Abstract
Vesicles mediate the trafficking of membranes/proteins in the endocytic and secretory pathways. These pathways are regulated by small GTPases of the Rab family. Rab proteins belong to the Ras superfamily of GTPases, which are significantly involved in various intracellular trafficking and signaling processes in the nervous system. Rab11 is known to play a key role especially in recycling many proteins, including receptors important for signal transduction and preservation of functional activities of nerve cells. Rab11 activity is controlled by GEFs (guanine exchange factors) and GAPs (GTPase activating proteins), which regulate its function through modulating GTP/GDP exchange and the intrinsic GTPase activity, respectively. Rab11 is involved in the transport of several growth factor molecules important for the development and repair of neurons. Overexpression of Rab11 has been shown to significantly enhance vesicle trafficking. On the other hand, a reduced expression of Rab11 was observed in several neurodegenerative diseases. Current evidence appears to support the notion that Rab11 and its cognate proteins may be potential targets for therapeutic intervention. In this review, we briefly discuss the function of Rab11 and its related interaction partners in intracellular pathways that may be involved in neurodegenerative processes.
Introduction
Rab proteins (20−25 kDa) are monomeric G proteins that form the largest group of the Ras superfamily, with 70 members in humans. Most of these proteins are expressed ubiquitously, and some are tissue-specific. Eleven Rab11 proteins have been found in yeast, 33 in Drosophila melanogaster (Zhang et al., 2007), 29 in Caenorhabditis elegans (Pereira-Leal & Seabra, 2000), and 57 in Arabidopsis thaliana (Vernoud et al., 2003). These proteins play an important role in regulating membrane trafficking during vesicle formation, budding, motility, tethering, and finally fusion (reviewed by Bhuin & Roy, 2014). The Rab11 subfamily consists of three members, Rab11a, Rab11b, and Rab11c/Rab25. Each Rab protein has a specific location related to its physiological functions. Rab11a is ubiquitously expressed, Rab11b is enriched in the brain, testis, and heart, and Rab25/rab11c is restricted to epithelia, as studied in mouse cells (Bhartur et al., 2000). Rab11 was first recognized as a marker for recycling endosomes. It regulates vesicles via the recycling endosome compartment and early endosomes to the trans-Golgi network and plasma membrane (Ullrich et al., 1996).
Rabs are molecular switches that regulate intracellular transport in eukaryotes. Rab11 cycles between an inactive GDP (guanosine diphosphate)-bound state and an active GTP (guanosine triphosphate)-bound state (Grosshan et al., 2006). In its inactive state, Rab11 forms a complex with RabGDI (Rab guanine dissociation inhibitor) and remains in the cytoplasm to avoid nonspecific activation. Once it receives signals for activation, the RabGDI complex is transported to the membrane and undergoes nucleotide exchange of GDP for GTP mediated by guanine nucleotide exchange factor (GEF). Activation is followed by recruitment of host effectors, which perform further functions in membrane trafficking. Once cycling is complete, GTP bound to Rab11 is hydrolyzed by GTPase-activating protein (GAP) and resumes its inactive state (Dirac-Svejstrup et al., 1997; Ingmundson et al., 2007; Machner & Isberg, 2007; Sivars et al., 2003). Rab11 directs various cargo proteins and membranes through the long loop of endosomal recycling, thereby regulating vesicle trafficking pathways and ensuring the transport of endosomes from the plasma membrane to the endosomal recycling compartment and vice versa (Stenmark, 2009). The functions of Rab11 are carried out through its interaction with various effector proteins. In this context, the Rab11-family interacting proteins (FIPs), which possess a Rab-binding domain (RBD) and mediate GTP-dependent interactions with Rab GTPases, have been attributed a special role. These proteins are known to facilitate Rab11-dependent vesicle recycling (Baetz & Goldenring, 2013). A schematic illustration of the Rab11 GTPase cycle is shown in Figure 1.
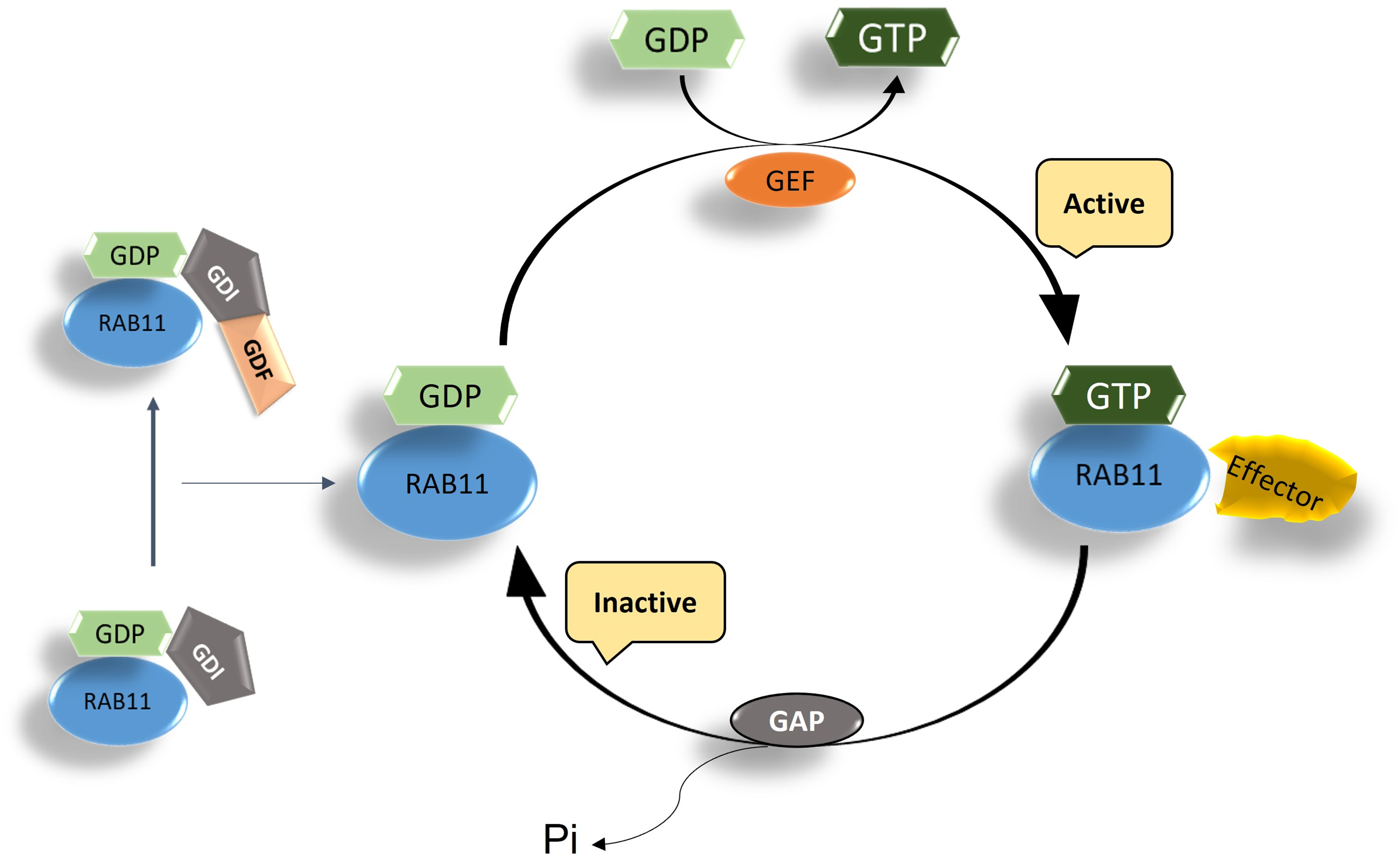
Schematic representation of Rab11 GTPase cycling between active and inactive states. GDF (GDI dissociation factor) interacts with GDI bound to Rab11GDP to free Rab11GDP and allow its activation. GEF then exchanges GDP for GTP, converting the inactive Rab11 to its active state. Rab11GTP is now ready to interact with the effector proteins.
In the past decade, Rab11 proteins and the components of Rab11-mediated intracellular trafficking and signaling pathways have emerged as molecules possibly involved in etiology of some neurodegenerative diseases. Therefore, the presumed role of these proteins in neurodegeneration should be better addressed by future studies.
Rab11 Proteins
The Rab11 subfamily consists of three members, Rab11a, Rab11b, and Rab11c/Rab25. A brief characterization of these proteins is given below.
Rab11a
Rab11a is ubiquitously expressed. It is a vastly studied and characterized member of the Rab11 family. It localizes in the endocytic recycling compartments (Ullrich et al., 1996). The role of Rab11a in controlling the trafficking of receptors from the endoplasmic reticulum compartment (ERC) to the plasma membrane has been extensively discussed by Maxfield & McGraw (2004). It transports cargoes from peripheral sorting endosomes (SEs) to recycling endosomes (REs) (Horgan et al., 2010). In the trans-Golgi network (TGN) Rab11a routes vesicle trafficking between the TGN and ERC or plasma membrane. It plays a vital role in controlling tissue homeostasis during embryonic development and the postnatal period. Total knockout of Rab11a was found to be embryonic lethal in a mouse model. Interestingly, brain-specific Rab11a knockout showed no overt abnormalities in brain architecture, but, on the other hand, intestine-specific Rab11a knockout resulted in mislocalization of apical proteins in the intestine (Sobajima et al., 2014). This study raises the question of whether other family members such as Rab11b or Rab11c/Rab25 might be able to compensate for the normal neuronal phenotype in the brain. In this context, a closer examination of the knockout mouse models of Rab11b or Rab11c might be an important consideration. It should also be mentioned here that Rab11a affects several cellular processes, including cytokinesis, phagocytosis, cell migration, immunological synapses, etc. (Assaker et al., 2010; Fielding et al., 2005; Gorska et al., 2009).
Rab11b
Rab11b is abundant in the brain, heart, and testis and is also enriched in the ERC, which recycles the transferrin receptor. In contrast to Rab11a, Rab11b is localized to the apical pericentriolar region in the gastric parietal cells (Lapierre et al., 2003). It also controls the trafficking of the cystic fibrosis transmembrane conductance regulator (CFTR) in polarized epithelial cells (Silvis et al., 2009). In addition, it regulates insulin granule exocytosis (Sugawara et al., 2009). Lamers et al. (2017) reported that certain dominant de novo missense mutations in Rab11b with a distinct brain phenotype impair intellectual abilities and are associated with absent speech, epilepsy, and hypotonia. This suggests the important role of Rab11b in the brain and may be consistent with the above speculations about its possible compensatory role in the case of Rab11a dysfunction.
Rab11c/25
Rab11c or Rab25 is highly expressed in polarized epithelial cells of the lung, kidney, and gastrointestinal mucosa (Goldenring et al., 1993). Loss of Rab25 promotes colorectal adenocarcinomas suggesting that it may play a critical role as a tumor suppressor or oncogene in intestinal epithelial cells (Nam et al., 2010). Rab25 promotes the development and neoplastic transition of skin squamous cell carcinoma through dysregulation of integrin trafficking (Jeong et al., 2019). In addition, it was found that downregulation of Rab25 is associated with a subtype of breast cancer, making this protein therapeutic target in luminal B breast cancer tumors (Belhadj et al., 2020). While it is evident that Rab25 plays an important role in cancer, its function in neurodegenerative diseases remains to be explored.
Rab11-FIPs and Their Functions
The downstream machinery of Rab11 GTPases identified an evolutionarily conserved protein family known as the Rab11-family interacting proteins (FIPs). They consist of five members: Rip11/FIP5/Gaf-1/Gaf1b, FIP2, RCP/FIP1/FIP1C (Rab coupling protein), FIP3/arfophilin/eferin, and FIP4/arfophilin-2 (Hales et al., 2001; Lindsay et al., 2002; Prekeris et al., 2000). The C-termini of the FIPs include a highly conserved Rab11-binding domain consisting of 20 amino acids. The FIPs are divided into two classes depending on the presence of a calcium-binding domain in the N-terminus. Class I consists of FIP1, FIP2, and FIP5, while class II consists of FIP3 and FIP4 (Wallace et al., 2002). These proteins can bind to motor proteins such as myosin V, kinesin, and dynein to regulate the endosomal recycling process, depending on their specificity of protein, lipid, and calcium binding abilities and their post-translational modification by phosphorylation.
As shown in Figure 2, Rab11-FIP proteins include protein domains/motifs such as the Rab-binding domain (RBD) that mediates GTP-dependent interactions with Rab GTPases. C2 or EF-hand domains of the FIPs confer sensitivity to calcium and phospholipid binding. An α helical coiled-coil structure of these proteins mediates homodimerization by binding the switch I and switch II regions of two Rab11a-GTP molecules (Eathiraj et al., 2006; Jagoe et al., 2006). The switch I and switch II regions are nucleotide-sensitive regions that are the major binding sites for Rab11 effectors (Pasqualato et al., 2004). These proteins regulate various membrane trafficking events, which can be classified into the following three actions: cargo recycling, membrane delivery, and associating motor proteins. The functions of the FIPs are listed in Table 1.
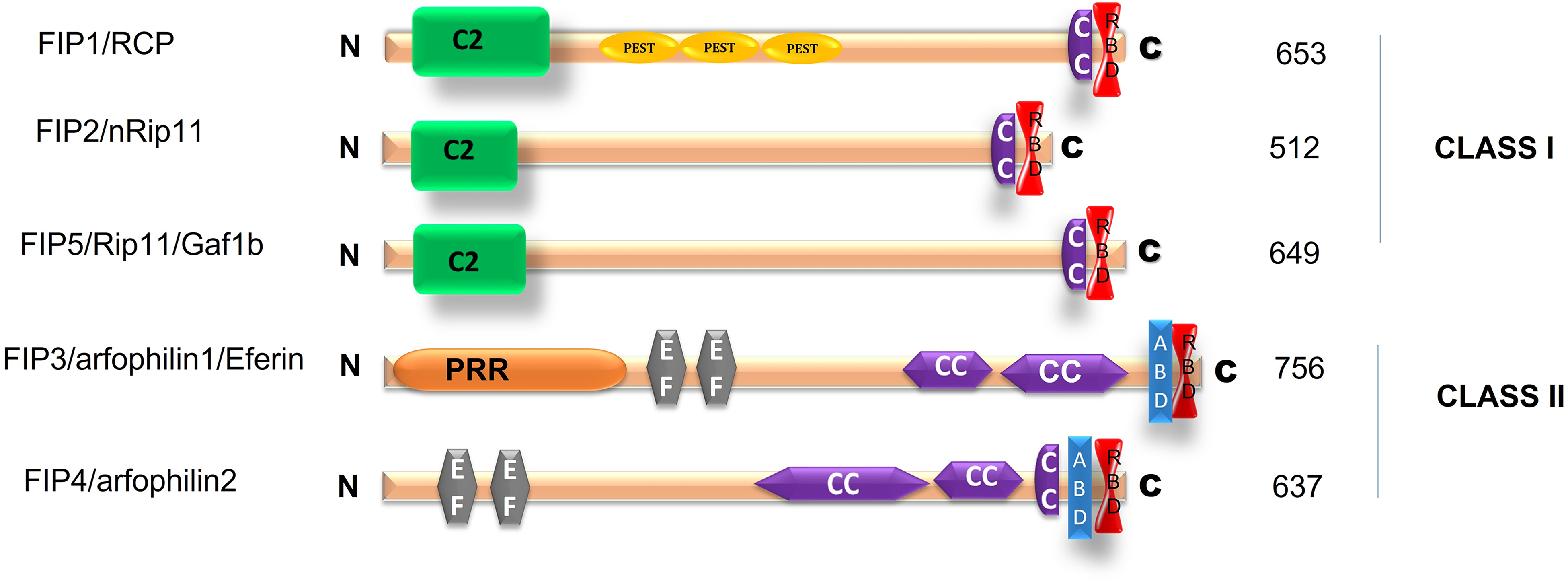
Schematic representation of Rab11-FIPs. C2 = C2 domain, EF = EF-hand domain, PRR = protein-rich region, CC = coiled coil domain, ABD = Arf6 binding domain, RBD = Rab11 binding domain.
The Role of Rab11-FIP Binding Domains
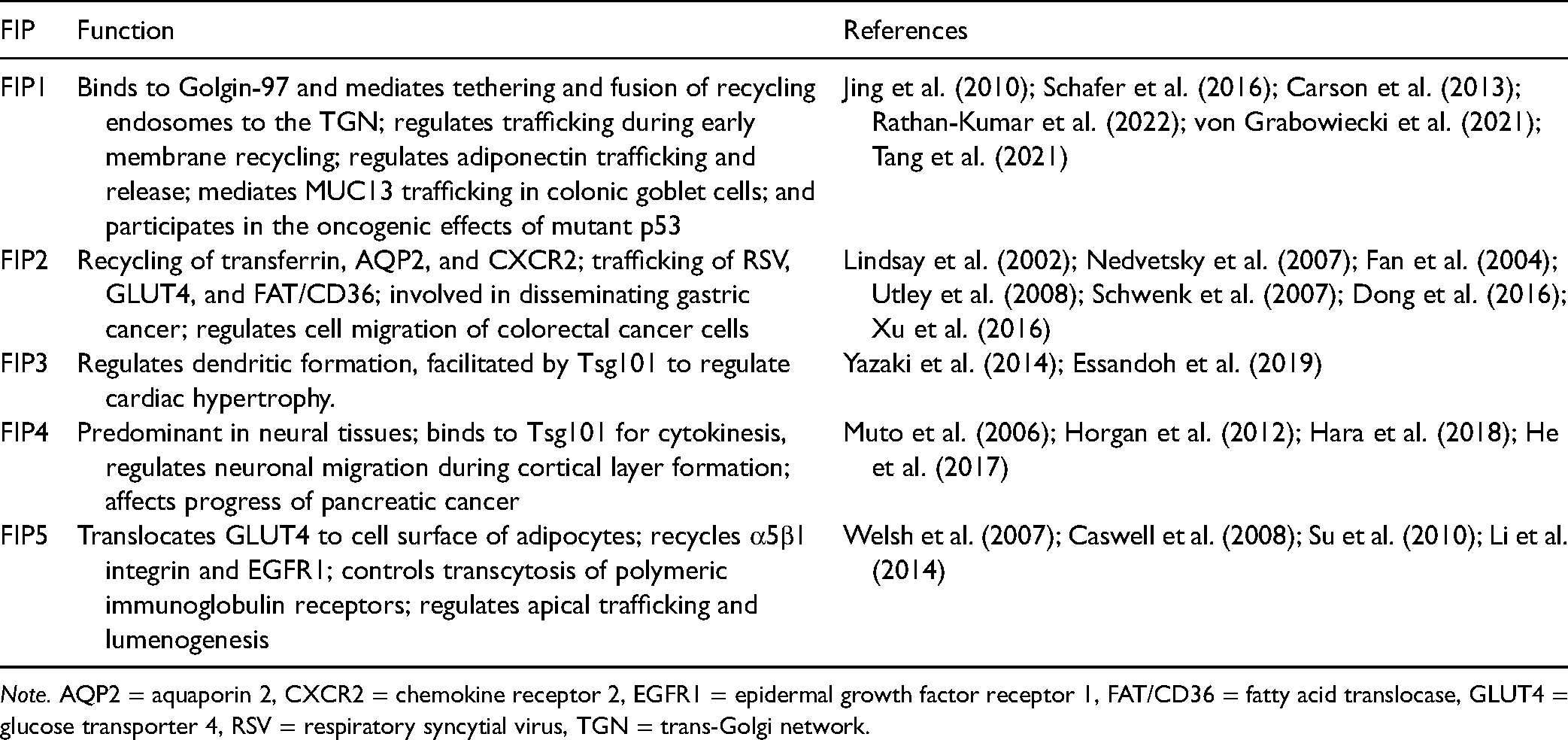
Note. AQP2 = aquaporin 2, CXCR2 = chemokine receptor 2, EGFR1 = epidermal growth factor receptor 1, FAT/CD36 = fatty acid translocase, GLUT4 = glucose transporter 4, RSV = respiratory syncytial virus, TGN = trans-Golgi network.
The N-terminal C2 domains of Class I Rab11-FIPs regulate most recycling cargoes to docking sites on the phospholipid-enriched plasma membrane (Lindsay & McCaffrey, 2004). Class II has been shown to play a key role in supplying endosomal material during cell division. Both members of class II bind to a member of the Arf6 GTPases. Arf6 recruits both FIP3 and FIP4 to the cleavage furrow and midbody during cytokinesis (Fielding et al., 2005; Simon et al., 2008). For transport from the endoplasmic reticulum to the plasma membrane or vice versa, FIPs associate motor proteins with members of the Rab11 subfamily (Figure 3). FIP1 interacts directly with the motor protein myosin Vb to regulate recycling of Rab11a vesicles. Mutation of Rab11-FIP1 causes trafficking between early and late endosomes to stop at certain points and may destabilize the recycling system. This may indicate that there are multiple pathways with recycling endosomes where dynamic trafficking decisions are made (Schafer et al., 2014). Interestingly, a PEST (Pro-Glu-Ser-Thr) motif exists in FIP1 that targets a protein for proteolytic degradation, and once the recycled endosome reaches the plasma membrane, FIP1 is inactivated by calpain (Marie et al., 2005). It would be important to determine how FIP1 interacts with the proteins using the PEST motif. FIP3 binds to kinesin 1 and dynein 1 to mediate endosomal recycling in the endosomal recycling compartment (ERC). However, it is not yet clear how this regulation is maintained (Horgan et al., 2010; Simon & Prekeris, 2008). FIP2 and FIP5 are known to be regulated by phosphorylation, and the C2 domain of FIP5 binds to phospholipids in a magnesium-dependent manner. FIP5 associates with kinesin 2 (a primary FIP5 endosome motor) along with several partner proteins to move endosomes from the centrosome to the cleavage furrow during apical lumen formation (Li et al., 2014). Rab11 interacts with several cargoes and interacting proteins listed in Supplemental Tables S1 and S2, respectively.
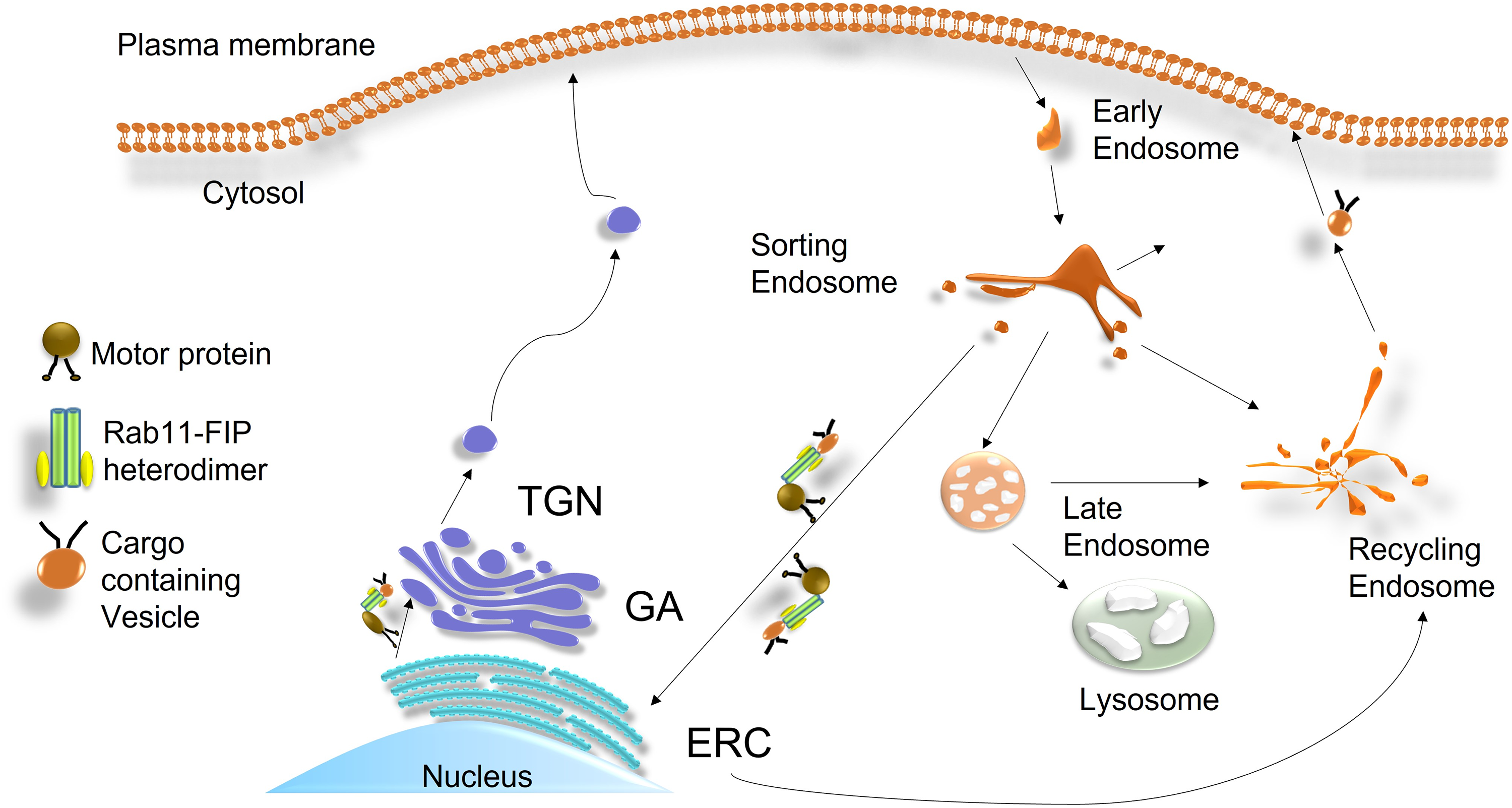
Illustration of the endocytic pathway and the role of Rab11-FIPs in vesicle transport. Rab11-FIP heterodimers containing cargo are bound to motor proteins for transport from the endoplasmic reticulum compartment (ERC) to the plasma membrane or from the plasma membrane to the ERC.
The Role of Rab11 in Recycling Endosomes
Rab11 protein is involved in several cellular trafficking pathways. It is localized in the TGN and post-Golgi vesicles of the secretory pathway, where it controls recycling endosomes. Dysfunctional Rab11 mutants cause inhibition of transferrin recycling from late recycling endosomes (Ullrich et al., 1996). In polarized epithelial cells, Rab11 has ben shown to be localized in the pericentriolar endosomal compartments (Casanova et al., 1999). A recent analysis suggests that Rab11 is dynamically distributed in neuronal cells to ensure proper protein delivery during neuronal development (Siri et al., 2020). It plays an important role in regulating the transport of numerous receptors/cargoes that are constitutively recycled (Supplemental Table S1). Some cargoes such as cholesterol and transferrin receptors are found in all cell types, cargoes such as AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid) receptors are important for neurons (Correia et al., 2008). Rab11-dependent pathways represent a key component involved in neuronal migration, which is critical for mammalian brain formation. In a study using in utero electroporation, the role of endocytic pathways in neuronal migration in the developing cerebral cortex was investigated in vivo (Kawauchi et al., 2010). These authors observed dysregulation of migration when the Rab11-dependent pathway was suppressed. They found that recycling of Rab11-dependent N-cadherin is important for neuronal migration during brain maturation in mammals. The developing mammalian brain requires AMPA receptor trafficking and dendritic spine growth. Rab11 forms complexes with motor proteins that direct vesicles to subcellular sites of different polarity via microtubule tracks. Actin-dependent myosin Vb has been shown to mobilize AMPA receptors for postsynaptic plasticity in neurons. Myosin Vb interacts with Rab11-linked recycling endosomes and disruption of this interaction prevents AMPA receptor insertion (Wang et al., 2008). AMPA receptors important for long-term potentiation (LTP) are trafficked via the Rab11 bound endocytic recycling pathway from dendritic shafts to spines using Rab11 effectors. To maintain LTP, exocytosis of AMPA receptors from recycling endosomes to extrasynaptic sites must occur (Correia et al., 2008; Park, 2018; Park et al., 2004). In addition, synaptic trafficking of AMPA receptors also plays an important role in long-term depression (LTD) (Bacaj et al., 2015). In general, Rab11 provides exocytic processes at the TGN and recycling processes at the pericentriolar recycling endosome. Rab11 is closely related to the exocyst complex (exocyst, a downstream effector of Rab11, is a protein complex that mediates vesicles at the plasma membrane for exocytosis). Rab proteins and their interaction with exocyst have been studied in yeast to understand the mechanism of polarized exocytosis. Das and Guo (2011) suggested that there are a number of protein interactions between Rab11 and the exocyst complex involving the tethering of secretory vesicles to the plasma membrane. The molecular mechanism involving the Rab cascade is still unclear for mammalian homologs.
The Role of GEFs and GAPs in the Secretory Pathway
It is known that the GDP/GTP binding state of Rab11 determines its interaction with its effectors. The GDP/GTP cycle is regulated by GAPs and GEFs. Whereas GAPs catalyze the hydrolysis of GTP to GDP, GEFs mediate the exchange of GDP for GTP and thus control the cellular functions of small GTPases.
Rab11 GEFs
GEFs for several Rab proteins are still unknown. One report suggests that huntingtin (Htt, a protein involved in Huntington′s disease), which is located at endosomal membranes, may participate in guanine nucleotide exchange on Rab11. However, it does not interact directly with Rab11 but in complex with Rab11 GEF. Moreover, the mutant form of Htt inhibits Rab11 activity and impairs vesicle formation on recycling endosomes (Li et al., 2008, 2009). In 2012, Xiong et al. reported that Crag is a Rab11 GEF important for trafficking of light-induced rhodopsin in Drosophila photoreceptor cells. Crag consists of the calmodulin-binding domain, and in the presence of calcium, its GEF activity promotes the release of GDP (Xiong et al., 2012). A motor protein known as myosin Vb plays a key role in recycling of endosomes to the plasma membrane. It binds to calmodulin via EF-hand calcium-binding motifs encoded by the class II of Rab11-FIPs. This also suggests that calcium is an essential element for Rab11 function (Welz et al., 2014). Crag has three mammalian homologs, DENND4A, B, and C, with DENND4B having the highest homology. Therefore, DENND4 proteins deserve further exploration. Parcas (Pcs), another GEF involved in rhodopsin transport in Drosophila photoreceptors, has been reported to play an important role in eye development (Otsuka et al., 2019). Rab11-interacting protein-1 (REI-1) was discovered to be the GEF for Rab11 in C. elegans, and it is important during cytokinesis in the embryo. REI-1 and its human homolog SH3-binding protein 5 (SH3BP5) were found to have strong GEF activity toward Rab11a in vitro (Sakaguchi et al., 2015). SH3BP5 interacts with the N-terminal region, switch I, interswitch, and switch II of Rab11a (Goto-Ito, Morooka et al., 2019). Interestingly, the Rab-GEF domains of REI-1 do not resemble DENN (Barr & Lambright, 2010). Figure 4 shows that the zinc-binding motif (Btk-type zinc finger) is conserved from C. elegans to humans.
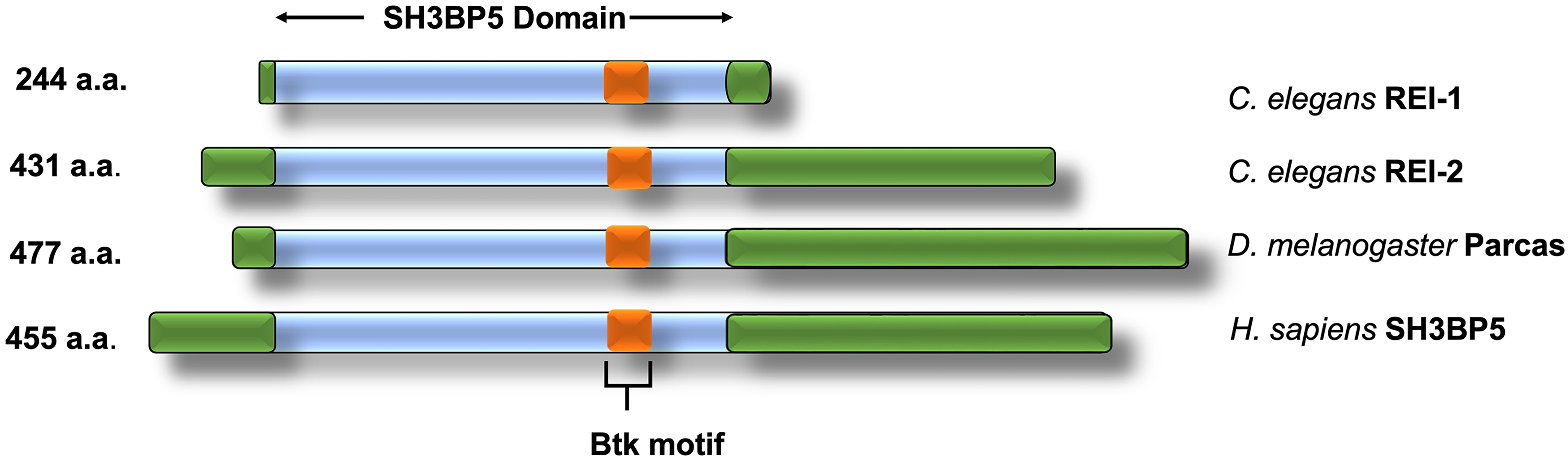
Representation of different Rab11 GEF protein domains and Btk-binding motifs. These sequences are highly conserved from C. elegans to H. sapiens.
Rab11 GAPs
A group of proteins with the TBC (Tre-2, Bub2, Cdc16) domain includes Rab GAPs, namely TBC1D11, TBC1D15, EVI5, and TBC1D9B (Dabbeekeh et al., 2007; Gallo et al., 2014; Zhang et al., 2005). TBC provides two catalytic residues (arginine, glutamine finger), thereby stabilizing the GTP-hydrolysis transition state (Pan et al., 2006). EVI5 has been extensively studied and shown to be an essential regulator of the proper Rab11 function. EVI5 GAP activity in Drosophila is required for border cell migration in a Rab11-dependent manner. Loss or increase in of EVI5 function impedes cell migration by disrupting Rab11-dependent polarization of active guidance receptors (Laflamme et al., 2012), suggesting tight regulation of recycling endosomes for spatial restriction of receptor tyrosine kinase at the edge of migrating cells. The precise roles of TBC1D15 and TBC1D11 remain unclear. In addition to Rab11, they also activate the GTPases Rab7 and Rab4. Interestingly, overexpression of TBC1D9B reduces the rate of basolateral to apical IgA transcytosis (Rab11a-dependent pathway) but shows no effect on basolateral recycling of transferrin receptors or epidermal growth factor receptor. It reduces the amount of GTP-bound Rab11 and disrupts its interaction with its effector Sec15A (Gallo et al., 2014).
The Role of Rab11 in Transcytosis
A transcellular transport process in polarized cells is referred to as transcytosis, which allows communication between apical and basolateral plasma membrane regions. In developing neurons, polarized membrane trafficking of signaling receptors is essential in response to stimuli such as growth and survival. Figure 5 illustrates transcytosis in polarized and neuronal cells. In polarized cells, transcytosis targets newly synthesized proteins toward the apical surface by internalizing basolateral membrane protein targeted to the apical surface (Anderson et al., 2005). In neuronal cells, newly synthesized molecules are inserted into the soma surface for endocytosis and are targeted to the axon via Rab11-bound recycling endosomes (Ascaño et al., 2009). The recycling endosome acts as a hub and sorts apically directed proteins. Silencing of Rab11FIP5 has been reported to disrupt transcytosis (Su et al., 2010). Another study showed that Rab11-FIP2 regulates transcytosis in MDCK cells through its phosphorylation at Ser-227 by MARK2 (Ducharme et al., 2006) and also controls epithelial cell polarity (Lapierre et al., 2012). In neurons, the transcytotic pathway directs proteins (e.g., L1/NgCAM, VAMP2, and TrkA) from the somatodendritic membrane to the axonal surface (Ascaño et al., 2009; Sampo et al., 2003). Importantly, the endocytic and transcytotic pathways may be impaired in Alzheimer's disease (AD). Reduction of lipoprotein transcytosis to the axon was found to lead to neuron-specific impairment of axonal delivery. Transcytosis of axonal amyloid-beta precursor protein (APP) and lipoproteins from the soma to the axon appears to be strongly dependent on Rab11. Defects in recycling endosomes and knockdown of Rab11 have been shown to reduce in APP density and lipoprotein transcytosis (Woodruff et al., 2016). In addition, axonal β-site APP-cleaving enzyme 1 (BACE1) was observed to initiate APP processing of endosomes in a Rab11-dependent manner by sorting endosomes to axons in hippocampal neurons (Buggia-Prévot et al., 2014), suggesting that manipulation of BACE1 may modulate amyloid-β peptide (Aβ) transport in a Rab11-dependent manner and serve as a potential therapeutic target for the treatment of Alzmeimer′s disease. Macromolecules are also transported from the axonal membrane to the somatodendritic surface via a partially retrograde axonal transport pathway. Glial cell-derived neurotrophic factor (GDNF) and brain-derived neurotrophic factor (BDNF) were found to be taken up by hypoglossal neurons and directed to cell bodies in small vesicles. The trophic factors are either degraded in the cell body or recycled for transcytosis and accumulate at dendritic synapses (Rind et al., 2005). These studies demonstrate the important role of Rab11 in transport processes in neurons.
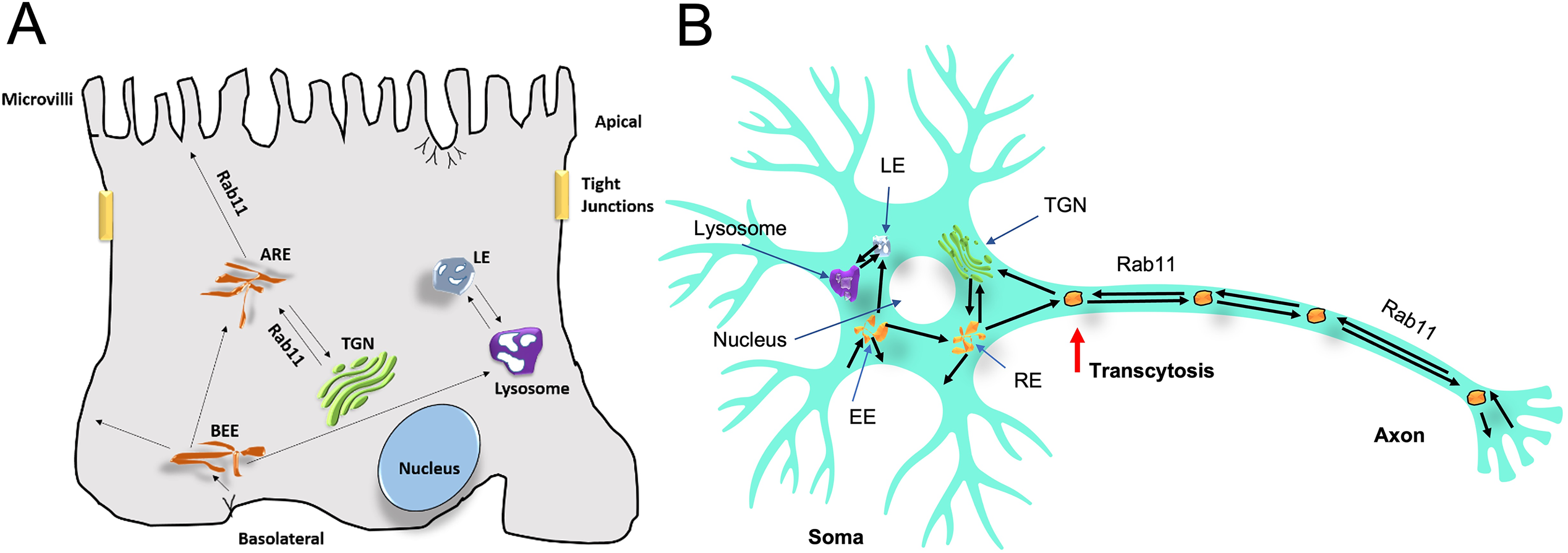
A schematic showing the crucial role of Rab11 in transcytosis in polar epithelial cells and neuronal cells. (A) In epithelial cells, new proteins from basolateral early endosomes (BEE) are transported toward apical recycling endosomes (ARE) and to lysosomes, where they are destined to form late endosome (LE). Proteins in ARE are bound to Rab11 to be either transported to the apical region or returned to the trans-Golgi network (TGN). (B) In neurons, proteins are endocytosed from the somatodendritic space into early endosomes (EE) and moved to late endosomes (LE), which fuse with lysosomes, or they are trancytosed anterogradely to the axon via Rab11-positive recycling endosomes (RE), where they fuse with the membrane or bud out and return to the TGN.
Autophagy
Autophagy is an intracellular, lysosome-dependent degradation process in eukaryotes that may also involve vesicular trafficking. Rab11 exerts a critical function in the formation of amphisomes, which are autophagosomes fused to multivesicular bodies (MVBs) or endosomes (Morvan et al., 2009). The crucial role of Rab11 in the formation of amphisomes during autophagy is illustrated in Figure 6. Rab11-GTP has been reported to be required for amphisome formation in starving K562 cells, and the Rab11-dependent autophagic pathway has been shown to help get rid of endocytic compartments and also remove other organelles (Fader et al., 2008). TBC1D14, a TBC domain protein (Rab-GAP) regulates autophagosome formation by Rab11-positive recycling endosomes. Interestingly, detailed characterization of TBC1D14 revealed that it has no GAP function but has effector function (Longatti et al., 2012). Overexpression of TBC1D14 may lead to misplacement of Rab11, resulting in disruption of recycling transport and autophagosome formation. Therefore, investigation of other Rab targets for TBC1D14 may provide an explanation for the mechanism of how it affects Rab11 function. TBC1D14 bound to Rab11 can also bind to the TRAPP III complex (a multisubunit tethering complex and also a GEF for Rab1) via its N-terminus and activates Rab1, converting the Rab11-positive membrane to a Rab1-positive membrane, facilitating the movement of endosomes to Golgi traffic. Together, they regulate ATG9 (autophagy protein) trafficking from the peripheral recycling endosome to the early Golgi, which is required for the initiation of autophagy (Lamb et al., 2016). It would be of interest to understand how Rab11-Rab1 exchange occurs and whether other cargos use this pathway and contribute to the initiation of autophagy. In addition, Rab11-positive membranes allow autophagy of the transferrin receptor and damaged mitochondria. Upon starvation, phosphatidylinositol-3-phosphate (PI3P) is formed on Rab11-positive membrane, which has been shown to recruit a protein WIPI2 that attaches to autophagic precursor membranes by binding to Rab11. It is worth noting that the loss of Rab11 impairs the autophagy machinery (Puri et al., 2018). This suggests that the Rab11-positive membranes are a preliminary platform for autophagosome formation.
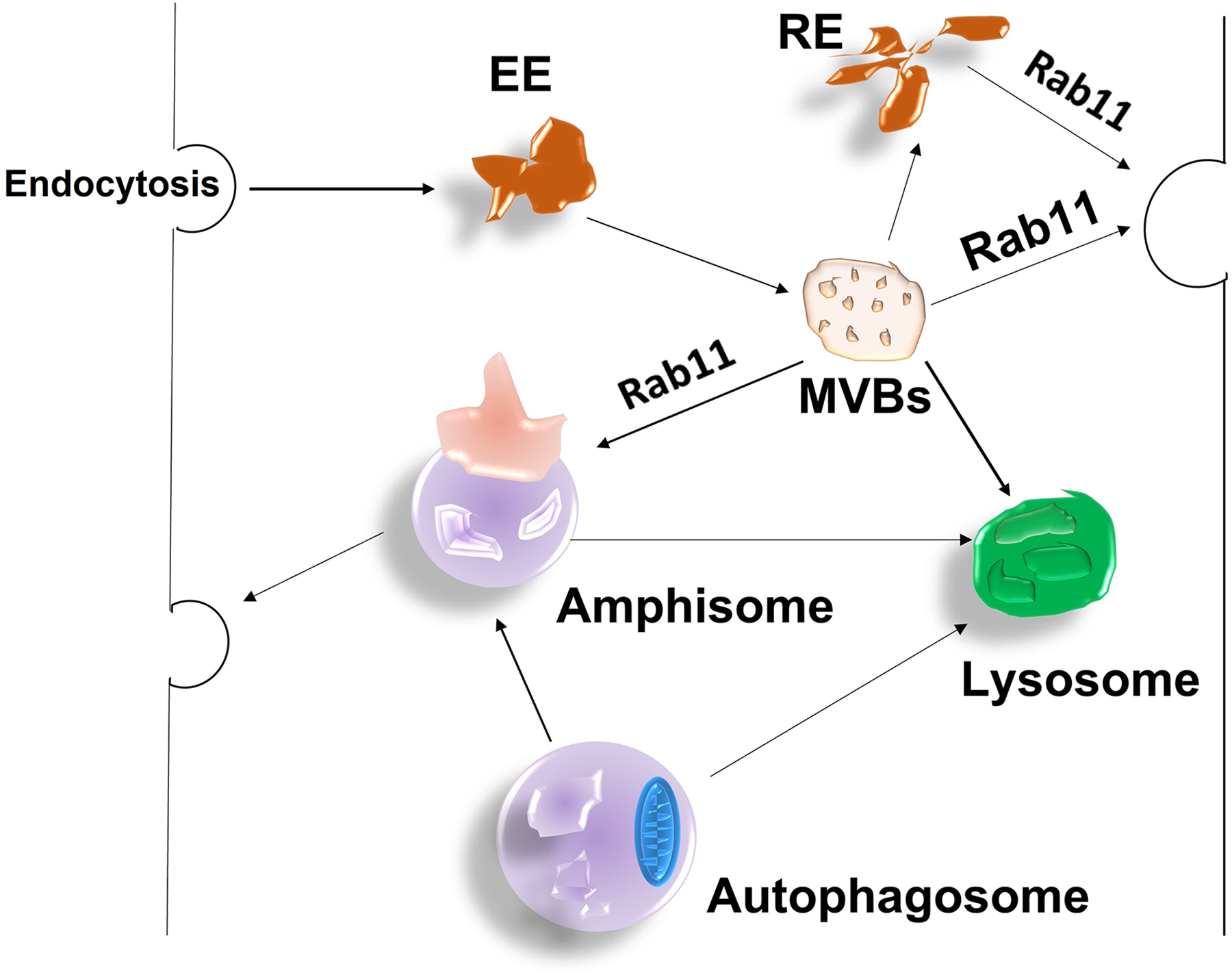
A schematic showing the crucial role of Rab11 in the formation of amphisomes during autophagy. Early endosomes form multivesicular bodies (MVBs), which then fuse with the autophagosome to form amphisome with the help of Rab11; the amphisome is either taken up by lysosomes or used to transport of membranes to the plasma membrane for recycling purposes.
The TRAPP Complex and Neurological Diseases
Transport protein particle (TRAPP), which was first identified in yeast, is a tethering factor consisting of several subunits. It comprises an entire family of complexes, namely I, II, and III. TRAPP complexes are conserved from yeast to humans. The core of each complex consists of seven subunits, namely Trs20, Bet5, Bet3 (2 copies), Trs23, Trs31, and Trs33, and only these subunits constitute TRAPP I. Besides that, TRAPP II and TRAPP III contain another four subunits (Tca17, Trs65, Trs120, and Trs130) and one subunit (Trs85), respectively. Although the subunits are conserved, only the human TRAPP II and TRAPP III have been described. The human TRAPP complexes contain similar core proteins named TRAPPC1, TRAPPC2, TRAPPC3, TRAPPC4, TRAPPC5, and TRAPPC6. Human TRAPP II additionally contains TRAPPC9 and TRAPPC10, while TRAPP III additionally contains TRAPPC8, TRAPPC11, TRAPPC12, and TRAPPC13 (Kim et al., 2016). All three TRAPP complexes show GEF activity toward the Rab1 homolog in yeast, Ypt1p. The TRAPP II complex acts as a GEF toward Ypt31, the Rab11 homolog in yeast (Thomas & Fromme, 2016). In Drosophila, the Trs120 homolog bru functions in the Golgi and interacts with Rab11 (Robinett et al., 2009). In Arabidopsis, TRAPP II is linked to the plant Rab11 ortholog Rab-A (Qi et al., 2011), suggesting that TRAPP II as a GEF is not restricted to yeast.
Based on the knockdown effect of mTrs130, Yamasaki et al. suggested that mammalian TRAPP II (mTRAPP II) functions in ER-to-Golgi transport (Yamasaki et al., 2009). mTRAPP II and Rab11 are associated together for transport from the Golgi to the plasma membrane in human cells during primary cilia assembly, indicating a link between Rab11 and mTRAPP II (Westlake et al., 2011). A recent study showed that a Rac1GEF kalirin associates with mTRAPP II and regulates Rab11 function in recycling endosomes (Wang et al., 2020). Depletion of kalirin resulted in downregulation of TRAPPC9 (the subunit of mTRAPP II) due to a decrease in Rab11 GEF activity on cellular membranes. TRAPPC9/NIBP (NIK and IKK2 binding protein) is a key player in the TRAPP II complex and is known for its important role in NF-|B signaling in the central nervous system (Hu et al., 2005). Rab11 has also been found to recruit Rabin8, which interacts with TRAPPC9 and causes activation of Rab8 to promote ciliogenesis (Knödler et al., 2010). Interestingly, Myo5B has been described as a new interacting partner for TRAPPC9 (Bodnar et al., 2020). Disruption of the interaction of Myo5b with Rab11-FIP2 can abolish exocytosis from recycling endosomes, thereby preventing AMPA receptor insertion and spine growth. These data suggest a direct link between TRAPPC9 and Rab11. It is important to note that a recent study demonstrated the interaction of mTRAPP II with inactive Rab11, proving that TRAPPC9 is a Rab11GEF. In addition, extensive behavioral testing was performed in a knockout (KO) mouse model that revealed a robust role of TRAPPC9 in memory and defects in the brain (Ke et al., 2020). For example, in an open field test the KO mice were less active and did not explore the arena. The female KO mice showed no interest in dealing with unfamiliar objects compared with their wild-type (WT) counterparts and the KO mice also had poor motor skill memory with impaired coordination. Although their vision and swimming ability were similar to WT, the KO mice found the submerged platform much more slowly. Dopamine receptor neurons were also affected by loss of TRAPPC9, explaining the contribution of impaired endocytic Rab11 recycling to poor performance on cognitive tasks. Interestingly, TRAPPC9 KO mice showed increased levels of glial fibrillary acidic protein in glial cells of the corpus callosum and dentate gyrus, suggesting the onset of astrogliosis (a defense mechanism of astrocytes against harmful stimuli), which in turn suggests a possible role of TRAPPC9 in neuropsychiatric disorders. Nevertheless, it is not clear how the loss of TRAPPC9 may affect the glial cells function. In general, these findings suggest the involvement of TRAPPC9 in brain function and learning ability.
Mutations in TRAPPC9 have been identified in patients with nonsyndromic autosomal recessive mental retardation (NS-ARMR). This phenotype has been associated with downregulated NF-κB activation and impaired trafficking function of TRAPPC9 (Mir et al., 2009). In addition, TRAPPC9 may be implicated in several other neurological diseases (Supplemental Table S3). The involvement of Rab11 protein in the mechanical pathway causing neurodegenerative disorder has become increasingly clear over the past decade. Disruption of the trafficking pathway is actively involved in several neurological disorders, which are described below.
Huntington's Disease
Huntington's disease (HD) is an autosomal inherited neurodegenerative disorder characterized by the accumulation of polyglutamine in the amino-terminal portion of Htt protein. Htt is distributed mainly in the cytoplasm. It localizes in vesicles, endosomes and plasma membranes. Htt plays an important role in the movement of Rab11 vesicles within axons (Power et al., 2012). Patients with HD suffer from loss of neurons in the striatum and cortex. Htt plays a functional role in multipolar-bipolar transition via a novel Htt-Rab11-N-cadherin pathway (Barnat et al., 2017). Importantly, mutant Htt has been shown to inhibit Rab11 function (Li et al., 2008). In addition, mutant Htt impairs endocytic recycling from the ERC to the plasma membrane and aggregation of mutant Htt disrupts trafficking of membrane materials to dendritic spines and eventually leads to loss of spines (Richards et al., 2011). It has been reported that overexpression of Rab11 can improve synaptic dysfunction and delay the progression of HD (Steinert et al., 2012). Using real-time 3D tracking of single synaptic vesicles, abnormal movements and vesicle pools were observed in HD mice, and these abnormal movements were dramatically rescued by overexpression of Rab11, suggesting that these phenomena may be an early stage of the pathogenic mechanism (Chen et al., 2021). A recent study by Akbergenova and Littleton (2017) showed that pathogenic Htt in the Drosophila model exhibits a defective synaptic endosomal trafficking associated with expansion of a recycling endosomal signaling compartment containing sorting nexin 16 and a decrease in late endosomes containing Rab11. In parallel with the disruption of endosomal compartments, pathogenic Htt enhanced bone morphogenetic protein signaling, leading to excessive synaptic growth. Apparently, pathogenic Htt directly induces neuropathology by impairing synaptic endosomal signaling. Changes in synaptic morphology caused by abnormal endosomal function may contribute to the lethality resulting from mutant Htt expression, indicating the important role of proper trafficking between endosomal compartments (Akbergenova & Littleton, 2017). Interestingly, HD mice showed significantly less Rab11 along with alterations in transferrin receptor recycling from recycling endosomes back to the plasma membrane. Considering that overexpression of Rab11 protects primary neurons from glutamate toxicity (Li et al., 2009), dysfunctional Rab11 could act as a vulnerability factor for Rab11 membrane-bound cargoes in the early onset of HD. It is known that glucose transporter 3 (GLUT3) and Rab11 are associated with glucose metabolism, and dysfunction of Rab11 is a major cause of glucose metabolism disorders in HD (McClory et al., 2014). In wild-type neurons, glucose uptake can be altered by the expression of dominant-negative/dominant-active Rab11, suggesting that Rab11 is required for the maintenance of normal neuronal glucose uptake. These results suggest that impaired Rab11 activity may be one of the main causes of glucose hypometabolism seen in HD (Li et al., 2012). Htt also regulates polarized vesicular transport by forming a complex with Rab11a (Elias et al., 2015). Because Htt plays a key role in Rab11-regulated vesicle movement within axons, mutant Htt causing impaired transport represents early neuropathology in HD. Interestingly, overexpression of Rab11 prevented synaptic dysfunction in the Drosophila model of HD by normalizing the size of synaptic vesicle which in turn improved locomotion defects in Drosophila larvae (Giorgini & Steinert, 2013). Moreover, overexpression of Rab4 together with Rab11 could rescue synaptic dysfunction and larval locomotion mediated by polyQ-HTT (White et al., 2020). The glutamate/cysteine transporter (EAAC1), which ensures the uptake of extracellular cysteine required for the de novo synthesis of glutathione in neurons, provides another interesting link between Rab11 function and HD pathology. Constitutive trafficking of EAAC1 from recycling endosomes depends on Rab11 activity, which is impaired in the brain of HD. Dysfunction of Rab11 slows trafficking of EAAC1 to the cell surface and impairs the uptake of cysteine, resulting in deficient synthesis of glutathione and inadequate clearance of reactive oxygen species (Li et al., 2010). It is evident that Rab11 may play a prominent role in the pathology of HD, but a detailed mechanism of how Rab11 mediates all its effects remains to be explored.
Alzheimer's Diseases
Alzheimer's disease (AD) is a common neurodegenerative disorder characterized by progressive memory loss and cognitive deficits. It is generally believed that AD pathogenesis is driven by formation of toxic clusters of Aβ. An abnormal amyloid precursor protein that produces a toxic Aβ leads to plaque accumulation, synaptic disruption, and neurodegeneration. RNAi screening studies of Rab GTPases have shown that Rab11 controls endosomal recycling of β-secretase in the plasma membrane, thereby affecting Aβ production. Endocytosed Aβ in neurons is transported through early to late endosomes and then to lysosomes for degradation via Rab11-positive recycling vesicles. Its clearance is regulated by apolipoprotein E (ApoE) and any disruption to this pathway causes accumulation of Aβ aggregates and neuronal toxicity (Li et al., 2012). Early studies reported that estrogen is a regulator in the trans-Golgi network (TGN). It has been shown that 17β-estradiol (17β-E2) plays a role in the formation of vesicles containing APP from the TGN and that recruitment of Rab11 from the cytosol is an important step in this process (Greenfield et al., 2002). This study sheds light on the role of Rab11 in AD via regulation of the secretory pathway. It has been observed that the endosomal Ser/Thr kinase called lemur tail kinase 1 (LMTK1) is involved in regulating the endosomal localization of the β-secretase BACE1 and its accumulation in presynaptic terminals (Komaki et al., 2022). BACE1 is important for the β-cleavage of APP, the initial and rate-limiting step of Aβ formation. In this context, it is worth noting that a novel modulator called LMTK1 controls the GAP activity of TBC1D9B on Rab11a and TBC1D9B mediates LMTK1 activity on Rab11a via the LMTK1-TBC1D9B-Rab11a signaling cascade. It therefore regulates dendritic spine formation in neurons (Nishino et al., 2019). Further studies may shed light on how LMTK1 regulates Rab11a-dependent trafficking and whether it affects brain cognitive functions. Impairment of Rab11 function has been reported to reduce total and endocytosed BACE1 in axons (Buggia-Prévot et al., 2014). Electrochemiluminescence assays indicated that BACE-1-mediated processing of APP is impaired by Rab11 silencing, demonstrating a robust role of Rab11 in APP processing, Aβ production, and Aβ exosome release (Udayar et al., 2013). It is clear that neuronal functions depend on endosomal pathways. The endosomal pathway consists of extracellular vesicles (EVs) including exosomes and microvesicles. Endosomal cargoes are incorporated into extracellular vesicles. Neuronal EVs are critical for intercellular communication, which has been extensively reviewed by Holm et al. (2018). Cargoes which are sorted in EVs at presynaptic vesicles depend on a balance between the endosomal retromer complex and Rab11-mediated recycling in the brains of AD patients. Loss of the endosomal retromer complex leads to accumulation of EV cargoes which include APP in presynaptic terminals of motor neurons in a drosophila model (Walsh et al., 2021). Postsynaptic trafficking also plays an important role in regulating the structure and function of synapses. In a study of the late onset of AD (LOAD), the protein BIN1 showed abundance in the postsynaptic compartment. BIN1 tightly colocalizes with clathrin (a key protein in endocytosis) and localize with the GTPases Rab11 and Arf, that is, in the exocytic pathway. When the Bin1 gene is knock out, its protein content in synapses decreases, leading to the LOAD. However, Bin1 manipulations has smaller effects suggesting more additional studies is required to explore the pathway for LOAD pathology (Schürmann et al., 2020). It is important to point out the role of phosphatidylinositol-binding clathrin assembly protein (PICALM), which has been found to regulate clathrin-dependent internalization of Aβ and to control the trafficking of this peptide to Rab11 for its transcytosis and clearance (Zhao et al., 2015). Endocytosis has been observed to be impaired in AD. Accumulation of APP slows vesicle formation from the endocytic compartment characterized by the presence of the transcytotic GTPase Rab11. Impairment of endocytic axonal delivery of lipoproteins is caused by decreasing levels of lipoprotein endocytosis and transcytosis to the axon. This may lead to further neuron-specific impairments (Woodruff, 2016), suggesting that impairment of transcytotic trafficking may be one of the main reasons for AD. Recently, M6a, a neuronal glycoprotein, has been reported to possibly play a role in several neurological diseases, including AD (Aparicio et al., 2020). This protein is involved in neurite elongation, synapse formation, and spine growth and is trafficked back to the neuronal membrane via Rab11-positive endosomes (Garcia et al., 2017). It was found that the number of synapses is positively influenced by an increased amount of M6a on the cell surface (Garcia et al., 2017). Considering that the number of synapses is crucial for proper brain functioning, Rab11-bound M6a might be important for the etiology of AD. The above data suggest that Rab11, as a key player that regulates the trafficking pathways involved in the modulation of Aβ levels, may possibly be associated with the pathogenesis of AD. Deciphering the molecular complexity associated with Rab11 function may provide new therapeutic ideas for the treatment of AD.
Parkinson's Disease
Parkinson's disease (PD) is a common neurodegenerative disorder caused by mutations in the gene encoding leucine-rich repeat kinase 2 (LRRK2) and the deposition of Lewy bodies composed mainly of α-synuclein (aSyn). The Rab11-rabin8-Rab8A cascade has been shown to regulate LRRK2-mediated inactivation of Rab8A, thereby disrupting endolysosomal trafficking (Rivero-Ríos et al., 2019). In Drosophila, LRRK2 regulates synaptic endocytosis together with Vps35 and Rab11 (Inoshita et al., 2017). In microglia, LRRK2 was found to negatively regulate the clearance of aSyn which was accompanied by downregulation of the endocytosis pathway (Maekawa et al., 2016). There is some evidence that Rab11 can participate in regulating aSyn secretion. Extracellular aSyn is resecreted out of the neurons via Rab11 recycling endosomes in which Rab11a interacts with heat shock protein 90 (Liu et al., 2009). The autophagy-lysosomal pathway (ALP) is involved in the degradation of extracellular aSyn, however, it also plays a role in the intercellular accumulation of aSyn. Inhibition of ALP reduced aSyn aggregation and increased secretion of smaller oligomers by exosomes and Rab11-associated pathways (Poehler et al., 2014). An RNAi-based screening of live cells revealed nine Rab GTPases that modulate aSyn aggregation, with Rab8b, Rab11a, and Rab13 playing a major role in eliminating aSyn inclusions (Goncalves et al., 2016). Silencing of Rab11 increased the secretion of aSyn, suggesting that the secretion and aggregation of aSyn is modulated by the involvement of Rab11 (Chutna et al., 2014). The size of synaptic vesicles was found to increase upon synaptic potentiation induced by aSyn. Overexpression of Rab11 reversed this effect in fruit flies, indicating the importance of Rab11 for synaptic function (Breda et al., 2015). Alterations in aSyn are known to disrupt neuronal vesicle formation and transport. Overexpressed Rab11 has been reported to enhance synaptic potentiation and modulate aSyn-dependent synaptic dysfunction and neurodegeneration. It decreases aSyn inclusions and cellular toxicity. By an undetermined mechanism, aSyn is actively secreted via endocytic, exosomal, and secretory pathways. Importantly, dominant-active Rab11 exhibits a clearance property as it eliminates toxic insoluble aSyn, indicating its therapeutic relevance to PD (Breda et al., 2015). Damaged mitochondria and oxidative stress are important factors contributing to age-related neurological diseases. Dysfunctional mitochondria have been studied in detail at PD. Rai and Roy (2022) have found a close relationship between Rab11 and clearance of damaged mitochondria. The clearance pathway links two important proteins called PARKIN and PINK1. In their study, using Drosophilla models of PD with mutant Park13 and Rab11 knockouts, they found that the brains of the mutant Park13 larvae had low Rab11 levels, which were associated with increased oxidative stress and loss of dopaminergic neuronal networks. Overexpression of Rab11 significantly enhanced these networks and vesicle trafficking, suggesting that Rab11 has a direct regulatory role in the mitophagy pathway. Rab11 is involved in the clearance of aSyn aggregates and dysfunctional mitochondria in PD models, suggesting that it may be a potential candidate for therapeutic intervention.
In the context of possible involvement of Rab11 in the pathogenesis of neurological disorders, it should be mentioned here that the disrupted Rab11-regulated endosomal recycling has recently been linked to neurodegeneration in amyotrophic lateral sclerosis (Mitra et al., 2019). Some other diseases in which Rab11 may play a role, are listed in Supplemental Table S4.
Conclusion
Rab11 as a master GTPase is a well-characterized molecule that contributes significantly to Rab11-mediated endosomal recycling and trafficking in neuronal cells. It is a key regulatory molecule involved in the coordinated trafficking of receptors important for neuronal growth, such as AMPA receptors, integrin, Trk, protrudin, etc. Rab11 has an extensive protein-protein network that transports various cargo molecules. It interacts with different effectors depending on the GDP/GTP binding state, which is an on/off switch that determines its function. GAPs and GEFs regulate the GDP/GTP binding state. Rab11-FIPs play an important role in neuronal transcytosis by transporting proteins from the somatodendritic membrane to the axonal surface. Rab11 mediates the fusion of MVBs with autophagosomes to form amphisomes that direct transferrin receptors and damaged mitochondria into autophagy, and also delivers membrane components to the plasma membrane. A tethering complex consisting of multiple subunits, termed the Trapp complex, has been shown to be a GEF for the Rab11 homolog in yeast, and TRAPPC9 has been determined to be a GEF for Rab11 in mammals. Rab11 may play an important neuroprotective role in several neurodegenerative diseases by ameliorating synaptic dysfunction at HD, clearing Aβ at AD, and maintaining synaptic vesicle size at PD. Some of the important questions that remain to be investigated are the mechanism behind Rab11-mediated signaling pathways, their regulation, and the molecular mechanism of upstream activation of Rab11 family members. It is essential to understand the potential of physical interactions between different complexes and their regulators in specific intracellular pathways. Studies investigating the regulation of the mitophagy pathway by Rab11 have so far been limited to PD. Therefore, it would be interesting to find out whether Rab11-regulated mitophagy is also relevant to other age-related neurodegenerative diseases. In recent years, the importance of glial cells as key partners in neuronal modulation has become increasingly clear, and studies focusing on the regulation of neuronal signaling by glial cells are ongoing. In this context, it could be of interest to investigate whether Rab11 may be implicated in maintaining neuronal network plasticity via glial cells. Because members of the Rab11 GTPase family are critically involved in intracellular trafficking processes in developing or aging neurons and in nerve repair, a possible contribution of Rab11 and its cognate partners to neurodegeneration should be further investigated in future studies.
Supplemental Material
sj-docx-1-asn-10.1177_17590914221142360 - Supplemental material for Rab11 and Its Role in Neurodegenerative Diseases
Supplemental material, sj-docx-1-asn-10.1177_17590914221142360 for Rab11 and Its Role in Neurodegenerative Diseases by Pinky Sultana and Jiri Novotny in ASN Neuro
Footnotes
Authors Note
Both authors were involved with the article preparation and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Charles University, Faculty of Science, (grant number SVV-260871/2020).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
