Abstract
Microglia play an important role in maintaining central nervous system homeostasis and are the major immune cells in the brain. In response to internal or external inflammatory stimuli, microglia are activated and release numerous inflammatory factors, thus leading to neuroinflammation. Inflammation and microglia iron accumulation promote each other and jointly promote the progression of neuroinflammation. Inhibiting microglia iron accumulation prevents neuroinflammation. Ferroptosis is an iron-dependent phospholipid peroxidation-driven type of cell death regulation. Cell iron accumulation causes the peroxidation of cell membrane phospholipids and damages the cell membrane. Ultimately, this process leads to cell ferroptosis. Iron accumulation or phospholipid peroxidation in microglia releases a large number of inflammatory factors. Thus, inhibiting microglia ferroptosis may be a new target for the prevention and treatment of neuroinflammation.
Keywords
Introduction
Neuroinflammation refers to an inflammatory response within the central nervous system (CNS) that is caused by exogenous and/or endogenous factors (Leng & Edison, 2021; Maccioni et al., 2018). Neuroinflammation shares common features with peripheral inflammation, but the unique glial cells and neurons in the CNS distinguish neuroinflammation from peripheral inflammation (Urrutia et al., 2021). Following CNS injury, early inflammation is beneficial to remove harmful substances and inhibit the development of injury, which is conducive to tissue regeneration and recovery (Russo & McGavern, 2016). However, ongoing neuroinflammation in chronic neurological diseases damages the nervous system (Stephenson et al., 2018). Owing to gradual improvement of the living environment and rapid development of modern medicine, the average life expectancy has been gradually increasing with a corresponding increase in the number of the elderly in society. Increased life expectancy may increase the risk of neuroinflammation-related neurodegenerative diseases. Microglia are the main functional cells that mediate neuroinflammation in the CNS. Activated microglia release inflammatory factors to promote neuroinflammation. Chronic inflammation mediated by microglia has been found to cause damage to healthy neural tissue in neurodegenerative diseases (Subhramanyam et al., 2019). Therefore, interventions that address chronic inflammation mediated by microglia may be a suitable strategy against neurodegenerative diseases (Subhramanyam et al., 2019). Microglia iron accumulation and inflammation have a bidirectional relationship and they jointly promote the progression of neuroinflammation. Ferroptosis is an iron-dependent and phospholipid peroxidation-driven process to regulate cell death (Jiang et al., 2021). Previous studies have shown that ferroptosis is closely related to neuroinflammation (Cao et al., 2021; Cui et al., 2021a; Ko et al., 2021). This article presents a new viewpoint that the prevention and treatment of a number of neurodegenerative diseases can be achieved by inhibiting microglia iron accumulation or microglia ferroptosis.
Microglia and Neuroinflammation
Microglia which originate from primitive macrophages of the embryonic yolk sac are the main immune cells in the CNS (Nayak et al., 2014). Microglia are the first line of defense of the CNS innate immune system (Orihuela et al., 2016). Microglia have important roles in neurogenesis (Xiong et al., 2016), synaptic density, and synaptic plasticity (Colonna & Butovsky, 2017). When the CNS is damaged, microglia are responsible for phagocytosis and clearance of pathological protein aggregates, dead cells, and other pathological particles (Colonna & Butovsky, 2017).
Inflammatory stimuli of lipopolysaccharide (LPS) and interferon γ (IFN γ) activate resting microglia to transform into classically activated M1 type which express the proinflammatory factors interleukin (IL)-1β, IL-6, and tumor necrosis factor (TNF)-α, leading to CNS damage (Orihuela et al., 2016). IL-4 and IL-13 induce microglia to transform into M2 type which express anti-inflammatory factors such as transforming growth factor-β, IL-4, IL-10, and IL-13 that promote nerve repair and regeneration to maintain CNS homeostasis (Orihuela et al., 2016; Subhramanyam et al., 2019). The morphology of microglia is highly dynamic. Resting microglia that show the branching morphology will polarize to the amoeboid morphology when stimulated (Colonna & Butovsky, 2017). In response to stimulation by different factors, microglia continuously polarize between their phenotypes M1 and M2 (Orihuela et al., 2016). M1 and M2 can be interconverted to maintain CNS internal environment homeostasis.
A study showed that hemoglobin induced microglia to express the anti-inflammatory factor IL-10 (Li et al., 2021b). Subsequently, IL-10 increased the phagocytic capacity of microglia by inducing the expression of leukocyte differentiation antigen 36, which accelerated the clearance of hematoma and reduced neuroinflammation. Another study showed that downregulating the expression of interferon regulatory factor (IRF) 5 in microglia led to the increase of IRF4 expression and M2 type activation in microglia, which released anti-inflammatory factors and ultimately improved stroke prognosis (Al Mamun et al., 2020). Conversely, downregulation of IRF4 expression resulted in the increase of IRF5 expression and M1 type activation, releasing proinflammatory factors (Al Mamun et al., 2020). Therefore, the microglia IRF5-IRF4 regulatory axis regulates neuroinflammation (Al Mamun et al., 2020). Scp2-1, a polysaccharide extracted from Schisandra chinensis, inhibited nuclear factor-κB (NF-κB) and c-Jun N-terminal kinase (JNK) pathways, which significantly reduced M1 type polarization of microglia induced by LPS and promoted the transformation of microglia to M2 type, thus inhibiting neuroinflammation (Xu et al., 2020). The triggering receptor expressed on myeloid cells-1 (TREM-1) expressed on microglia is a transmembrane immune receptor. In a previous study, the interaction between TREM-1 and spleen tyrosine kinase activated the downstream inflammatory pathway and demonstrated that blocking TREM-1 was a target for the treatment of stroke (Xu et al., 2019). In conclusion, microglia are closely related to neuroinflammation (Rodríguez et al., 2022).
Neuroinflammation and Microglia Iron Accumulation
Neuroinflammation Induces Microglia Iron Accumulation
Coculture of heparan sulfate oligosaccharides isolated from the urine of patients with Sanfilippo syndrome (SP) and microglia reduced the content of ferroportin 1 (FPN1) in microglia, leading to microglia iron accumulation and the release of inflammatory factors with a severe neuroinflammatory response (Puy et al., 2018).
Microglia iron accumulation is involved in the occurrence and development of Parkinson's disease (PD). Chronic inflammation in the substantia nigra (SN) was accompanied by progressive neurodegenerative lesions (Thomsen et al., 2015). Significant iron accumulation occurred in microglia and the expression of ferritin (FRN) was significantly increased during the progression of inflammation (Thomsen et al., 2015). Treatment with 6-hydroxydopamine (6-OHDA) induced the expression of divalent metal transporter 1 (DMT1) and iron influx, resulting in iron overload in microglia (Xu et al., 2022). After the injection of 6-OHDA into the rat brain to establish a PD model, a large number of iron-rich microglia appeared in the damaged SN, indicating that microglia iron accumulation was associated with the progression of PD (Olmedo-Díaz et al., 2017). Pathological accumulation of α-synuclein (α-syn) in the brain promoted the progression of PD, and the results of the study indicated that intranasal injection of α-syn preformed fibrils caused iron deposition in microglia, suggesting that microglia iron deposition may be involved in the progression of PD (Guo et al., 2021).
Inflammatory factors and inflammatory substances cause microglia iron accumulation. LPS increased the expression of heme oxygenase-1 (HO-1) in microglia, which catalyzed heme to release Fe2+, thereby increasing the content of iron in cells (McCarthy et al., 2018). LPS and β-amyloid (Aβ) separately upregulated the expression of DMT1 and FRN in microglia, increased the uptake of iron by microglia, and led to microglia iron accumulation (McCarthy et al., 2018). LPS treatment and oxygen-glucose deprivation/reperfusion upregulated the expression of hepcidin (Hepc) and FRN in microglia and caused microglia iron accumulation (Shin et al., 2018). Stimulation of microglia by either LPS, TNF-α, or IL-6 promoted the expression of DMT1 and Hepc, and ultimately caused microglia iron accumulation (Urrutia et al., 2013). Simultaneous stimulation with IFNγ and Aβ induced microglia iron accumulation and glycolysis, which increased the content of reactive oxygen species (ROS), TNF-α, and inducible NO synthase (iNOS) in microglia (McIntosh et al., 2019). IFNγ treatment alone also induced microglia iron accumulation and significantly increased the mRNA expression of TNF-α and iNOS (Holland et al., 2018). TNF-α upregulated the expression of DMT1 and downregulated the expression of FPN1 in microglia, thus increasing the uptake of iron and iron retention in microglia (Rathore et al., 2012).
Microglia Iron Accumulation Leads to Neuroinflammation
Microglia iron accumulation aggravates neuroinflammation and the development of PD. A clinical autopsy study found a close link between iron accumulation and microglia proliferation in the SN sections of patients with PD (Martin-Bastida et al., 2021). Microglia exposure to high concentrations of ferric ammonium citrate (FAC) led to iron overload in microglia, which increased the expression of ROS and promoted the release of TNF-α and IL-1β (Angelova & Brown, 2018b). Microglia iron accumulation led to an increased release of TNF-α in microglia and subsequent TNF-α activation of the NF-κB and JNK pathways in SH-SY5Y neurons, resulting in an increased expression of α-syn in SH-SY5Y neurons (Angelova & Brown, 2018b).
Microglia iron accumulation promotes the progression of neuroinflammation and disease in multiple sclerosis (MS). Many iron-rich microglia accumulated around chronic active demyelinating lesions and continuously released proinflammatory factors, leading to tissue damage and MS progression (Gillen et al., 2018). The brain sections of patients with MS showed that iron deposition occurred in the marginal area of MS demyelinating lesions, and the microglia in the marginal area exhibited M1 polarization (Mehta et al., 2013). After macrophages were exposed to ferric chloride in vitro, macrophage iron accumulation drove macrophage transformation into M1 type, leading to the release of proinflammatory factors (Mehta et al., 2013). Studies investigating patient brain sections and in vitro research results showed that the microglia at the edge of chronic active white matter demyelinating lesions accumulated iron, thus promoting the transformation of microglia into M1 type which released proinflammatory factors and resulted in the expansion of the lesion (Mehta et al., 2013). A clinical study showed that in the case of demyelinating lesion with slow expansion, iron-rich microglia accumulated at the edge of the lesion and stayed in an activated state, which promoted the progression of MS (Dal-Bianco et al., 2017). Another clinical study found that a large number of iron-rich microglia accumulated around MS demyelinating lesions, which promoted the progression of MS and demonstrated the potential for the development of more serious disease (Dal-Bianco et al., 2021).
Microglia iron accumulation promotes the progression of Alzheimer's disease (AD) and Huntington's disease (HD). The clinical autopsy results of patients with AD showed that iron accumulation occurred in the microglia of the frontal cortex (van Duijn et al., 2017). Coculture of microglia with FAC resulted in microglia iron overload, establishing an in vitro model of aged microglia for culture with SH-SY5Y neurons and a subsequent increase in the level of Aβ in the medium (Angelova & Brown, 2018a). A later mechanistic study showed that microglia iron overload reduced the secretion of the insulin-degrading enzyme that degraded Aβ followed by extracellular Aβ accumulation-caused neurotoxicity (Angelova & Brown, 2018a). The activation of the kynurenine pathway (KP) for tryptophan degradation promoted the progress of HD. Indoleamine-2,3-dioxygenase (IDO) expressed in microglia was the key enzyme of the KP (Donley et al., 2021). After neonatal HD mice were supplemented with iron, microglia were activated and IDO activity was increased, thus activating KP pathway and promoting the progress of HD (Donley et al., 2021).
Microglia iron accumulation drove microglia transformation into the M1 phenotype and glycolysis phenotype. Microglia iron accumulation significantly increased TNF expression, and TNF drove the increase of microglia M1 phenotype and the decrease of M2 phenotype, which was detrimental to the recovery after spinal cord injury (Kroner et al., 2014). Microglia iron accumulation induced by ferric chloride increased the expression of TNF-α and IL-6 and drove microglia transformation into the glycolytic phenotype (Holland et al., 2018). The polarization of microglia to M1 phenotype is often accompanied by the transformation of cells to glycolytic phenotype (Orihuela et al., 2016).
Fe2+ in microglia activates reduced nicotinamide adenine dinucleotide phosphate oxidase (NOX)2 in microglia. In a coculture of rat neurons and astrocytes with microglia and Fe2+ compared to that with Fe2+ only, the number of dopaminergic neurons were significantly lower, indicating that microglia plays a major role in Fe2+ induced neurotoxicity (Zhang et al., 2014). The Fe2+ in microglia activated NOX2, which increased the production of superoxide and ROS, thus promoting neuroinflammation and producing neurotoxicity (Zhang et al., 2014).
Inflammation and Microglia Iron Accumulation Jointly Promote Neuroinflammation
Microglia iron accumulation and inflammatory substances jointly aggravate the progression of neuroinflammation. After FAC induced microglia iron overload and subsequent LPS treatment, microglia were induced to release more IL-1β and TNF-α (Wang et al., 2013). Compared with that in LPS alone, the combined treatment of Fe2+ and LPS significantly increased the production of ROS in microglia (Yauger et al., 2019). Aβ binding to toll-like receptors (TLRs) on microglia activated the NF-κB pathway and subsequently led to an increased expression of IL-1β. Additionally, costimulation of microglia with iron and Aβ significantly increased the expression of IL-1β (Nnah et al., 2020). Microglia iron accumulation increased the expression of IL-6 mRNA (Zhou et al., 2017). IL-6 activated the JNK pathway to upregulate the expression of iron regulatory protein 1 and DMT1 while downregulating the expression of FPN1, which led to further microglia iron accumulation (Zhou et al., 2017).
The simultaneous occurrence of iron accumulation and neuroinflammation amplify the production of ROS and promote iron overload, resulting in neuronal death (Urrutia et al., 2021). Microglia iron accumulation and neuroinflammation also interact to lead to iron overload and a strong inflammatory reaction, which further aggravates the brain damage and its disorders.
Inhibition of Microglia Iron Accumulation Prevents Neuroinflammation
DMT1 inhibition, knockdown of DMT1 gene, and iron chelation significantly reduced the expression of IL-1β in microglia induced by Aβ and FAC (Nnah et al., 2020). Noggin, an antagonist of bone morphogenetic protein (BMP), blocked the BMP/Hepc pathway promoting the release of iron in microglia and improving the prognosis of cerebral ischemia injury (Shin et al., 2018). Deferoxamine (DFO) provided neuroprotection by enhancing the expression of HO-1 in microglia and chelating iron ions after subarachnoid hemorrhage (SAH) (LeBlanc et al., 2016). HO-1 decomposed heme to produce toxic Fe2+ and neuroprotective carbon monoxide (CO) in microglia (LeBlanc et al., 2016). Because DFO chelates Fe2+ and raises the content of CO, it provides neuroprotection by increasing the expression of HO-1 in microglia.
Fe2+ raises the production of ROS in microglia in a NOX-dependent manner. Exposure to Fe2+ and LPS raised the content of ROS in microglia, while inhibition of NOX2 and NOX4 significantly reduced the production of ROS (Yauger et al., 2019). The use of NOX2 inhibitor or NOX2 knockout mice reduced the production of superoxide and ROS in microglia induced by Fe2+, thereby reducing neurotoxicity (Zhang et al., 2014). Microglia iron accumulation activated NOX and then drove the progression of neuroinflammation. Therefore, inhibition of microglia iron accumulation could inhibit the progression of neuroinflammation. (Figure 1)
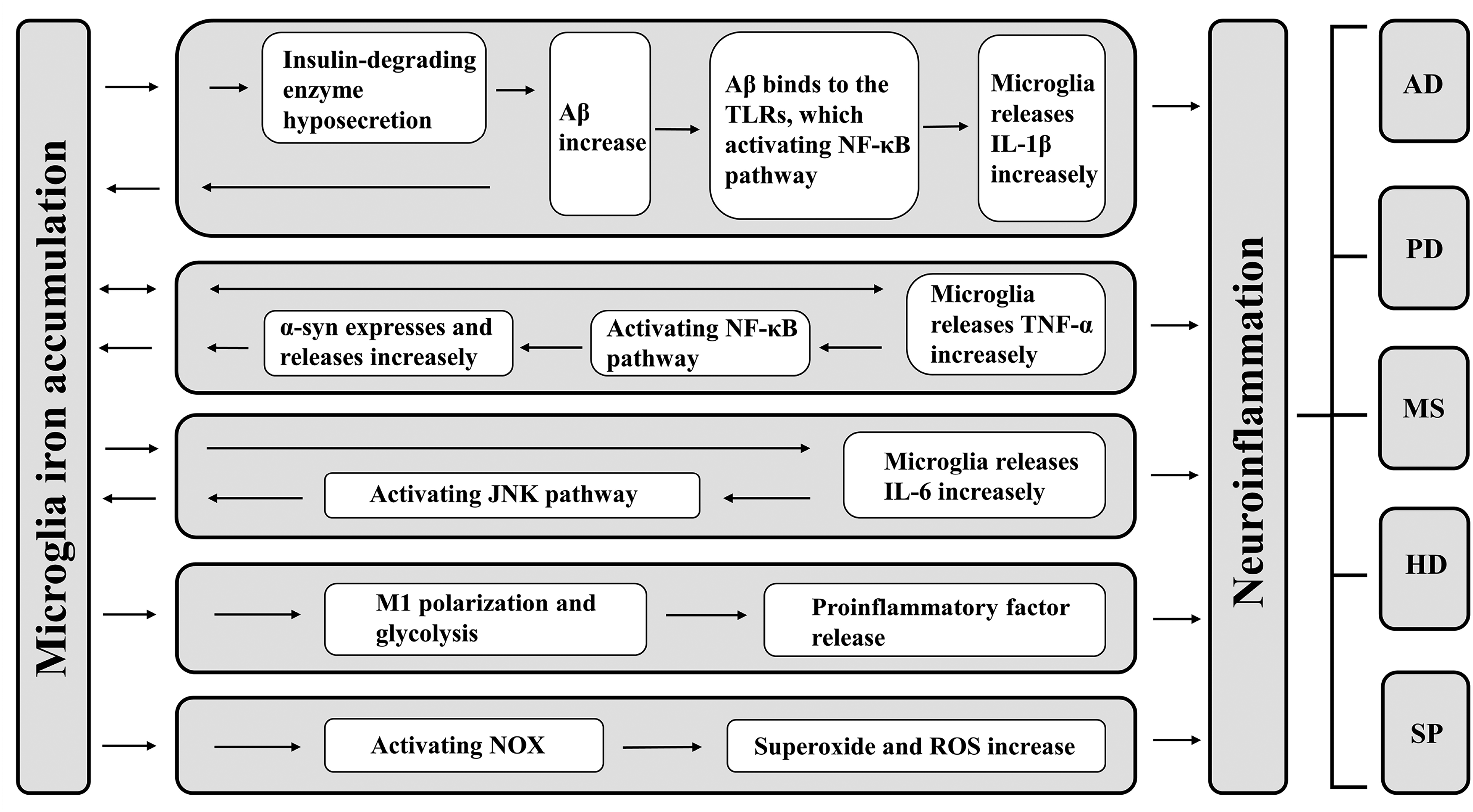
Relationship between microglia iron accumulation and neuroinflammation. Iron accumulation promotes microglia transformation into M1 type, leading to an increased release of the proinflammatory factors IL- 1β, IL-6, and TNF-α. In addition, either IL- 1β, IL-6, or TNF-α induced microglia iron accumulation. Thus, microglia iron accumulation and inflammation promote each other, exacerbating the progression of neuroinflammation. interleukin (IL), tumor necrosis factor (TNF), nuclear factor-κB (NF-κB), c-Jun N-terminal kinase (JNK), Sanfilippo syndrome (SP), Parkinson's disease (PD), α-synuclein (α-syn), β-amyloid (Aβ), reactive oxygen species (ROS), multiple sclerosis (MS), Alzheimer's disease (AD), Huntington's disease (HD), reduced nicotinamide adenine dinucleotide phosphate oxidase (NOX), toll-like receptors (TLRs).
Ferroptosis and its Regulation Mechanism
Ferroptosis is a type of iron-dependent cell death (Dixon et al., 2012); iron accumulation promotes phospholipid peroxidation and drives cell ferroptosis (Jiang et al., 2021; Stockwell et al., 2020). Ferroptosis is a form of cell death with morphological and immunological characteristics different to necroptosis, apoptosis, and autophagy (Xie et al., 2016). During the process of cell ferroptosis, the size of the nucleus is normal, and the chromatin does not agglutinate; mitochondria become significantly smaller, mitochondrial cristae decrease significantly or even disappear, mitochondrial membrane density increases, and mitochondrial outer membrane is ruptured (Stockwell et al., 2017; Xie et al., 2016). Cell ferroptosis is accompanied by the presence of iron, ROS, and polyunsaturated fatty acid-containing phospholipids (PUFA-PLs). Phospholipid peroxidation will destroy the cell membrane integrity, ultimately resulting in cell ferroptosis (Jiang et al., 2021).
Iron Metabolic Part of Cell Ferroptosis
Iron is an essential trace element required by the human body for normal life activities. It is also indispensable for the normal functioning of the brain. Iron takes part in oxygen transport, neurotransmitter synthesis, myelination, mitochondrial energy metabolism, cell division, and other processes (McCarthy et al., 2018; Urrutia et al., 2021). The main sources of iron in the body are iron released by erythrocyte degradation and iron absorbed in food (Brown et al., 2020). Iron exhibits redox activity and has two forms, ferrous (Fe2+) and ferric (Fe3+), which can be interconverted (Nnah & Wessling-Resnick, 2018).
The continuous accumulation of iron ions in cells triggers ferroptosis. There are four main sources of iron ions in the process of cell ferroptosis (Salvador, 2010): 1. After transferrin (Tf) receptor 1 on the cell membrane binds to Tf which is rich in Fe3+, the cell forms an endosome through endocytosis and Tf releases Fe3+ in the acidic environment of the endosome; subsequently, Fe3+ is transformed into Fe2+ which enters the cytoplasm; 2. Extracellular free Fe2+ is transported into the cell mediated by DMT1; 3. FRN which stores Fe3+ is degraded by autophagy mediated by nuclear receptor coactivator 4 (NCOA4); subsequently, Fe3+ is transformed into Fe2+ which is released into the cytoplasm (Hou et al., 2016); 4. The heme in cells is degraded by catalysis of HO-1, and the generated Fe2+ increases the content of iron ions in the cytoplasm. Free Fe2+ in the cytoplasm is transformed into Fe3+ by ceruloplasmin; then, Fe3+ is discharged from the cell to the outside through FPN1 (Xu et al., 2017). The increase of free Fe2+ in the cytoplasm forms a labile iron pool, which has a toxic effect on cells (Salvador, 2010). Free radical HO• is produced by the Fenton reaction (Fe2+ + H2O2 = Fe3+ + OH− + HO•) and ROS is produced by mitochondria oxidizing the cell membrane, which then cause cell ferroptosis (Bu et al., 2021).
Phospholipid Peroxidation Part of Cell Ferroptosis
Catalyzed by acyl-CoA synthetase long-chain family member 4 (ACSL4), polyunsaturated fatty acids (PUFAs) and coenzyme A produce PUFA-CoA; subsequently, PUFA-CoA reacts with phosphatidylethanolamine through lysophosphatidylcholine acyltransferase 3 (LPCAT3) to produce PUFA-PLs (Jiang et al., 2021; Kagan et al., 2017). In response to the action of free radicals produced by the Fenton reaction or lipoxygenases (LOXs), PUFA-PLs are transformed into phospholipid hydroperoxides (PLOOHs) and the continuous accumulation of PLOOHs will cause irreversible damage to the cell membrane and ultimately cause cell death by destroying the function of the cell membrane (Jiang et al., 2021). LOXs which contribute to lipid peroxidation are deemed to be crucial for cell ferroptosis (Shah et al., 2018; Yang et al., 2016). Vitamin E prevents cell ferroptosis by inhibiting LOXs (Kagan et al., 2017).
The cystine/glutamate antiporter (System XC−) is an antiporter for transporting amino acids, and the two peptide chains of the solute carrier family 7 member 11 (SLC7A11) and SLC3A2 interact to constitute System XC− (Bridges et al., 2012). In response to the action of System XC−, intracellular glutamate is transported outside the cell while extracellular cystine is transferred into the cell (Jiang et al., 2021). After cystine receives a hydrogen proton in the cell, it is converted into cysteine which provides raw materials for the next step of glutathione (GSH) synthesis (Jiang et al., 2021). Glutathione peroxidase 4 (GPX4) action results in GSH changing PLOOHs to corresponding alcohols (PLOHs) (Jiang et al., 2021). Hence, in the presence of GPX4, GSH protects the cell membrane by reducing the accumulation of PLOOHs. Therefore, System XC−-GPX4-GSH is extremely important to inhibit cell ferroptosis (Jiang et al., 2021). In addition, ferroptosis suppressor protein 1 (FSP1) increases ubiquinol and α-tocopherol content, inhibiting phospholipid peroxidation (Jiang et al., 2021). (Figure 2)
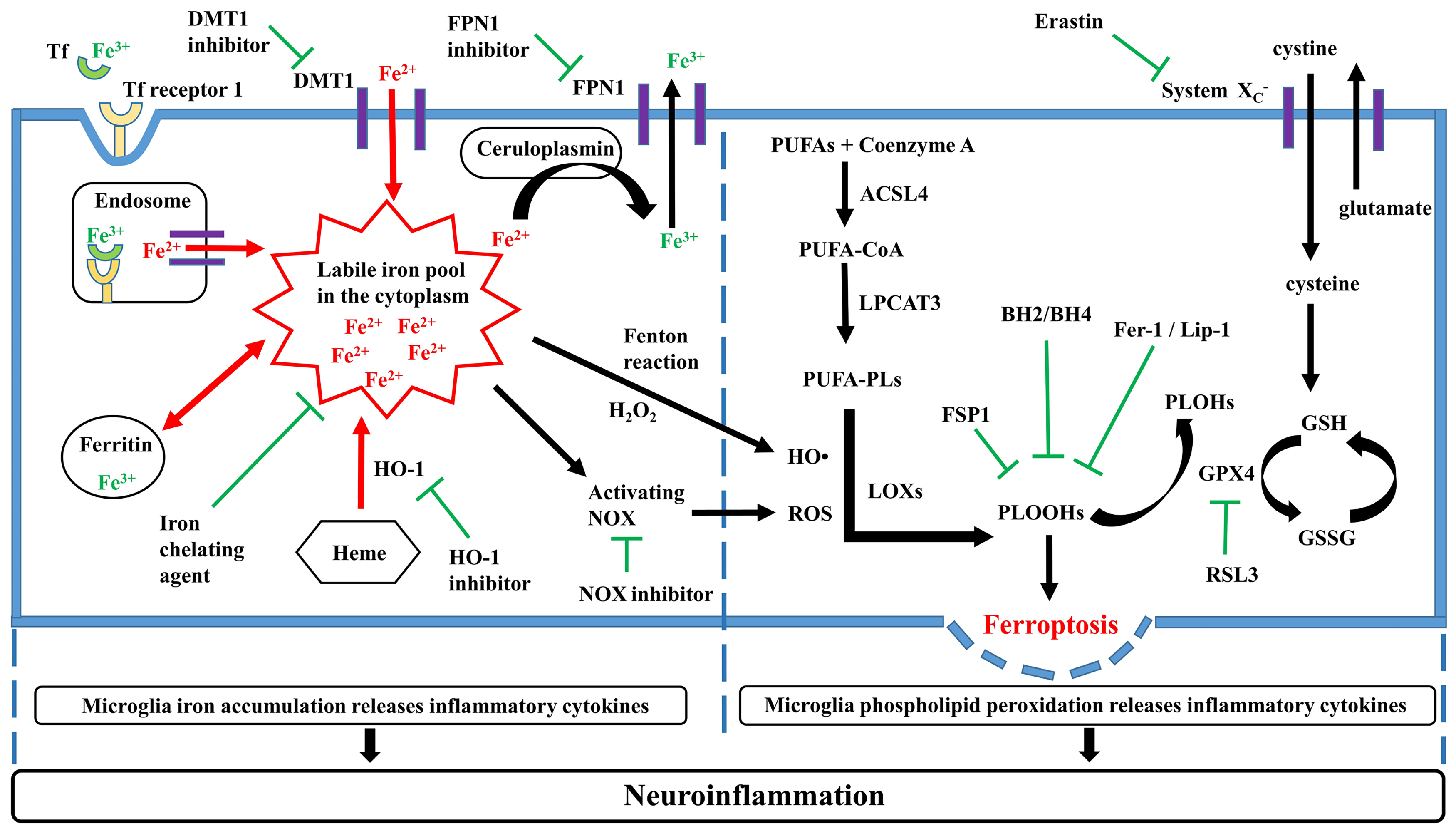
Molecular regulatory mechanism of ferroptosis. Either Tf degradation, ferritinophagy, or heme degradation releases Fe2+. Extracellular free Fe2+ is transported into the cell mediated by DMT1. The increase of free Fe2+ in the cytoplasm forms a labile iron pool. Fe3+ is discharged from the cell to the outside through FPN1. Fe2+ in the cytoplasm significantly increases the production of HO• and ROS. PUFAs is converted to PUFA-PLs under the stepwise catalysis by ACSL4 and LPCAT3. Either HO•, ROS, or LOXs promotes PUFA-PLs transforming into PLOOHs. PLOOHs cause the phospholipid peroxidation of cell membrane, which leads to cell ferroptosis. GSH reduces PLOOHs content under the catalysis by GPX4, thereby inhibiting cell ferroptosis. Either microglia iron accumulation or phospholipid peroxidation increases the release of inflammatory cytokines, leading to neuroinflammation. ferroportin 1 (FPN1), divalent metal transporter 1 (DMT1), heme oxygenase-1 (HO-1), reactive oxygen species (ROS), reduced nicotinamide adenine dinucleotide phosphate oxidase (NOX), polyunsaturated fatty acid-containing phospholipids (PUFA-PLs), transferrin (Tf), acyl-CoA synthetase long-chain family member 4 (ACSL4), polyunsaturated fatty acids (PUFAs), lysophosphatidylcholine acyltransferase 3 (LPCAT3), lipoxygenases (LOXs), phospholipid hydroperoxides (PLOOHs), cystine/glutamate antiporter (System XC−), glutathione (GSH), oxidized glutathione (GSSG), glutathione peroxidase 4 (GPX4), ferroptosis suppressor protein 1 (FSP1), ferrostatin-1 (Fer-1), liproxstatin-1 (Lip-1), dihydrobiopterin (BH2), tetrahydrobiopterin (BH4).
Microglia Ferroptosis may be a new Target for the Prevention and Treatment of Neuroinflammation
Cell Iron Accumulation Causes Cell Ferroptosis
After knockdown of neuronal FPN1, mitochondria in neurons exhibited signs of ferroptosis such as rupture and atrophy (Bao et al., 2021). Concurrently, iron content and the level of lipid peroxidation were increased, indicating that cell iron accumulation promoted cell ferroptosis (Bao et al., 2021). Erastin induced the increase of iron and ROS content in SH-SY5Y neurons and the decrease of cell viability (Geng et al., 2018). Upregulation of FPN1 expression significantly reduced iron content induced by Erastin in SH-SY5Y neurons and increased cell viability (Geng et al., 2018). Knockdown of FPN1 genes further promoted the ferroptosis process of SH-SY5Y neurons induced by Erastin, demonstrating that the iron accumulation in SH-SY5Y neurons drove the ferroptosis process (Geng et al., 2018). Ectopic endometrial stromal cells (EESCs) exhibited ferroptosis induced by Erastin, which decreased the content of FPN1 and led to iron accumulation (Li et al., 2021c). Further in vitro studies found that knockdown of FPN1 expression increased the level of lipid peroxidation and ROS content in EESCs induced by Erastin (Li et al., 2021c). However, overexpressed FPN1 inhibited the impact of Erastin-induced EESC ferroptosis, indicating that EESC iron accumulation significantly affected ferroptosis progression (Li et al., 2021c). Ubiquitin specific protease 35 (USP35) participates in cell proliferation. Knockdown of the USP35 gene resulted in lung cancer cell ferroptosis and overexpression of USP35 inhibited ferroptosis induced by Erastin/RSL3 (Tang et al., 2021). Further research found that knockdown of the expression of USP35 significantly reduced the expression level of FPN1 in lung cancer cells. These findings demonstrated that knockdown of USP35 expression induced cell ferroptosis, which may be achieved by reducing the content of FPN1 in lung cancer cells (Tang et al., 2021). Therefore, cell iron accumulation caused by reduced cell iron output promotes cell ferroptosis.
Erastin directly inhibits System XC− to cause the reduction of GSH production, thus promoting cell ferroptosis (Dixon et al., 2012). Erastin-induced cell ferroptosis in mouse embryonic fibroblasts or human fibrosarcoma cells with autophagy-related gene knockout significantly reduced intracellular Fe2+ content and cell death and inhibited lipid peroxidation compared to that in wild-type cells (Hou et al., 2016). Knockdown of the NCOA4 gene in human pancreatic cancer cells or human fibrosarcoma cells also significantly reduced cell death and intracellular Fe2+ content and inhibited lipid peroxidation after Erastin treatment (Hou et al., 2016). Knockout of autophagy-related genes in primary pulmonary fibroblasts (PPF) or treatment of PPF with an autophagy inhibitor inhibited cell death and reduced intracellular ROS and lipid peroxidation levels induced by Erastin (Park & Chung, 2019). These results demonstrate that ferritinophagy enhances cell ferroptosis and is a key step in cell ferroptosis.
After iron overload in primary cortical neurons treated with FAC, ROS accumulation and lipid peroxidation level were increased and the content of GPX4 were decreased in neurons, mitochondria shrunk significantly, while these were all improved after FAC-chelating DFO treatment (Feng et al., 2021). In vitro studies showed that bleomycin promoted iron accumulation, increased ROS content in the lung epithelial cell (MLE-12), and caused ferroptosis characteristics such as mitochondrial atrophy and increased membrane density (Cheng et al., 2021). DFO inhibited the action of bleomycin on MLE-12, indicating that MLE-12 iron accumulation led to cell ferroptosis (Cheng et al., 2021).
In conclusion, the iron accumulation in the cell triggers the process of cell ferroptosis, followed by accumulation of PLOOHs in the phospholipid peroxidation steps of ferroptosis, ultimately resulting in cell ferroptosis. After intraperitoneal LPS injection in aged mice, overexpression of HO-1 in microglia led to iron accumulation (Fernández-Mendívil et al., 2021). Moreover, overexpression of HO-1 in microglia increased the content of ROS and decreased the expression of GPX4 (Fernández-Mendívil et al., 2021). Thus, microglia iron accumulation also promotes the process of cell ferroptosis and causes the lethal accumulation of PLOOHs, which eventually leads to the ferroptosis of microglia.
The accumulation of iron in cells occurs in the initial steps of ferroptosis. Inhibiting microglia iron accumulation as described above prevent and treat neuroinflammation. This action may be due to the inhibition of iron accumulation in the process of microglia ferroptosis. Moreover, inhibiting microglia iron accumulation may decrease PLOOHs accumulation, thereby reducing the ferroptosis of microglia. The decrease of iron and PLOOHs significantly reduce the expression and release of inflammatory factors in microglia. Therefore, inhibition of microglia iron accumulation to prevent and treat neuroinflammation may be ultimately achieved by inhibiting the ferroptosis of microglia.
Inhibiting Microglia Phospholipid Peroxidation Prevents Neuroinflammation
Nitrogen-doped graphene quantum dots (N-GQDs) are a nanomaterial used widely in the fields of biomedicine and neuroscience. In vitro experimental studies found that N-GQDs treatment decreased the content of GSH, SLC7A11, and GPX4 in microglia and increased the content of Fe2+, ROS, and lipid peroxide, indicating that N-GQDs induced microglia ferroptosis. Simultaneously, N-GQDs upregulated the expression of proinflammatory factors IL-1β and TNF-α in microglia (Wu et al., 2022). Therefore, proinflammatory factors were released and caused neuroinflammation when microglia underwent ferroptosis (Wu et al., 2020, 2022).
Additionally, inhibiting phospholipid peroxidation in the process of microglia ferroptosis prevents neuroinflammation. ACSL4 is an enzyme that catalyzes PUFAs, which affect the sensitivity to cell ferroptosis. Knockdown of the ACSL4 gene in microglia decreased the sensitivity of microglia to ferroptosis and reduced the production of proinflammatory cytokines (Cui et al., 2021a). RSL3 resists lipid peroxidation by inhibiting the catalytic activity of GPX4, thereby promoting cell ferroptosis (Jiang et al., 2021). After treatment of microglia with RSL3, the mRNA expression of proinflammatory factor TNF-α, IL-6, and IL-1β were increased, revealing that the development of microglia ferroptosis is accompanied by an increased expression of inflammatory factors (Cui et al., 2021b).
Inhibition of phospholipid peroxidation during microglia ferroptosis alleviates the damage of intracerebral hemorrhage. Ferrostatin-1 (Fer-1) and liproxstatin-1 (Lip-1) capture unstable free radicals and exert an anti-lipid peroxidation effect,so they are efficient inhibitor of ferroptosis (Zilka et al., 2017). Fer-1 drove microglia transformation to the M2 phenotype and enhanced the phagocytosis of microglia, thereby reducing the inflammatory response after intracerebral hemorrhage (Huang et al., 2022). Lip-1 significantly decreased the number of microglia M1 phenotype after SAH (Cao et al., 2021). Additionally, the content of IL-6, IL-1β, and TNF-α was significantly reduced in the ipsilateral cortex (Cao et al., 2021). After SAH, mitochondria in microglia showed signs of ferroptosis such as increased mitochondrial atrophy and mitochondrial membrane density (Gao et al., 2022). However, cepharanthine (CEP) reduced the damage to microglia mitochondria after SAH, indicating that CEP inhibits microglia ferroptosis (Gao et al., 2022). Further study found that CEP reduced the lethal accumulation of PLOOHs by inhibiting the expression of 15-lipoxygenase-1, thereby inhibiting microglia ferroptosis which was conducive to the repair of nerve injury after SAH (Gao et al., 2022). SLC7A11 encodes for a light amino acid light chain in the System XC− (Bridges et al., 2012). The upregulation of SLC7A11 expression is beneficial to the synthesis of GSH which inhibits cell ferroptosis. Resveratrol increases the expression of SLC7A11 by activating the nuclear factor E2-related factor 2 (Nrf2) signal pathway to suppress rotenone-induced microglia ferroptosis (Li et al., 2021a). Thus, resveratrol plays an antioxidant and anti-inflammatory role.
The process of microglia ferroptosis is composed of two parts: microglia iron accumulation and phospholipid peroxidation. Neuroinflammation could be prevented by inhibiting iron accumulation or phospholipid peroxidation in the process of microglia ferroptosis. Therefore, inhibiting microglia ferroptosis could be a new target in the prevention and treatment of neuroinflammation. (Figure 3)
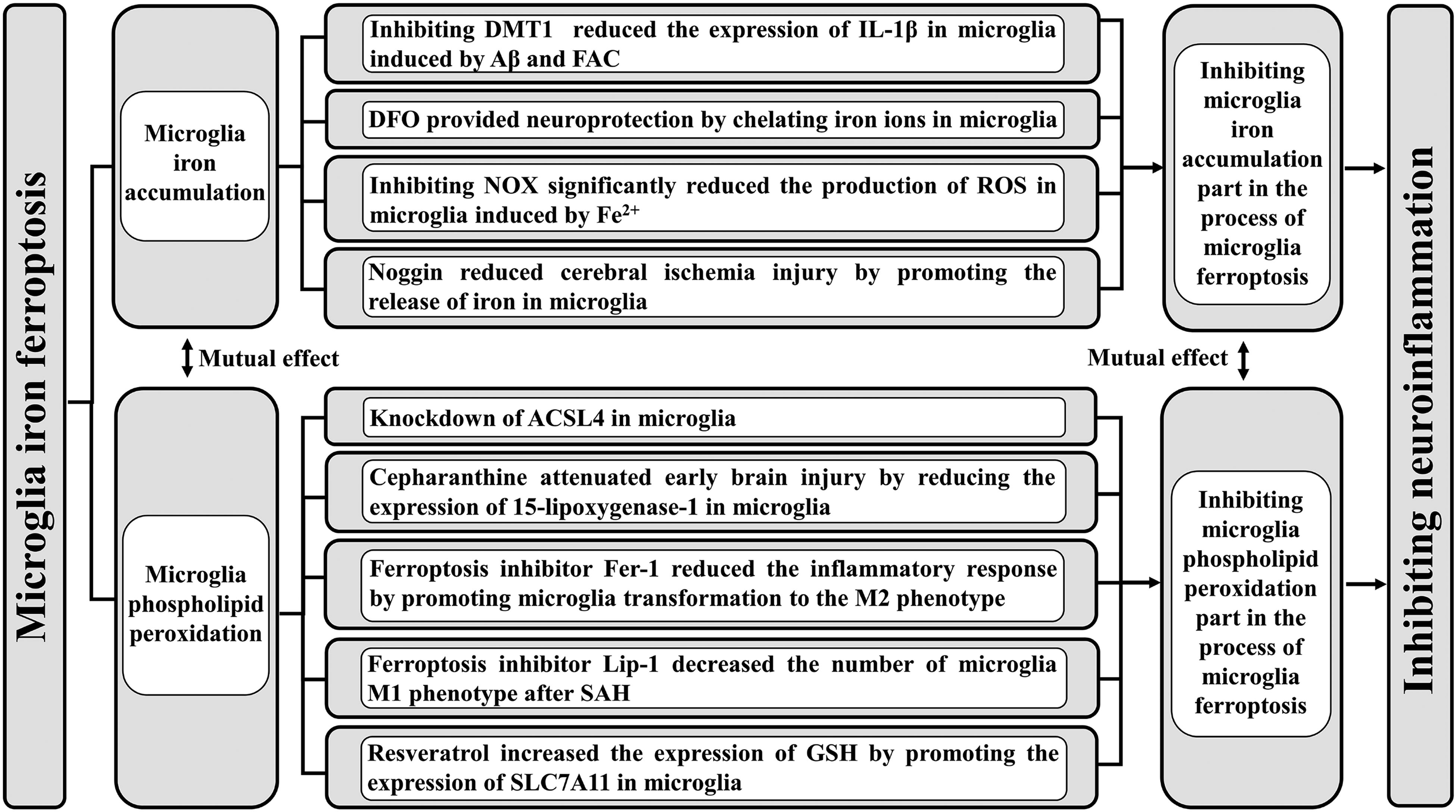
Relationship between microglia ferroptosis and neuroinflammation. The process of microglia ferroptosis is composed of two parts: microglia iron accumulation and phospholipid peroxidation. Either inhibiting microglia iron accumulation or phosphololipid peroxidation restrains neuroinflammation. Therefore, inhibiting microglia ferroptosis is a novel target for the prevention and treatment of neuroinflammation. divalent metal transporter 1 (DMT1), interleukin (IL), β-amyloid (Aβ), ferric ammonium citrate (FAC), deferoxamine (DFO), reduced nicotinamide adenine dinucleotide phosphate oxidase (NOX), reactive oxygen species (ROS), acyl-CoA synthetase long-chain family member 4 (ACSL4), ferrostatin-1 (Fer-1), liproxstatin-1 (Lip-1), subarachnoid hemorrhage (SAH), solute carrier family 7 member 11 (SLC7A11), glutathione (GSH).
Summary and Outlook
Microglia iron accumulation is intertwined with neuroinflammation, and inflammation drives microglia iron accumulation. Microglia iron accumulation, almost involved in the occurrence of neuroinflammation, is accompanied by the polarization of microglia into the M1 phenotype and glycolysis phenotype, resulting in an increase of the expression and release of inflammatory factors. Hence, microglia iron accumulation and inflammation interact to jointly drive the development of neuroinflammation and aggravate CNS damage.
The iron accumulation and the phospholipid peroxidation part of the microglia ferroptosis process both activate microglia to release inflammatory cytokines and subsequently lead to neuroinflammation. Microglia iron accumulation triggers the process of microglia ferroptosis and could enhance the effect of phospholipids peroxidation. Inhibiting microglia iron accumulation could dampen neuroinflammation, which may be achieved by blocking iron accumulation in the process of microglia ferroptosis. In addition, a large amount of iron, free radicals, and cell debris from cells are released after microglia ferroptosis. Other resting microglia continue to remove these substances. As a result, more resting microglia are activated and further aggravate neuroinflammatory damage to tissues.
In conclusion, inhibiting microglia iron accumulation may be a new target for the prevention and treatment of neuroinflammation. Inhibiting microglia ferroptosis may also be a new target for the prevention and treatment of neuroinflammation. The relationship between microglia ferroptosis and neuroinflammation-related diseases needs further exploration. Using ferroptosis inhibitors Lip-1 and Fer-1 to investigate the relationship between microglia ferroptosis and neuroinflammation-related diseases could have great potential.
Footnotes
Acknowledgment
Author Contribution
SHZ, SFL, and XG outlined the manuscript. SHZ and SFL wrote the manuscript. SHZ and SFL critically revised and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 82060250), Guangxi Science and Technology Plan Project (grant number AD20238035), and Hunan Provincial Natural Science Foundation Project (grant number 2021JJ30614).
