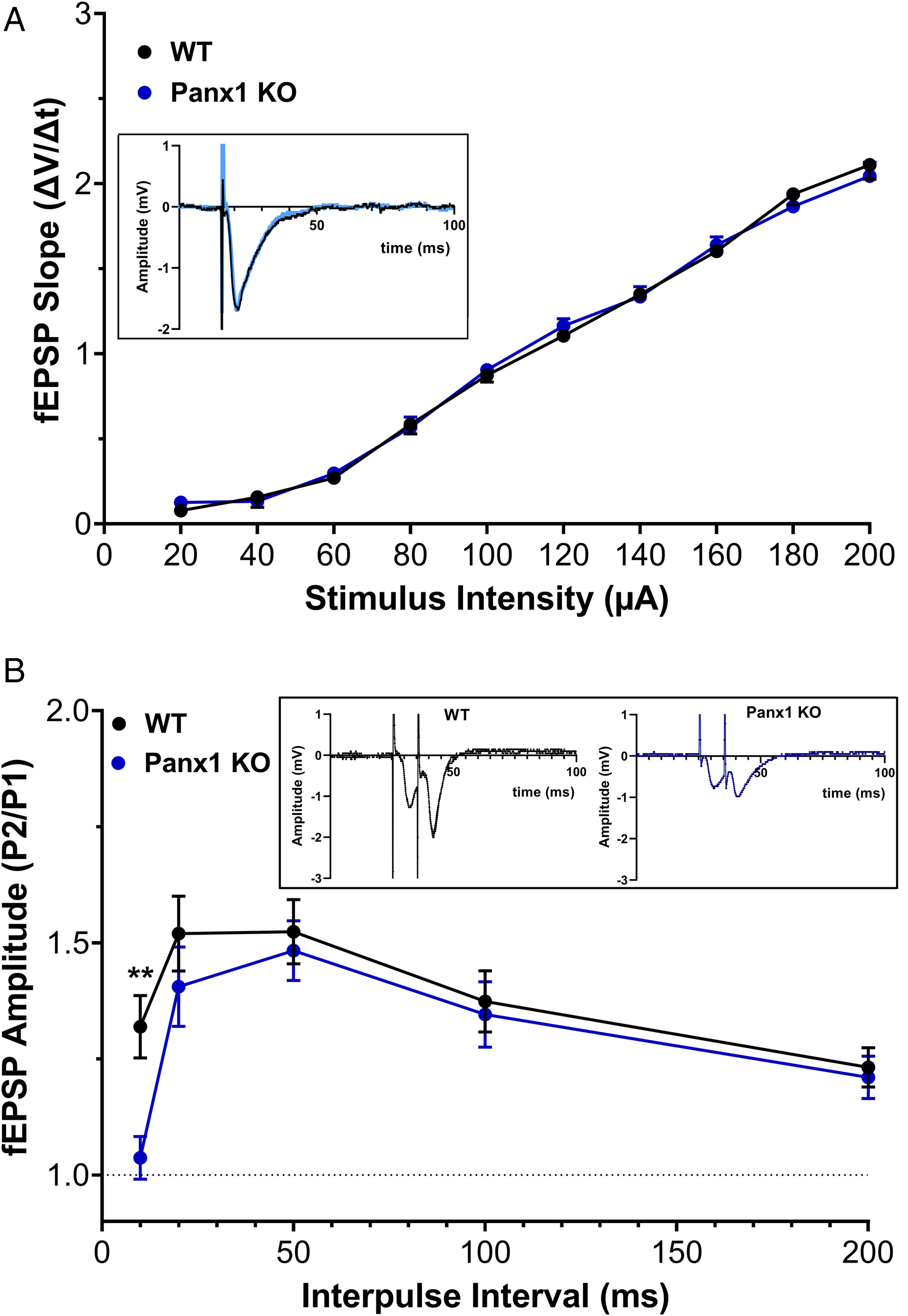
Basal synaptic transmission in Panx1-deficient mice. (A) Input–output (I/O) relations showing normalized fEPSP slopes at different stimulus intensities at CA3-CA1 synapses of hippocampal slices from control (WT) versus Panx1-null (Panx1 KO) mice. No significant difference in basal transmission was detected (two-way RM ANOVA: Fgenotype (1, 40) = 0.001400, p = 0.9703). (B) Paired-pulse facilitation ratio of the fEPSP at various inter-stimulus intervals. Significant difference was detected between WT and Panx1 KO mice but only at the 10 ms inter-pulse interval (p = 0.007; two-way RM ANOVA followed by Šidák's multiple comparison test).
To further characterize excitatory synaptic transmission in the hippocampal CA1 apical dendritic region in stratum radiatum, we measured paired-pulse facilitation (PPF; Andersen, 1960), a short-lasting form of synaptic plasticity primarily attributed to changes in presynaptic Ca2+ homeostasis (Zucker and Regehr, 2002), with 10–200 ms inter-pulse stimulus intervals. Statistical analysis (two-way RM ANOVA with Geisser-Greenhouse correction) of PPF ratio did reveal a significant difference between the groups (Finteraction (4, 144) = 0.9094, p = 0.4603; Fgenotype (1, 36) = 3.704, p = 0.0622; Finterpulse interval (3.028, 109) = 6.212, p = 0.006). Post-hoc analysis indicated that only at 10 ms interval the PPF ratio was significantly lower in KO than in WT slices (p = 0.009, Šídák's multiple comparisons test; Figure 3B). This result indicates that Panx1 deletion affects neurotransmitter release probability.
Panx1 Contributes to Hippocampal Synaptic Plasticity
Since deficits in long-term reference memory in spatial tasks such as the radial maze are often associated with impairments in hippocampal long-term activity-dependent synaptic plasticity (Martin et al., 2000), we sought to investigate whether the spatial learning deficits observed in Panx1 deficient mice (Figure 2) were correlated with alterations in hippocampal long-term synaptic plasticity. To that end, we compared LTP and LTD at the Schaffer collateral-CA1 synapses in hippocampal slices of Panx1-null and Panx1f/f mice. In slices from Panx1-null mice, the magnitude of TBS-induced LTP was significantly decreased compared to hippocampal slices from Panx1f/f mice (Student's t-test for unpaired data, p < 0.0001; Figure 4A). Similar to LTP, slices from Panx1-null mice also showed impaired LTD induced by low-frequency (2 Hz/10 min) stimulation when compared to Panx1f/f mice (two tailled Student's t-test for unpaired data, p < 0.0001; Figure 4B). There was no significant difference in 50% maximal fEPSP amplitudes recorded between the genotypes (Panxf/f: 0.7271 ± 0.1221 mV, N = 29 slices; Panx1-null: 0.6926 ± 0.0702 mV, N = 13 slices; Student's t-test, p = 0.81).
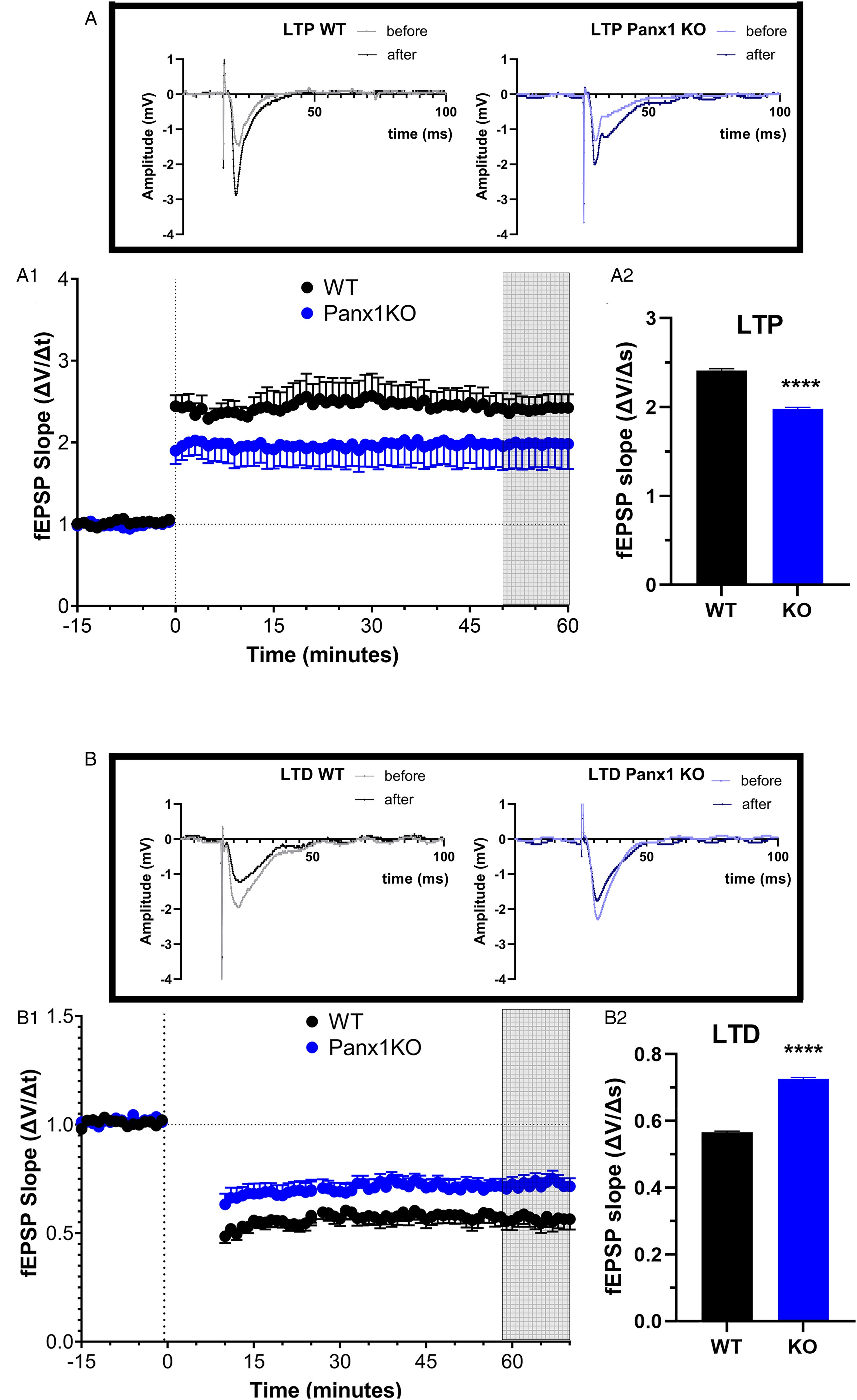
Altered hippocampal synaptic plasticity in Panx1-deficient mice. (A-B) Time courses (A1, B1) and magnitudes (A2, B2) of long-term potentiation (LTP) and of long-term depression (LTD). LTP was elicited by theta burst (TBS) and LTD by low frequency (LFS) stimulation of hippocampal Schaffer collateral-CA1 synapses. Panx1-null mice (blue symbols, n = 17 slices from six male and six female mice) expressed significantly lower LTP compared to Panx1f/f mice (black symbols, n = 13 slices from five male and four female mice) (two tailled Student's t-test: t = 55.69, df = 20, p < 0.0001), and Panx1-null (blue symbols, n = 15 slices from six male and six female mice) exhibited diminished LTD compared to Panx1f/f mice (black symbols, n = 12 slices from four male and four female mice) (two-tailled Student's t-test: t = 29.58, df = 20, p < 0.0001). Each data point was normalized to the averaged baseline and expressed as mean ± sem. The first set of vertical dashed lines indicate application of TBS and LFS, respectively, and the second set indicates the 50th–60th minutes post-stimulation period used for LTP and LTD analyses.
These data suggest that Panx1 is a significant contributor that modulates the magnitude of LTP and LTD in young adult mice, a role that is correlated with the necessity for Panx1 for consolidation of long-term spatial reference memory in a hippocampus-dependent task.
Discussion
The present study shows that long-term spatial reference memory, but not spatial working memory, is deficient in young adult mice lacking Panx1 and that both astrocyte and neuronal Panx1 contribute to sustain long-term spatial memory. Deficits in hippocampal dependent memory in Panx1-null mice were associated with decreased LTP and LTD of synaptic transmission at Schaffer collateral-CA1 synapses. These impairments were not related to altered neuronal excitability as indicated by the similar input-output relations between the two genotypes, but related to presynaptic mechanisms involved in neurotransmitter release probability, as the PPF ratio obtained for the shortest inter-pulse interval (10 ms) was decreased in the KO compared to wild-type slices. This is in contrast with a previous report showing that Panx1 deletion did not affect PPF ratio (Ardiles et al., 2014). This discrepancy is likely related to the inter-pulse intervals used (50–500 ms), which were longer than the ones (10–200 ms) tested in the present study. Indeed, we did not detect any significant difference in PPF ratio between the two genotypes for inter-pulse intervals above 10 ms.
The hippocampus plays crucial roles in encoding and consolidating spatial memory (Morris et al., 1982; Squire, 1992). Activity-dependent plasticity of hippocampal glutamatergic synapses, particularly NMDAR-dependent LTP and LTD, has been proposed as necessary cellular substrates for fulfilling these cognitive functions (Bliss and Collingridge, 1993; Ge et al., 2010; Martin and Shapiro, 2000).
The physiological contribution of Panx1 channels to synaptic plasticity, particularly to LTP and LTD, has been previously reported in older mice with global deletion of Panx1 (Ardiles et al., 2014; Gajardo et al., 2018; Prochnow et al., 2012). These studies showed that in adult (6–12-month old) mice, global deletion of Panx1 resulted in increased LTP and reduced LTD at Schaffer collateral-CA1 synapses, compared to age-matched wild-type mice (Ardiles et al., 2014; Gajardo et al., 2018). However, peripubertal Panx1-null mice (1 month old) did not display alterations in LTP and LTD (Ardiles et al., 2014). In contrast to these previous reports, our results show that both forms of long-term, activity-dependent synaptic plasticity at CA3-CA1 synapses in hippocampus are attenuated in 2 months old Panx1-null mice. Whether these differences are age-related and/or due to the distinct genomic fabrics between the different Panx1 transgenic lines used (current study: Panx1tm1a(KOMP)Wtsi—UCDavis KOMP repository; Prochnow et al., 2012: (Panx1Vsh—Romanov et al., 2012), and Ardiles et al., 2014: Panx1−/−—(Anselmi et al., 2008; Bargiotas et al., 2012)) needs further investigation. Given that aberrant LTP and LTD have been associated with age-related cognitive decline (Bach et al., 1999; Barnes, 1979; Burke and Barnes, 2006) and that the expression of LTP-specific genes and of Panx1 declines during aging (Ardiles et al., 2014; Ryan et al., 2015; Vogt et al., 2005), it is possible that the degree by which Panx1 contributes to age-dependent synaptic plasticity will also depend on changes in LTP-specific proteins, particularly with those that Panx1 has been shown to interact/modulate, such as NMDA receptors (Bialecki et al., 2020; Weilinger et al., 2012).
Alternatively, discrepancies between our results and others may be related to differences in phenotype caused by mouse genetic background (Gerlai, 1996; Simpson et al., 1997) and/or effects of passenger mutations in Panx1 congenic mice (Vanden Berghe et al., 2015). An example from a previous study found that two Panx1-null mouse lines (Panx1Vsh and Panx1Vmd, derived, respectively, from 129 and C57Bl/6 embryonic stem cells) expressed the presence or absence of a phenotype that coincided with the presence or absence of one passenger mutation (Vanden Berghe et al., 2015). Thus, it is possible that the potentiation of LTP and absence of LTD found in the two 129-derived Panx1 mouse lines (Panx1−/− and Panx1Vsh (Ardiles et al., 2014; Prochnow et al., 2012) and the attenuation of LTP and LTD we observed for the Panx1KOMP (C57BL/6-derived ECS)) could be due to a passenger mutation. Even considering this possibility, the impact of altered synaptic plasticity on the phenotype of the different mouse lines was similar, as both blockade and saturation of LTP have been found to lead to impaired spatial learning (Morris et al., 1986; Moser et al., 1998).
Our results associating reductions in both LTP and LTD with deficits in hippocampal-dependent learning are generally supported by literature implicating these forms of synaptic plasticity in spatial learning. Our findings that long-term reference, but not working, memory is impaired in Panx1-deficient mice are particularly interesting. The CA1 region of the hippocampus has been implicated in virtually all forms of hippocampus-dependent learning. However, evidence concerning its role and that of the hippocampus at large in working memory is more limited. Highly selective genetic lesions (Brun et al., 2002) or optogenetic silencing (Nakashiba et al., 2008) of glutamatergic medial entorhinal cortex layer 3 inputs to CA1 impaired trace fear conditioning and spatial working memory, but did not perturb spatial reference memory, contextual fear conditioning, or place-cell firing. These studies allow for the proposal of dichotomous transmission pathways for working and reference memory information to CA1 pyramidal cells. That is, hippocampal-dependent reference memory may be maintained by the CA3 Schaffer collateral synaptic inputs to CA1 pyramidal neurons, whereas entorhinal cortical inputs onto apical dendrites of CA1 pyramidal neurons may play a primary role in transmitting information concerning working memory, as suggested by our results.
In light of the memory consolidation theory (Alvarez & Squire, 1994), we propose that Panx1-dependent synaptic plasticity at CA1 pyramidal neurons facilitates the ability of these neurons, the major output of the hippocampus, to present or activate memories in cortical synapses for long-term storage for later retrieval. The finding that long-term reference, but not working, memory is impaired in Panx1-deficient mice suggests that activation of Panx1 is selectively involved in the long-term plasticity in multiple brain regions that is necessary for reference memory storage.
Activity-dependent plasticity has long been emphasized as neuronal and largely synaptic alterations. However, astrocytes have been highlighted as playing important roles in the modulation of synaptic plasticity, neural circuits, and networks (Kastanenka et al., 2020; Wang et al., 2021). Besides their metabolic functions, as part of the “tripartite synapse” (Araque et al., 1999), astrocytes increase their cytoplasmic calcium levels in response to numerous synaptic signals, which induces the release of neuroactive substances (“gliotransmitters”), which in turn modulate synaptic activity and plasticity. The involvement of astrocytes in modulation of activity-dependent synaptic plasticity appears to be important for learning and memory, and thus is of behavioral relevance. For instance, astrocyte-specific genetic deletion of CB1 receptors impaired LTP and recognition memory, both of which were completely rescued by elevation of extracellular concentration of the NMDAR co-agonist D-serine (Robin et al., 2018). Moreover, a recent study showed that astrocyte p38α MAPK signaling is required for induction of LTD at CA3-CA1 synapses that contributes to contextual fear conditioning (Navarrete et al., 2019). Although more investigation will be needed to determine the mechanism(s) by which astrocyte Panx1 modulates synaptic plasticity, our data are the first to indicate that these cells have a significant role in long-term spatial reference memory. Release of ATP and D-serine through astrocytic Panx1 channels (Iglesias et al., 2009; Pan et al., 2015) represents an important mechanism for activity-dependent neuron-glia interaction. ATP acting on P2X receptors can affect the efficacy of all elements of the tripartite synapse via interaction with postsynaptic NMDA and GABA receptors, by triggering trafficking of AMPA receptors and modulating the release of neuro- and gliotransmitters (reviewed in (Pankratov et al., 2009)). D-serine stored and released from astrocytes has been shown to act on neuronal NMDARs and thus modulates NMDAR-dependent synaptic plasticity (Henneberger et al., 2010; Mothet et al., 2000; Mothet et al., 2005; Panatier et al., 2006; Yang et al., 2003).
Our behavioral studies indicating that deletion of Panx1 from either astrocytes or neurons results in similar memory deficits as those seen in the global Panx1 KO certainly requires further evaluation to disclose the mechanisms by which Panx1 in these two cell population leads to similar behavioral outcomes. In addition, although unlikely, we cannot exclude the possibility that the similar behavioral defects seen between mice lacking Panx1 in astrocyte and neuronal resulted from their different genetic backgrounds (Panx1f/f: originally 129S1/Sv * 129S6/SvEvTac * 129X1/SvJ * C57BL/6 * SJL; GFAP-Cre:Panx1f/f: originally 129S1/Sv * BALB/c * C57BL/6 * C57BL/6NHsd, and NFH-Cre:Panx1f/f: originally 129S1/Sv * 129X1/SvJ * C57BL/6 * FVB/N).
The presence of a causal relationship between Panx1 deletion, LTP/LTD, and learning, or a correlation due to shared compensatory mechanisms, remains to be determined. In addition, future experiments aimed to evaluate the detailed mechanism by which astrocyte and neuronal Panx1 impact LTP/LTD should also provide some insights of why deleting Panx1 from astrocytes or neurons results in similar behavioral defects as seen in the global Panx1 knockout. Nevertheless, our findings that a Panx1 deletion induces impairments in the induction of both LTP and LTD in the hippocampus have significant implications for Panx1 modulation of learning and memory.
Main Points
(1) Long-term reference memory, but not long-term working memory is compromised in mice deficient of Panx1. (2) Both astrocyte and neuronal Panx1 contribute to long-term spatial memory. (3) Deficits in hippocampal dependent memory were associated with decreased long-term potentiation (LTP) and long-term depression (LTD).
Supplemental Material
sj-docx-1-asn-10.1177_17590914231184712 - Supplemental material for Astrocyte and Neuronal Panx1 Support Long-Term Reference Memory in Mice
Supplemental material, sj-docx-1-asn-10.1177_17590914231184712 for Astrocyte and Neuronal Panx1 Support Long-Term Reference Memory in Mice by Price Obot, Galadu Subah, Antonia Schonwald, Jian Pan, Libor Velíšek, Jana Velíšková, Patric K. Stanton and Eliana Scemes in ASN Neuro
Footnotes
Acknowledgments
This work was supported by the National Institute of Neurological Disorders and Stroke of the National Institutes of Health (5R01NS092786).
Author note
Price Obot is currently affiliated to Department of Emergency Medicine, Penn State Hershey Medical Center, Hershey, PA, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by NIH (RO1-NS09786) to ES, JV, LV, PO and by NIH (1R21NS118337) to LV.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
