Abstract
There is a critical need for small molecules capable of rescuing pathophysiological phenotypes induced by alpha-synuclein (aSyn) misfolding and oligomerization. Building upon our previous aSyn cellular fluorescence lifetime (FLT)-Förster resonance energy transfer (FRET) biosensors, we have developed an inducible cell model incorporating the red-shifted mCyRFP1/mMaroon1 (OFP/MFP) FRET pair. This new aSyn FRET biosensor improves the signal-to-noise ratio, reduces nonspecific background FRET, and results in a 4-fold increase (transient transfection) and 2-fold increase (stable, inducible cell lines) in FRET signal relative to our previous GFP/RFP aSyn biosensors. The inducible system institutes greater temporal control and scalability, allowing for fine-tuning of biosensor expression and minimizes cellular cytotoxicity due to overexpression of aSyn. Using these inducible aSyn-OFP/MFP biosensors, we screened the Selleck library of 2684 commercially available, FDA-approved compounds and identified proanthocyanidins and casanthranol as novel hits. Secondary assays validated the ability of these compounds to modulate aSyn FLT-FRET. Functional assays probing cellular cytotoxicity and aSyn fibrillization demonstrated their capability to inhibit seeded aSyn fibrillization. Proanthocyanidins completely rescued aSyn fibril-induced cellular toxicity with EC50 of 200 nM and casanthranol supported a 85.5% rescue with a projected EC50 of 34.2 μM. Furthermore, proanthocyanidins provide a valuable tool compound to validate our aSyn biosensor performance in future high-throughput screening campaigns of industrial-scale (million-compound) chemical libraries.
Introduction
Misfolding and self-association of alpha-synuclein (aSyn) is at the epicenter of pathophysiological challenges in alpha-synucleinopathies, such as Parkinson's disease, dementia with Lewy bodies, and multiple systems atrophy (Emadi et al., 2004; Gallardo et al., 2008; Lorenzen et al., 2014; Power et al., 2015; Rockenstein et al., 2014; Spillantini et al., 1998; Uversky, 2008; Uversky & Eliezer, 2009; Wirths et al., 2000). Identifying small molecules that can rescue these aSyn-associated cellular insults has been the focus of multiple studies (Caruana et al., 2012; Casalino et al., 2022; Herva et al., 2014; Höllerhage et al., 2017; Macchi et al., 2016; Moussaud et al., 2015; Perni et al., 2017; Pujols et al., 2017; Sahihi et al., 2021; Tóth et al., 2019), including our recent live-cell high-throughput screens (HTS) that monitored aSyn oligomerization and misfolding via fluorescence lifetime (FLT) detection of Förster resonance energy transfer (FRET) within cellular biosensor constructs (Braun et al., 2021) and computational docking HTS targeting aSyn fibril structures (Nathan Kochen et al., 2022). With both approaches, we identified compounds that modulate aSyn fibrillization.
Our initial aSyn FLT-FRET biosensors were engineered using the GFP/RFP (G/R) FRET pair with either single XFP fusion proteins (GFP-aSyn and aSyn-RFP) or double XFP fusion (GFP-aSyn-RFP) transiently expressed in HEK293 cells (Braun et al., 2021). These biosensors monitor aSyn-aSyn protein-protein interactions through changes in donor FLT when either the single-fusion donor and acceptor constructs are co-expressed (FRET resulting from changes in aSyn oligomerization or conformation) or a double-fusion donor–acceptor construct (FRET resulting from both changes in oligomer state as well as changes in monomer conformation) (Braun et al., 2021). The use of FLT-detected FRET provides a significant increase in signal-to-noise relative to intensity based FRET approaches, and our HTS campaigns are made possible by a specialized FLT plate-reader (FLPR; Gruber et al., 2014; Schaaf, 2016; Schaaf et al., 2018) and spectral-unmixing plate-reader (SUPR;Schaaf et al., 2016). With our aSyn-G/R biosensor, we ran an HTS of the 1280-compound LOPAC library with the FLPR monitoring the aSyn-G FLT and the SUPR identifying interfering compounds that manifest in the biosensor's emission spectra. Biochemical characterization and cell-seeded aggregation experiments demonstrated that overexpression of the aSyn-XFP fusion constructs resulted in soluble, oligomeric aSyn assemblies that were not able to seed in vitro aSyn fibrilization (Braun et al., 2021).
We have successfully employed the GFP/RFP FRET pair on multiple HTS campaigns that targeted an array of different protein targets and protein-protein interactions (e.g., aSyn, tau, Huntington protein, TNFR1 and DR5) (Braun et al., 2021; Lo et al., 2017, 2019a, 2019b, 2020, 2021; Young et al., 2023). However, advancements in fluorescent fusion protein donor–acceptor pairs for FRET have yielded improved spectral characteristics (e.g., increased spectral overlap, reduced maturation time, reduced self-association) that have shown promise for improving the FLT-FRET HTS assay quality (Bajar, 2016).
In this study, we have incorporated the red-shifted mCyRFP1/mMaroon1 (OFP/MFP; O/M) FRET pair into our genetically encoded aSyn FRET biosensors. Previous implementation of this OFP/MFP FRET pair in a biosensor targeting SERCA2a (the human cardiac calcium pump) has resulted in nearly double the FRET signal window and improved the fluorescence signal-to-noise (S/N) (Schaaf et al., 2018). We created two inducible aSyn biosensor cell lines for an HTS campaign with the 2684-compound Selleck library. With a ± 4SD hit-selection threshold, we have identified 36 reproducible hits, of which 31 compounds passed validation via FLT dose-response assays. Secondary functional assays were used to determine each compound's ability to rescue aSyn fibril induced cytotoxicity (in cellulo MTT assay) and modulate aSyn fibrillization (cell-free in vitro aggregation).
Results
mCyRFP1/mMaroon1 (O/M) FRET Pair Results in Increased FRET Signal for Cellular aSyn Biosensors
Initial FLT-FRET experiments were performed to evaluate the FRET signal for the new O/M aSyn FRET biosensors, relative to our original G/R aSyn FLT-FRET system, using both single-fusion (i.e., where donor constructs [N-terminal aSyn fused with GFP or OFP] and acceptor constructs [C-terminal aSyn fused with RFP or MFP] are co-expressed) and double-fusion biosensors (i.e., aSyn has two fusion proteins, one N-terminal and one C-terminal; FRET can occur from both conformational changes within a monomer as well as protein-protein interactions). HEK293 cells were transiently transfected for 48 hours with donor-only (G-aSyn or O-aSyn) or donor–acceptor (single-fusion FRET pair: [G-aSyn + aSyn-R]; [O-aSyn + aSyn-M]; double-fusion: G-aSyn-R; O-aSyn-M) biosensor constructs. The single-fusion, intermolecular FRET biosensors, were transfected at a 1:8 donor:acceptor ratio as previously reported (Braun et al., 2021) whereas the double-fusion constrains a donor:acceptor ratio of 1:1.
The FLPR instrument is configured with two laser lines, at 473-nm and 532 nm. Our G/R biosensor utilized only the 473-nm laser for excitation whereas the red-shifted O/M biosensor can be excited with either the 473-nm or 532-nm lasers, providing two key advantages over the original G/R platform (Schaaf et al., 2018). Excitation of the donor with the 532-nm laser line bypasses a significant amount of cellular autofluorescence that is observed with 473-nm excitation. Furthermore, using a combination of 473-nm and 532-nm provides an additional detection channel for identification and exclusion of interfering fluorescent compounds (Schaaf et al., 2018). Figure 1a highlights the increased FRET efficiency for the O/M biosensors, which was observed at both wavelengths, relative to the original G/R biosensor. For both single-fusion (monitoring distinct protein-protein interaction) and double-fusion (monitoring both protein–protein interactions and protein conformation) biosensors, we achieved a 3–4-fold increase in the FRET E from 0.072 to 0.266 with 473-nm excitation [Ex:473] and 0.229 at 532-nm excitation [Ex: 532] for single-fusion; 0.043 to 0.262 [Ex:473] and 0.201 [Ex:532] for double-fusion biosensors. Figure 1b illustrates a 3–4-fold increase in donor fluorescence relative to untransfected cells when the O/M biosensor is excited at 532-nm relative to the O/M or G/R biosensor when excited at 473-nm and significantly improves the S/N.
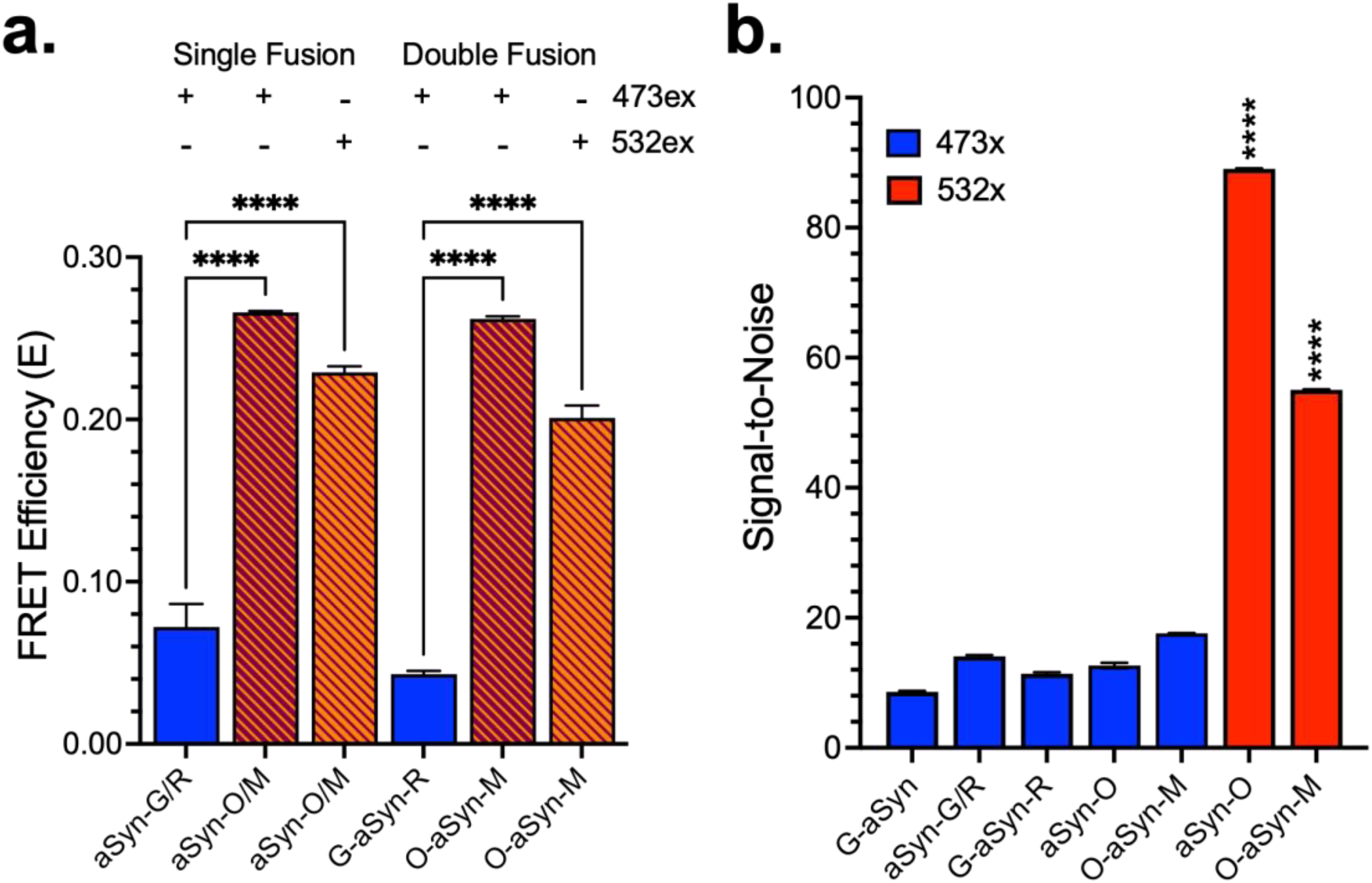
Asyn biosensors expressing the O/M red-shifted FRET pair have improved FRET properties relative to the G/R while still maintaining low background signal and significantly improving the signal-to-noise (S/N). (a) Transient transfection of HEK293 cells demonstrates increased FRET from the mCyRFP1/mMaroon1 (OFP/MFP; O/M) biosensors relative to GFP/RFP (G/R) for both single-fusion and double-fusion aSyn FRET biosensors. (b) mCyRFP1 (OFP; O) can be excited at either 473-nm or 532-nm wavelength. The red-shifted 532-nm excitation results in significantly reduced background signal and corresponding increase in signal-to-noise.
Engineering a Stable Inducible Cellular Biosensor
We previously observed nonlinear expression with cotransfection of the single-fusion biosensor constructs relative to the DNA ratios used (Braun et al., 2021). As the ratio of acceptor-to-donor increased to achieve higher FRET efficiency, there was a reduced donor signal and subsequent decrease in FLT-FRET S/N. This was exacerbated when attempting to create stable cell lines expressing consistent but different amounts of donor and acceptor. Ideally, a double-fusion biosensor would be used for stable cell line generation because it constrains expression of the XFPs to equimolar levels of donor and acceptor. However, with our previous aSyn-G/R platform, the double-fusion biosensor's FRET efficiency was only 2-fold above background FRET, resulting in low S/N for HTS, and attempts to generate stable biosensor expressing cells with sufficient expression and FLT-FRET signal were unsuccessful. The significant improvements in FRET efficiency and signal-to-noise achieved with the O-aSyn-M double-fusion construct have enabled it to stand-alone as a biosensor for HTS, thus simplifying the initial screening step of our drug discovery pipeline.
Using the aSyn-O/M biosensor constructs, we generated inducible HEK293 cell lines expressing aSyn-O (donor) or O-aSyn-M (donor–acceptor) under tet-On control. Inducible aSyn-O (iaSyn-O) and O-aSyn-M (iO-aSyn-M) constructs were subcloned into the pTT22 empty vector (Supplemental Figure S1). The iaSyn-O and iO-aSyn-M plasmids were then transfected into HEK293 cells stably expressing the tetracycline transactivator (tTa) and placed under puromycin selection (1.0 μg/ml) for 30 days to generate a population of cells with high (>95%) aSyn biosensor expression. The pTT22 vector includes two tet-operators downstream of a minimal CMV promoter, which exploits tTA regulation to induce expression upon addition of doxycycline (Dox). Using tTA as a repressor element results in low-level, leaky biosensor expression, facilitating the selection for stable transfected cells without the need to overexpress the biosensor to levels that would lead to cellular toxicity.
We next performed a 48-hour Dox induction concentration response with the iaSyn-O cell line to determine optimal Dox concentrations for biosensor expression. Supplemental Figure S2 highlights the Dox effects on cell proliferation (S2a), fraction of fluorescent cells (S2b), and average positive cell fluorescence intensity (S2c). For all subsequent experiments, biosensor induction was performed at 0.5 μg/ml Dox due to the robust increase in fluorescence (both cell population and intensity) and consistent FLT signal, while also observing no reduction in overall cell proliferation. Flow cytometry analysis of the iaSyn-O and iO-aSyn-M cell lines with and without Dox demonstrate both the low-level leaky expression inherent in the pTT22/tTa regulation system and the robust and complete population shift upon Dox addition (i.e., biosensor induction; see Supplemental Figure S3).
We then optimized the induction time for our inducible biosensor cell lines. Supplemental Figure S4a presents the FLT induction time course for the iaSyn-O cell line. We observe an increase in FLT from approximately 2.3 ns up to a plateau at approximately 3.0 ns after 4-days of induction. The low initial FLT arises from the high background fluorescence due to minimal, leaky donor expression. We began to see increased FLT variability beyond 5 days of induction. Induced biosensor expression for both iaSyn-O and iO-aSyn-M cell lines was confirmed via Western blots probed for aSyn expression (Supplemental Figure S4b). Both iaSyn-O and iO-aSyn-M cells showed an increase in biosensor expression over induction time, with a plateau that parallels the timing of FLT increase. From these experiments, we determined that 5-day induction at 0.5 μg/ml Dox for the iaSyn-O and iO-aSyn-M cells would be used for all subsequent FLT-FRET experiments.
Red-Shifted O/M Biosensors Have Low Background FRET
We next characterized the background FRET signal for the O/M system. iaSyn-O cells were induced for 4 days then transiently transfected for 24-hour with two different amounts of either: (1) MFP only (+M), or (2) aSyn-MFP (aSyn-M) pcDNA3.1 plasmids. Figure 2a highlights the extremely low basal FRET signal observed for the +M transfection which resulted in FRET efficiencies indistinguishable from the donor-only, no FRET condition. In contrast, when an aSyn-M accepter is co-expressed, there is a strong DNA-dependent increase in the FRET signal. This confirms that the FRET signal observed for our iO-aSyn-M biosensor is truly dependent on aSyn's conformation and/or oligomerization and is not a result of nonspecific interactions between OFP and MFP.
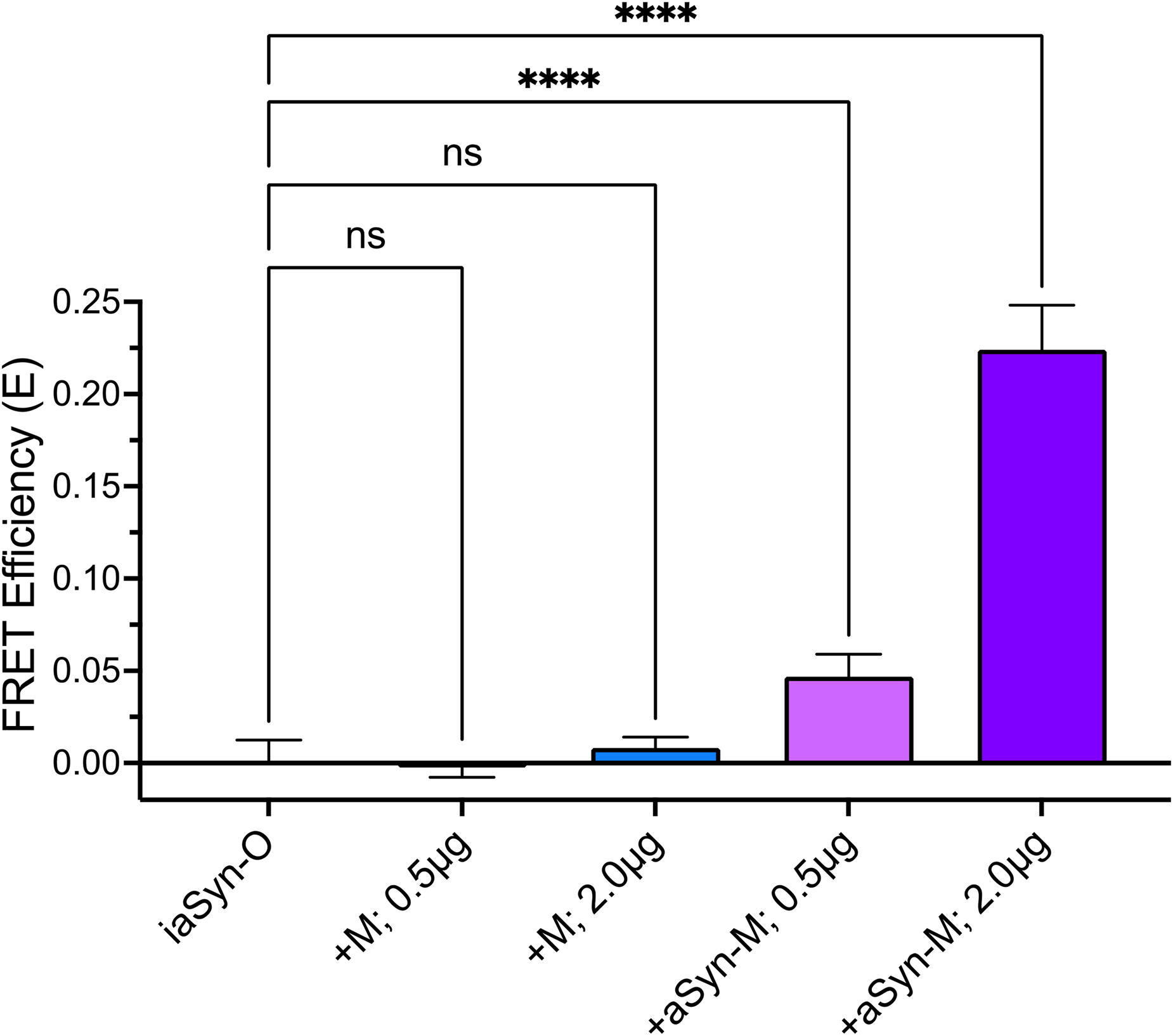
Characterization of aSyn O/M biosensor background. Transient transfection of iaSyn-O donor-only biosensor cells, with MFP (M) or aSyn-M at two concentrations, illustrates a +10-fold increase of FRET signal for the aSyn-M biosensor relative to M only and is confirmation that the FRET signal is due to aSyn conformation and/or oligomerization and not the result of nonspecific interactions between OFP and MFP. Data are mean and SEM from N = 3 experiments with 5 replicates per experiment. One-way ANOVA of N = 3 independent experiments, ****p < .001.
HTS of SELLECK Library Using iaSyn-O and iO-aSyn-M Biosensor Cell Lines
Noninduced iaSyn-O and iO-aSyn-M biosensor cells were plated into 15-cm culture dishes and treated with 0.5 μg/ml Dox for 5-days to induce aSyn-O or O-aSyn-M expression. Prior to applying biosensor cells to SELLECK library assay plates, FLT was measured to confirm biosensor expression, FRET, and the coefficient of variance (CV = SD/mean). Cells were dispensed (5 μl/well at 1e6 cells/ml) into 1536-well compound plates (10 μM final compound concentration) and incubated in the dark for 120 minutes prior to reads using the FLPR (to monitor the donor FLT) and SUPR (to monitor the full emission spectrum to identify fluorescent compounds) instruments. Figure 3a and b presents the baseline FLT and FRET signal for all CV plates read on the day of screening. The resulting CV of 0.79 ± 0.046% is on par with our previous aSyn HTS campaigns, whereas the O/M double-fusion biosensor's FRET signal is nearly 3-fold greater than our previous G/R, providing greater signal window to detect compound responses.
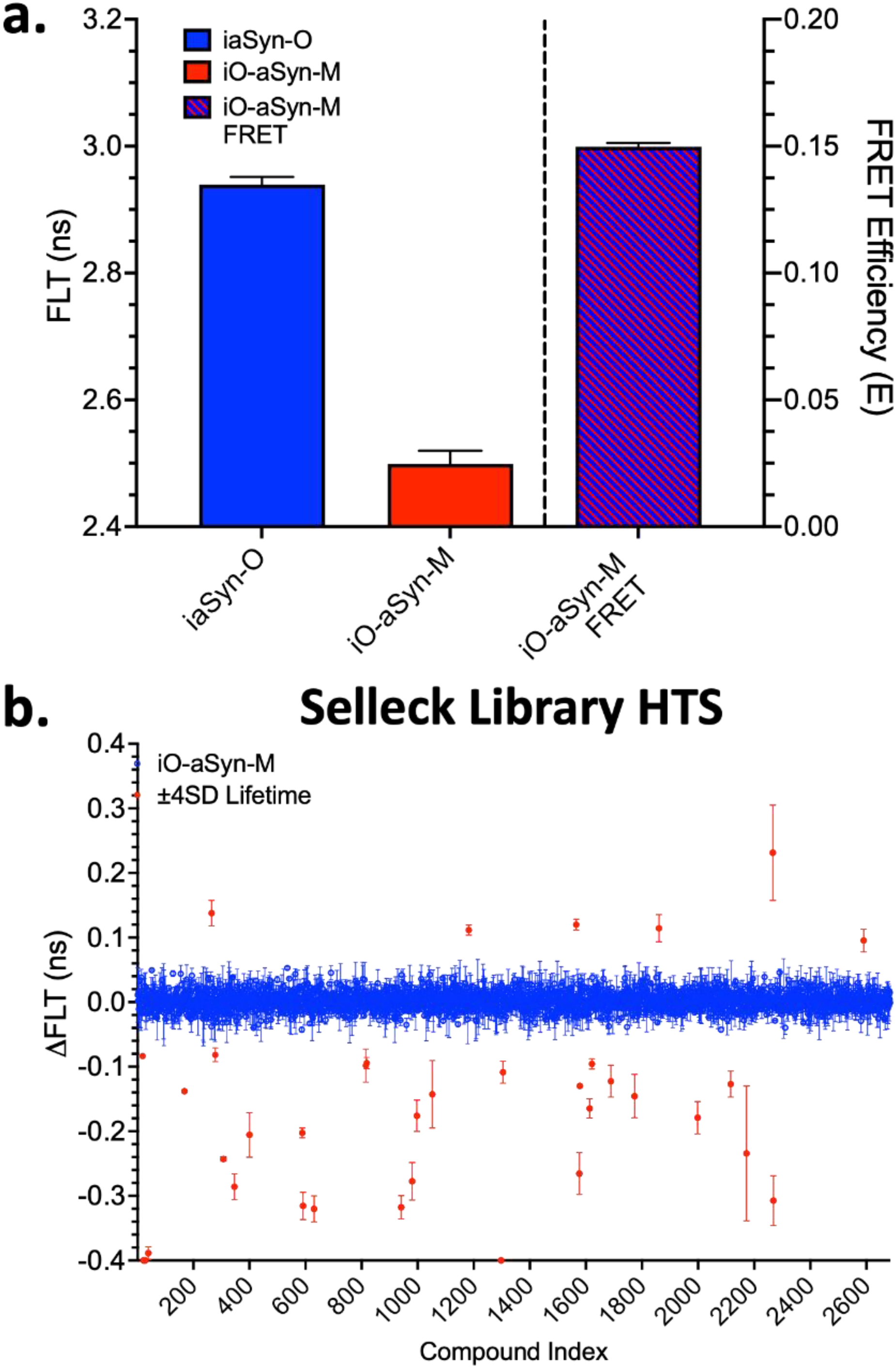
HTS for inducible aSyn O/M double-fusion biosensor. (a) Baseline FLT and FRET measurements from control plates are used to determine CV and confirm FLT-FRET signal prior to each replicate HTS. (b) Screens of the 2682 compound SELLECK library were performed in triplicate and resulted in 36 reproducible hits (red) at a ±4SD threshold. Data are mean and SEM from N = 3 HTS replicates.
In total we carried out four independent screens of the SELLECK library. Of these, three runs were done with the double-fusion iO-aSyn-M, with the fourth run using the iaSyn-O as a donor-only control. We processed all FLT HTS data for fluorescent compounds (FC) using a spectral similarity index filter (Schaaf et al., 2016, 2018; Stroik et al., 2018), which evaluates the ratio of intensity from two regions of the donor emission spectrum. The combined HTS data for all compounds that passed the FC filter are presented in Figure 3c (blue) with hits identified as the compounds with ±4 SD ΔFLT (red). In total, 36 reproducible hits were identified that passed FC filters. The ±4 SD cutoff is an arbitrary threshold, based on the number of hits that meet that criterion, the throughput of secondary assays, and resources available for purchasing compounds.
FLT Concentration Response Curves (CRC) of Hits With iaSyn-O and iOaSynM FRET Biosensors
In total, 31 of the 36 hits were available to order from commercial suppliers, and assay plates for CRCs were prepared as detailed in section “Methods” (see Table 1 for a summary of the HTS hits that were tested). In addition to the hit compounds, EGCG was used as a positive control in the FLT CRC assay because it has been shown to modulate aSyn oligomerization (Lorenzen, 2014). FLT CRCs were performed using pre-plated 8-point serial dilutions in 384-well plates (black, low-binding). Both iaSyn-O and iO-aSyn-M cell lines were induced and processed using the same conditions as used in the HTS phase. Cells were dispensed 50 μl per well at 1e6 cells/ml, then incubated in the dark for 120 minutes prior to FLPR reads.
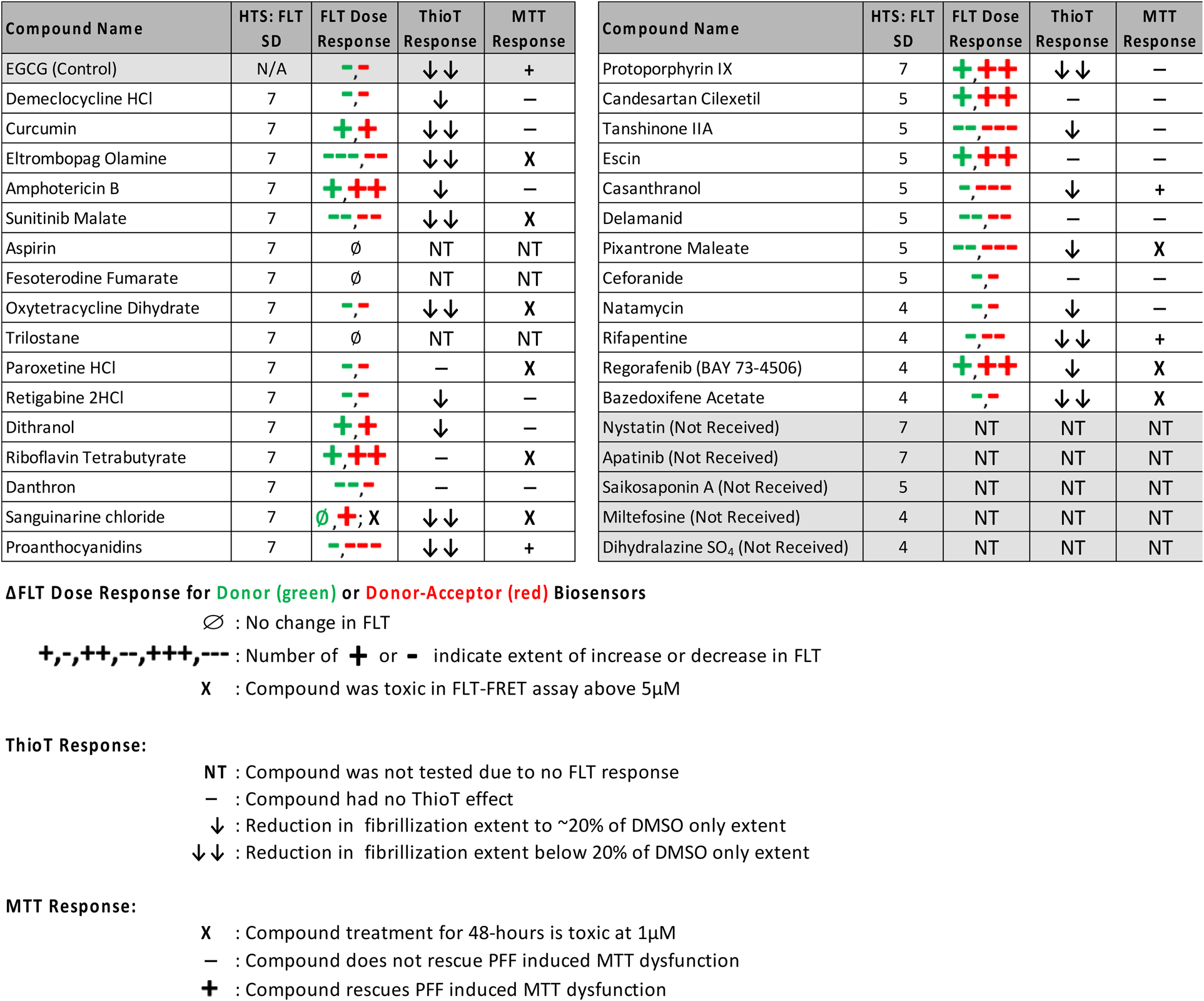
List of Hit Compounds and Assay Response. Hit Compounds are Listed in Order of Reducing Standard Deviation from the FLT-FRET HTS. for Each Hit Tested the FLT Dose Response Direction and Magnitude of Change are Indicated as a “+” (Increased FLT) or “-” (Decreased FLT) for Donor–Acceptor (Red) and Donor Only (Green) Biosensors. No Dose–Response is Indicated by ∅. Only Compounds with a FLT-FRET Dose Response Were Tested in Subsequent Assays, Those That Were not Tested are Indicated with “NT”. Attenuation of ThioT Aggregation to ∼20% of DMSO (↓), Greater Than 20% (↓↓) or no Response “-.” Lastly MTT Response is Indicate as Rescue “+,” no Change “-,” or Toxic “X.”
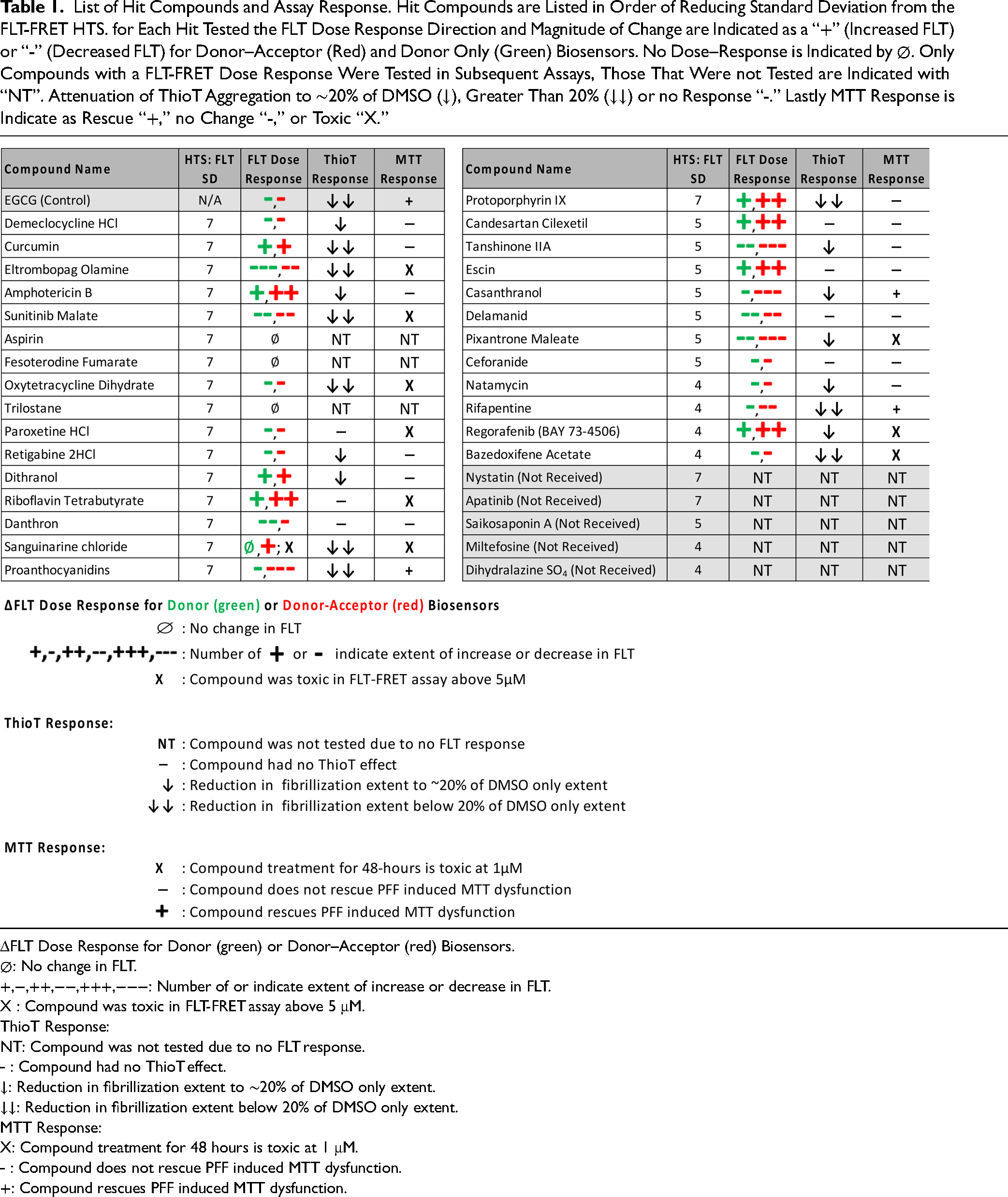
ΔFLT Dose Response for Donor (green) or Donor–Acceptor (red) Biosensors.
∅: No change in FLT.
+,−,++,−−,+++,−−−: Number of or indicate extent of increase or decrease in FLT.
X : Compound was toxic in FLT-FRET assay above 5 μM.
ThioT Response:
NT: Compound was not tested due to no FLT response.
⎯ : Compound had no ThioT effect.
↓: Reduction in fibrillization extent to ∼20% of DMSO only extent.
↓↓: Reduction in fibrillization extent below 20% of DMSO only extent.
MTT Response:
X: Compound treatment for 48 hours is toxic at 1 μM.
⎯ : Compound does not rescue PFF induced MTT dysfunction.
+: Compound rescues PFF induced MTT dysfunction.
Figure 4 presents summary data and corresponding CRCs for all hit compounds with the iO-aSyn-M (donor–acceptor, DA) and iaSyn-O (donor-only, D) biosensors. Of the 31 compounds that we received, 25 compounds displayed a dose-dependent response for the FLT-FRET biosensors, with 21 having significant DA ΔFLT relative to DMSO at 10 μM concentration in the larger, 384-well CRC assay format. The other six compounds either did not produce any titratable response or displayed a strong background fluorescence signal and were consequently eliminated from subsequent characterization (Figure 4a–c, compounds were grouped by direction and magnitude of response). By using both aSyn-O/M biosensors to determine the FLR-FRET CRC we can contrast compounds that are likely to directly act on aSyn aggregation (those affecting the iO-aSyn-M biosensor only) or those that are acting in an indirect mechanism (due to effects on both iO-aSyn-M and iaSyn-O). Figure 4d presents an example CRC comparison with a full complement of compound CRC presented in Supplemental Figures S5 and S6.
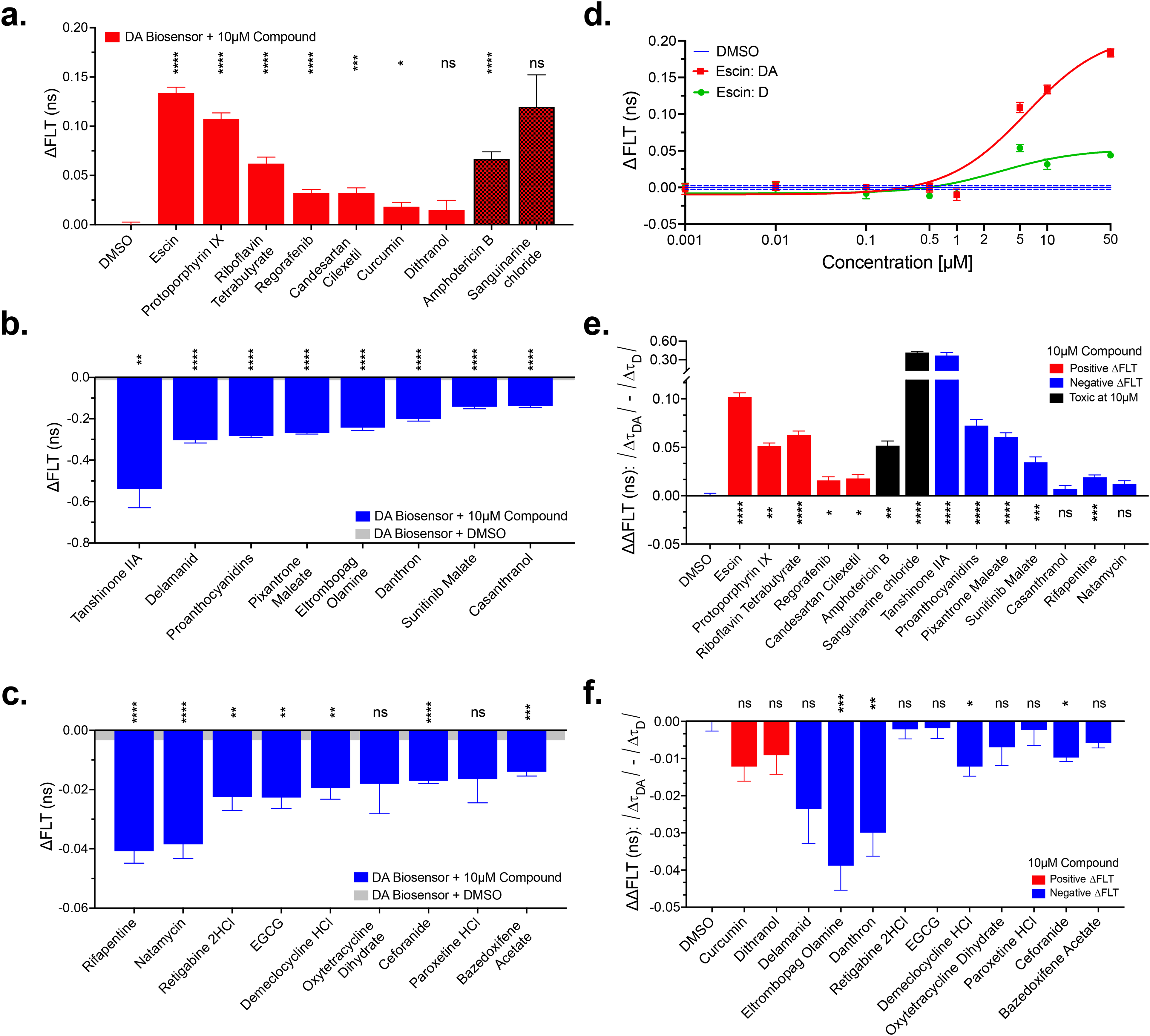
FRET dose response summary. Hit compounds were evaluated in an 8-point dose response (0.001–50 μM) with both the iaSynO (donor-only, D) and iO-aSyn-M (donor–acceptor, DA) cellular biosensors. The 25 different compounds ΔFLT response at 10 μM in the iO-aSyn-M biosensor are broken down as compounds that (a) produce a positive ΔFLT (red bars), (b) a large magnitude negative ΔFLT (blue bars), and (c) a small negative ΔFLT (blue bars). (d) An example FLT-FRET CRC for iO-aSyn-M (red) and iaSyn-O (D, green) with all compound CRC presented in Supplemental Figures S5 and S6. The difference between donor–acceptor and donor-only signal at 10 μM drug for (e) compounds with positive ΔΔFLT values (e.g., stronger DA (iO-aSyn-M) effect relative to D (iaSyn-O)) and (f) compounds with a negative ΔΔFLT (D response is greater than DA). Compounds with an overall positive ΔFLT (a) are in red and negative ΔFLT (b and c) are in blue. Data are mean and SEM from N = 3 experiments; with 4 replicate wells per experimental condition. One-way ANOVA statistical analysis with, nonsignificant (ns), *p < .05, **p < .01, ***p < .005, and ****p < .001.
Figure 4e and f summarizes the 10 μM compound response by presenting the difference between the donor–acceptor (iO-aSyn-M) and donor (iaSyn-O) ΔFLT where an overall negative or positive ΔFLT are illustrated in blue or red respectively. Figure 4e presents 12 compounds the had a large ΔΔFLT (stronger DA effect relative to D): 5 increased FLT, decreasing FRET (escin, protoporphyrin IX, riboflavin tetrabutyrate, regorafenib, and candesartan cilexetil); and 7 decreased FLT, increasing FRET (tanshinone IIA, proanthocyanidins, pixantrone maleate, sunitinib malate, rifapentine, natamycin, and casanthranol). Two of the compounds, sanguinarine chloride and amphotericin B (red checkered bars), with large ΔΔFLT resulted in acute toxicity at 10 μM. Seven hits and the control EGCG had similar effects on both DA and D and were not significantly different than DMSO controls (Figure 4f) for the hit compounds: 2 increased (curcumin and dithranol) and 5 hits plus control EGCG decreased FLT (delamanid, retigabine 2 HCl, oxytetracycline dihydrate, paroxetime HCl, and bazedoxifene acetate). A subset of the compounds with negative ΔΔFLT (D response is greater than DA) had a significantly greater D-only ΔFLT response (eltrombopag olamine, danthron, demeclocycline HCl, and ceforanide). Compounds that affect only the iO-aSyn-M FLT suggest a mechanism of action (MOA) that directly targets the iO-aSyn-M construct, whereas hits that affect both iaSyn-O and iO-aSyn-M biosensors could be acting through an indirect MOA or could potentially be toxic to the cells. The control, EGCG, had a similar response for both iaSyn-O and iO-aSyn-M biosensors suggesting its effect is not specific to aSyn and highlights the benefit of running donor-only counter screen. It is known that the MOA of EGCG is not aSyn specific and it can inhibit fibrillization of many proteins (Lorenzen, 2014).
Proanthocyanidins and Casanthranol are Protective in a PFF-Induced Toxicity Model
All compounds with an FLT-FRET dose response were tested in an in cellulo aSyn pre-formed fibril (PFFs) toxicity model in differentiated Neuro-2a (N2a) cells. To mimic the pathophysiology of synucleinopathies (which selectively damage dopaminergic neurons) we utilized dibutyryl cyclic adenosine monophosphate (dbcAMP) to differentiate N2a cells into dopaminergic-like neurons. Their differentiation is evidenced by increased expression of tyrosine hydroxylase and increased production of dopamine (Tremblay et al., 2010).
Differentiated N2a cells were treated with 1 µg/ml sonicated aSyn PFF (with and without DMSO vehicle) to establish a baseline for the PFF induced MTT effect. Figure 5a shows that PFF treatment resulted in a apprximately 40% reduction in MTT signal, consistent with other reports of fibril-induced toxicity (Ardah et al., 2020; Li et al., 2018). We next evaluated all 25 compounds (with EGCG as positive control) using a prophylactic pretreatment paradigm at a single dose of 1 µM. Briefly, compounds were added to differentiated N2a cells and incubated for 1.5 hours. aSyn PFF were then added at 1 µg/ml and cells were incubated for an additional 48 hours. Figure 5b presents the response of all compounds (with and without PFF treatment). Three compounds were identified (proanthocyanidins, casanthranol and rifapentine) as potentially rescuing PFF induced toxicity. Figure 5c and Supplemental Figure S7 present dose titrations of EGCG and the three MTT hits using the same PFF treatment model. Of the drugs tested, only EGCG and proanthocyanidins showed a complete rescue of PFF toxicity, EC50 of 7.2 nM and 200 nM, respectively. Casanthranol showed a dose-dependent, incomplete rescue of viability (recovering up to 85.5%) with a projected EC50 of 34.2 μM. EGCG, proanthocyanidin and casanthranol did not affect the MTT signal when evaluated with vehicle-treated N2a cells alone (e.g., no PFFs), suggesting that their effect is PFF-dependent or at the least dependent on a PFF-induced mitochondrial deficit. In contrast, rifapentine showed a slight increase (nonsignificant) in viability at lower concentrations but either became toxic or confounded the absorbance measurement at higher concentrations, failing to rescue the PFF-induced toxicity.
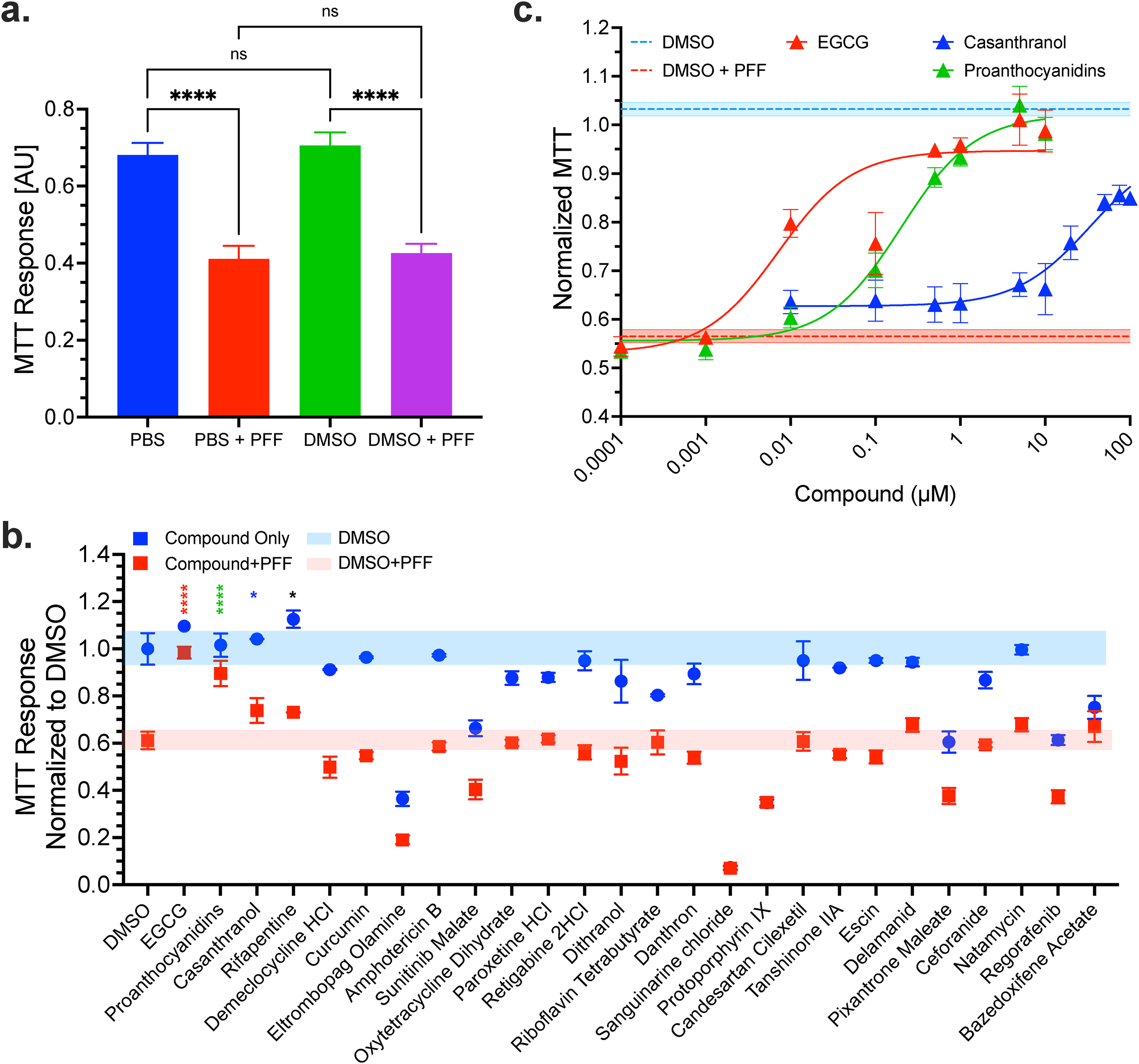
Proanthocyanidins and casanthranol rescue PFF induced cellular toxicity. (a) N2a cells have a robust MTT response, with an approximate 40% reduction, due to 1 μg/ml PFF treatment that is unaffected by treatment with vehicle DMSO. (b) EGCG (control) and all 25 hit compounds with FLT-FRET dose response were evaluated at 1 μM with (red) or without (blue) PFF treatment to determine compounds that both rescued the PFF induced deficit and had little to no effect on baseline cells. Three compounds, proanthocyanidins, casanthranol, and rifapentine (in addition to EGCG) had a significant rescue. (c) MTT dose response curves for each compound with PFF treatment. Supplemental Figure S7 presents the compound dose response for both with and without PFF as well as for Rifapentine, which demonstrated little to no concentration dependent effects. Data are mean and SEM from N = 3 experiments; with 4 replicates per experiment.
We further evaluated EGCG, proanthocyanidins, and casanthranol using four different treatment paradigms where compounds were administered 1.5 hour pre-PFF treatment, simultaneous treatment, 1.5-hour and 24-hour post-PFF treatment. Each compound was used at concentrations just below their maximum observed MTT efficacy (1 μM EGCG, 1 μM proanthocyanidins and 50 μM casanthranol, see Supplemental Figure S7). Supplemental Figure S8 shows that EGCG, proanthocyanidins and casanthranol have the highest potency at pre and simultaneous administration with PFF and lose potency in a time-dependent manner after PFF treatment.
Direct Effect on aSyn Aggregation: Seeded Thioflavin-T Assays
All 25 compounds were then tested using in a cell-free in vitro PFF seeded aSyn aggregation assay monitored via Thioflavin T (ThioT) fluorescence. Although our FLT biosensors monitor soluble oligomeric species (Braun et al., 2021), the seeded aggregation assay resolves which drugs can target both early- and late-stage processes in the aggregation cascade of aSyn, while providing more reproducible experimental replicates. In brief, 15 µM monomeric aSyn with or without 0.75 µM (monomer equivalent) PFF (i.e., 5% seed) were incubated in a sealed 384-well plate under continuous agitation. Figure 6a illustrates the typical seeded aSyn ThioT trace (e.g., an increase in ThioT fluorescence without any discernable lag phase) and demonstrates that 15 µM monomer-only aSyn does not undergo any significant primary nucleation throughout the duration of the assay and highlights that the vehicle control, (1% DMSO) does not alter the aggregation profile of aSyn + PFF.
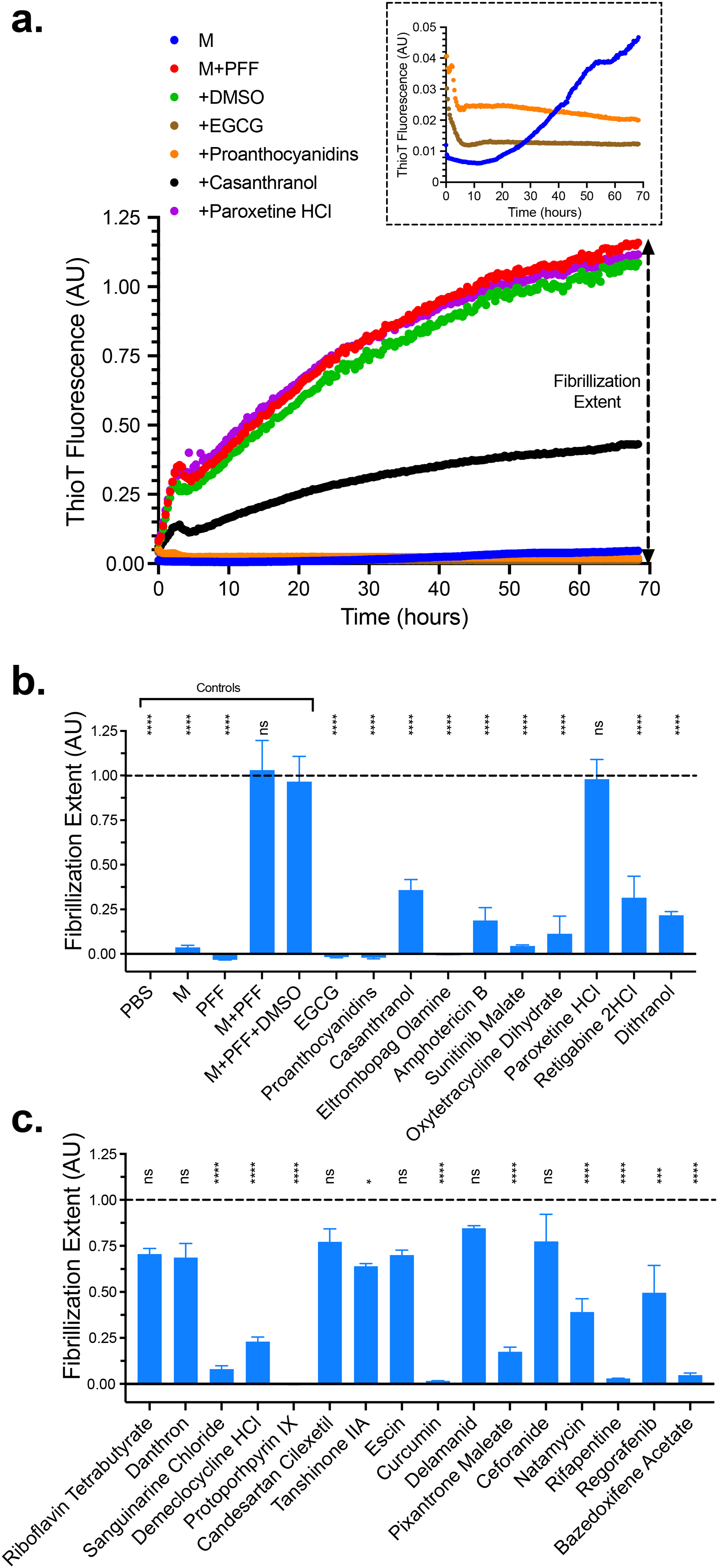
Thiot fibrillization extent. All 25 compounds with FLT-FRET dose response plus the control EGCG were tested in a seeded ThioT aggregation assay. (a) A representative ThioT trace showing vehicle (DMSO, green), hit compounds EGCG, proanthocyanidins, casanthranol, (inhibitors of aggregation), and Paroxetine HCl (compound with no effect, purple). Inset is a magnification of the dashed box showing rapid inhibition by EGCG and proanthocyanidins at early time points. (b and c) In total, 18 compounds resulted in significant attenuation of aSyn seeded fibrillization (1 at p < .05; 1 at p < .005; and 16 at p < .001). Full kinetic traces for each hit are presented in Supplemental Figure S10. Data represents mean fibrillization extent and SEM from N = 2 independent experiments with three replicate wells per experiment.
We then tested our positive control (EGCG) and the 25 hit compounds, all at 50 µM. Supplemental Figure S10 presents the kinetic traces for each, along with aSyn + PFF + DMSO for direct comparison. The effects on the seeded aggregation kinetics curve were wide-ranging with most drugs only altering the plateau level (completely blocking fibrillization, Supplemental Figure S10a and b; or attenuating fibrilization Supplemental Figure S10c and d). Some drugs greatly altered the morphology of the curve (most notably candesartan cilexetil, regorafenib and bazedoxifene acetate; Supplemental Figure S10e), and some drugs had little to no effect on fibrillization (Supplemental Figure S10f). To interpret the effects observed more clearly, we calculated the fibrillization extent by subtracting the average ThioT signal of the first and last hours of aggregation, as reported previously (Nathan Kochen et al., 2022) (Figure 6b and c). In total, 18 of the 25 tested compounds induced a significant attenuation of fibrillization extent, demonstrating a strong correlation with compounds that modulate aSyn FLT-FRET in cells, compared with those that attenuate cell-free fibrillization.
TEM Imaging of Seeded Aggregation Products for EGCG, Proanthocyanidins, and Casanthranol
Next, we used transmission electron microscopy (TEM) imaging to confirm the ThioT assay results for the three compounds with positive MTT response (EGCG, proanthocyanidins, and casanthranol). Although all three compounds displayed significant attenuation of the ThioT signal, each compound displayed a different type of remodeling of the seeded aSyn fibrils. Supplemental Figure S11 presents a representative image for each condition with the DMSO-treated control displaying the characteristic long twisting aSyn fibrils consistent with efficient aSyn monomer templating and elongation from PFF seeds (Bae et al., 2010; So et al., 2021). EGCG treatment resulted in only short fibrils (likely residual sonicated PFF seeds) whereas both proanthocyanidins and casanthranol still maintained some fibril elongation. Proanthocyanidins treated fibrils were shorter than those in the DMSO control and were clustered into bundles surrounded by spherical-like particles (likely insoluble proanthocyanidins aggregates). Conversely, casanthranol-incubated reactions resulted in fibrils of intermediate length relative to DMSO, but with a distinct fibril morphology (reduced fibril twist and almost double the fibril width).
Due to the proanthocyanidins aggregates observed in the TEM images we used dynamic light scattering (DLS) measurements at concentrations that span both in cellulo and in vitro assays conditions to confirm proanthocyanidins’ solubility. Supplemental Figure S12 shows that proanthocyanidins do not aggregate to significant levels in complete medium up to approximately 5 μM, which is above the MTT treatment concentration. A similar titration was done in PBS to replicate HTS (10 μM) and ThioT (50 μM) assay conditions and particles were only detectable at 50 μM, suggesting solubility issues were present for proanthocyanidins in the ThioT experiment only.
We then performed seeded-aggregation assays of EGCG, proanthocyanidins, and casanthranol titrated across a range of concentrations where the compounds had no DLS signature in PBS. Supplemental Figure S13 shows that each compound has a dose-dependent attenuation of ThioT fluorescence. Due to the minimal response of proanthocyanidins at 1 μM, it is likely that its effect on rescuing PFF-induced cellular deficits in the MTT assay is not due to direct PFF interactions. In contrast, EGCG has a significant effect on seeded aggregation at or below the EC50 observed in the MTT assay.
Proanthocyanidins as Viable Tool Compounds for Future aSyn-O/M FLT-FRET HTS Campaigns
The creation of a stable-inducible aSyn-O/M FLT-FRET biosensor cell line facilitates scaling up of biosensor production for HTS campaigns of large compound libraries. Having consistent plate-to-plate variability is essential to maintain good assay quality. We determined the aSyn-O/M HTS assay quality by calculating the Z-factor (Equation 2; where a Z-factor >0.5 indicates excellent assay quality) using the functionally active hit compound proanthocyanidins as the positive control and DMSO as negative control. Proanthocyanidins FLT response results in a Z-factor of 0.65, indicating excellent assay quality. Proanthocyanidins will provide a useful tool compound for future HTS campaigns with the aSyn-O/M cellular biosensors, monitoring the plate-to-plate biosensor response and assay quality throughout the screen.
Discussion
Transitioning the original aSyn-GFP/RFP cellular FLT-FRET biosensor to the mCyRFP1/mMaroon1 red-shifted FRET pair provides key improvement to the HTS platform, including (1) reduced background fluorescence, yielding improved signal-to-noise; (2) longer donor FLT, further increasing signal-to-noise; and (3) a larger FRET signal window, providing increased dynamic range for compound response. Most importantly, these biosensor improvements resulted in a significantly larger FRET efficiency for the double-fusion (single-construct) aSyn biosensor, allowing us to generate the iO-aSyn-M stable inducible cell line as a stand-alone biosensor cell model that can be readily scaled for large library HTS.
In our HTS campaign with the Selleck library, there was notable overlap between hit compounds that displayed an aSyn FLT-FRET dose response and compounds that attenuated aSyn fibrillization. Two compounds in particular—proanthocyanidin and casanthranol—also demonstrated rescue of aSyn PFF-induced cellular pathology. Screening drug libraries that have known MOA have the added benefit of providing insight into potential pathways that are being affected within the biosensor.
Proanthocyanidins are a heterogeneous mixture of oligomeric and polymeric polyphenols generated via the flavonoid biosynthesis pathway (Dixon et al., 2005). Proanthocyanidins have been shown to improve pathological phenotypes associated with Parkinson's in MPTP cell and mouse models, via inhibition of ROS generation and subsequent c-Jun N-terminal kinase 1 (JNK) activation (Chen et al., 2018). In addition, proanthocyanidins have also shown neuroprotective effects in oxidative stress models induced by rotenone in SH-SY5Y cells and rats (Ma et al., 2018; Witucki et al., 2022) and have been shown to cross the blood–brain barrier (BBB) in in vivo models of neurodegeneration (Ahn et al., 2011; Zhen et al., 2014). The second-most potent functional hit, casanthranol, is a mixture of polyphenolic compounds that are all part of the anthraquinone glycosyl family, which include emodin and aloin. Casanthranol has a known MOA as a stimulatory laxative with limited bioavailability through the GI tract. Emodin and aloin have been shown to be neuroprotective and display anti-inflammatory properties, acting through the PI3 K/AKT/mTOR and NF-κB pathways (Park et al., 2009; Pengjam et al., 2016; Xian et al., 2021). There is limited pharmacokinetic characterization on casanthranol beyond oral administration. However, emodin has also been shown to be BBB penetrant in in vivo studies (Xian et al., 2021). Lastly, both proanthocyanidins and casanthranol have been shown to activate the fast-acting antioxidant Keap1/Nrf2 axis, which could explain the protective effects of these two compounds on prophylactic and simultaneous treatment with PFFs (Kaspar et al.; Ren et al., 2023; Tambe et al., 2023).
Due to their capacity to disrupt aSyn fibrillization, rescue cellular viability, and favorable BBB penetrance, both proanthocyanidins and casanthranol warrant further investigation as potential therapeutic interventions for alpha-synucleinopathies. Although beyond the scope of this study, characterization of the substituents of both polyphenolic mixtures could improve the potency and extent of rescue as well as elucidate compound properties required for anti-aggregation and cytoprotective effects.
We are not certain of the specific oligomeric states that are present in our cellular aSyn biosensors. FLT-FRET provides an ensemble average of FLT from all assemblies and conformations that contain a donor XFP. There are currently no existing cellular biosensors that can distinguish between different aSyn oligomeric states beyond the dimer (through biomolecular fluorescence complementation, BIFC) (Dimant, 2014; Moussaud et al., 2015) or fibril (via ThioT/S staining). In our previous aSyn FLT-FRET study we demonstrated that our cellular biosensors monitored nonfibril aSyn assemblies (Braun et al., 2021). This uncertainty is compounded by the use of a double-fusion biosensor, where changes in FLT-FRET can be driven by either conformational changes within a monomer, conformational changes within an oligomer, or modulation of oligomer formation. Nevertheless, the sensitivity afforded by FLT-FRET allows us to use our platform as a first-pass screening filter to identify molecules that perturb the ensemble of aSyn assemblies. As shown here, further secondary, and functional assays provide us with insight into whether hits are anti-fibrillogenic and are able to rescue aSyn induced disease associated phenotypes.
Further considerations need to be taken into account to address the temporal resolution of compound-induced effects. In our FLT-FRET HTS platform, we currently monitor changes in aSyn FRET following a 2-hour incubation in the drug assay plates. Although this treatment duration is relatively short compared to the many cellular mechanisms affected in alpha-synucleinopathies, it allows us to focus on a shorter time window for the identification of compounds directly modulating the aSyn assemblies. However, it is important to acknowledge that this approach may potentially overlook compounds with interesting MOA that require a longer timescale to manifest. In future HTS campaigns, the integration of our target-based HTS (FLT-FRET) with a phenotypic measure would be advantageous, increasing the likelihood of finding functional, validated hits.
Methods
Molecular Biology
Plasmids encoding aSyn-O, aSyn-M, and O-aSyn-M were designed and purchased from (VectorBuilder, Chicago, IL) and included the removal of aSyn's internal BamH1 restriction site to facilitate downstream subcloning (see Supplemental Figure S1). Inducible biosensor constructs were created by subcloning aSyn-O and O-aSyn-M into an empty vector pTT22 plasmid via restriction enzyme digestion using Nhe1 (5′-end) and BamH1 (3′-end) sites. All biosensor plasmids constructs were sequenced for confirmation (ACGT, Wheeling, IL).
Cell Culture
HEK293 (ATCC, USA) were cultured in phenol red-free Dulbecco's modified Eagle medium (DMEM, Gibco) supplemented with 2 mM
Selleck Library and Liquid Handling
The Selleck library contains 2684 compounds that span commercial FDA approved drugs that have well-characterized activities with widely described biological effects. The library is originally formatted in 96-well mother plates and dispensed across three 1536-well flat, black-bottom polypropylene plates at 10 μM final concentration/well (5 μl for 1536-well) using an automated Echo acoustic liquid dispenser from Labcyte (Sunnyvale, CA, USA). DMSO (matching %v/v) was loaded as in-plate no-compound negative controls to make a total of 704 wells in the 1536-well plate format. The plates were sealed and stored at -20 °C until use. Cells were harvested and diluted to 1e6 cells/ml.
One week prior to screening, iaSynO and iOaSynM cells were plated into 3 × 15 cm plates at 3 × 106 cells per plate. After 24 hours medium cells were induced through medium changed supplemented with Dox (0.5 μg/ml). Medium was changed again after 3 days, and induced cells were harvested after 5 days total induction time. Expression of O-aSyn and O-aSyn-M biosensors were confirmed by fluorescence microscopy prior to each screen (Leica DMI3000B brightfield microscope).
The cells were harvested from the 15 cm plates by incubating with TrypLE (Invitrogen) for 2 minutes, washed three times in PBS by centrifugation at 200g for 5 minutes and filtered using 70 μm cell strainer (BD Falcon). Cell viability prior to screen was assessed using a trypan blue assay and was confirmed to be at least 80%. Cells were then counted and diluted to 1.0 × 106 cells/ml. After resuspension and dilution in PBS, the biosensor cells were constantly and gently stirred using a magnetic stir bar at room temperature, keeping the cells in suspension, and evenly distributed to avoid clumping.
On each day of screening, the compound plates were equilibrated at room temperature (25 °C). During screening, cells (5 μl/well for 1536-well) were dispensed by a Multidrop Combi liquid dispenser from Thermo (Pittsburg, PA, USA) into the assay plates containing the compounds and allowed to incubate at room temperature for 90 minutes before readings were taken by the fluorescence lifetime plate reader and spectral unmixing plate reader (Fluorescence Innovations, Inc), which were provided by Photonic Pharma LLC (Lo et al., 2017; Schaaf et al., 2016). Reproducible FRET hits for retesting were purchased from Tocris (Minneapolis, MN), Sigma (St. Louis, MO), or Invitrogen (Carlsbad, CA), depending on availability.
FLPR Experiments, HTS, and Fluorescence Lifetime Data Analysis
As described previously (Lo et al., 2017; Schaaf et al., 2016, 2018), time-resolved fluorescence waveforms for each well were fit with single-exponential decays using least-squares minimization global analysis software to give donor–acceptor lifetime (τDA) and donor-only lifetime (τD). FRET efficiency (E) was then calculated based on Equation (1).
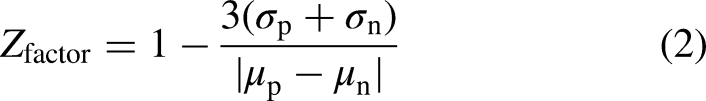
False positives were flagged based on interference from compound fluorescence by a set of stringent filters based on analysis of the spectral waveforms of each well from the Selleck HTS (Lo et al., 2017; Schaaf et al., 2016). After removal of fluorescent compounds, a histogram of the FRET distribution from all compounds in the screen was plotted and fit using a Gaussian function to obtain the mean (µ) and standard deviation (σ, SD). A hit was defined as a compound that decreased the FRET efficiency by 4SD (4σ) relative to the mean (µ).
FRET Concentration-Response Assay
All hit compounds were tested in a FRET concentration-response assay. Each compound was dissolved to 10 mM stock in DMSO which was serially diluted in 96-well mother plates. Each hit was re-screened for FRET dose response across eight concentrations (1 nM to 50 μM). Compounds (0.5 μl) were transferred from mother plates into assay plates using a Mosquito HV liquid handler (TTP Labtech Ltd, UK). The preparations for aSyn FRET biosensors were carried out as above.
Western Blot Analysis
To test the expression of aSyn FRET biosensors, iaSynO and iOaSynM biosensor cells were plated in a six-well plate at a density of 1 × 105 cells/well and induced for 1 to 7 days with 0.5 μg/ml Dox. Medium was changed every 3 days. Cells were lysed for 30 minutes on ice with radioimmunoprecipitation assay (RIPA) lysis buffer (Pierce RIPA buffer, Thermo Fisher Scientific) containing 1% protease inhibitor (Clontech, Mountain View, CA) and 1% phosphatase inhibitors (Millipore Sigma), and centrifuged at 15,000g at 4 °C for 15 minutes. The total protein concentration of lysates was determined by bicinchoninic acid (BCA) assay (Pierce), and equal amounts of total protein (60 μg) were mixed with 4 × Bio-Rad sample buffer and loaded onto 4% to 15% Tris–glycine sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) gels (Bio-Rad, Hercules, CA). Proteins were transferred to supported nitrocellulose membrane and probed using Syn101 antibodies against aSyn (BD labs, San Jose, CA). Blots were imaged on using a ChemiDoc MP imager and analyzed using Image Lab (Bio-Rad, Hercules, CA).
PFF-Induced Toxicity Model in dbcAMP Differentiated N2a Cells Monitored via MTT
N2a cells were plated at 5000 cells/well in a 96-well plate (Cell+) in full media (EMEM, 10% FBS, 1% penicillin/streptomycin). After 24 hours, media was switched to differentiation media (DMEM, 0.5% FBS, 1 mM dbcAMP) and incubated for 24 hours. After the differentiation period, cells were pre-treated with drugs at the different final concentrations (1 µM for all drugs and titrations for hits that showed significant rescue in this assay) and DMSO control. After 1.5 hours of drug pre-treatment, cells were treated with 1 µg/ml of sonicated aSyn PFFs (sonicated for 1 minute with 1 second ON and 1 second OFF pulses at 50% amplitude) or PBS control. After 48 hours of incubation, 10 µl of 5 mg/ml MTT reagent was dispensed into each well of the 96-well plates. Plates were incubated for 4 hours; media was removed, and formazan crystals were dissolved in 100 µl of DMSO. Plates were then incubated while shaking for 15 minutes, and absorbance was measured at 570 nm using a Molecular Devices Spectramax i3 plate reader. MTT measurements were normalized to the global PBS-only treated controls across all plates.
For treatment paradigm experiments (pre-PFF, simultaneous and post-PFF), N2a cells were plated using the same conditions described above, with the only difference in timing of drug addition to cells (−1.5 hours for pre-PFF, 0 hours for simultaneous and +1.5 and +24 hours for post-PFF, relative to PFF addition). EGCG, proanthocyanidins and casanthranol were tested at 1 µM, 1 µm and 50 µM, respectively.
PFF-Seeded Aggregation Monitored via Thioflavin T
Full-length monomeric aSyn was expressed and purified as described previously (Kamboj et al., 2021; Nathan Kochen et al., 2022). PFFs were generated by incubating 2 mg/ml monomeric aSyn and a single 3 mm borosilicate glass bead for 90 hours (orbital shaking conditions and 37 °C) (Nathan Kochen et al., 2022).
Aliquots of monomeric aSyn were quick thawed from −80 °C and centrifuged at 21,000g for 30 minutes and top 95% supernatant volume transferred to new tubes. Monomeric aSyn was diluted to 15 µM (0.21 mg/ml), 25 µM Thioflavin T (EMD Millipore) and 5% PFF seeds (0.75 µM) in PBS. 50 µl of the aSyn seeded master solution was then pipetted in triplicate into a 384-well flat black-bottom polypropylene plate (PN 781209 Greiner Bio-One) containing drugs (final concentration of 50 µM, 1:3.33 monomer to drug ratio) and DMSO controls. The 384-well plate was then covered with optically transparent plastic film (Excel Scientific) to prevent sample evaporation. Plates were incubated at 37 °C under continuous orbital shaking (medium setting). Fluorescence was measured using a Spectramax i3 plate reader (Molecular Devices) every 20 minutes with a 440/10 nm excitation and 490/10 nm emission. Fibrillization extent was calculated by subtracting the average ThioT signal of the initial and last hour of aggregation.
TEM Imaging of Seeded Aggregation Reactions With Hit Compounds
Seeded aggregation reactions with 15 μM monomer and 0.75 μM PFF were supplemented with DMSO control or 50 μM EGCG, 50 μM proanthocyanidins or 50 μM casanthranol. 3 μl sample was deposited onto a glow discharged EM grid and followed by negative staining with 2% uranyl acetate. The images were acquired with a FEI Tecnai Spirit BioTWIN TEM operated at 120 kV.
Dynamic Light Scattering (DLS) Analysis of Proanthocyanidins
DLS measurements of proanthocyanidins were performed at 0.1, 1, 5, 10, and 50 μM concentrations in DMEM + 10% FBS (constant 0.5% final DMSO across all samples) using a Brookhaven 90 Plus particle size analyzer. Measurements were conducted after incubating samples for 30 minutes at room temperature (covered to protect from dust particles) using a viscosity of 0.94 cP (Fröhlich et al., 2013), refractive index of 1.345 (Alwani et al.), wavelength of 658 nm and scattering angle of 90°.
Statistical Analysis
Data are shown as mean ± standard deviation unless stated otherwise. Statistical analysis for FLT and FRET experiments were conducted with one-way ANOVA using Prism v9.0 (GraphPad, USA) to determine statistical significance for all experiments. Values of p-value < .05 were considered statistically significant.
Supplemental Material
sj-docx-1-asn-10.1177_17590914231184086 - Supplemental material for Advancements in a FRET Biosensor for Live-Cell Fluorescence-Lifetime High-Throughput Screening of Alpha-Synuclein
Supplemental material, sj-docx-1-asn-10.1177_17590914231184086 for Advancements in a FRET Biosensor for Live-Cell Fluorescence-Lifetime High-Throughput Screening of Alpha-Synuclein by Anthony R. Braun, Noah Nathan Kochen, Samantha L. Yuen, Elly E. Liao, Razvan L. Cornea, David D. Thomas and Jonathan N. Sachs in ASN Neuro
Footnotes
Abbreviations
Acknowledgements
The authors thank Dr. Nagamani Vunnam from the Sachs Group for technical support and discussions. Compound dispensing was performed at the UMN Institute of Therapeutic Drug Discovery and Development (ITDD) High-Throughput Screening Laboratory. Fluorescence spectroscopy was performed at the UMN Biophysical Technology Center and at the adjacent Photonic Pharma laboratory. TEM imaging was performed at the University of Minnesota's Characterization Facility. This research uses technology patented by the University of Minnesota, with an exclusive commercial license to Photonic Pharma LLC. This study was supported by U.S. National Institutes of Health (NIH) grants to J.N.S. (NINDS R01NS117968) and to J.N.S and D.D.T (NIA SBIR 1R43AG063675).
Author Contributions
A.R.B. designed and conducted the FLT-FRET experiments and co-directed the study. NNK conducted the aggregation, MTT, TEM, and DLS experiments. A.R.B and N.N.K wrote and edited the manuscript. A.R.B and S.L.Y. conducted transient aSyn-O/M FLT-FRET experiments. E.E.L consulted on functional assay optimization and edited the manuscript. R.L.C. and D.D.T. consulted on the FLT-FRET biosensor engineering and experiments and assisted with manuscript editing. J.N.S. co-directed the study and edited the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.D.T. and R.L.C. hold equity in and serve as executive officers for Photonic Pharma LLC, a company that owns intellectual property related to technology used in part of this project. These relationships have been reviewed and managed by the University of Minnesota in accordance with its conflict of interest polices. The remaining authors declare no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Neurological Disorders and Stroke, National Institute on Aging (grant number R01NS117968, SBIR 1R43AG063675).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.