Abstract
Glial cells perform important supporting functions for neurons through a dynamic crosstalk. Neuron–glia communication is the major phenomenon to sustain homeostatic functioning of the brain. Several interactive pathways between neurons and astrocytes are critical for the optimal functioning of neurons, and one such pathway is the ephrinA3–ephA4 signaling. The role of this pathway is essential in maintaining the levels of extracellular glutamate by regulating the excitatory amino acid transporters, EAAT1 and EAAT2 on astrocytes. Human immunodeficiency virus-1 (HIV-1) and its proteins cause glutamate excitotoxicity due to excess glutamate levels at sites of high synaptic activity. This study unravels the effects of HIV-1 transactivator of transcription (Tat) from clade B on ephrinA3 and its role in regulating glutamate levels in astrocyte–neuron co-cultures of human origin. It was observed that the expression of ephrinA3 increases in the presence of HIV-1 Tat B, while the expression of EAAT1 and EAAT2 was attenuated. This led to reduced glutamate uptake and therefore high neuronal death due to glutamate excitotoxicity. Knockdown of ephrinA3 using small interfering RNA, in the presence of HIV-1 Tat B reversed the neurotoxic effects of HIV-1 Tat B via increased expression of glutamate transporters that reduced the levels of extracellular glutamate. The in vitro findings were validated in autopsy brain sections from acquired immunodeficiency syndrome patients and we found ephrinA3 to be upregulated in the case of HIV-1-infected patients. This study offers valuable insights into astrocyte-mediated neuronal damage in HIV-1 neuropathogenesis.
Introduction
Central nervous system (CNS) infection by human immunodeficiency virus-1 (HIV-1) leads to HIV-associated neurocognitive disorders (HAND), mostly due to astrocyte-mediated neuronal death. HAND is characterized by neurocognitive and motor deficits in acquired immunodeficiency syndrome (AIDS) patients. The HIV-associated neurocognitive disorder is a major concern in >50% of HIV patients (Sacktor et al., 2016). Albeit advances in the widely used combinational antiretroviral therapy (cART) have significantly increased the life expectancy of people living with HIV by reducing the virus replication in blood and immune reconstitution, clearance of the virus from the brain remains a challenge. Harboring of the virus in the brain, particularly in astrocytes, causes neurological and cognitive defects (McArthur et al., 2010). Neurons in the brain of HIV-infected patients are not infected, but glial cells, although seemingly more infected due to their large numbers in CNS, serve as HIV reservoirs. Astrocytes increase the risk of HIV neuropathology via indirect mechanisms causing neuronal death (Fatima et al., 2017). Secretory protein HIV-1 transactivator of transcription (Tat) is released by HIV-infected non-neuronal cortical cells (astrocytes and microglia) (Bagasra et al., 1996; Brack-Werner, 1999) causing neuronal injury (Liu et al., 2000). As the proviral HIV-1 integrates into the host genome, Tat is the first viral protein to be transcribed and translated that helps in the recruitment of host elongation factor on the HIV-1 RNA and thus increase the rate by 100 folds, compared to the normal rate of viral transcription (Bagashev & Sawaya, 2013; Spector et al., 2019). Most studies are focused on HIV-1 Tat from subtype B, which is 101 amino acids long. HIV-1 subtype B, which is common in America and Europe, has been involved in neurotoxicity and brain damage in various ways. HIV-1 Tat has been detected in the cerebrospinal fluid of the patients where the viral load is almost undetectable in AIDS patients due to cART (Marino et al., 2020a; Taylor et al., 2008).
Neuron–glia interplay is quintessential for maintaining homeostatic brain functioning. Among the juxtacrine signaling in nervous system development, Eph:Ephrin is a contact-dependent bidirectional signaling between neighboring glia cells and neurons, and neighboring axons and dendrites in the CNS (Egea & Klein, 2007; Hruska & Dalva, 2012). Ephrin family members are ligands that coordinate signaling with their cognate Eph receptors, a family of receptor tyrosine kinases. The Eph receptors and its ligand Ephrins are constitutively expressed in the developing brain in areas such as the retina, optic tectum/superior colliculus, thalamus, neocortex (Wang et al., 2016) and the adult nervous system (Tuzi & Gullick, 1994). Cell contact-dependent Eph:Ephrin signaling is crucial for excitatory synaptic structure (Murai & Pasquale, 2002), embryonic CNS development, axon guidance and axonal connections as well as pathway selection and neural patterning (Egea & Klein, 2007; Flanagan & Vanderhaeghen, 1998). In addition to adult neurogenesis (Yamaguchi & Pasquale, 2004), neuroblast migration and cell proliferation in the adult subventricular zone (Conover et al., 2000), Eph:Ephrin can also regulate cell adhesion or cell movement, cell survival and differentiation (Genander & Frisen, 2010). Ephrin interaction has been implicated in long-term potentiation (LTP) during adulthood and is a modifier in amyotrophic lateral sclerosis (Van Hoecke et al., 2012). In doing so, these juxtacrine signaling plays a major role in the recruitment, formation and functioning of the N-methyl-D-aspartate (NMDA) receptors (Nolt et al., 2011). Among ligands, EphrinAs are also expressed in dopaminergic brain areas (Wang et al., 2016). EphrinA3 and ephA4 interaction needs cell–cell contact as they are tethered on the plasma membrane of astrocytes and neurons respectively (Thompson, 2003). Astrocytic glutamate transporters EAAT1 and EAAT2 are the human homologs of murine glutamate-aspartate transporter (GLAST) and glutamate transporter-1 (GLT-1), respectively, and serve as a major role in transport by reuptake of glutamate released at excitatory synapses (Jimenez et al., 2014). Detailed analysis of ephrinA3 knockout mice and ephA4 conditional knockout mice demonstrate that astrocytes regulate the expression of excitatory amino acid transporters, GLAST/EAAT1 and GLT-1/EAAT2 (Carmona et al., 2009; Filosa et al., 2009) which controls the long-term synaptic changes, function and plasticity. Astrocytes play a multifunctional role in brain physiology that includes homeostasis, metabolism, release and uptake of glutamate, gamma-aminobutyric acid and adenosine, fundamental neurotransmitters within the CNS. Glutamate has neurotoxic potency, thus uptake of excess glutamate and its clearance by their transporters expressed on the astrocytes is essential (Choi et al., 1987). Among the five types of glutamate transporters, GLAST/EAAT1 and GLT-1/EAAT2 are widely distributed in the brain (Gegelashvili & Schousboe, 1998). Moreover, astroglia is a primary site that bears the brunt of clearing excess glutamate in the brain areas with high glutamate activity thereby preventing glutamate neuroexcitotoxicity (Hindeya Gebreyesus & Gebrehiwot Gebremichael, 2020).
HIV- 1 mediated neuronal toxicity is essentially indirect and glial mediated, as these cells harbor the virus in the brain (Pandey & Seth, 2019). Glutamate excitotoxicity is the major cause of neuronal death in HIV-infected patients. Previous literature suggests that HIV-1 exposure regulates the glutamate transporter EAAT2 in human primary astrocytes (Cisneros & Ghorpade, 2014), whereas autopsy study of HAND patients indicates that HIV-1 Tat protein can reduce the expression of EAAT2 in astrocytes (Ye et al., 2017). As ephrinA3 is known to regulate glutamate transporters in astrocytes (Carmona et al., 2009), we aimed to study the role of Ephrin A3 in the regulation of EAAT1 and EAAT2 glutamate transporters and its role in HIV induced neuropathology.
Materials and Methods
Cell Culture
Neural progenitor cells were isolated from aborted human fetal brain, collected after informed consent of mothers, following protocols approved by institutional human ethics committee and guidelines laid by Indian Council for Medical Research, New Delhi, India. The stem cells were cultured as monolayers on poly-D-Lysine (PDL) (Cat. #P7280, Sigma-Aldrich, St. Louis, MO) coated flasks in neurobasal media (Cat. #21103-049, Invitrogen, San Diego, CA) containing pen strep (Cat. #15140122, Invitrogen, USA), neural survival factor (Cat. #CC-4323, Lonza, Charles City, IA), L- glutamine (Cat. #G7513, Sigma-Aldrich, St Louis, USA), N2 supplement (Cat. #17502-048, Invitrogen, USA) and gentamicin (Cat. #G1522, Sigma-Aldrich, USA), supplemented with 20 ng/ml of epidermal growth factor (Cat. #AF-100-15, Peprotech, USA) and 25 ng/ml of basic-fibroblast growth factor (Cat. #100-18B, Peprotech, USA) (Fatima et al., 2016). More than 95% of cells expressed stem cell markers, Nestin and Sox-2. For neuronal differentiation, the growth factors were replaced with brain-derived neurotrophic factors (Cat. #450-02, Peprotech, USA) and platelet-derived growth factor-AB (Cat. #100-00AB, Peprotech, USA). Post differentiation, cells were positive for neuronal marker microtubule-associated protein 2 (MAP2). Neural progenitor cells were differentiated into mature astrocytes for 21 days by replacing the media with minimal essential medium (MEM) (Cat. #M0268-10X, Sigma-Aldrich, USA) with 10% fetal bovine serum (Cat. #10270-106, Invitrogen, SA). Differentiated cells stained positive for glial fibrillary acidic protein (GFAP) marker. Passaging of cells was done with 0.1% trypsin (Cat. #T3924, Sigma-Aldrich, USA). Differentiated astrocytes and neurons were used to prepare neuron–glia co-cultures by overlaying the astrocytes with neurons at the density of 2:1 in astrocytic and neuronal media (1:1). As all the experiments were transfection based, and to ensure that only the astrocytes express HIV-1 Tat B, the astrocytes were first seeded to the density of 0.5 × 106 cells in T25 flasks and then transfected with the plasmid or small interfering RNA (siRNA), followed by overlaying the neurons at a density of 0.25 × 106 cells in a 2:1 astrocyte to neuron ratio, to mimic the normal brain environment.
Immunocytochemistry
For immunocytochemistry (ICC), astrocytes and neurons were seeded separately in 8-well chambered slide with a density of 10,000 and 25,000 cells per well, respectively. For co-culture staining astrocyte and neuron were seeded in 2:1 ratio. The cells were fixed with 4% paraformaldehyde (PFA). Blocking and permeabilization were done with 4% bovine serum albumin (BSA) (Cat. #A9418, Sigma-Aldrich, USA) and 0.3% Triton-X 100 for 1 h at room temperature (RT). Primary antibodies of EphrinA3 (Cat. #ab64814, RRID: AB_2246429, 1:1000, Abcam, Cambridge), ephA4 (Cat. #ab5389, RRID:AB_304853, 1:1000, Abcam, Cambridge) and MAP2 (Cat. #MAB3418, RRID:AB_94856, 1:1000, Chemicon, USA, Cat. #ab5392, 1:500, RRID:AB_2138153, Abcam, Cambridge) were incubated at 4 °C overnight whereas GFAP (Cat. #MAB360, RRID:AB_11212597, 1:1000, Millipore, USA) was incubated at RT for 1 h. Cells were then labelled with secondary antibodies, Alexa Fluor 488 goat anti-mouse and Alexa Flour 594 goat anti-rabbit (Cat. #A-11001, RRID:AB_2534069, #A-11037, RRID:AB_2534095, Invitrogen, USA) and goat anti-Chicken Cy3 (Cat. #ab97145, RRID:AB_10679516
Immunohistochemistry
Autopsy paraffin-embedded brain sections (5 µm thick) of HIV-infected patients and their age-matched controls were obtained from the Human Brain Tissue Repository at the National Institute of Mental Health and Sciences, Bangalore, India (Table 1). The sections were from the hippocampal regions of HIV patients and age-matched controls, who succumbed to accidental deaths. The age group ranged from 22–48 years. The sections were deparaffinized using xylene and decreasing concentration of alcohol and finally hydrated in 1 × phosphate-buffered saline (1 × PBS). Antigen retrieval was done using citrate-based antigen unmasking solution (Cat. #H-3300, RRID: AB_2336226, Vector Labs, USA). The sections were then treated with 3% hydrogen peroxide to eliminate any endogenous peroxidase activity. Permeabilization of the sections was done with 0.1% Triton-X 100. Sections were kept in blocking solution containing 2% BSA (Cat. #A9418, Sigma-Aldrich, USA), 5% Normal goat serum (Cat. #S-1000, RRID:AB_2336615, Vector Labs, USA) and 0.1% Triton-X 100 for 3 h at RT. Post blocking, the primary antibody of ephrinA3 (Cat. #ab239067, 1:100, Abcam, Cambridge) and ephA4 (Cat. #ab5389, RRID:AB_304853, 1:1000, Abcam, Cambridge)was incubated overnight at 4 °C. The next day the sections were washed with 1 × PBS followed by secondary goat anti-rabbit biotinylated antibody (Cat. #BA-1000, RRID: AB_2313606, 1:500, Vector Labs, USA) for 3 h at RT. The sections were then exposed to avidin–biotin complex (ABC) peroxidase solution using the Vectastain Elite ABC kit (Cat. #PK-6100, RRID: AB_2336819, Vector Labs, USA) and developed the sections with peroxidase DAB substrate kit (Cat. #SK-4100, RRID: AB_2336382, Vector Labs, USA). The sections were imaged in Leica DMRXA2 (Germany). 10–12 random images were taken from the hippocampal region by a person blinded to the experiment. The staining was quantitated using the ImageJ software (NIH, Bethesda, USA) by measuring the intensity of the ephrinA3 stain. The images were converted to 8-bit grayscale and the intensity was quantified by measuring the mean puncta count of ephrinA3 staining by subtracting the background image.
Details of Post-Mortem Brain Sections of Age-Matched Control and HIV-1 Subjects.
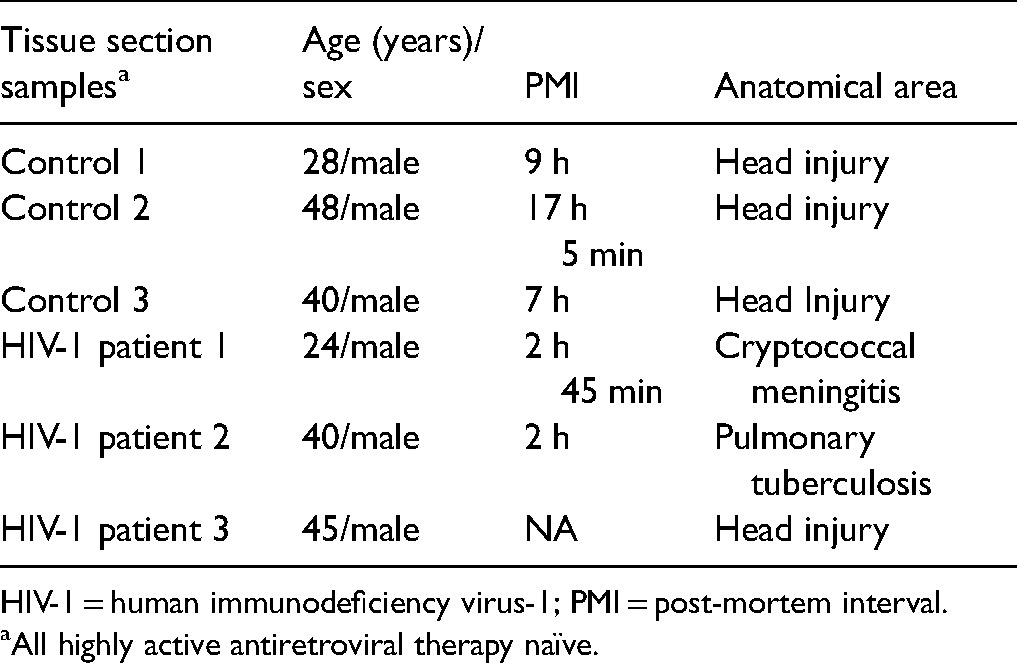
HIV-1 = human immunodeficiency virus-1; PMI = post-mortem interval.
All highly active antiretroviral therapy naïve.
Knockdown and Transfection
Knockdown of ephrinA3 was done with Mission esiRNA (Cat. #EHU029191, Sigma-Aldrich, USA). The concentration of the siRNA was standardized at 40 pmol. Scrambled Mission esiRNA (Cat. #SIC001, Sigma-Aldrich, USA) was used as control. The pcDNA 3.1 expressing full-length Tat B plasmid was used to transfect co-cultures along with pcDNA 3.1 empty vector as control (gift from Dr. Udaykumar Ranga, JNCASR, India). All transfections were carried out with Lipofectamine 2000 (Cat. #11668027, Invitrogen, USA) as per the manufacturer's protocol in opti-MEM media (Cat. #31985-070, Invitrogen, CA) with the incubation time of 24 or 48 h at 37 °C.
RNA and Protein Isolation
RNA and protein isolation were done by phenol-chloroform extraction principle using TRIzol reagent (Cat. #15596018, Invitrogen, USA). The middle aqueous phase was processed for RNA extraction and the lower pink phase was processed for protein extraction. The protein pellet was resuspended and dissolved in 1% sodium dodecyl sulfate (Cat. #L3771, Sigma-Aldrich, USA). After assessing the RNA concentration, the samples were treated with DNase1 (Cat. #18068-015, Invitrogen, USA) to eliminate any DNA contamination and then used for the synthesis of complementary DNA (cDNA) using the High-Capacity cDNA Reverse transcription kit (Cat. #4368814, Applied Biosystems, USA). Protein samples were processed for quantification and western blotting.
Western Blotting
The concentration of the isolated proteins was determined by bicinchoninic acid (Cat. #B9643, Sigma-Aldrich, USA) and 4% copper sulfate with absorbance measured at 540 nm in Tecan Spark plate reader (Switzerland). 60–80 µg of protein samples were loaded in 10% acrylamide gels. The gel was transferred on the nitrocellulose membrane (Cat. #SCNJ, MDI, India) and the integrity of protein was checked by 0.1% ponceau. Blots were blocked with 5% skimmed milk (Cat. #1.15363.0500, Millipore, USA) in 1 × PBS-Tween20 for 2 h at RT and then incubated with primary antibodies, EphrinA3 (Cat. #ab64814, RRID:AB_2246429, 1:1000, Abcam, Cambridge), EAAT1 (Cat. #NBP1-42587, RRID:AB_2190584, 1:1000, Novus biological, USA), EAAT2 (Cat. #NBP1-59632, RRID:AB_11005463, 1:1000, Novus biological, USA) NMDAR2B (Cat. #ab65875, 1:500, RRID:AB_1141850), β-tubulin (Cat. #T4026, RRID:AB_477577, 1:4000, Sigma-Aldrich, USA) and Tat B antibody raised in rabbit in our lab at 4 °C overnight. The blots were then washed with 1 × Tris-buffered saline-Tween20 (1 × TBST) thrice and probed with horseradish peroxide (HRP)-conjugated secondary antibody (Cat. #PI-1000, RRID: AB_2336198, #PI-2000, RRID: AB_2336177, 1:4000, Vector Labs, USA) for 2 h at RT. Blots were washed with 1 × TBST and then developed with chemiluminescent HRP substrate reagent (Cat. #WBKLS0500, Millipore, USA) and imaged using Uvitec Imaging system (Cambridge, UK). The protein bands were densitometrically quantified using the ImageJ software (NIH, Bethesda, USA) and β-tubulin was used as the normalization control.
Real-Time Polymerase Chain Reaction:
The cDNA samples prepared from RNA were used for quantitative-polymerase chain reaction (PCR) analysis. Real-time PCR was setup in ViiATM7 (Applied Biosystems, USA) using a Power SYBR green master mix (Cat. #4367659, Applied Biosystems, USA) and manually designed gene-specific primers as per the following: EphrinA3 forward 5′-CTACTACATCTCCACGCCCA-3′ and EphrinA3 reverse 5′-TCCCTCAAAGTCTTCCAGCA-3′, EAAT1 forward 5′-GAATGGCGGCGCTAGATAGT-3′ and EAAT1 reverse 5′-CCAGGCTTCTACCAGATTTG-3′, EAAT2 forward 5′-CAGGGAAAGCAACTCTAATC-3′ and EAAT2 reverse 5′-CAAGGTTCTTCCTCAACA-3′, glyceraldehyde 3-phosphate dehydrogenase (GAPDH) forward 5′-CAAGAGCACAAGAGGAAGAGAG-3′ and GAPDH reverse 5′-CTACATGGCAACTGTGAGGAG-3′. PCR cycle conditions were 1 cycle of 95 °C for 10 min followed by 95 °C for 20 s, 60 °C for 30 s and 72 °C for 40 s for 40 cycles. The specificity of the primers was detected by a single peak in the melt curve.
Glutamate Assay
Supernatant collected post 24 h of transfection was utilized immediately to minimize any glutamate degradation for the detection of extracellular glutamate. Glutamate assay was performed using the Glutamine and Glutamate Determination Kit (Cat. #GLN1, Sigma-Aldrich, USA) as per the manufacturer's protocol. Absorbance was measured in the Tecan Spark plate reader (Switzerland) at 340 nm. Concentration was calculated by plotting the standard curve for concentration against the absorbance.
Terminal Deoxynucleotidyl Transferase dUTP Nick End Labeling Assay
Supernatant collected 24 h post-transfection from the co-cultures with transfected astrocytes, was added on neurons seeded in 8-well chambered slides in a density of 25,000 cells per well (with neuronal media and supernatant in 1:1 ratio). The slides were then fixed with 4% PFA post 24hr incubation. Blocking and permeabilization were done with 4% BSA (Cat. #A9418, Sigma-Aldrich, USA) and 0.2% Triton-X 100 for 1 h at RT then processed for terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay using the In Situ Cell Death Detection Kit, TMR red (Cat. #12156792910, Roche, Germany) as per the manufacturer's protocol. Cells were then washed with 1 × PBS and mounted with the hardest DAPI (Cat. #H-1500, RRID: AB_2336788, Vector Labs, USA). Random images were taken across the slide using AxioObserver.Z1 Apotome (Carl Zeiss, Germany) by a person blinded to the experimental groups. Neuronal death was quantified as the percentage of TUNEL positive cells, by counting TUNEL positive cells coinciding with DAPI positive cells divided by the total DAPI positive cells, by using the ImageJ software (NIH, Bethesda, USA).
Statistical Analysis
All the experimental control and treated groups were analyzed for their statistical significance using the Student's t-test. Multiple transfection groups were analyzed by one-way analysis of variance. P-value< 0.05 was considered to be statistically significant. Each experiment was performed and repeated three to five times independently as indicated in the legends of each figure.
Results
Establishing and Characterization of the Neuron–Glia Co-Cultures
From previous literature, it is known that ephrinA3 is majorly expressed on astrocytes whereas its receptor ephA4 is expressed on the neurons in the adult brain (Thompson, 2003). To confirm this in our culture system, we relied on ICC tools. Before studying the effects of HIV-1 Tat B, the expression profile of ephrinA3 and ephA4 was checked on astrocytes and neurons respectively. Primary human neural progenitor cells differentiated into astrocytes and neurons over a period of 21 days were stained individually to check the expression of ephrinA3 on astrocytes and ephA4 on neurons along with their characteristic markers GFAP (for astrocytes) and MAP2 (for neurons). Upon immunostaining, it was confirmed that the differentiated astrocytes positively expressed ephrinA3 (Figure 1A) and ephA4 was highly expressed on the differentiated neurons (Figure 1B). Insets clearly show the punctate staining.
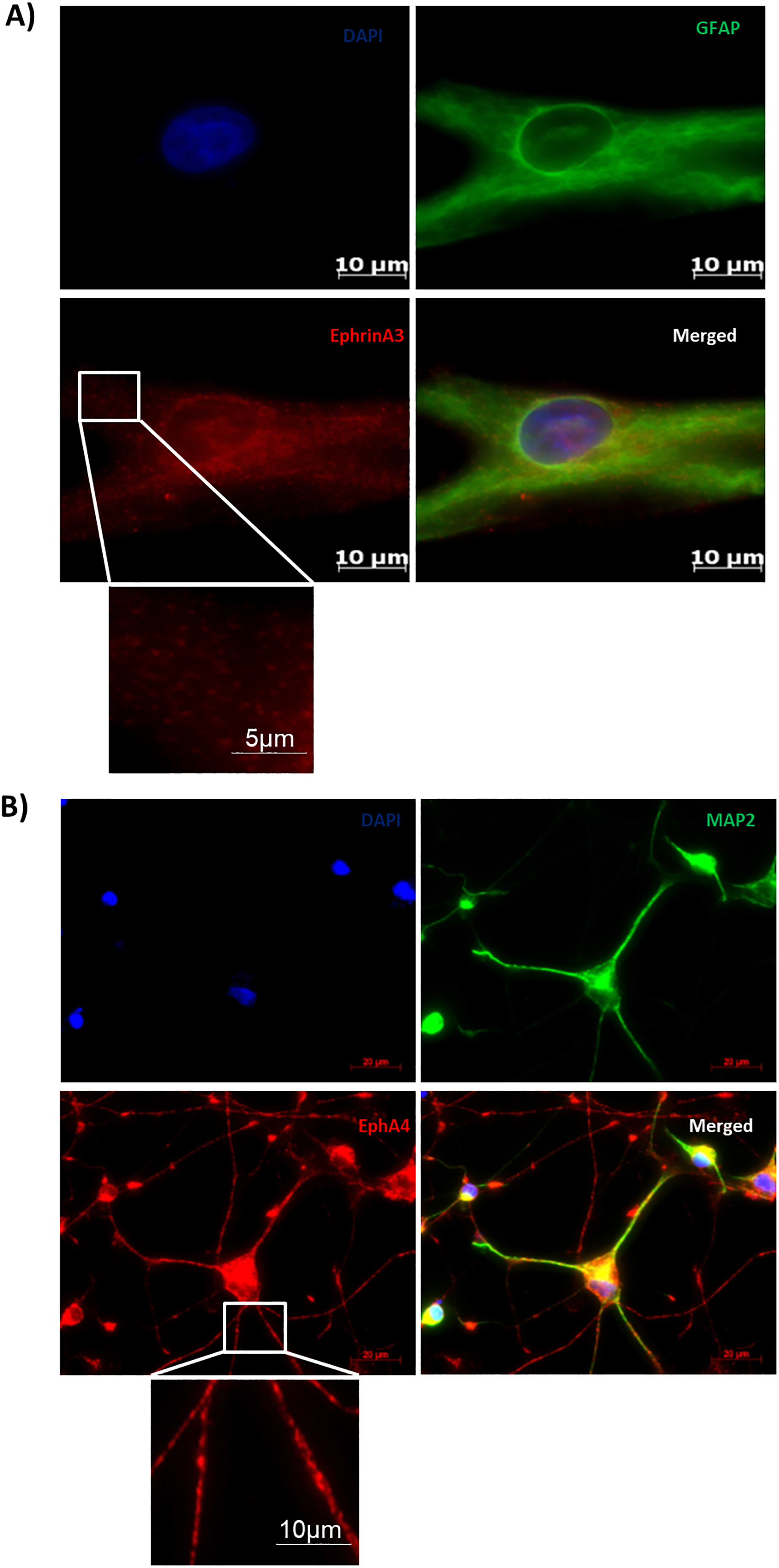
Ephrina3 is expressed on astrocytes and ephA4 is expressed on neurons: astrocytes and neurons were differentiated from human fetal derived neural progenitors and then stained with ephrinA3 and ephA4 antibodies, respectively, to check their expression on these cells, (A) astrocytes are fixed and stained with ephrinA3 (red) and GFAP (green, marker for mature astrocytes) and mounted with DAPI (blue) (scale bar: 10 µm), (B) neurons are fixed and stained with ephA4 (red) and MAP2 (green, marker for mature neurons) (scale bar: 20 µm). The insets are a magnification of the areas indicated to show punctate staining.
EphrinA3 ligand expressed on astrocytes, when binds to its receptor epha4, expressed on the neurons, elicits a reverse signaling in the astrocytes which is known to regulate glutamate transporters on astrocytes (Carmona et al., 2009). For this specific reason, all the experiments were performed in neuron–glia co-cultures, which mimic the brain environment in vitro. The differentiated astrocytes were first plated on PDL-coated flasks and were then over-layered with mature neurons to form neuron–glia co-culture in a 2:1 astrocyte to neuron ratio. We were successful in establishing neuron–glia co-cultures as observed in the live-cell image where the neuron could be seen making connections with the astrocytes (Figure 2A). The co-culture was stained to check for the specific markers of both cell types to confirm the development of neuron–glia co-culture. The astrocytes expressed GFAP and neurons expressed MAP2 positively as seen in immunostaining, hence affirming successful establishment of neuron–glia co-culture (Figure 2B). Also, astrocytes and neurons expressed very low levels of ephA4 and ephrinA3 respectively (Supplementary Figure 1). All further experiments were performed using this as the model system, where the astrocytes were first transfected with specific plasmids or siRNA to ensure their expression only in astrocytes and then overlaid with neurons.
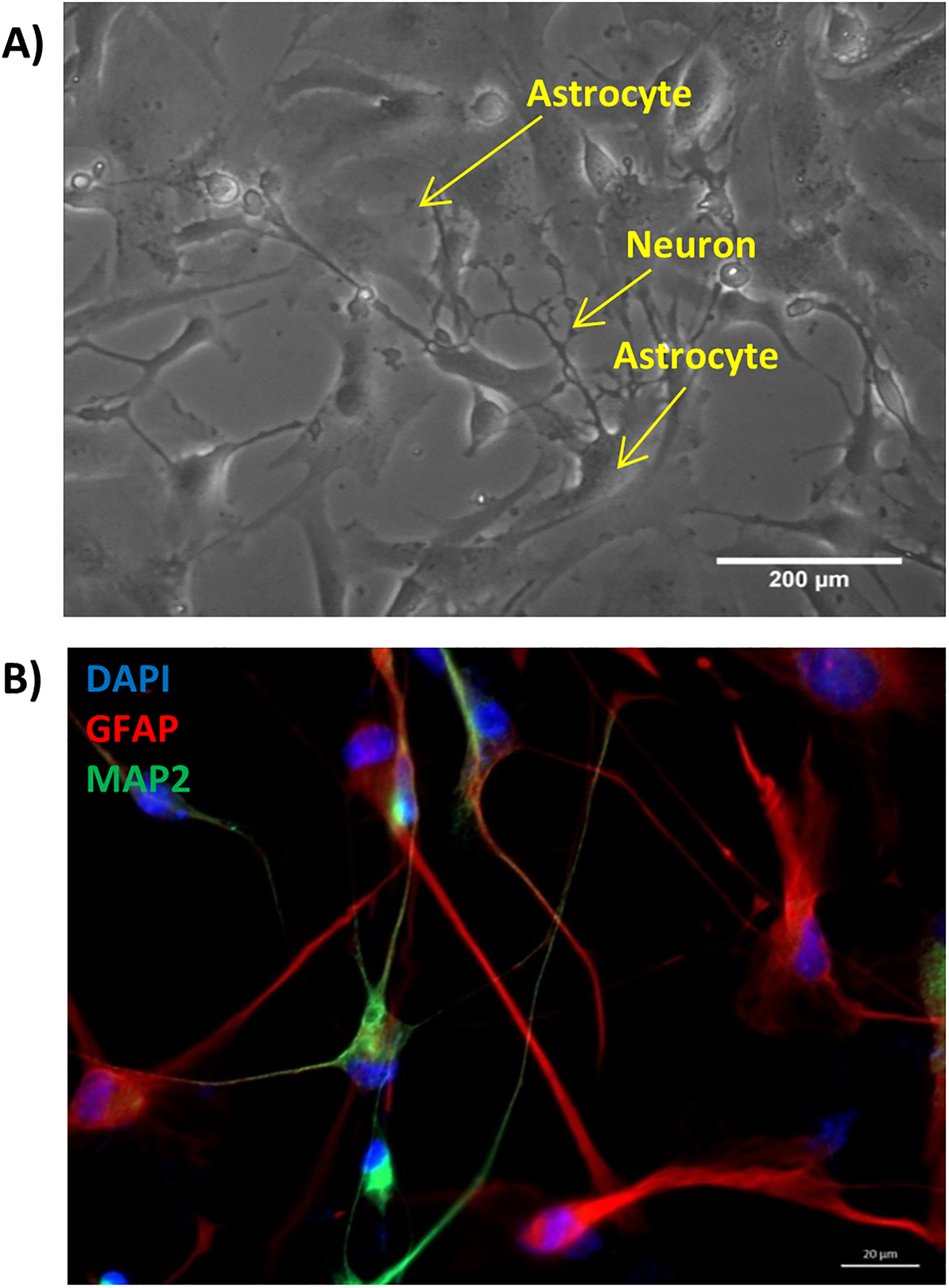
Neuron–glia co-cultures established as the model system—human fetal derived neural progenitor cells were differentiated into astrocytes and neurons for a period of 21 days, neurons were overlaid on astrocytes (in a 1:2 ratio) to create neuron–glia co-cultures, (A) a live-cell bright field image of established neuron–glia co-culture (scale bar: 200 µm), (B) neuron–glia co-culture fixed and stained with GFAP (red, marker for mature astrocytes) and MAP2 (green, marker for mature neurons) and mounted with DAPI (blue), (scale bar: 20 µm).
EphrinA3 is Upregulated by Overexpression of HIV-1 Tat B and in Autopsy Tissues of HIV-1-Infected Patients
Previous studies from our lab have shown the effects of HIV-1 Tat B protein on alterations in proliferation and differentiation in neural progenitor cells (Malik et al., 2011) and its toxic effects on purinergic receptors in astrocytes (Tewari & Seth, 2015). Astrocytes act as a viral reservoir since they harbor HIV, they also cause neuronal toxicity via indirect pathways due to dysregulated glutamate uptake (Pandey & Seth, 2019). As ephrinA3 regulates glutamate transporters, we investigated whether its expression is altered under the effects of HIV-1 Tat B. Therefore, we transfected the astrocytes in the neuron–glia co-cultures with HIV-1 Tat B expressing plasmid for indigenous production of Tat B by astrocytes and analyzed its effect on ephrinA3 expression. Transfection with HIV-1 Tat B elevated ephrinA3 expression up to 64% post 24 h and 26% post 48 h of transfection at the protein level as compared to the expression in the vector control (*p < .05) (Figure 3). 72 h transfection showed a 34% increase in ephrinA3 expression (*p < .05) (Supplementary Figure 2A). The indigenous production of Tat B and its presence was confirmed at the protein level as observed in the representative blot images (Figure 3). As ephrinA3 signaling regulates glutamate transporters, this result suggests that upregulation of ephrinA3 in the presence of HIV-1 Tat B may hinder glutamate transport in astrocytes. In line with this observation, we first assessed the alteration in the levels of glutamate transporters, EAAT1 and EAAT2 by overexpression of HIV-1 Tat B.
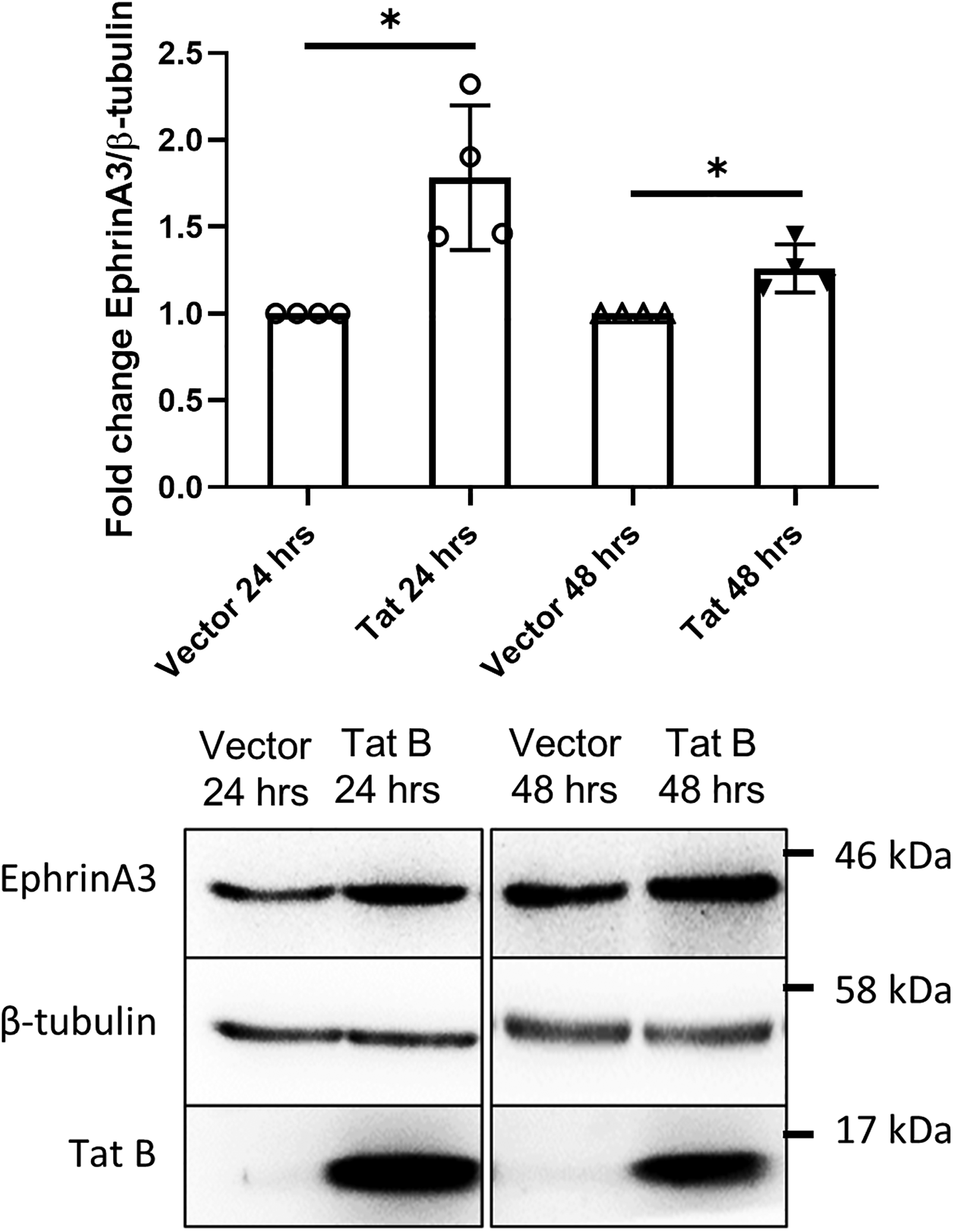
Expression of ephrinA3 is upregulated when transfected with HIV-1 Tat B: ephrinA3 protein expression was checked by transfecting astrocytes in co-cultures with vector control and HIV-1 Tat B expressing plasmids and protein was isolated by TRIzol method 24 and 48 h post-transfection and separated by SDS-PAGE. Blots were probed with ephrinA3, β-tubulin and Tat B antibodies. β-tubulin was used for normalization control. The bottom panel shows representative blot images and the top panel shows the bar graph depicting the densitometric analysis of the blots. This data is representative of four different independent experiments, n = 4, *p < .05.
To validate the upregulation of ephrinA3 in case of HIV infections, we checked the expression of ephrinA3 in autopsy brain sections of HIV patients and control subjects. Sections from the hippocampus and frontal cortex were stained for ephrinA3 as these two regions in the brain are amongst the highly affected in HIV-1 patients (Everall et al., 1991; Kruman et al., 1998). Both the hippocampus and frontal cortex showed 3.3 folds and 3.7 folds significant increase respectively in the expression of ephrinA3 in HIV-1 patients as compared to their age-matched controls (Figure 4). This data confirms the upregulation of ephrinA3 and therefore the possibility of ephrinA3 regulating the glutamate transporters and causing neuronal toxicity in HIV-1-infected patients is highly likely. The ratio of ephrinA3 and ephA4 is checked in normal subjects and ephA4 is expressed more as compared to ephrinA3 in the hippocampus of post-mortem brain sections (Supplementary Figure 5).
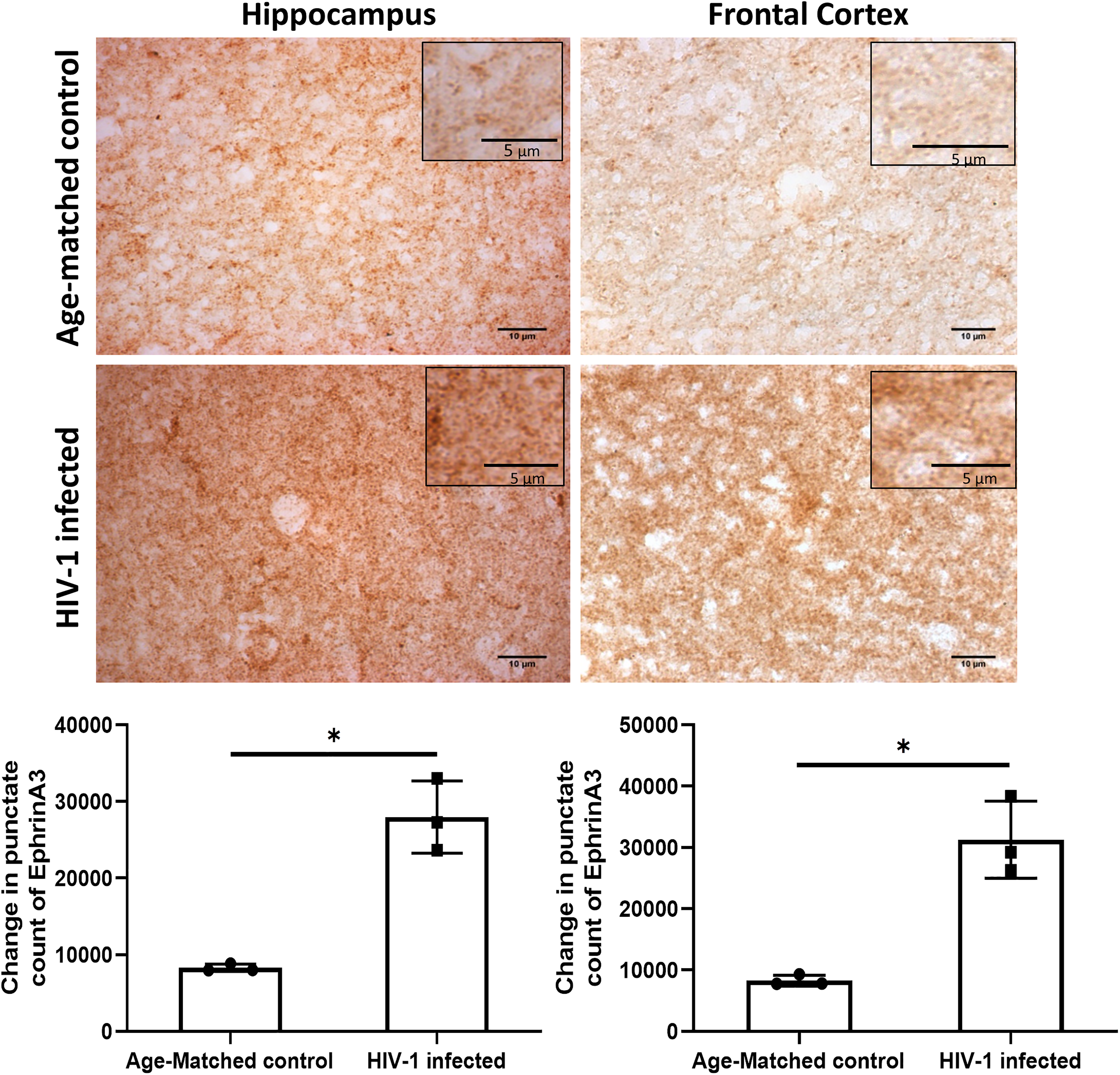
EphrinA3 overexpression in brain autopsy tissues of human immunodeficiency virus-1 (HIV-1) infected patients: the top panel shows representative images of ephrinA3 staining in age-matched controls and HIV-1-infected brain sections of the hippocampus and frontal cortex region (scale bar: 10 µm) with their inset images depicting punctate staining (scale bar: 5 µm). The lower panel shows the bar graph depicting the mean punctate count of ephrinA3. The data is representative of three different HIV-1-infected subjects and three different age-matched controls, n = 3, *p < .05.
HIV-1 Tat B Decreased Glutamate Transporters, EAAT1 and EAAT2, in Human Astrocytes
Glutamate excitotoxicity due to impaired glutamate uptake by astrocytes is one of the major contributors of astrocyte-mediated neuronal death in HIV-1 induced neurodegeneration (Marino et al., 2020b). This lowered uptake of glutamate by astrocytes is owed to the disruption of the glutamate transporters, EAAT1 and EAAT2 on astrocytes. As ephrinA3 regulates glutamate transporters, the expression of glutamate transporters, particularly EAAT1 and EAAT2, was assessed in co-cultures by transfecting the astrocytes with HIV-1 Tat B expressing plasmid. EAAT1 showed a decrease of nearly 51% and EAAT2 showed almost a 39% decrease in the presence of Tat B as compared to the vehicle control (*p < .05) (Figure 5A and B). This further confirmed our hypothesis that HIV-1 Tat B leads to downregulation of glutamate transporters, causing glutamate accumulation, which may be via alteration in ephrinA3 levels on astrocytes. To confirm the role of ephrinA3 in the regulation of glutamate transporters and glutamate uptake, we performed a knockdown of ephrinA3. Neurons express NMDA receptors and are therefore susceptible to glutamate excitotoxicity. We also observed a modest yet significant increase in the NMDA expression in co-cultures (*p < .05) (Supplementary Figure 3).
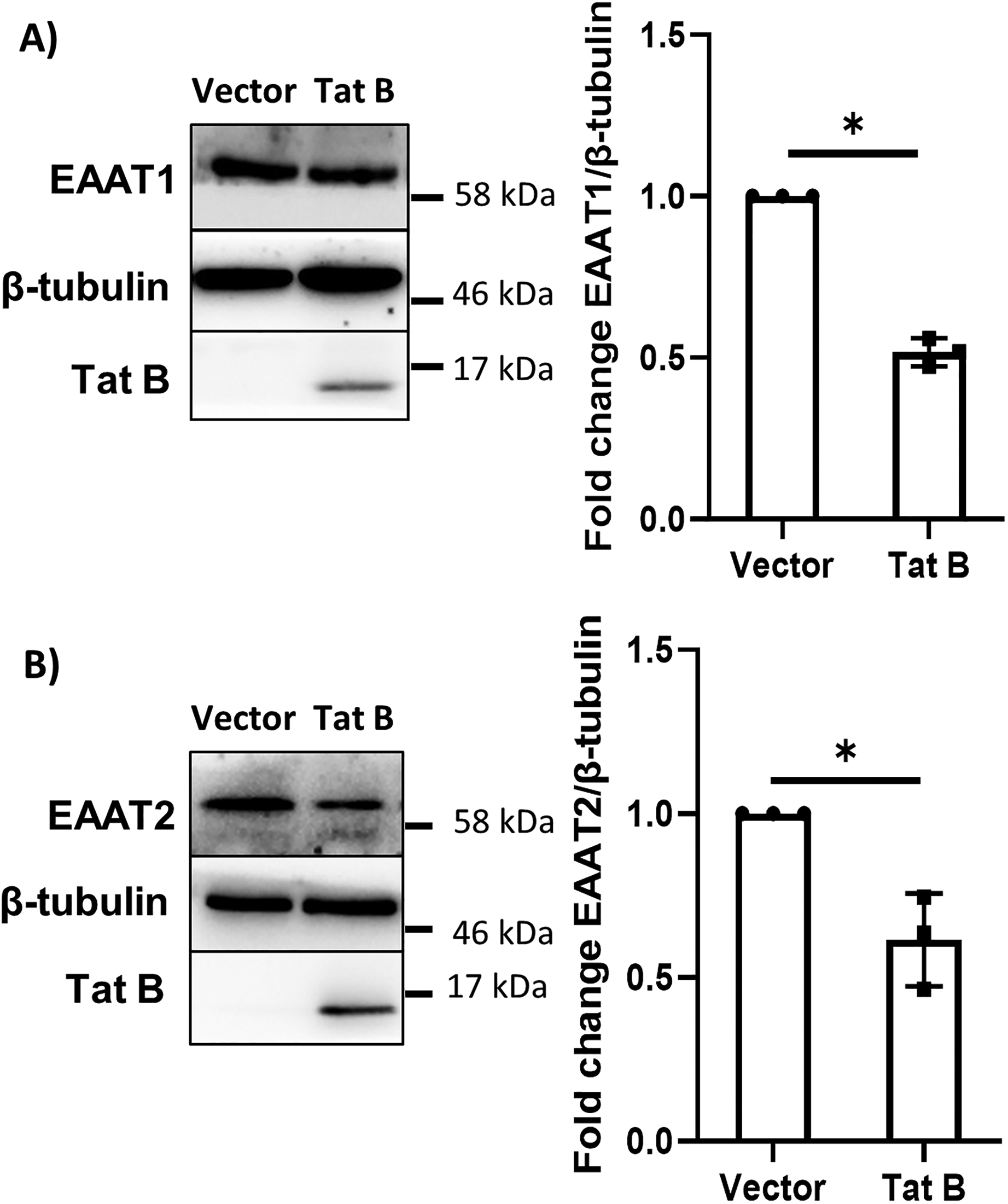
Expression of glutamate transporters EAAT1 and EAAT2 in neuron–glia co-cultures is reduced when transfected with Tat B: astrocytes in co-cultures were transfected with vector control and HIV-1 Tat B expressing plasmids, the protein was isolated and run on SDS-PAGE. The blots were then probed with EAAT1, EAAT2, β-tubulin and Tat B antibodies, (A) the expression of EAAT1 and (B) EAAT2 transporters decreased under the effect of Tat B as compared to that of control. The right panel shows the bar graphs depicting densitometric analysis of the blots and the left panel shows representative blot images. β-tubulin was used as a normalization control and the data represents cumulative results of three different independent experiments, n = 3, *p < .05.
EphrinA3 Knockdown in the Astrocytes Reverses the Tat B-induced Decrease in Glutamate Transporters
As ephrinA3 regulates glutamate level around synapses by regulation of glutamate transporters on the astrocytes and prevents glutamate excitotoxicity under physiological conditions, it was important to investigate its role in the presence/absence of HIV-1 Tat B. To evaluate the role of ephrinA3 in regulating glutamate transporters on astrocytes and to confirm its functionality; and to show that the decrease in the expression of glutamate transporters is in fact regulated by ephrinA3, ephrinA3 was knocked down using siRNA in astrocytes of the co-culture. First, the concentration of siRNA for efficient knockdown was standardized on the astrocytes and we found that 40 pmol showed more than 70% knockdown as compared to that of scrambled control at the messenger RNA (mRNA) level and more than 60% knockdown at the protein level in astrocytes. EphrinA3 knockdown was not affected in the presence of Tat B (Figure 6A and B). Hence further experiments were carried out with a siRNA concentration of 40 pmol. EphrinA3 knockdown showed a significant increase of 47% in EAAT1 and an increase of 80% in EAAT2 expression at the mRNA level as compared to the control (***p < .001) whereas Tat B showed a significant decline of 30% in EAAT1 and 52% in EAAT2 expression as opposed to the vector transfections (*p < .05, **p < .01). Upon co-transfection with ephrinA3 siRNA and Tat B (EphrinA3si + Tat B), knockdown of ephrinA3 resulted in reversal of Tat B-induced decrease in EAAT1 and EAAT2. Co-transfection led to a significant increase of 13% in EAAT1 and an increase of 39% in EAAT2 expression as compared to that of only Tat B transfection (**p < .01) and a 20% increase in EAAT1 and 38% increase in EAAT2 as compared to TatB + Scr (*p < .05). EphrinA3 knockdown hence thwarted the Tat B-induced decrease of the glutamate transporters (Figure 6C and D). These findings suggest a role of ephrinA3 in astrocytes in regulating the alteration of glutamate transporter expression in the presence of HIV-1 Tat B.
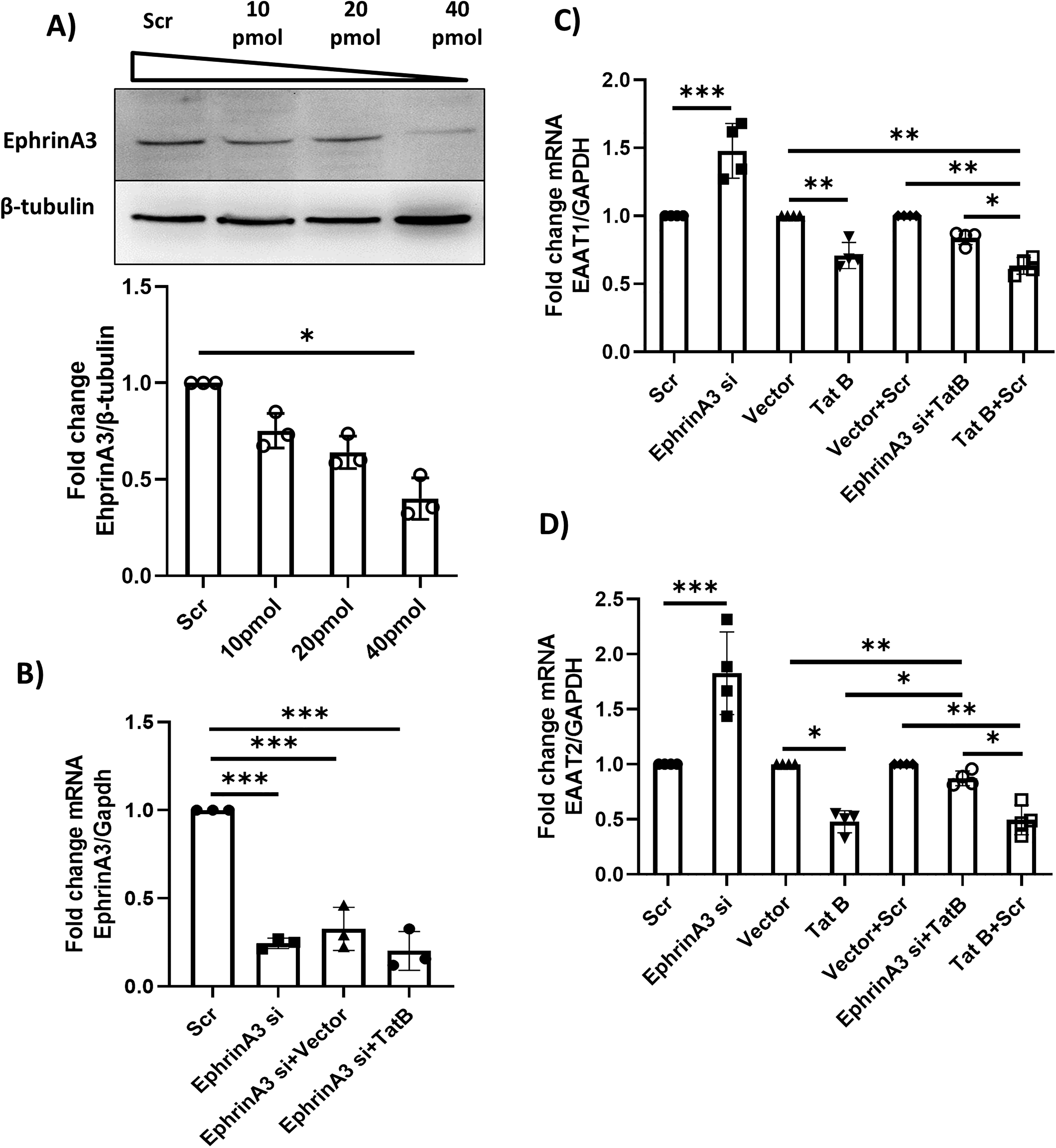
EphrinA3 siRNA and HIV-1 Tat B co-transfection reverse the effects of HIV-1 Tat B transfection: efficient knockdown concentration of ephrinA3 by siRNA transfection was standardized on astrocytes and its levels were checked post 24 h of transfection, (A) protein was isolated post-transfection and separated on SDS-PAGE. The blots were probed with ephrinA3 and β-tubulin antibody and significant knockdown was observed at 40 pmol concentration as compared to that of scrambled control. The top panel depicts referral images of blots and the bottom panel shows the densitometric analysis of the blots depicted by the bar graph. β-tubulin was used as the normalization control. Data is representative of three independent experiments, n= 3, *p < .05. (B) Bar graph showing ephrinA3 knockdown at the mRNA level and 40 pmol siRNA showed a significant reduction in the expression of ephrinA3 compared to scrambled control. EphrinA3 knockdown is not affected by Tat B transfection. GAPDH was used as the normalization control. Data represented is of three independent experiments, n = 3, ***p < .001. (C, D) Bar graph representing EAAT1 and EAAT2 expression at mRNA level post ephrinA3 knockdown and Tat B transfection. An increase in EAAT1 and EAAT2 expression is seen in the case of ephrinA3 knockdown and a decrease in Tat B transfection as compared to the controls. Co-transfection shows an increase in both EAAT1/EAAT2 as compared to that of Tat B and Tat B + scr. GAPDH was used as the normalization control and the data represents four independent experiments, n = 4, *p < .05, **p < .01, ***p < .001.
There was a decrease of 69% in the expression of Tat B, in the presence of ephrinA3 knockdown as compared to only Tat B transfection (Supplementary Figure 4A). We observed an increase in the expression of ephrinA3 when under Tat B transfection (63%) but the expression level reduced to 68% in the presence of ephrinA3 knockdown (Supplementary Figure 4B).
Extracellular Glutamate Levels are Reversed in Tat B and EphrinA3 siRNA Co-Transfection
According to our hypothesis, altered levels of glutamate transporters under ephrinA3 knockdown and Tat B transfections should have an effect on the levels of extracellular glutamate. The ideal way to study the efficiency of glutamate transporters in the uptake of excess glutamate was to measure the level of glutamate in the extracellular environment. Hence, we checked if there was any difference in extracellular glutamate levels in the supernatant of the co-cultures with transfected astrocytes. We observed an increase of 25% in the level of extracellular glutamate in the case of Tat B as compared to that of control (**p < .01), however, the same was reduced by 21% although not significant, when siRNA-induced knockdown of ephrinA3 was carried out in astrocytes. In the case of co-transfection with Tat B and ephrinA3 siRNA, the level of extracellular glutamate significantly decreased to 39% when compared with Tat B transfected alone (**p < .01). There is a significant increase in glutamate levels in Tat B when compared to ephrinA3 knockdown (***p < .001) (Figure 7).
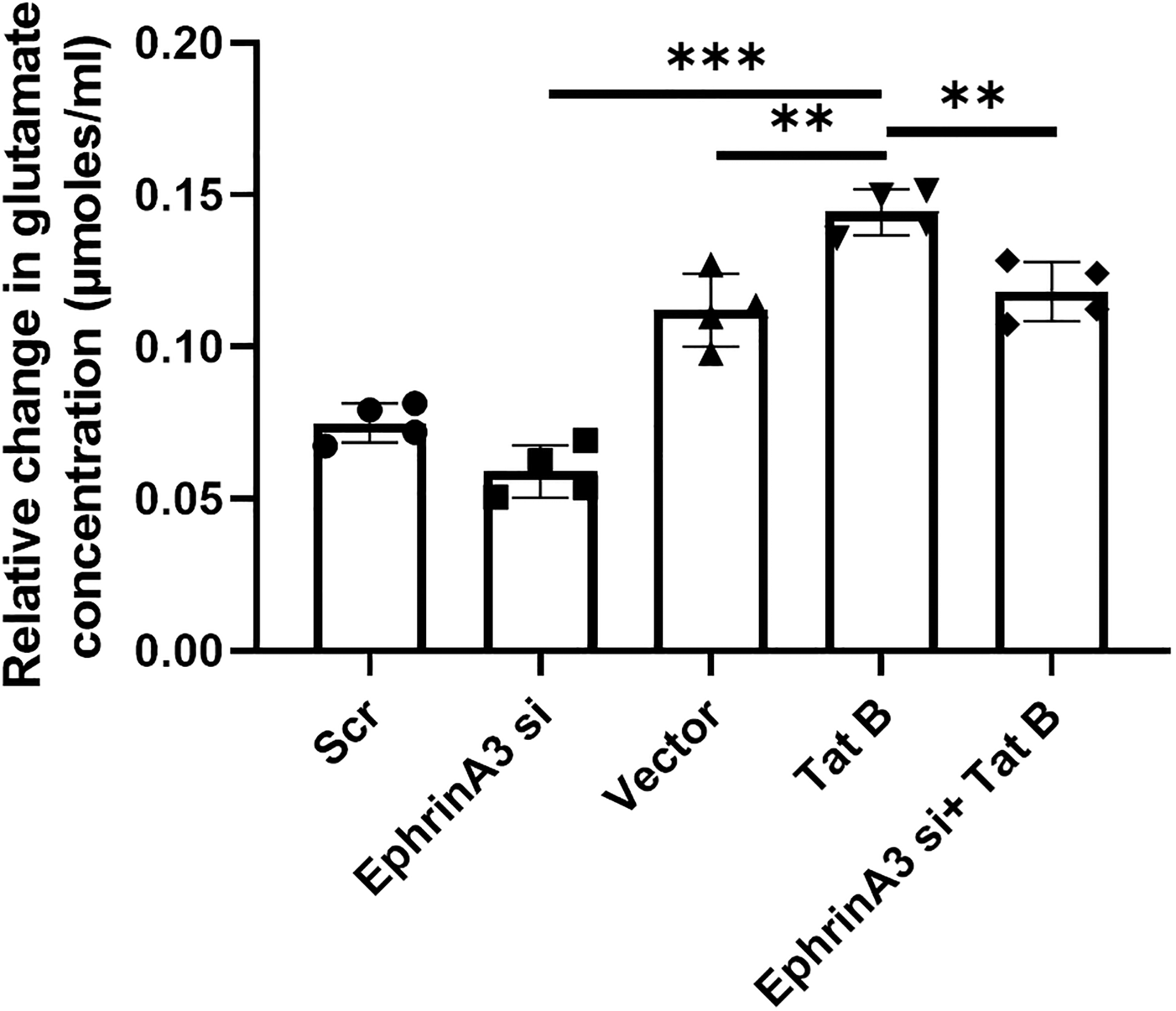
Bar graph showing extracellular glutamate levels post ephrinA3 and Tat B transfections: extracellular glutamate concentration was measured from the supernatant of co-cultures with transfected astrocytes post 24 h of transfection. Transactivator of transcription (Tat) B transfection showed a higher concentration of glutamate levels as compared to that of controls. Co-transfection showed a decline in the extracellular glutamate levels as compared to that of only Tat B. Tat B transfection showed a significant increase in glutamate levels when compared to ephrinA3 knockdown. Data is representative of four independent experiments, n = 4, **p < .01, ***p < .001.
This experiment proved that the changes in the expression of glutamate transporters lead to variation in extracellular glutamate due to the efficiency of uptake by the astrocytes and the importance of ephrinA3 in maintaining the extracellular glutamate levels via EAAT1/EAAT2.
EphrinA3 Knockdown Reverses the Tat B-induced Neuronal Death
An increase in the level of extracellular glutamate, which is considered to be one of the major causes of neuronal toxicity in HIV-1 patients (Gorska & Eugenin, 2020). To assess whether alterations in glutamate levels are critical for the astrocyte-mediated neuronal damage, and to deduce whether the increase in the level of extracellular glutamate has any effects on neuronal death, we performed a TUNEL assay on neurons. Supernatant from the co-cultures with transfected astrocytes that contains extracellular glutamate was added on mature neurons and neuronal death via an indirect pathway was assessed by the TUNEL assay. EphrinA3 knockdown did not show any significant neuronal death as compared to scrambled control whereas Tat B transfection showed a significant increase (2.5-fold) in neuronal death as opposed to that of vector control (*p < .05). Also, Tat B and ephrinA3 co-transfection showed a decline (3-fold) (**p < .01) in neuronal death as compared to that of Tat B transfection alone (Figure 8). We also observed moderate yet significant neuronal death at 72 h post-transfection (Supplementary Figure 2B).
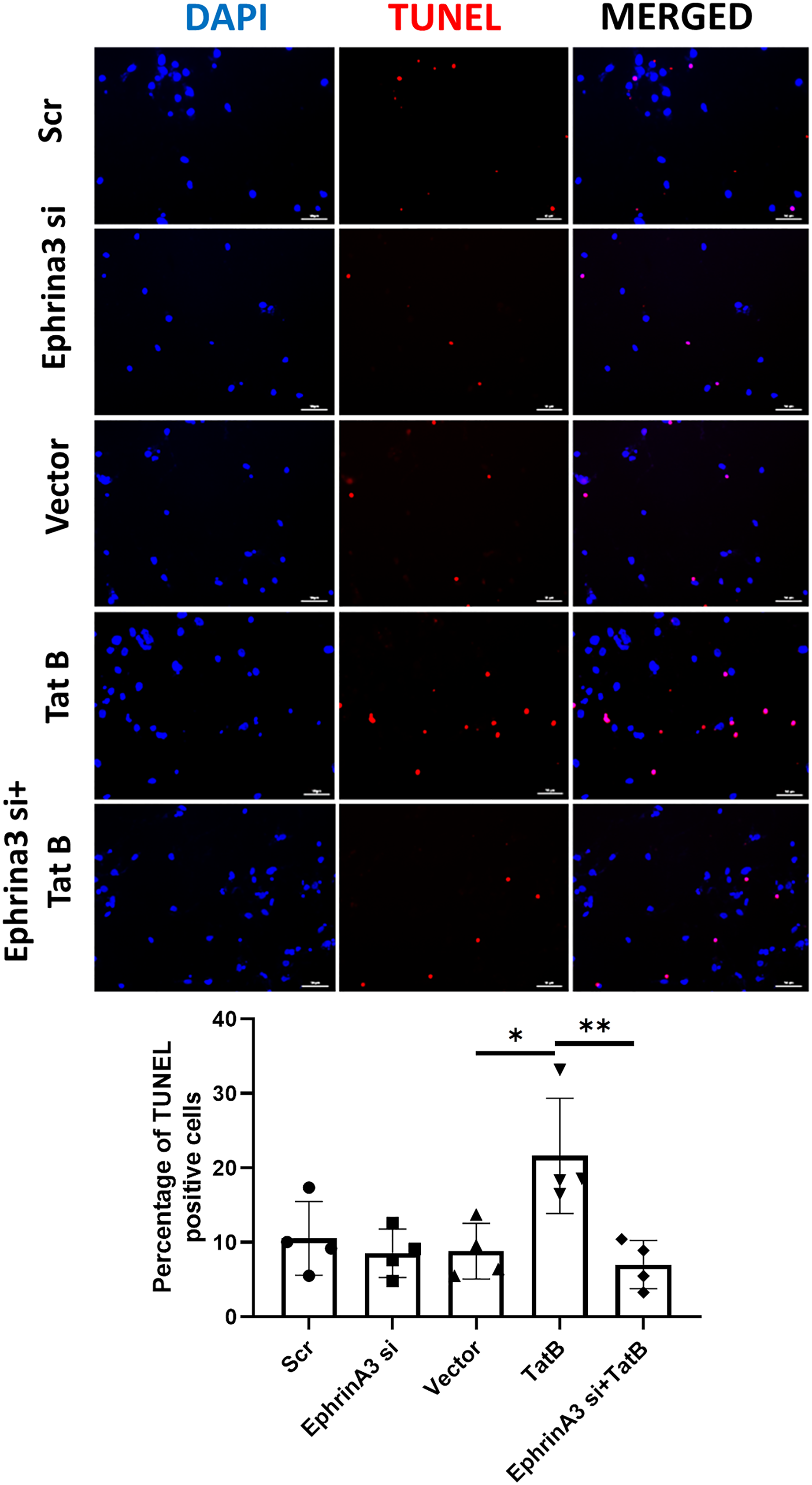
EphrinA3 knockdown attenuates HIV-1 Tat B-induced neuronal death as seen by TUNEL assay: supernatant from co-cultures with transfected astrocytes, 24 h post-transfection was added on neurons. Neuronal apoptosis was analyzed by TUNEL assay after 24 h incubation with astrocyte supernatant. The TUNEL positive cells (red) coinciding with DAPI (blue) were counted and the percentage was calculated by dividing double positive with the total number of DAPI positive cells. EphrinA3 knockdown did not show any significant change in neuronal death as opposed to the vector, but the Tat B transfection showed a significant increase in neuronal death compared to its control. Co-transfection with ephrin A3 siRNA and Tat B showed a decline in neuronal death as opposed to only Tat B transfection. The top panel shows the representative images of each group (scale bar: 100 µm) and the bottom panel shows the bar graph for the percentage of TUNEL positive cells in each group. The data represents four independent experiments. n = 4, *p < .05, **p < .01.
This data highlights the fact that ephrinA3 not just controls the glutamate transporters and glutamate uptake, but also has a significant effect in rescuing neurons from the toxic effects of excess extracellular glutamate that is persistent in the case of HIV-1 Tat B.
Discussion
Neuron and glia co-functioning are indispensable for the proper functioning of the brain. Any change or disruption in communication between these cells may lead to pathological effects. EphrinA3 ligand expressed on the astrocytes and its receptor ephA4 expressed on the neurons in the adult brain is one of the many that regulate neuron–glia communication and homeostasis. EphrinA3–ephA4 interaction leads to a two-way signaling, forward signaling in neurons expressing ephA4, and reverse signaling in astrocytes expressing ephrinA3 (Carmona et al., 2009). The reverse signaling in astrocytes is known to regulate extracellular glutamate levels whereas the forward signaling modulates spine density in neurons. Previous literature has pointed out the importance of ephrinA3 on astrocytes in glutamate uptake. It has been demonstrated that the ephrinA3–ephA4-axis in neuron–glia is critical for LTP formation and glutamate transporters were upregulated in ephrinA3 knockout mice whereas transgenic overexpression of ephrinA3 in mice leads to lower uptake of glutamate due to the reduced expression of glutamate transporters (Filosa et al., 2009). The major cause of dementia in HIV patients is due to neuronal death which can happen directly or indirectly, but the most important pathway being indirectly through astrocytes, majorly due to glutamate excitotoxicity (Pandey & Seth, 2019). Glutamate excitotoxicity in HIV patients occurs due to the inefficiency of the astrocytes to uptake excess glutamate from the synapses that have been released by the neurons. Our lab has previously highlighted the importance of purinergic receptors and VDAC1 axis in astrocytes that mediate neuropathogenesis in the case of HIV-1 Tat B (Fatima et al., 2017; Tewari et al., 2015). As glutamate uptake is regulated by ephrinA3 in astrocytes and glutamate being the major cause of neuronal death in HIV patients, in this study, we ventured to study the effects of HIV-1 Tat B on ephrinA3 and its role in maintaining glutamate homeostasis in primary cultures of human neuron–glia co-cultures.
As the ephrinA3–ephA4 signaling demands cell to cell contact, we successfully established neuron–glia co-cultures by differentiating human neural progenitor cells separately into astrocytes and neurons each, followed by overlaying the neurons on astrocytes. This was done to ensure pure astrocyte–neuron co-cultures as opposed to mixed brain cultures, with no contamination of any other cell types (Figure 2). The co-cultures expressed the cell-specific markers, GFAP by astrocytes and MAP2 by neurons, and are seen making physical connections. Our differentiated cells showed significant expression of ephrinA3 on astrocytes and ephA4 on neurons (Figure 1). Although at very low levels, astrocytes and neurons also express ephA4 and ephrinA3, respectively (Supplementary Figure 1). Although co-culture being the optimum model system for this study, it has its own pros and cons. The major feature of this model system is that it closely mimics the brain environment. Since we want to maintain a homogenous population of only astrocytes and neurons, without contamination of any other cell types, we differentiated them from neural progenitor cells which take 21 days of period and is therefore time-consuming, which is a drawback of this model system. As ephrinA3 and ephA4 are expressed interchangeably on astrocytes and neurons at low levels, the results depict a cumulative of all of them and it is difficult to segregate the two cell types. Signaling in ephrinA3 and ephA4 can be cis (activated by the receptor on the same cell) and trans (activated by the receptor on different cells) (Egea & Klein, 2007).
In accordance with the hypothesis, we first checked the effects of HIV-1 Tat B on ephrinA3 in neuron–glia co-cultures which was found to be elevated (Figure 3). In addition to co-cultures, we also confirmed the increase in the expression of ephrinA3 in autopsy brain sections of HIV-1-infected patients. The increase in expression of ephrinA3 in co-cultures is modest that compared to the increase seen in autopsy brain sections (Figure 4). This may be because the HIV-1 patients have fully progressed into the disease and hence show the terminal stage of disease progression.
As per previous reports, where higher expression of ephrinA3 showed a decline in the potency of astrocytes to uptake excessive synaptic glutamate, due to lowered levels of glutamate transporters (Filosa et al., 2009), we next confirmed whether the increased expression of ephrinA3 in HIV- Tat B is responsible for decreased expression of glutamate transporters. Therefore, the functionality of ephrinA3 was confirmed by knocking down its expression using siRNA. In our knockdown experiments, we observed that decreased levels of ephrinA3 increased the expression of glutamate transporters while HIV-1 Tat B showed a decline in the expression of glutamate transporters. But co-transfection with ephrinA3 siRNA and HIV-1 Tat B increased the expression of glutamate transporters as compared to the decline seen in HIV-1 Tat B alone, confirming our hypothesis that the glutamate transporters are fundamentally regulated by ephrinA3 expression in the case of HIV-1 Tat B (Figure 6). The effects were seen more on EAAT2 than EAAT1, the former being the major transporter for glutamate uptake due to its high expression on astrocytes (Wang et al., 2003).
If the glutamate transporters are being regulated by ephrinA3, then so should the glutamate uptake by astrocytes. We examined the levels of extracellular glutamate in neuron–glia co-cultures in ephrinA3 siRNA transfected, HIV-1 Tat B transfected and co-transfection with both. We observed a significant increase in the concentration of glutamate in HIV-1 Tat B whereas a decrease in the glutamate concentration in ephrinA3 siRNA knockdown. However, in the co-transfection groups, the extracellular glutamate concentration decreased as compared to that of HIV-1 Tat B transfection (Figure 7). This supported our hypothesis that ephrinA3 not only regulates the glutamate transporters but eventually controls glutamate uptake by astrocytes and its knockdown reverses the effects of HIV-1 Tat B. Astrocytes are not only known for the task of clearing out excessive glutamate from the synapses but also contribute to the release of glutamate through different mechanisms (Malarkey & Parpura, 2008). But this phenomenon is still debatable as to whether or not astrocytes release glutamate in vivo, and if they do release glutamate then at what conditions and how (Mahmoud et al., 2019). Although it is still an unexplored territory, ephrinA3 has not been reported to be a part of any glutamate release mechanisms by astrocytes. But we cannot discard the possibility of other factors playing a role in astrocytic glutamate release in HIV-1 Tat B that might contribute to the glutamate accumulated in the synapses in addition to the glutamate that is released by the neurons and has not been taken up by the astrocytes. Astrocytes express more than 90% of glutamate transporters in the brain but we also cannot totally eliminate the possibility of neurons playing a very minor role in excess glutamate clearance from the synapses as they have also been known to express very low levels of glutamate transporters as compared to astrocytes (Sheldon & Robinson, 2007).
Neuronal toxicity and death in the case of HIV patients draw them to HIV-associated dementia and the foremost reason for neuronal death is glutamate excitotoxicity. In our study, we observed a significant increase in neuronal death in the presence of HIV-1 Tat B. The neurotoxicity was reversed in the case of ephrinA3 knockdown in the presence of HIV-1 Tat B and there was less neuronal death as compared to the one seen in only HIV-1 Tat B (Figure 8). As discussed that neuronal glutamate transporters might also have a minor role in excess glutamate uptake but the significant toxicity and death of neurons might not help in a larger and cumulative uptake as compared to that of astrocytes. This data clearly establishes the importance of ephrinA3 in maintaining neuronal viability through the clearance of glutamate uptake. As ephrinA3 binds to ephA4 and its forward signaling in neurons is known to modulate spine density (Carmona et al., 2009), there is a possibility that the death of neurons might be caused due to compromise in the forward signaling in neurons in addition to the glutamate excitotoxicity and this area needs further exploration. Other factors that cause neuronal toxicity due to Tat B cannot be ruled out. Activation of NMDA receptors has also been a contributing factor to glutamate excitotoxicity (Haughey et al., 2001; Lipton, 1994) and our cells express this receptor making them highly susceptible to glutamate excitotoxicity. Various other pathways such as extracellular signal-regulated kinase/mitogen-activated protein kinase, nuclear factor-kB and protein kinase C cause neurotoxicity due to Tat B (King et al., 2006). These pathways are implicated in tat-mediated neuropathogenesis and may also be contributing to neuronal death but there are no reports to link any of these pathways to ephrinA3 downstream signaling. EphrinA3 downstream signaling is still a blank slate and will need further exploration. Recently, ephrinA1–A3 have been observed to have a role in neuroinflammation in brain ischemia. With this study, we can speculate that ephrinA3 might be involved in activating proinflammatory cytokines that lead to neuronal death (Thundyil et al., 2013). We also observed a decrease in Tat B expression in the case of ephrinA3 knockdown, hinting at the possibility of ephrinA3 regulating Tat B expression in transfected astrocytes.
Owing to this data, ephrinA3 functioning is found to be ruptured in the case of HIV-1 Tat B (Figure 9). The exact pathway of reverse signaling in astrocytes is not yet known and it would be interesting to explore this area for further studies. Also, we did not check the role of ephA4 in the forward signaling in neurons and this territory still needs to be investigated in the case of HIV-1 Tat B. This axis, if explored in more detail, may be a potential target for therapeutic strategies to resolve the adverse outcomes of glutamate excitotoxicity on neurons and prevent neuronal death.
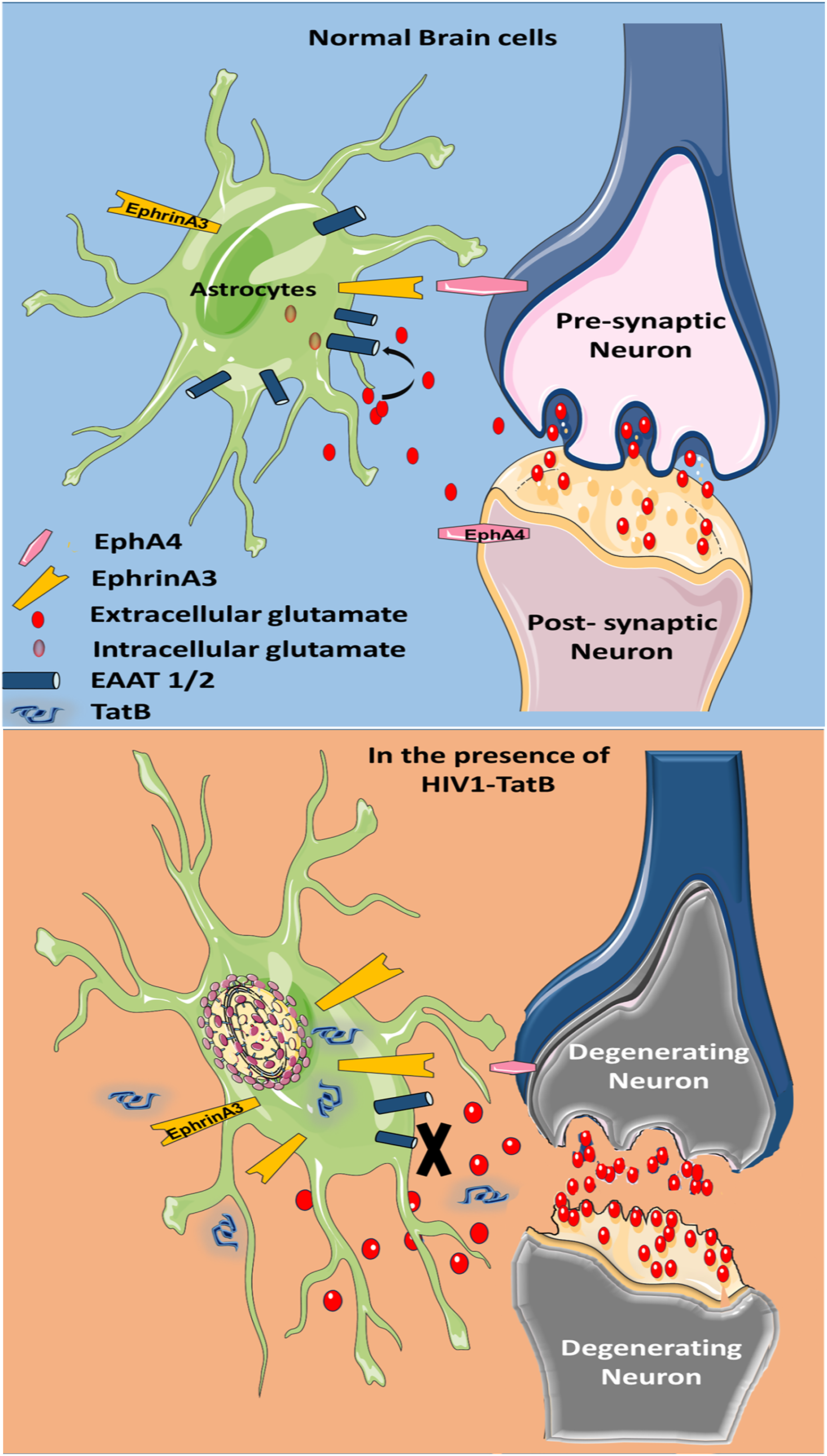
Summary: glutamate uptake is regulated by ephrinA3 by maintaining the levels of glutamate transporters (EAAT1 and EAAT2) in normal brain cells. In the presence of HIV-1 Tat, ephrinA3 expression increases which decrease the expression of glutamate transporters on astrocytes and hence decreases glutamate uptake. This causes glutamate excitotoxicity and leads to neuronal toxicity and degeneration and ultimately neuronal death. (This figure was created using images from Servier Medical Art by Servier, which are licensed under a Creative Commons Attribution 3.0 Unported License; https://smart.servier.com
Footnotes
Acknowledgments
Technical assistance garnered by Naushad Alam and Durgalal Meena is highly appreciated for this study. Funding for this project from NBRC core funds and project grant (BT/PR27512/MED/122/146/2018) from the Department of Biotechnology (DBT), New Delhi are highly acknowledged. The authors also acknowledge the support of the facilities provided under the Biotechnology Information System Network (BTISNET) grant, Department of Biotechnology, India and Computing facility as well as Distributed Information Centre at NBRC, Manesar, India. We highly acknowledge the Human Brain Tissue Repository for Neurobiological Studies, Department of Neuropathology, National Institute of Mental Health and Neurosciences, Bangalore, India for providing well-characterized autopsy brain sections.
Author’s Contribution
C.M.S.S. has contributed to the conceptualization, investigation, methodology, data acquisition, analysis, validation and writing of this manuscript. P.J. has contributed to the investigation, data acquisition, analysis and writing of the manuscript. K.S. and A.M. have contributed to the investigation and data acquisition and analysis. P.S. has contributed to acquiring project funding, project management, conceptualization of the project, intellectual inputs, supervision, and writing of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Biotechnology, Ministry of Science and Technology (grant number BT/PR27512/MED/122/146/2018).
Supplemental Material
Supplemental material for this article is available online.
