Abstract
Multiple sclerosis (MS) is a chronic autoimmune disease of the central nervous system. Recent clinical study suggested that MS patient exhibited acute heart failure. Further, 12-lead electrocardiographic study showed a longer QTc interval in both MS patient and experimental autoimmune encephalomyelitis (EAE) Lewis rat. However, there is limited study regarding the effect of sex on cardiac injury in EAE. To our knowledge, sex effect on cardiac damage in mice with EAE has not yet been published. Herein, we examined the role of the immune system in mediating cardiac dysfunction after EAE in female and male mice. Neurological function was subsequently evaluated and cardiac function was assessed by echocardiography at multiple time points after EAE. EAE mice exhibited severe neurological deficit and significant cardiac dysfunction, including decreased left ventricular ejection fraction (LVEF) and left ventricular fractional shortening (LVFS) at 1 and 2 months after EAE induction. Meanwhile male EAE presented increased expression of the oxidative stress (e.g., nicotinamaide adenine dinucleotide phosphate oxidase-2; NOX-2) in heart, as well as cardiac hypertrophy, increased left ventricle (LV) mass and more severe cardiac fibrosis compared with male control mice. In addition, male EAE mice showed significantly increased cardiac canonical inflammatory mediator (e.g., monocyte chemoattractant protein-1; MCP-1, transforming growth factor-β; TGF-β and toll-like receptor 2; TLR-2) compared with female EAE mice at 2 months after EAE induction. In conclusion, EAE increases inflammatory factor expression and aggravates cardiac dysfunction in male mice compared with female mice, which may contribute to different cardiac outcome in EAE mice.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic inflammatory condition of the central nervous system (CNS) characterized by axonal demyelination. Experimental autoimmune encephalomyelitis (EAE) is the most common experimental model of MS. MS patient was reported to exhibit acute heart failure, including left ventricular hypokinesis measured by echocardiography.(Androdias et al., 2017) Further, both multiple sclerosis patient and EAE Lewis rat showed longer QTc interval on 12-lead electrocardiography than control.(Drouin et al., 1998; Turri et al., 2017) In addition, P wave duration and dispersion were significantly higher in MS patient than control subject.(Razazian et al., 2014) Many studies indicated cardiac dysfunction with a variety of underlying mechanisms after brain injury, such as the hypothalamic–pituitary–adrenal axis, catecholamine surge, and systemic inflammation. However, difference of cardiac damage in different sex EAE model remains unclear.
Gonadal hormone is known to slow the progression of MS by down-regulating CNS inflammation in female compared with male.(Laffont et al., 2015) Estrogen treatment also protected against EAE by up-regulating multiple subtypes of regulatory B cell (e.g., CD5+CD1dhi on CD19+ cell, programmed death-ligand 1 expressing CD19+ cell, and gene expression of the regulatory B cell marker programmed death-ligand 1, interferon regulatory factor-4 and transforming growth factor-β) in the spleen, and promoted the migration of regulatory B cell into the CNS.(Benedek et al., 2016) Serum testosterone level was decreased in male passive EAE, while estrogen level was normal in female EAE.(Foster et al., 2003)
Estrogen receptor α (ERα) and receptor β (ERβ) ligand were reported to exert differential neuroprotective and anti-inflammatory effect in EAE, suggesting different cellular target of estrogen-mediated neuroprotection.(Spence et al., 2013) Estrogen and ERα have anti-inflammatory action in EAE. Further, lentiviral-mediated overexpression of ERα inhibited inflammatory response (e.g., matrix metalloproteinase 9, tumor necrosis factor-α, interferon-γ, interleukin-17, and interleukin-23) and reduced myelin sheath damage in MS patient, suggesting that ERα may be a potential therapeutic target. (Hu and Qin, 2013) The selective ERβ modulator 5-androsten-3β, 17β-diol was also reported to suppress toll like receptor (TLR)-mediated inflammatory response and limit EAE progression in an ERβ-dependent manner. (Laffont et al., 2015)
Given the reported data of sex on cardiac dysfunction in MS and EAE, the aim of the present study is to investigate the role of immune and inflammatory response in mediating cardiac dysfunction after EAE in different sex mice.
Materials and Methods
Experimental Procedure
Female and male C57BL/6J mice (6−8 weeks –old) were obtained from Beijing Huafukang Biotechnology Co., LTD. (Beijing, China). Animals were maintained with standard rodent chow and ad libitum water. This study was conducted in accordance with the National Institutes of Health guidelines for the use of experimental animals. All experimental protocols were approved by the Tianjin Medical University General Hospital Animal Care and Use Committee. Adequate measures were taken to minimize the number of experimental animal used and to ensure minimal pain or discomfort in animal.
EAE Model
To induce EAE, mice were subcutaneously (lower back) inoculated with 200 µg of MOG35-55 (sequence: MEVGWYRSPFSRVVHLYRNGK) (GenScript, Nanjing, China) in Freund’s complete adjuvant (100 μL) (day 0). Pertussis toxin (salt free; 250 ng; List Biological Laboratories, Inc.) was injected intraperitoneally on day 0 and day 2 post-immunization. Freund’s complete adjuvant was made by addition of Tuberculosis Des.H37Ra (BD) to Freund’s incomplete adjuvant (Sigma) at a final concentration of 5 mg/mL.
Neurological Function Score
Neurological deficit score (female EAE, n = 15; male EAE, n = 9) was assessed on alternate days for 2 months after induction of EAE using 0−6 scoring system: 0 = asymptomatic, 1 = loss of tail tone or staggering gait with a strong tail, 2 = staggering gait with loss of tail tone, 3 = paralysis of one hind limb, 4 = paralysis of two hind limbs, 5 = forelimb limb paralysis.(Kadowaki et al., 2019; Yasuda et al., 2019)
Measurement of Cardiac Function
Animal was anesthetized with 2% isoflurane in 0.5 L/min of 100% oxygen, the chest was shaved, and the animal placed in the supine position on a 37 °C heating pad. Steady-state sedation was maintained throughout the procedure with 1.0%–1.5% isoflurane in 0.5 L/min of 100% oxygen. Cardiac function was evaluated in real time using transthoracic echocardiography (Vevo 2100 High Resolution Ultrasound System; Visual Sonics, Canada) with an MS-250 ultrasound scanning transducer (model C5) at the 1st and 2nd month after EAE induction. Left ventricular ejection fraction (LVEF) (female control, n = 10; female EAE, n = 15; male control, n = 10; male EAE, n = 13), left ventricular fraction shortening (LVFS) (female control, n = 10; female EAE, n = 15; male control, n = 10; male EAE, n = 13), left ventricular internal diameter diastolic (LVID;d) and left ventricular internal diameter systolic (LVID;s) (female control, n = 10; female EAE, n = 15; male control, n = 10; male EAE, n = 13), and left ventricular mass (LV mass) (female control, n = 6; female EAE, n = 5; male control, n = 6; male EAE, n = 7) were recorded (LVEF = [(LV VOL; d-LV VOL; s)/LV VOL; d × 100], LVFS = [(LVID; d-LVID; s)/LVID; d × 100]). Echocardiography was performed by an investigator blinded to the experimental groups. All primary measurements were digitized by goal-directed, diagnostically-driven software and three beats were averaged for each measurement.
Measurement of Heart Fibrosis and Hypertrophy
Mice were sacrificed at 2 months after EAE, heart and tibia isolated, weighed after dried with wipe and measured length with vernier caliper, respectively (n = 6/group). Cardiac hypertrophy was evaluated by the ratio of heart weight to tibia length: HW/TL (mg/mm). Then heart was fixed with 4% paraformaldehyde and embedded in paraffin. And we selected the coronal sections of the heart from the base, mid and apex regions.(George et al., 2020) Coronal section was cut (thickness, 7 μm) and stained with picros sirius red (1:1,000 dilution, Sigma) for calculation of the interstitial collagen fraction (female control, n = 7; female EAE, n = 7; male control, n = 6; male EAE, n = 6).(Zhou et al., 2018) Hematoxylin and eosin staining was used for measurement of cardiomyocyte cross-sectional area (female control, n = 9; female EAE, n = 8; male control, n = 10; male EAE, n = 7).(Arimoto et al., 2006; Chen et al., 2018) Three slides from each heart, 5 fields of view with each slide were digitized under 200× magnification using a light microscope (Olympus, Japan). The cardiomyocyte cross-sectional area of no less than 20 cells per view field was counted (more than 300 cells per one mouse heart).(Zhang et al., 2019) The picros sirius red-positive area and cardiomyocyte cross-sectional area were calculated (Image Pro Plus 6.0). Immunohistochemical analysis was performed by an investigator blinded to the experimental groups.
Immunostaining
Mice were sacrificed at 2 months after EAE induction and the heart and brain were isolated, fixed with 4% paraformaldehyde for 24 h, and then embedded in paraffin. And we selected the coronal sections of the heart from the base, mid and apex regions. Paraventricular slice of the brain showing basal ganglia was collected (start at bregma). Coronal heart and brain tissue sections were cut (thickness, 7µm), heated at 60°C for 30 min, deparaffinized with pure xylene I, pure xylene II, 100% ethanol I, 100% ethanol II, 95% ethanol, 90% ethanol, and 80% ethanol, and washed with deuterium-depleted water for 3 × 5 min. Tissue sections were then incubated in sodium citrate solution at 95 °C for 30 min for antigen retrieval, followed by 3% bovine serum albumin for 1 h at room temperature.
Primary antibodies against monocyte chemoattractant protein-1 (MCP-1; 1:50 dilution; Abcam), nicotinamide adenine dinucleotide phosphate oxidase-2 (NOX-2; 1:200 dilution; Abcam), transforming growth factor-β (TGF-β; 1:250 dilution; Santa Cruz Biotechnology, INC), toll-like receptor-2 (TLR-2; 1:100 dilution; Abcam), and myelin basic protein (MBP; 1:1,000 dilution; Biolegend) were diluted with 3% bovine serum albumin. Heart sections were incubated in anti-MCP-1 (n = 5/group), anti-NOX-2 (female control, n = 5; female EAE, n = 6; male control, n = 6; male EAE, n = 6), anti-TGF-β (female control, n = 6; female EAE, n = 5; male control, n = 6; male EAE, n = 6), and anti-TLR-2 (female control, n = 6; female EAE, n = 5; male control, n = 6; male EAE, n = 6) primary antibodies overnight at 4 °C, while brain sections were incubated in the anti-MBP primary antibody (female control, n = 6; female EAE, n = 7; male control, n = 6; male EAE, n = 8) overnight at 4 °C. Sections were then washed with cold phosphate buffer saline for 3 × 10 min, followed by fluorophore-conjugated secondary antibodies for 1 h at room temperature. Slides were counterstained with 4′, 6-diamidino-2-phenylindole (Abcam) to stain cellular nuclei. Three slides from each tissue, 5 fields of view with each slide were digitized under 200× (for heart sections) or 100× (for brain sections) magnification (Olympus) using a three-CCD color video camera (Sony DXC-970MD) interfaced with an MCID image analysis system (Imaging Research Inc., St. Catharines, ON, Canada). The mean integrated optical density value (IOD (sum)/AREA (sum)) for MCP-1, NOX-2, TGF-β, TLR-2 and MBP were measured by Image Pro Plus 6.0. Immunofluorescence staining analysis was performed by an investigator blinded to the experimental groups.
Quantitative Real-Time PCR
Mice (female control, n = 6; female EAE, n = 5; male control, n = 6; male EAE, n = 6) were sacrificed at 2 months after EAE induction and total RNA was isolated from the whole heart with 1 mL of TRIzol Reagent (Ambion) and 200 μL of chloroform. Samples were shaken, incubated for 15 s at room temperature, and centrifuged at 12,000 g for 15 min at 4 °C. The upper phase was mixed with the same volume of isopropanol, incubated for 10 minutes at room temperature, and then centrifuged at 12,000 g for 20 min at 4 °C. The upper liquid phase was removed and the pellet containing nucleic acid was suspended with 1 mL 75% ethanol. The solution was then centrifuged at 12,000 g for 5 min at 4 °C and the upper liquid phase removed. The pellet was air dried for 5−10 min, dissolved in 20 μL diethyl pyrocarbonate treated water, and the total RNA concentration quantified by ultraviolet spectrophotometry at 260/280 nm.
cDNA was synthesized from 1 µg RNA with the TransScript First-Strand cDNA Synthesis SuperMix Kit (Transgen). Quantitative PCR was performed in a total volume of 20 μL, containing 10 μL of the SYBR green PCR Master Mix (Roche Diagnostics, Basel, Switzerland), 2.5 μL of forward and reverse primer, 2.5 μL template DNA and 5 μl of DNase free water on an Opticon 2 Real-Time PCR Detection System (BioRad, Hercules, CA, USA) as follows: 10 min at 95 °C, followed by 40 cycles of 95 °C for 15 s and 60 °C for 1 min. Each sample was performed in triplicate and normalized to GAPDH. Analysis of relative gene expression data was performed using the 2–ΔΔCt method. The following primer sequences were used: GAPDH: Forward primer 5′-GCCAAGGCTGTGGGCAAGGT-3′, reverse primer 5′-TCTCCAGGCGGCACGTCAGA-3′; TLR2: Forward primer 5′-CTCCCACTTCAGGCTCTTTG-3′, reverse primer 5′-TTATCTTGCGCAGTTTGCAG-3′.
Statistical Analysis
Statistical analysis was measured by two-way ANOVA followed by Bonferroni post-tests with use of Graph Pad Prism 5 (Graph Pad Software Inc., San Diego, CA). P < 0.05 was considered statistically significant. Data in all figures are presented as mean ± SEM.
Results
Neurological Function Scores of Female and Male Mice With EAE
The study showed a monophasic disease course with no recurrence and no sex difference in the manifestation of EAE. Nevertheless, we observed a trend towards more serious symptom in female EAE mice compared with male (Figure 1).
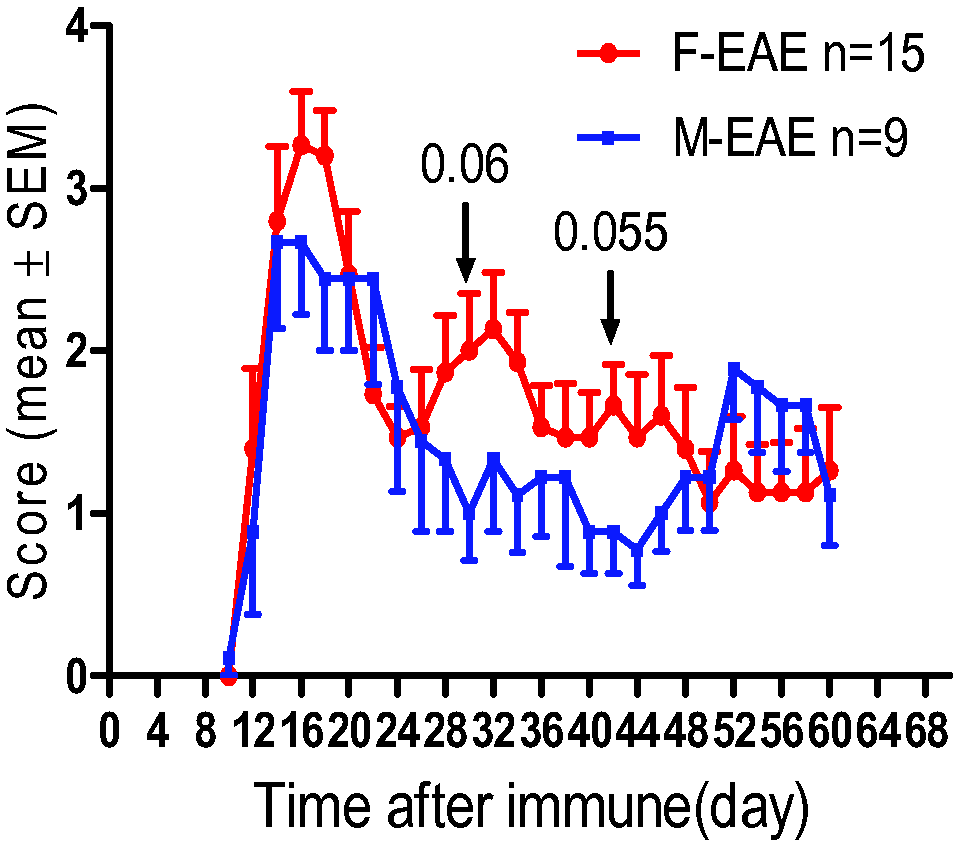
Neurological Function Scores in Female and Male Mice With Experimental Autoimmune Encephalomyelitis (EAE). Female EAE mice (F-EAE) are shown in red. Male EAE mice (M-EAE) are shown in blue. p = 0.06 (30th day after EAE) and p = 0.055 (42nd day after EAE) for female compared with male mice.
EAE-Induced Marked CNS Demyelination
In the present study, there was a significant decrease in MBP expression in the brain at 2 months after EAE compared with control mice (Figure 2). EAE induced marked CNS demyelination. And there was no sex difference in the degree of demyelination following EAE in C57BL/6.
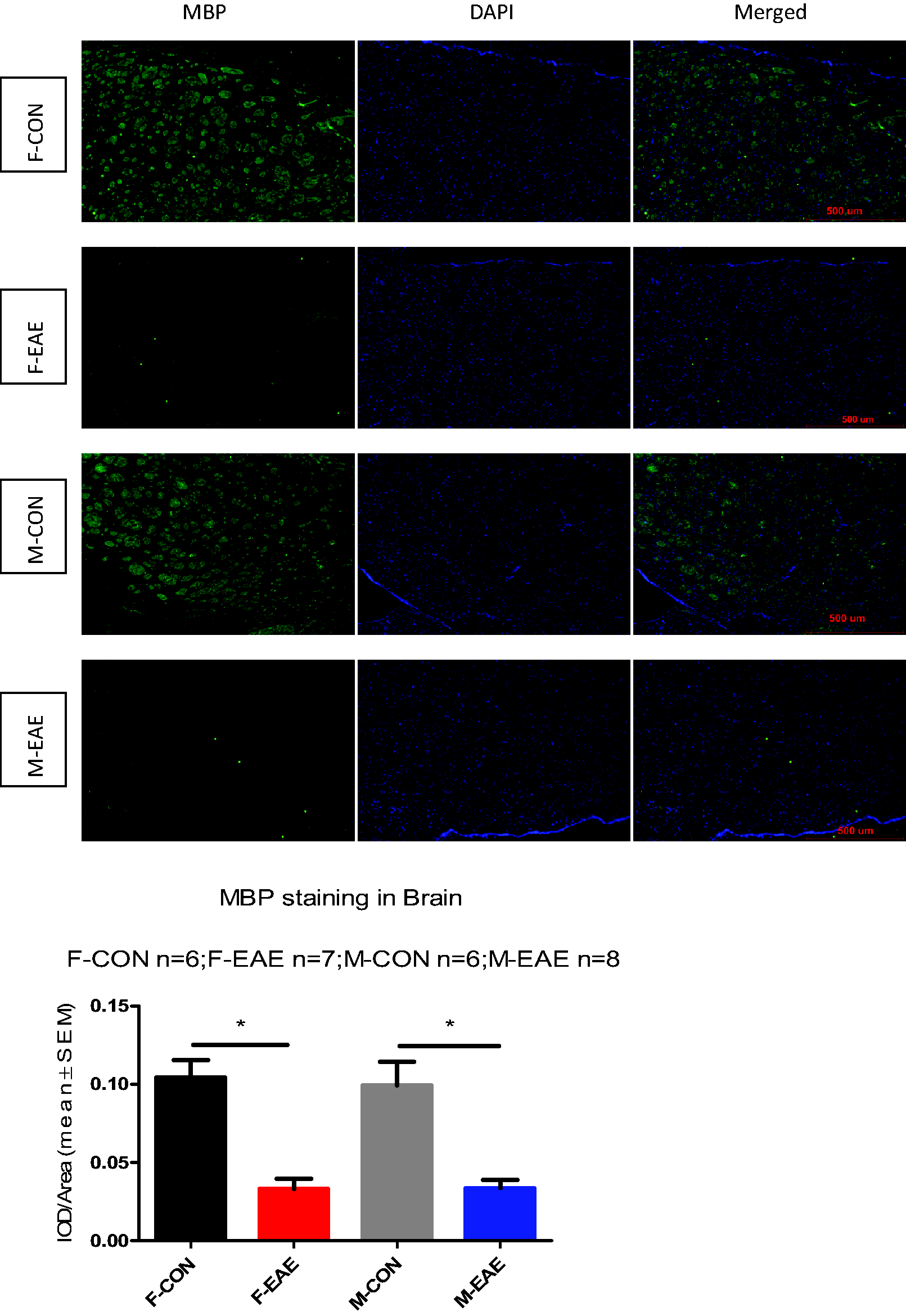
Myelin Basic Protein (MBP) Staining (×100) Showing Central Nervous System Demyelination in Experimental Autoimmune Encephalomyelitis (EAE). MBP staining is shown in the paraventricular (MBP, green; nucleus, blue). *P < 0.05 was considered statistically significant. (F-CON: female control; F-EAE: female EAE; M-CON: male control; M-EAE: male EAE).
EAE Induces More Severe Cardiac Dysfunction in Male
To test whether EAE regulates cardiac dysfunction in the absence of primary cardiac disease, we performed echocardiograph at 1 and 2 months after immunization. Both male and female EAE mice showed significant cardiac deficits involving decreased LVEF and LVFS at 1 and 2 months after EAE compared with male or female sham control mice, respectively (Figure 3A and B). There was no difference in cardiac deficits between 1 and 2 months of recovery in female EAE mice. Male EAE mice also showed worse cardiac function (decreased LVEF and LVFS) at 1 and 2 months after EAE compared with female EAE mice. Further, male EAE mice showed a trend towards worse cardiac function at 2 months of recovery compared with at 1 month (LVEF: 49.53 ± 2.063 vs 54.57 ± 1.700, respectively; p = 0.07; LVFS: 24.82 ± 1.263 vs 27.82 ± 1.063, respectively; p = 0.08). And, male EAE showed increased LV mass (Figure 3C) measured by echocardiograph at 2 months after EAE compared with male sham control mice (LV mass: 80.60 ± 2.680 vs 69.57 ± 1.418, respectively; p = 0.0054). Male EAE also showed increased LV mass at 2 months of recovery compared with at 1 month (LV mass: 80.60 ± 2.680 vs 67.78 ± 1.656, respectively; p = 0.0016). The values of LVID;d and LVID;s were significant increased in male EAE compared with male control as well as compared with female EAE, the values of the both were increasing tendency in female EAE compared with control (Figure 3D). Overall, these data suggest that EAE induces chronic cardiac deficits in both male and female mice, while it may be more severe in male.
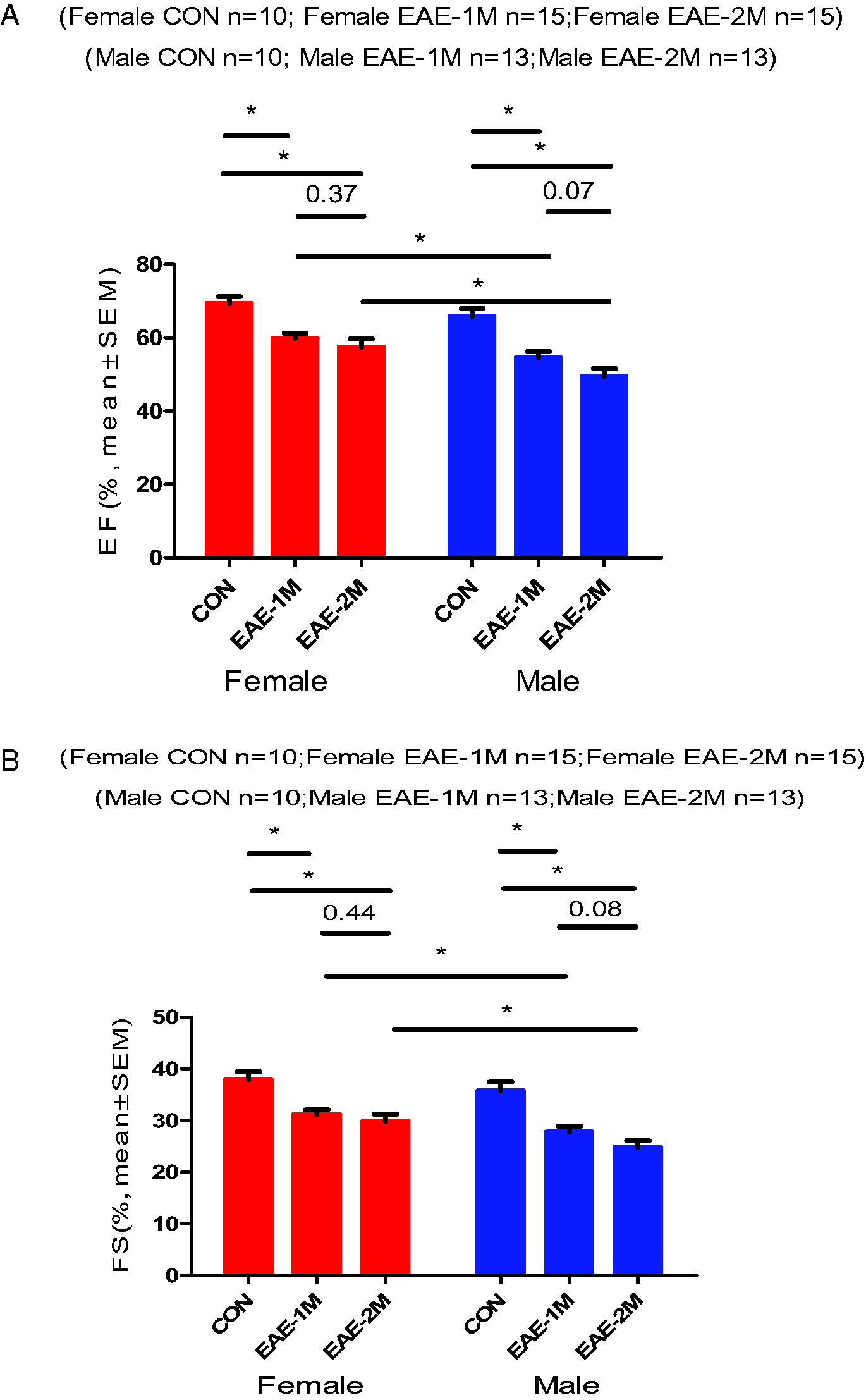
Assessment of Cardiac Function by Echocardiography. (A) LVEF (left ventricular ejection fraction) of the control and EAE mice. (B) LVFS (left ventricular shortening fraction) of the control and EAE mice. (C) LV (left ventricular) mass recorded by echocardiography in the control and EAE. (D) Left ventricular internal diameter diastolic (LVID;d) and left ventricular internal diameter systolic (LVID;s). (E) The long-axis Echocardiography images of control and EAE mice. The red bar represents the female, the blue represents the male. *P < 0.05 was considered statistically significant. (CON: control; EAE-1M: EAE-1 month; EAE-2M: EAE-2 month).
Male EAE Mice Show Increased Cardio Hypertrophy and Cardiac Fibrosis
The effects of EAE on cardiac hypertrophy and fibrosis were observed at 2 months of recovery. Male EAE mice showed evidence of cardiac hypertrophy compared with wild type control mice (cross-sectional area of myocardial cell in male EAE compared with wild type control: 226.2 ± 19.03 vs 169.4 ± 14.30, respectively; p = 0.0279) as shown in Figure 4(A), while there was no difference between female EAE and control mice (204.9 ± 15.40 vs 178.0 ± 18.2, respectively) or between female and male EAE mice (204.9 ± 15.40 vs 226.2 ± 19.03, respectively). Figure 4(B) shown that the ratio of heart weight to tibia length (HW/TL: mg/mm) was increased in male EAE compared with male control mice. Picro sirius red (PSR) staining also demonstrated severe cardiac fibrosis in male EAE mice compared with male control mice (ratio of myocardial fibrosis: 1.389 ± 0.1127 vs 0.9955 ± 0.1248, respectively; p = 0.0413) as shown in Figure 5, while there was no difference between female EAE and female control mice (ratio of myocardial fibrosis: 1.171 ± 0.1049 vs 1.058 ± 0.1048, respectively) or between female EAE and male EAE mice. Overall, these data suggest that EAE induces cardiomyocyte hypertrophy and severe cardiac fibrosis in male but not female mice.
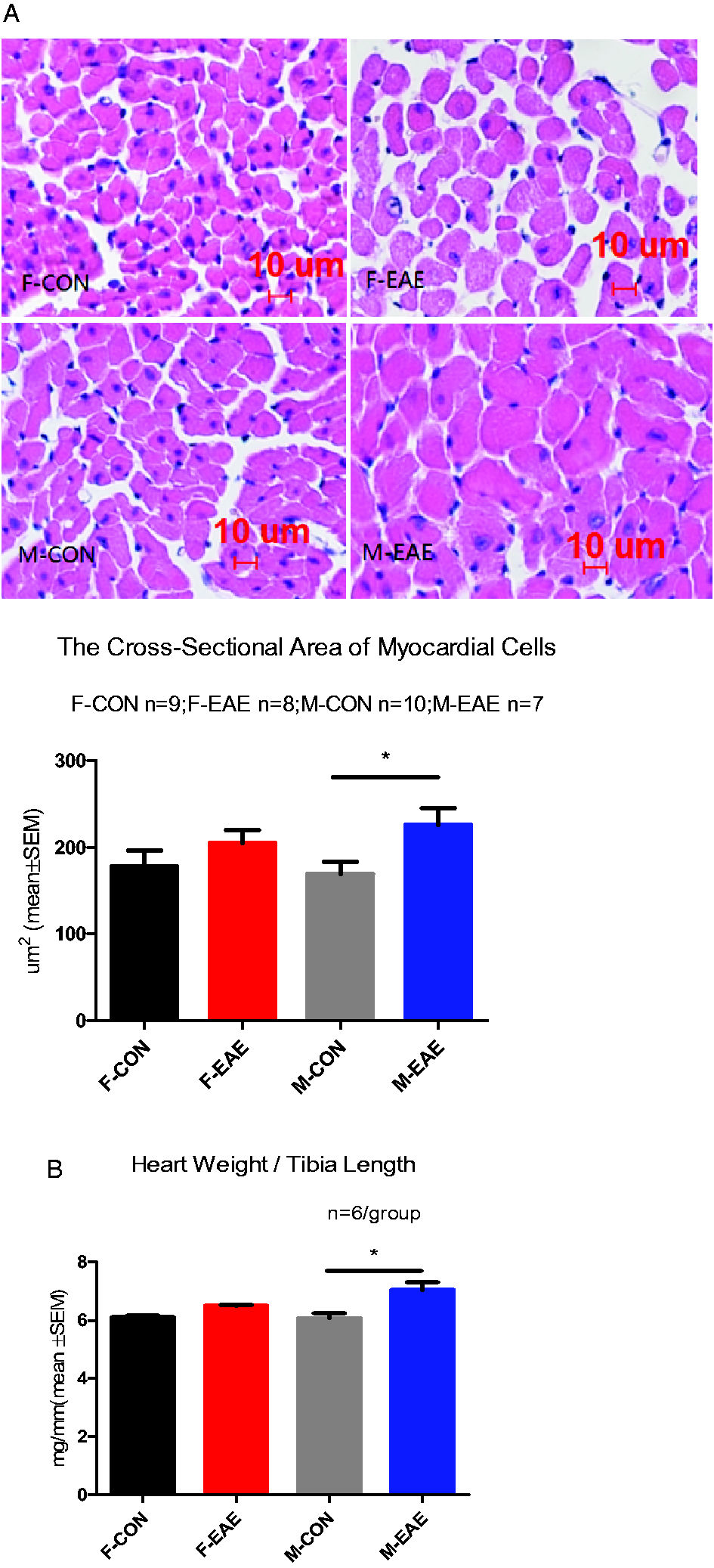
Hematoxylin and eosin (H&E) staining in heart and the ratio of heart weigh to tibia length. (A) Hematoxylin and eosin (H&E) staining in heart coronal section (H&E staining ×200). The cardiomyocyte cross-sectional area was measured by Image Pro Plus 6.0. (B) The ratio of heart weight (mg) to tibia length (mm). *P < 0.05 was considered statistically significant. (F-CON: female control; F-EAE: female EAE; M-CON: male control; M-EAE: male EAE).
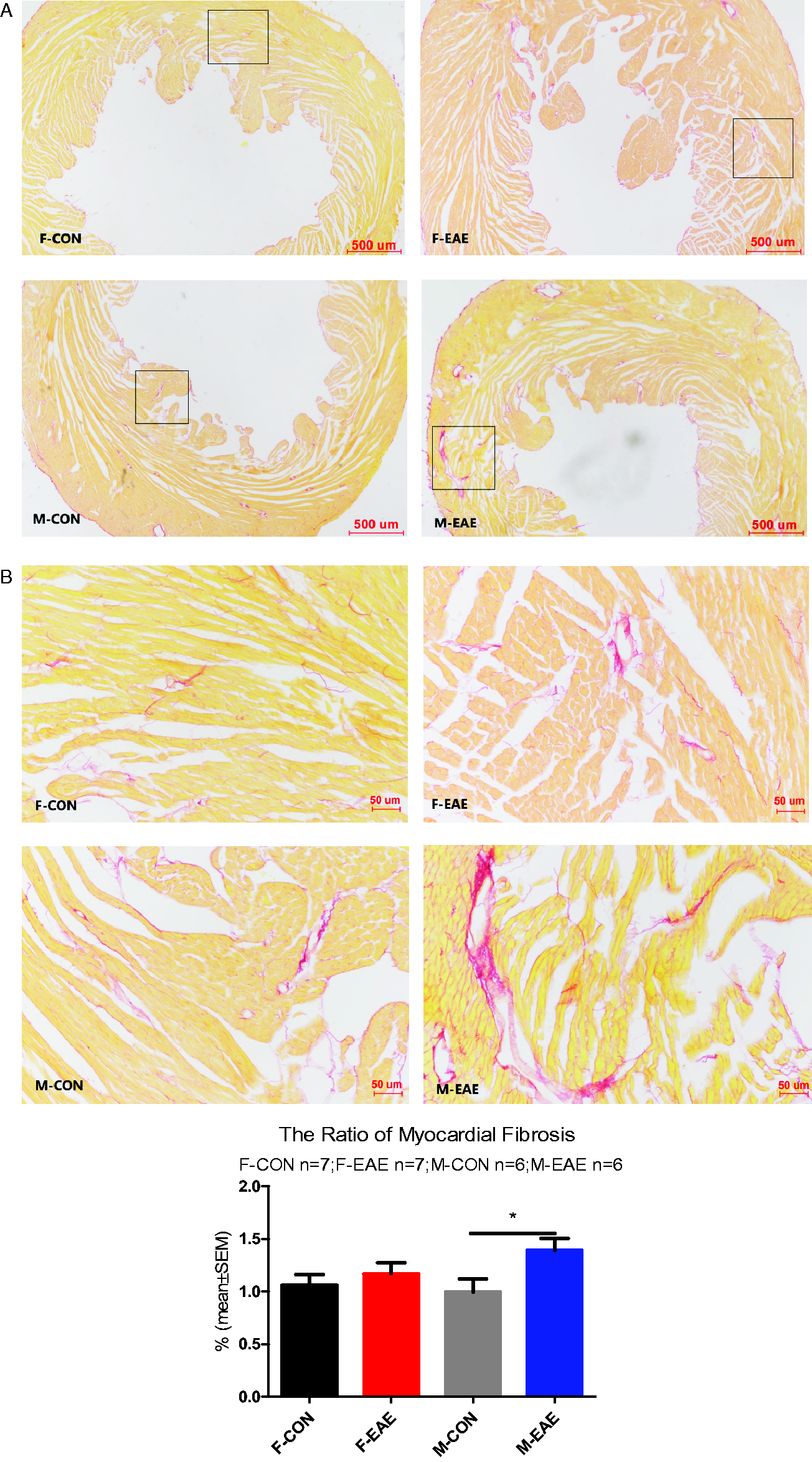
Picro sirius red staining in heart. (A) Picro sirius red (PSR) staining for assessment of the interstitial collagen fraction (ICF) in heart (PSR staining ×40). (B) The black square of (A) image was collected on ×200. Heart coronal sections were cut and stained with picros sirius red for calculation of the interstitial collagen fraction. *P < 0.05 was considered statistically significant. (F-CON: female control; F-EAE: female EAE; M-CON: male control; M-EAE: male EAE).
Male EAE Mice Show Increased Cardiomyocyte Inflammatory Factor Expression and Cardiac Oxidative Stress
To examine the mechanism of EAE induces cardiac dysfunction, we measured the expression of the inflammatory cytokines MCP-1 and TGF-β and the oxidative stress marker NOX-2 at 2 months after EAE induction. The cardiomyocyte inflammatory factors (MCP-1 and TGF-β) and cardiac oxidative stress marker (NOX-2) were increased in male EAE mice. Male EAE mice showed a significant increased in cardiac MCP-1 expression (Figure 6A) compared with female EAE mice. Further, male EAE mice showed a significant increased in cardiac NOX-2 expression compared with male control mice (Figure 6B), while there was no difference between female EAE and female control mice. TGF-β expression was also increased in cardiac of male EAE compared with male control as well as compared with female EAE as shown in Figure 6C.
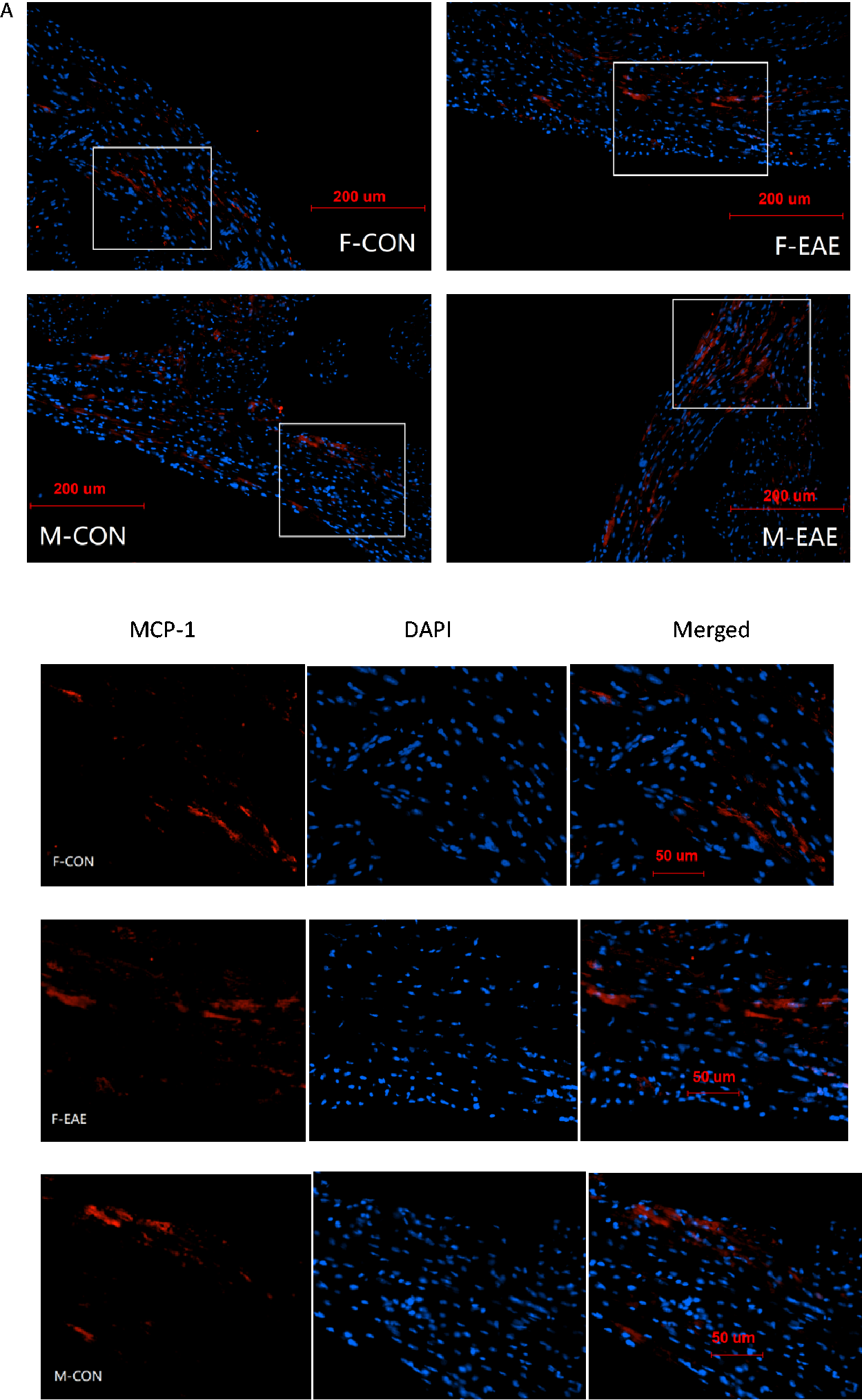
Immunofluorescence staining of MCP-1, NOX-2, and TGF-β in heart. (A) Immunofluorescence of monocyte chemoattractant protein-1 (×200). Heart coronal sections were cut and immunofluorescence stained with anti-MCP-1 antibody (MCP-1, red; nucleus, blue). The top four images are merged images, the white square was enlarged shown as followed in the lower part. (B) immunofluorescence of nicotinamide adenine dinucleotide phosphate oxidase-2 (×200). Heart coronal sections were cut and immunofluorescence stained with anti-NOX-2 antibody (NOX-2, green; nucleus, blue). The top four images are merged images, the white square was enlarged shown as followed in the lower part. (C) transforming growth factor-β immunofluorescence (×200). Heart coronal sections were cut and immunofluorescence stained with anti- TGF-β antibody (TGF-β, green; nucleus, blue). The top four images are merged images, the white square was enlarged shown as followed in the lower part. *P < 0.05 was considered statistically significant. (F-CON: female control; F-EAE: female EAE; M-CON: male control; M-EAE: male EAE).
To further test whether EAE induces cardiac inflammatory mediator, we measured TLR-2 expression by immunostaining and quantitative PCR in the heart at 2 months after EAE induction. TLR-2 expression was significant increased in male EAE cardiac compared with male control as well as compared with female EAE as shown in Figure 7. These data suggest that EAE induces increased cardiomyocyte inflammatory factor expression and oxidative stress in male mice.
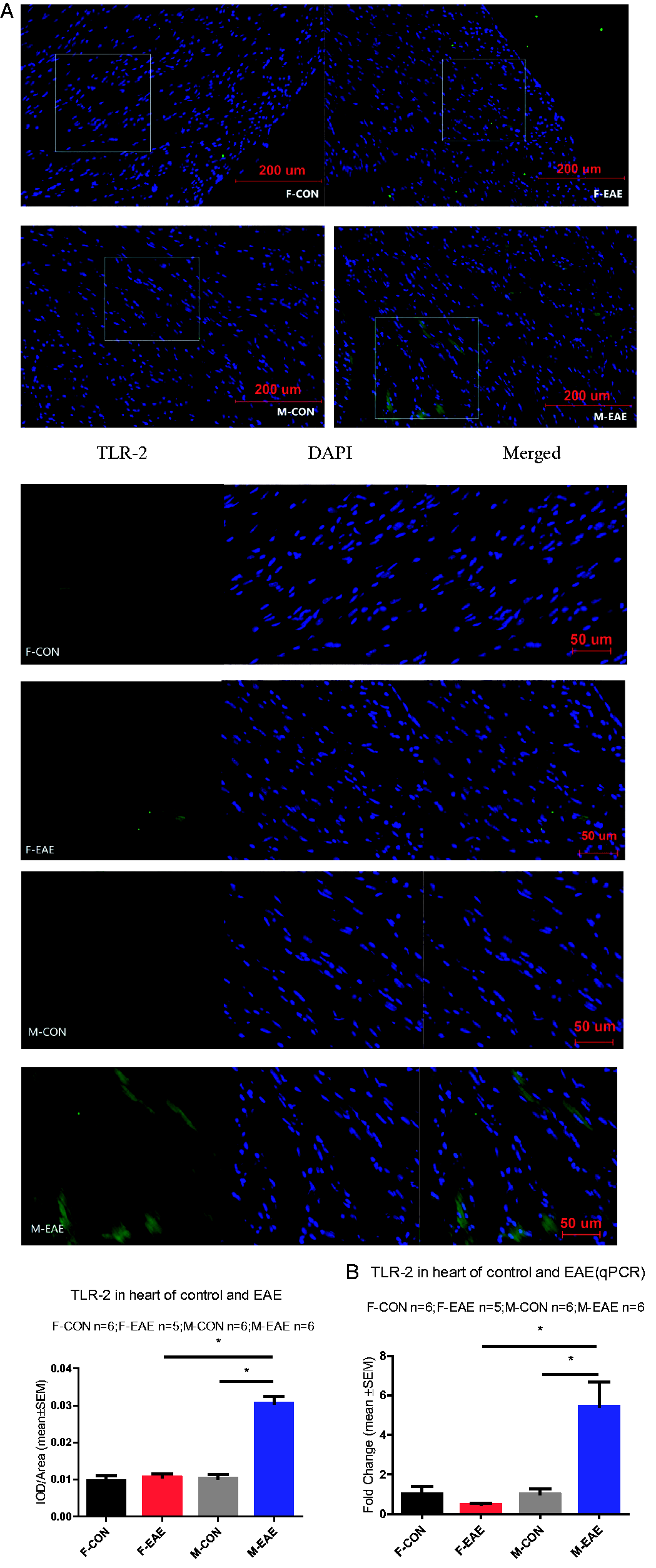
The immunofluorescence staining of TLR-2 and TLR-2 gene expression in heart. (A) Toll-like receptor-2 immunofluorescence in heart (×200). Heart coronal sections were cut and immunofluorescence stained with anti-TLR-2 antibody (TLR-2, green; nucleus, blue). The top four images are merged images, the white square was enlarged shown as followed in the lower part. (B) TLR-2 gene expression in heart measured by qPCR. *P < 0.05 was considered statistically significant. (F-CON: female control; F-EAE: female EAE; M-CON: male control; M-EAE: male EAE).
Discussion
MOG35-55-induced EAE C57BL/6 mice were reported to show a monophasic disease course with no recurrence and no sex difference in C57BL/6.(Yu and Whitacre, 2004; Stark and Cross, 2006) It was previously reported that there was no sex difference in the degree of neurological injury following EAE in C57BL/6.(Yu and Whitacre, 2004) The result showed a monophasic disease course with no recurrence and no sex difference in the manifestation of EAE in our study. Previous study showed that the protein expression of MBP in brain was significant decreased by the quantitative analysis of immunohistochemistry in EAE mice compared with control mice.(Yang et al., 2017) The result showed that the MBP expression was decreased in the brain of EAE compared with control mice in our study.
There is limited study regarding cardiac injury in EAE. A previous research observed longer QTc interval on 12-lead electrocardiography in EAE Lewis rat than control.(Drouin et al., 1998) Increased P wave dispersion, prolonged QT interval, decreased heart rate variability (measured as R-R interval on electrocardiogram), left ventricular systolic function, takotsubo-like cardiomyopathy, and sudden death with no history of antecedent cardiac disease, orthostatic hypotension and postural orthostatic tachycardia syndrome have been reported in MS patient.(Kaplan et al., 2015) In addition, Takotsubo syndrome was considered as an uncommon extra-neurological manifestation of multiple sclerosis relapse, which may indicate the intricate brain-heart connection.(Dell'Aquila et al., 2020) Cardiovascular autonomic dysfunction presenting a shift of cardiovascular sympathetic-parasympathetic balance toward increased sympathetic modulation was related with lesion of the central autonomic network area.(Winder et al., 2019) However, autonomic cardiovascular dysfunction was not caused solely by brain stem lesion in MS, because heart rate, very low rate, low rate and the low rate/high rate quotient are higher in MS (even present brain stem lesion) than patient with isolated brain stem lesion of a non inflammatory origin.(Monge-Argilés et al., 2002)
In addition, female EAE showed less inflammatory cell infiltration by the semiquantitative evaluation of the inflammation score in the lumbar tract of the spinal cord than male EAE.(Giatti et al., 2010) There was sex difference presenting that superoxide dismutase activity was lower, whereas O2 – concentration of advanced oxidation protein product was higher in spinal cord of male than female rat.(Dimitrijević et al., 2017) Change in synaptic protein is sex specific in EAE, for example the expression of gephyrin (inhibitory postsynaptic membrane scaffold protein) showed significant reduced in female EAE cortex, but in male EAE hippocampus.(Murphy et al., 2020) The level of neuroactive steroid showed sex difference in EAE nervous system and plasma, such as increased pregnenolone, progesterone, dihydroprogesterone, testosterone, dihydrotestosterone, 3a-diol and isopregnanolone in male EAE and decreased tetrahydroprogesterone in female EAE.(Giatti et al., 2010)
A key finding of the present study was that EAE mice exhibited different degree cardiac dysfunction between female and male, which may be related to difference of gonadal hormone level. As shown that sex-biased expression existed in mouse cardiac development.(Deegan et al., 2019) Female and male presented diverse responses to different diseases, for example that male had a higher CRP peak in circulating inflammatory response to endotoxemia than female, while higher level cytokines (IL-6 and TNF-α) were associated with lower bilateral ventral striatum activity in female but not male participant in depression.(Ferguson et al., 2013; Moieni et al., 2019) Male showed more obvious change of cardiac autonomics stress than female in athlete.(Abad et al., 2017) The autonomic vagal component played a greater role in the cardiac modulatory balance in women, while the sympathetic component played a greater role in men.(Dutra et al., 2013) There was gender difference in the rate of stress-heart disease, mental stress-induced myocardial ischemia was more likely to be woman, while exercise/pharmacological stress-induced ischemia was more likely to be man.(Bacon, 2018) The change of mitochondrial deacetylases Sirt3 (Sirtuin 3) expression may result in vascular dysfunction such as endothelial dysfunction, vascular hypertrophy and inflammation.(Dikalova et al., 2020) Difference of mitochondrial biology may result in cardiovascular disease gender difference.(Vona et al., 2018) Early inflammation response and NOD-like receptor pyrin domain-containing protein 3 inflammasome activation initiated by Ca2+/calmodulin-dependent protein kinase II δ signaling in cardiomyocyte may play important role in cardiac remodeling.(Suetomi et al., 2018) The Cav1.3 was responsible for L-type inward calcium current, and its high expression may result in increased intrinsic heart rate of the sinoatrial node in female rat.(Doris et al., 2019) The study focusing on the right auricle from patient undergoing aortic valve replacement or aortocoronary bypass operation showed female fiber was significantly more force than male.(Bening et al., 2013) Male heart was thinner and more fibrotic than female in senescent rat.(Forman et al., 1997)
Previous study shown the sex chromosome complement may cause sex difference in the function of non-gonadal tissue independent of the influence of sex hormone in mice.(Colafella and Denton, 2018) According to the Voskul laboratory, in the four core genotypes (FCG) mouse model with EAE after gonadectomized, expressions of a cluster of 5 X genes (Msl3, Prps2, Hccs, Tmsb4x, and Tlr7) were higher in CD4+ T lymphocyte from XY than from XX, and higher protein expression of TLR7 in CD19+ B lymphocyte from XY than from XX.(Golden et al., 2019) In the absence of TLR7, lymphocyte activity and serum IgG level were decreased in mouse model of systemic lupus erythematosus.(Christensen et al., 2006) According to the Teuscher laboratory, flow cytometry analysis of spleen cell from FCG mice shown higher T regulatory (CD4+ FoxP3+) cell number at day 7 post coxsackievirus B3 infection in XY than XX mice.(Robinson et al., 2011) The T regulatory cell played positive role in promoting Th17 cell differentiation.(Pandiyan et al., 2011) Encephalitogenic Th17 cell mediated proinflammatory response in the pathogenesis of multiple sclerosis and its animal model EAE.(Rus et al., 2017; Zhang and Kiapour, 2019) Sex difference in immune response or organ function is dependent on the combined effects of sex chromosomal gene and sex hormone in intact mouse.
In addition, EAE mice induced cardiac dysfunction, which may be related to the increase of cardiac canonical inflammatory mediator. TGF-β is an important profibrotic mediator since it plays an important role in regulating fibroblast proliferation and myofibroblast differentiation.(Guo et al., 2019) TGF-β may also contribute to cardiac fibrosis and LV dysfunction.(Zhou et al., 2018) MCP1 can activate TGF-β production and increase the fibrogenic potential by stimulating TGF-β and collagen synthesis.(Yaghchiyan et al., 2019) Further, MCP-1 was recently reported to enhance TGF-β signaling via increasing expression of MMP-9.(Youn et al., 2019) The silence of NOX-2 in cardiomyocyte can attenuate hypoxia-induced oxidative stress and apoptosis indicating that NOX-2 may cause cardiac dysfunction.(Yu et al., 2016) Oxidative stress was also found to regulate myocardial hypertrophy and trigger pro-inflammatory signaling pathway.(Frantz et al., 2001) TLR-2 exists on the membrane of various inflammatory cell and endothelial cell, and activation of TLR-2 can lead to reactive oxygen species-induced inflammation and leukocyte infiltration, ultimately lead to cardiomyocyte and coronary endothelial injury.(Favre et al., 2007) Activation of TLR-2/MyD88 pathway by TLR-2 agonist (e.g., bacterial peptidoglycan-associated lipoprotein) can depress cardiac contractility via induction of cardiomyocyte dysfunction (e.g., altered calcium homeostasis in cardiomyocyte) and inflammation.(Zhu et al., 2007) TLR-2 is also present in monocyte, polymorphonuclear neutrophil, and dendritic cell.(Muzio et al., 2000) In the present study, the expressions of MCP-1 and TLR-2 were increased in male EAE mice, suggesting a potential contribution of MCP-1 and TLR-2 to cardiac dysfunction.
Limitations
We did few studies about CNS inflammation differences among mouse strains, or changes in MHC and complement, which may also contribute to autoimmune disease dysfunction.(Yu and Whitacre, 2004) Further, we did not examine inflammatory mechanism of cardiac dysfunction in female EAE mice. Thus, further study is required to fully determine the mechanism of cardiac dysfunction in EAE and the effects of sex and immune response on cardiac dysfunction.
Conclusion
We found that EAE induce cardiac dysfunction, with male mice showing more severe cardiac dysfunction and the expression of canonical inflammatory mediator (e.g., MCP-1, TGF-β and TLR-2) than female EAE. Meanwhile male EAE showed the increase of the oxidative stress (NOX-2) expression in heart, as well as cardiac hypertrophy, left ventricular dilatation, left ventricle (LV) mass increase and more severe fibrosis compared with male control mice. Sex difference of the immune response to pathogens has been reported, (Rizzetto et al., 2018) moreover vascular inflammation can induce neuroinflammation via systemic inflammation.(Hashizume et al., 2019) We suggest that increased cardiac canonical inflammatory mediator following EAE may be induced via a systemic inflammatory response. Further, increased cardiac canonical inflammatory mediator may be involved in EAE-induced cardiac dysfunction. Thus, regulation of inflammation is potential therapeutic strategy to improve the systemic and CNS pathology in EAE.
Ethics
All experiments were conducted in accordance with and were approved by the National Institutes of Health guidelines for the Animal Care and Use Committee of Tianjin Medical University General Hospital.
Footnotes
Acknowledgments
Author Contributions
Ruixia Wu: experimental design, wrote the manuscript, analyzed data and gave final approval of manuscript; Yue Su: performed experiments; Quan Yuan: performed experiments, performed immunostaining; Linlin Li: performed experiments; Jimusi Wuri: performed experiments; Xiaoxuan Liu: performed experiments; Tao Yan was involved in experimental design, wrote the manuscript, analyzed data and gave final approval of manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China grant (81671144 and 91746205) and Tianjin Natural Science Foundation key Project (17JCZDJC36100).
