Abstract
The loss of spinal sensorimotor pathways following spinal cord injury (SCI) can induce retrograde neurodegeneration in the primary motor cortex (M1). However, the effect of thoracic SCI on forelimb motor skills has not been studied clearly. So, herein we aimed to examine the effects of the thoracic SCI model on forelimb motor skills learning, parallel with dopaminergic and oscillatory changes in hindlimb and forelimb areas (HLA and FLA) of M1 in rats. Male Wistar rats were randomly subjected to laminectomy (Control) or contusion SCI at the thoracic (T10) level. Oscillatory activity and motor skills performance were evaluated for six consecutive days using local field potential (LFP) recording and skilled forelimb reaching task, respectively. Dopamine (DA) levels and expression of dopamine receptors (D1R and D2R) were determined in HLA and FLA by ELISA and western blotting. LFP recording results showed a sustained increase of LFP power in SCI rats compared with uninjured rats through skilled reaching training. Also, the SCI group had a lower reaching performance and learning rate in contrast to the Control group. Biochemical analysis of HLA and FLA showed a reduction in DA levels and expression of D1R and D2R after SCI. According to these findings, thoracic SCI causes aberrant changes in the oscillatory activity and dopaminergic system of M1, which are not restricted to HLA but also found in FLA accompanied by a deficit in forelimb motor skills performance.
Summary statement: The reorganization of the primary motor cortex, following spinal cord injury, is not restricted to the hind limb area, and interestingly extends to the forelimb limb area, which appears as a dysfunctional change in oscillations and dopaminergic system, associated with a deficit in motor skills learning of forelimb.
Keywords
Introduction
Increasing evidence from both experimental and human research suggests that the loss of spinal sensorimotor pathways following spinal cord injury (SCI) can promote progressive retrograde neurodegeneration, including gray matter atrophy, diminished white matter integrity, and to a degree cell death in different parts of the brain, especially the primary sensorimotor cortex (Hou et al., 2014; Nardone et al., 2013; Solstrand Dahlberg et al., 2018). Nevertheless, the majority of SCI studies have been focused on pathophysiological changes in the affected area and sensorimotor deficits below the injury site. The effect of isolated SCI on the brain has recently been considered as one of the affecting mechanisms involved in sensorimotor dysfunction (Moxon et al., 2014). Based on functional MRI (fMRI) and transcranial magnetic stimulation (TMS), extensive changes in the primary motor cortex (M1) activation during rest and specific motor tasks of the non-affected upper limb have been shown in paraplegic SCI patients (Curt et al., 2002; Lotze et al., 1999; Saturno et al., 2008). However, the exact underlying mechanism of this issue has not been well addressed experimentally. A former study under anesthesia has suggested that thoracic SCI in the rat can increase the spontaneous single-neuron activity of the M1 not only in the hindlimb area (HLA) innervating below the injured spinal level but also in the forelimb area (FLA) that innervates above the injured spinal level (Frost et al., 2015). To our best knowledge, no studies have investigated the effect of isolated thoracic SCI on the oscillatory activity of both HLA and FLA along with functional neurological assay in conscious animal models.
The M1 as an executive motor area is also in charge of preparing the sequence of movement and motor skills learning (Li et al., 2017; Papale & Hooks, 2018). There is sufficient evidence supporting the pivotal role of the M1 in motor skills learning upon high-level cognitive mechanisms such as acquisition and storage of motor memory (Mutha & Haaland, 2014). Recent clinical and experimental investigations have reported that SCI results in cognitive impairments, such as spatial memory, processing speed, and learning ability associated with adverse reorganization in the hippocampus (Wu et al., 2014a; Wu et al., 2014c). Therefore, it is hypothesized that reorganization in FLA after thoracic SCI could have a negative impact on related motor skills learning. Dopaminergic (DAergic) projections from the ventral tegmental area (VTA) to M1 play an essential role in M1 oscillatory activity (Ozkan et al., 2017) and are required for successful motor skills learning (Hosp & Luft, 2013). Interestingly, altered dopamine (DA) concentration in Parkinson disease (PD) and traumatic brain injury (TBI) is associated with alternation in M1 oscillatory activity, and motor skills deficit (Gaspar et al., 1991; Pearson-Fuhrhop et al., 2013). It has also been documented to some extent, SCI-induced changes in the brain are similar to those observed after TBI (Wu et al., 2014a), so examining the M1 oscillatory activity and related DAergic, and functional changes following thoracic SCI can provide a great deal of information to understand the possible mechanisms of related M1 reorganization.
As a result, this study aimed to determine whether thoracic SCI affects the oscillatory activity and DAergic system within M1. If yes, how do these changes correlate with motor skills learning of the non-affected forelimb?
Materials and Methods
Animals
All the adult Wistar male rats (3–4 months old, weighing 250–300 g) were provided by the experimental animal center of Tabriz University of Medical Sciences. Rats were maintained under environmentally controlled conditions and subjected to a 12 h/12 h light/dark cycle with water available ad libitum and familiarized to the facility for seven days before starting the experiments. Rats were single-housed in Plexiglas home cages throughout the study, and all experiments were done during the light phase. Animals were randomly divided into two main groups: Control (the laminectomized rats without SCI) and SCI (the rats with Thoracic contusion SCI).
Ethical Approval
All procedure and handling techniques were approved by the Ethical Committee for Animal Research of Tabriz University of Medical Sciences (IR.TBZMED.REC.1396.874), and surgical procedures were conducted under aseptic conditions.
Experimental Design and Procedures
The sequence of procedures is presented in Figure 1.
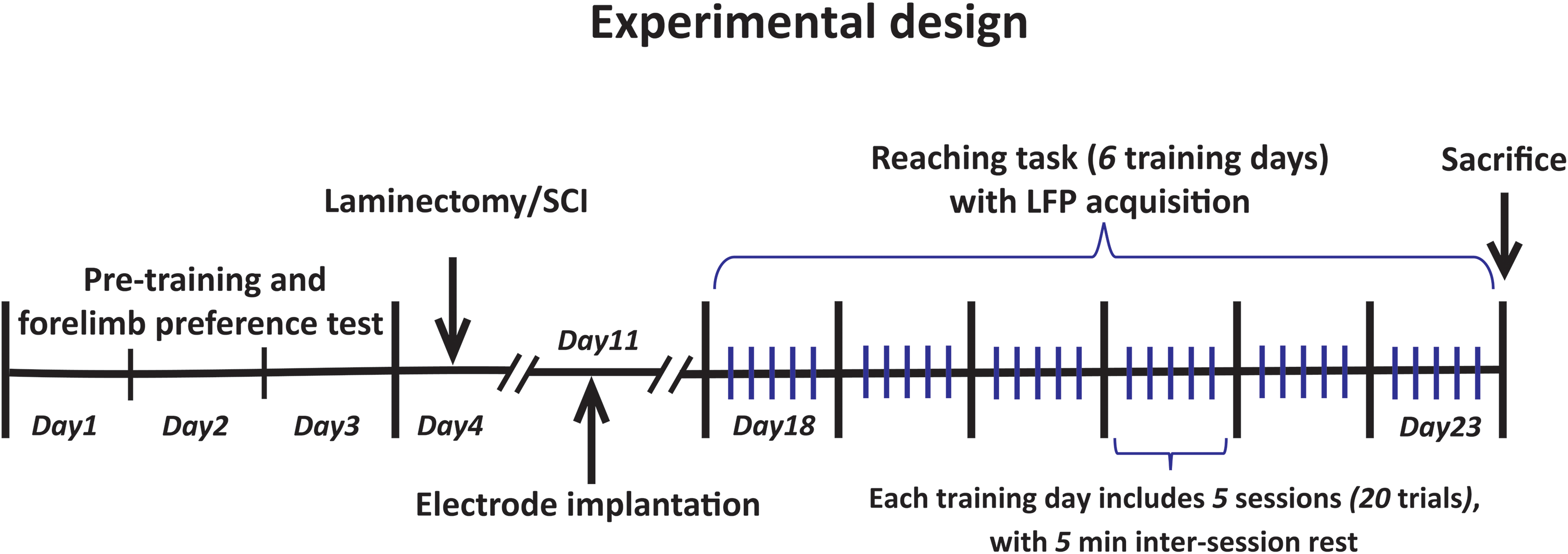
Experimental design.
Rats were anesthetized with an initial dose of 5% isoflurane/oxygen mixture in the induction box and were maintained at 1–3% isoflurane/oxygen mixture supplied via a nose cone. Then, rats were placed in the prone position, and an appropriate midline skin incision was made over the lower thoracic vertebral region. A laminectomy was then carefully performed at T10 level to expose the spinal cord without damaging the dura mater. Control rats were only subjected to laminectomy without SCI. In the SCI group, rats were placed in the Neurosciences research center (NSRC) impactor (Ghorbani et al., 2018). The lateral processes of thoracic vertebra 9 and 11 were clamped with two stabilization forceps to fix and flat the vertebral column during impact. The impactor tip was adjusted 3 mm above the center of the spinal cord, and a contusion injury was induced with a force of 150 kilodyne (kdyn). Induced impact reported by the impactor software for each rat was recorded, and a maximum deviation of 10% from the set impact force of 150 kdyn was considered as an acceptable range. At the conclusion of surgery, ketoprofen (0.05 mg/kg, subcutaneously) was administrated immediately and 24 h later for pain relief. On the first week after surgery, animals received daily intraperitoneal injections of 8 mg/kg ciprofloxacin in 2 mL saline to inhibit infections and dehydration. The bladders were manually expressed twice daily until recovery of micturition reflex (7–14 days post-injury).
Post-SCI Locomotor Assessment
Hindlimb locomotion was evaluated weekly from day 1 to three weeks post-injury, using the modified Tarlov's motor scale (Liu et al., 2011). The scoring was performed for each rat as follows: 0, no visible movement; 1, movement in the hip and/or knee; 2, movement in the all major joints of the hindlimb without weight-bearing; 3, uncoordinated gait with incomplete weight-bearing; 4, coordinated gait and improved postural support with poor foot control; 5, normal locomotion (Supplemental Figure 1A).
The Surgical Procedure of Recording Electrode Placement
One week post-SCI, all rats underwent stereotaxic surgery to implant recording electrodes (Teflon coated stainless steel wire, 50.8 µm diameter, A-M Systems, Inc.) in the FLA and HLA of M1. The location of the electrodes implantation was contralateral to the preferred forelimb in the reaching task (see below). Initially, rats were anesthetized with isoflurane (Minrad, Canada) in 100% oxygen (initial dose: 5%, maintenance dose: 1–3%) and placed in a stereotaxic device (Stoelting, USA). An appropriate sagittal incision was performed, and the entire skull surface was exposed and freed from connective tissue. The incisor bar was adjusted until the bregma, and lambda skull points were in equal heights (flat skull position). The coordinates for M1 recordings comprised 1.5 mm anterior to bregma and 3 mm lateral to the midline for the contralateral FLA (Cont-FLA) recording electrode (Li et al., 2017), and 2 mm posterior to bregma and 2.6 mm lateral to the midline for the contralateral HLA (Cont-HLA) recording electrode (Frost et al., 2013, 2015). Two holes (1 mm diameter) were drilled at this coordination, and both electrodes were lowered deep into 1.5 mm below the dura. On the contralateral side, two stainless steel screws soldered to the copper wire were screwed into the skull to serve as reference electrodes and anchors for the acrylic cement. All electrodes were connected to a female connector and fixed to the skull with dental acrylic cement. The targeted coordinates for HLA and FLA recording electrodes were confirmed by epidural stimulation-induced hindlimb and forelimb movement in a pre-study pilot experiment.
Skilled Forelimb Reaching Task
Rats were trained on the skilled forelimb reaching task, to assess motor skills learning, as previously described (Whishaw & Pellis, 1990; Zemmar et al., 2015). Single pellet reaching chamber was made of transparent Plexiglas (14 cm wide, 34 cm long, and 29 cm high) with a vertical narrow slot (1 cm wide × 23 cm high) in the middle of the front wall to get access a uniform food pellet placed on a shelf 3 cm above the floor of the chamber. For holding the pellet, aligning with the slot borders, two small indentations were created 1.5 cm away from the inside of the frontal wall. Twenty-four hours before pre-training, rats underwent food restriction to 90% of their baseline body-weight, which was maintained throughout the training (50 mg of standard lab diet per 1 g body-weight per day).
Pre-training was performed 20 min daily for three days before the surgical operations. In the first two days, the pellets were placed near the slot on the shelf until the rats retrieve them by using the tongue. On the third day, rats were permitted to use either forelimb to retrieve a centrally placed pellet 1.5 cm away from the slot. Once the rat executed more than 80% of the reaching attempts (i.e., 40 out of 50) with the same forelimb, this limb was defined as the rat's preferred forelimb.
After recovery from both surgeries (14 days), rats were trained with the preferred forelimb for six consecutive days. One pellet per trial was placed in the indentation contralateral to the preferred forelimb so that the rats were allowed to make reaching attempts with the preferred limb only. Each daily training day consisted of 100 trials, divided into five sessions (20 trial bins or maximum of 10 min) with 5 min intervals for each rat.
Analysis of Skilled Forelimb Reaching Task
Reaching performance was recorded using a digital video camera (Logitech QuickCam Pro 9000) and analyzed as follows. First reach success: was defined as trials in which the rat completed the reaching attempt and retrieved the pellet successfully on the first monolithic advance of the forelimb toward the food without repetition of movement components. To analyze the reaching performance between or within days, the first reach success rate was calculated for each training day and session, as the percentage of first reach success trials out of the total number of trials (i.e., 100 per day; 20 per session).
Total success was defined as trials in which the rat retrieved the pellet successfully on the one or more reaching attempts required, without knockout of the pellet away or dropping it during retrieval. The total success rate was calculated for each training day as the percentage of total success trials out of the total number of trials (i.e., 100).
Learning rate: The learning rate was calculated for both total success and first reach success of each rat by subtracting the average success rate in the first two days from the average that of in the last two days, and divided by the number of intervals between days [(mean days 5 and 6)– (mean days 1 and 2)/5] (Qian et al., 2013).
Electrophysiological Data Acquisition and Processing
After recovery from both surgical operations (14 days), the electrophysiological recording was performed for six days in Cont-FLA and Cont-HLA of control and SCI rats. All experiments were conducted in a faraday cage to minimize external electrical noises. Behavioral video recording was synchronized with electrophysiological recording and saved in a computer for later visual inspection. local field potential (LFP) activity in FLA was recorded under resting state and subsequent forelimb reaching task. LFP activity in HLA was only recorded at rest. LFP signals were amplified with a gain of 1000 and bandpass filtered at 0.1–10,000 Hz by an extracellular amplifier (DAM80, World Precision Instruments, Sarasota, FL, USA). The amplified signals were then digitized at a sampling rate of 20,000 Hz using the analog to digital converter (NI USB-6221, National Instruments) and stored for further offline analysis in MATLAB 2017b (The Mathworks, USA).
For electrophysiological data analysis, using an open-source MATLAB toolbox (EEGLAB), the raw signals were digitally bandpass filtered (1–80 Hz), and notch filtered (45–55 Hz) to the reduction of background noise to biological signal ratio. Motion artifact was also reduced as much as possible by visual inspection. User-defined MATLAB scripts were written, and LFP activity was quantified using Fast Fourier Transform (FFT) analysis. The mean FFT power spectra were computed for delta (1–4), theta (4–12), beta (12–30), and gamma (30–80) frequency bands (Jia & Kohn, 2011; McCracken & Kiss, 2014) with 20,000 samples and windowing with 50% of overlapping. The representative time–frequency spectrograms of the LFPs were plotted by visualization of the time-varying powers at each frequency band.
Tissue Preparation
At the conclusion of the last training day, some rats were decapitated under ketamine (100 mg/kg) anesthesia for biochemical analysis, while others were perfused for histological processing (see Histology). The fresh brains were rapidly removed and placed in ice-cold saline. Cont-FLA, ipsilateral FLA (Ipsi-FLA), and Cont-HLA were isolated, aliquoted, and stored at −70°C for later quantitative analysis of D1R and D2R protein contents and DA levels. The target territories of FLA ( + 0.5 to + 3 mm anterior and ± 1 to ± 4 mm lateral relative to bregma) and HLA (0 to − 3 mm posterior and ± 0.5 to ± 4 lateral relative to bregma) were based on previous motor mapping studies in the rat (Neafsey et al., 1986; Tandon et al., 2008; Zilles, 2012).
Biochemical Analysis
Weighted aliquots of tissue samples were kept at 2–8 °C and homogenized in ice-cold phosphate-buffered saline (PBS) with a protease inhibitor cocktail (100 mg tissue/1 mL buffer). The tissue homogenates were centrifuged (at 6,000 RPM) for 10 min at 4 °C, and supernatants were collected. The DA levels of the supernatants were estimated according to the manufacturer's instructions using Rat DA ELISA Kit (ZellBio GmbH, Germany, Cat.No: ZB-10219C-R9648). According to the specifications provided by the manufacturer, the sensitivity of assay was 2.5 ng/L, and intra-assay and inter-assay precisions were <10% and <12%, respectively.
Western Blot Analysis
Total proteins from tissues were extracted in ice-cold RIPA lysis buffer containing 50 mM Tris–HCl, pH 7.5, 150 mM NaCl, 1% Nonidet P-40, 0.1% sodium dodecyl sulfate, 0.5% sodium deoxycholate in the presence of a protease inhibitor cocktail tablet as previously described (Fallahi et al., 2019). Tissue homogenates were centrifuged (at 10,000 RPM) for 10 min at 4 °C, and supernatants were stored at −20 for western blot analysis. Briefly, total protein concentration was determined by the Bradford method. Protein bands were separated by electrophoresis on the SDS–page gel and then transferred to a polyvinylidene fluoride (PVDF) membrane. The membrane was gently shaken with blocking buffer containing 2% non-fat dry milk in TBS-T (50 mM Tris–HCl, 0.9% NaCl, 0.1%Tween-20, PH 7.5) for 1 h at room temperature and then incubated with monoclonal primary antibodies raised against D1R (mouse, 1:200; Cat.No: sc-33660, RRID: AB_668813, Santa Cruz Biotechnology, USA) and D2R (mouse, 1:200; Cat.No: sc-5303, RRID: AB_668816, Santa Cruz Biotechnology, USA). β-actin antibody (mouse, 1:200; Cat.No: sc-69879, RRID: AB_1119529, Santa Cruz Biotechnology, USA) was performed as a loading control. After this step, the membranes were washed three times with TBS-T and incubated with anti-rabbit secondary antibody conjugated with horseradish peroxidase (HRP) for 1 h at room temperature. After washing three times, protein bands were visualized by enhanced chemiluminescence (ECL) advanced reagent kit. Densitometric quantification of protein bands was done using Image J software and normalized to β-actin expression levels in each sample.
Histology
Rats (N = 3 per group) were deeply anesthetized by ketamine (100 mg/kg), and perfused transcardially with saline, followed by 4% paraformaldehyde in a PBS (pH 7.4). The brains and spinal cords were carefully removed, and placed in the same solution overnight. After fixation, paraffin blocks were taken from the brain, and the spinal cord. Coronal brain sections (5 μm) were cut, using a microtome (DS-8402), through the level of the HLA and FLA, and stained with cresyl violet to confirm the recording electrode placement (not shown).
For histological assessment of the spinal cord, H&E-stained cross-sections (5 μm) were prepared at the lower thoracic level (T9-T11). In a blinded manner, the histopathological analysis was performed based on the presence of inflammatory and morphological changes, including lymphocyte infiltration, axonal swelling, pyknotic nuclei, vacuolation, and cystic degeneration (Ghorbani et al., 2018) (Supplemental Figure 1B).
Statistical Analysis
Statistical analyses were performed using the GraphPad Prism software (version 8.0). All electrophysiological experiments were analyzed using two-way repeated-measures of variance (ANOVA) to assess the changes over the days, and differences between the groups or cortical areas. For the behavioral experiment, two-way repeated-measures ANOVA was used to compare the reaching performance between control and SCI rats over the training days. Additional linear regression and unpaired parametric or non-parametric t-test (two-tailed) were performed to assess the within-day performance and learning rate, respectively. Data from biochemical studies were analyzed using two-way mixed ANOVA to determine the changes of DA, D1R, and D2R levels between groups and cortical areas. Sidak's post hoc test was performed when significant ANOVA effects were found. The correlation among the variables was estimated with Pearson's correlation coefficients or non-parametric Spearman correlation when appropriate. P <.05 was considered statistically significant. All data are presented as means ± standard error of the mean (SEM).
Results
The Effect of Thoracic SCI on Skilled Forelimb Reaching Performance
The performance of the Control and SCI (n = 8 per group) rats in the skilled reaching task throughout six days is shown in Figure 2. Two-way repeated-measures ANOVA showed significant effects of group (First reach success (%): F(1, 14) = 24.00, P = .0002; Total success: F(1, 14) = 10.59, P = .0058), training day (First reach success (%): F(5, 70) = 75.98, P <.0001; Total success: F(5, 70) = 39.09, P <.0001) and group × day interaction (First reach success (%): F(5, 70) = 17.64, P <.0001; Total success: F(5, 70) = 15.96, P <.0001). Both groups started the first training day with almost the same performance; however, multiple comparisons test confirmed that the first reach success rate achieved by the SCI rats was significantly lower than that achieved by control rats from day 2 onward (day 2: P = .046; day 3: P = .0003; days 4–6: P <.0001). First reach performance in the control group was significantly improved over all training days with respect to the first day (day 1 vs. day 2: P = 0024; day 1 vs. days 3–6: P < .0001), Whereas the SCI rats displayed relative improvement over the last three days (day 1 vs. day 4: P = .0007; day 1 vs. days 5 and 6: P <.0001) (Figure 2A). As such, the difference of performance for total success demonstrated that the SCI rats had significantly lower scores from day 3 until day 6 compared with control rats (day 3: P = .031; day 4: P = .002; days 5 and 6: P <.0001). The total success rate achieved by the control rats was significantly improved over the training days (day 1 vs. day 2: P = .015; day 1 vs. days 3–6: P < .0001). In contrast to the Control group, SCI rats only showed slightly significant improvement in the last two days of training (day 1 vs. day 5: P = .045; day 1 vs. day 6: P = .011) (Figure 2B). Moreover, SCI rats had a significantly lower learning rate for first reach success (P < .0001; Figure 2C) and total success compared with control rats (P = 0003; not shown). Within-days assessment of first reach success was analyzed by linear regression that was significant in the control group for days 2 (R2 = .84, P = .029) and 4 (R2 = .90, P = .012) (Figure 2D). On the first training day, both control and SCI groups achieved approximately the same improved performance during five daily sessions, which was continued by the control group over subsequent training days. But in the SCI group, the achieved performance at the end of the first day was not maintained overnight, and they started the second day with a lower success rate. This dysfunctional cycle was repeated in the second 24 h interval until the low plateau in success rate was reached after three training days. The SCI rats achieved relatively good performance at the end of the first day, but it did not maintain overnight and started the second day at a lower success rate, which reached a plateau at day 4.
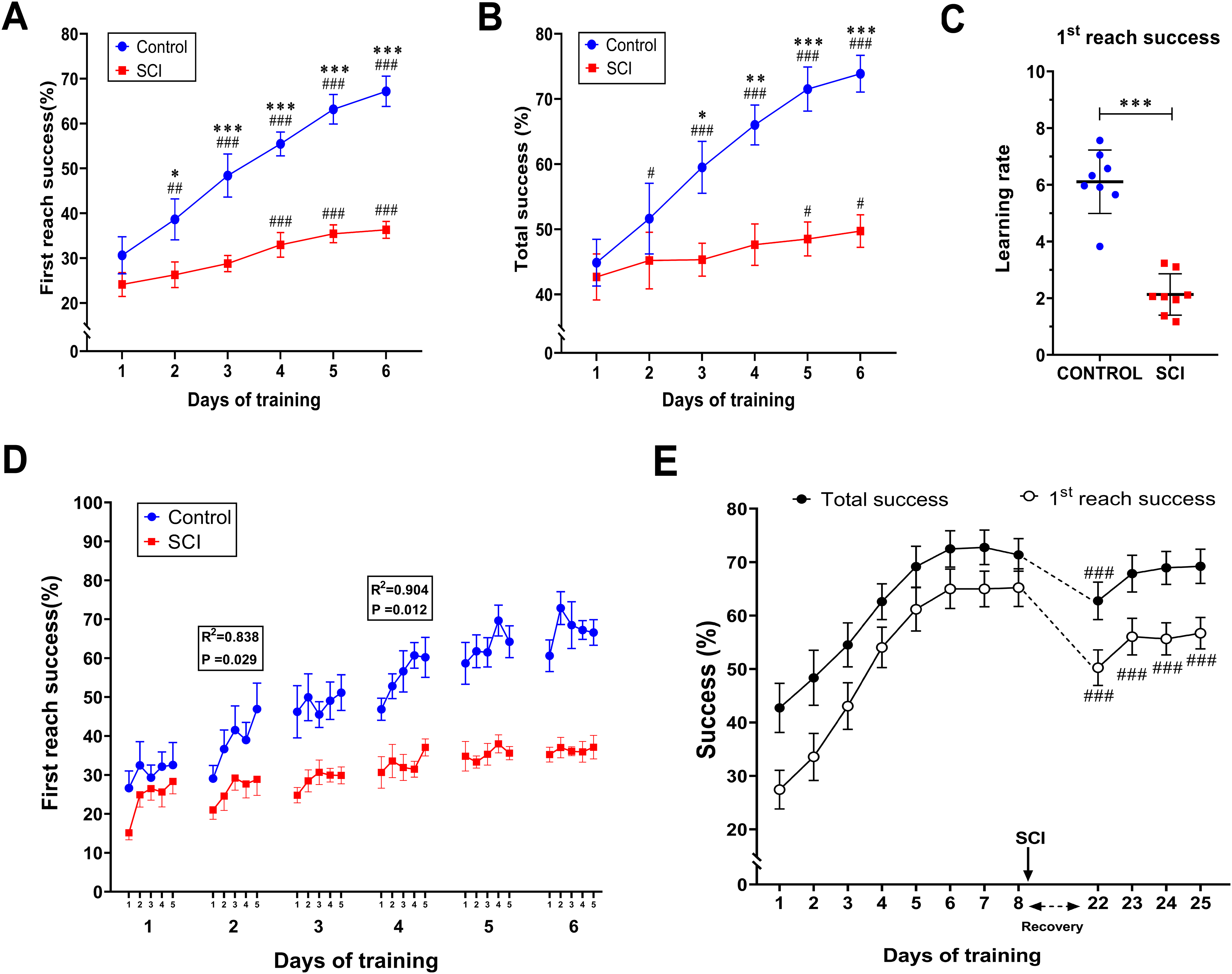
Thoracic spinal cord injury (SCI) impairs forelimb reaching skill. The performance of the Control and SCI (n = 8 per group) rats was evaluated in the skilled forelimb reaching task throughout six training days. First reach success rate (%): the percentage of first reach success trials out of the total number of trials (A) Total success rate (%): the percentage of total success trials out of the total number of trials (B). Learning rate comparison between Control and SCI groups for first reach success: [(mean success rate in days 5 and 6)–(mean success rate in days 1 and 2)/number of intervals between days] (C). Within-day reaching performance of Control and SCI groups over five sessions per day (D). Denotation of significant differences between Control and SCI groups (*P <.05, **P <.01, ***P <.001). Denotation of significant differences within-group comparison with respect to first day (#P <.05, ##P <.01, ###P <.001). PRE–POST experiment; Reaching performance of PRE–POST group, prior and post-injury (n = 6; ###P <.001 recalling days vs. day8) (E).
A PRE–POST group was defined to determine whether the thoracic SCI-induced motor skills deficiency has been interfered by loss of the supportive role of the hindlimb through weight-bearing or not. For this purpose, six intact rats were trained for eight consecutive days until their motor skill learning was saturated. On the following day, thoracic SCI was induced, and after recovery from surgery (14 days), motor skill training was recalled for four days. The results (one-way repeated-measures ANOVA followed by Dunnett's multiple comparisons test) showed that the previously acquired motor skill was significantly lowered when recalled for the first time after SCI (training days: P <.0001, F(11, 55) = 63.72; day 8 vs. day 22: P <.0001); however, in the subsequent recalling days, the total success rate was reached to the pre-SCI acquired level, with no significant differences compared to eighth day. The first reach success rate was significantly reduced in all recalling days compared to the pre-SCI acquired level (training day: P <.0001, F (11, 55) = 123.5; day 8 vs. days 22–25: P <.0001), indicating that induction of thoracic SCI, once skilled reaching performance was well acquired, significantly affect the first reach success rate (Figure 2E). In a separate comparison between the SCI group and PRE–POST group, it has been shown that the first reach success rate, which was achieved by the PRE–POST group over the post-SCI recalling days, was significantly higher compared to the achieved rate of the SCI group in the last training day. (Multiple unpaired t-test; day 6 (SCI group) vs. day 22 (PRE–POST group): P = .002, day 6 vs. day 23: P = .0002, day 6 vs. days 24–25: P <.000; not shown.)
LFP activity was recorded in HLA and FLA contralateral to preferred forelimb from control and SCI rats (n = 8 in both groups) throughout six days while the rats were at rest (Figure 3A). The time–frequency spectrogram of recorded signals in HLA and FLA revealed a marked increase of LFP power in SCI rats (Figure 3B). Two-way repeated-measures ANOVA followed by Sidak's multiple comparisons test for each frequency band demonstrated that the LFP power in HLA of SCI rats was significantly higher at delta (group effect: F(1, 14) = 36.94, P <.0001), theta (group effect: F(1, 14) = 17.57, P = .0009), beta (group effect: F(1, 14) = 12.23, P = .0036) and gamma frequency bands (group effect: F(1, 14) = 10.43, P = .0061) throughout all six days when compared with control rats (Figure 4A). As such, the LFP power in FLA of SCI rats was significantly higher at delta (group effect: F(1, 14) = 83.27, P <.0001; throughout all six days), theta (group effect: F(1, 14) = 23.27, P = .0003; on all days except day 2), and beta frequency bands (group effect: F(1, 14) = 28.25, P = .0001; on all days except day 5) when compared with control rats (Figure 4B). In terms of differences over time, we only found a significant reduction of delta LFP power in the HLA (day 1 vs. day 3: P = .040; day 1 vs. day 4: P = .0005; day 1 vs. day 5: P = .004; day 1 vs. day 6: P <.0001) and FLA (day 1 vs. day 6: P = .0041) of the SCI group. The ratio of LFP power in HLA to FLA was tended to increase following SCI compared to the control group, and it was statistically significant at gamma frequency band (group effect: F (1, 14) = 9.06, P = .009; Figure 4C).
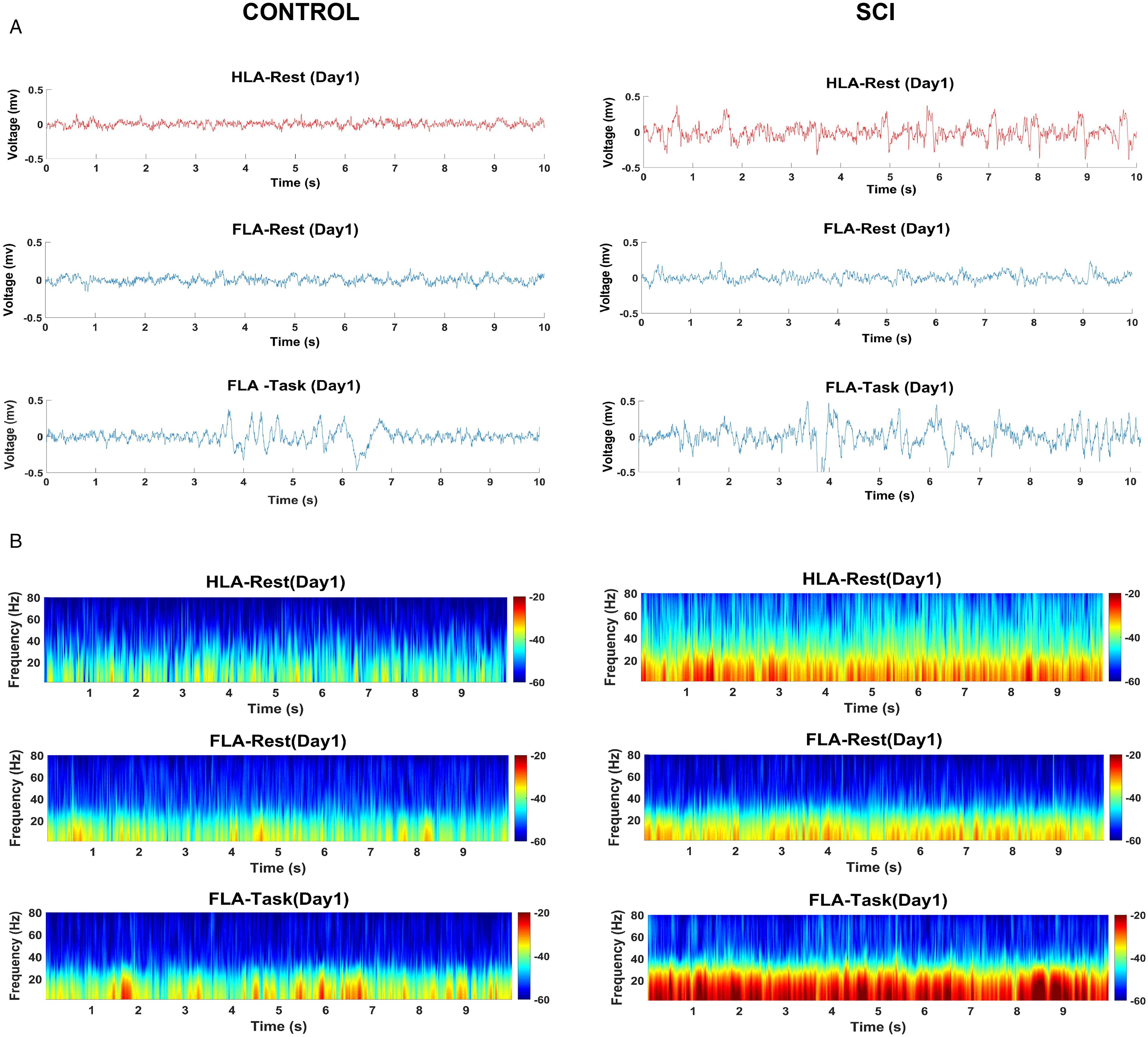
Representative examples of LFP signals in HLA (in resting state) and FLA (in resting state and reaching behavior) from a control rat (left) and a SCI rat (right) (A). Representation of LFP power as a time-frequency spectrograms in HLA (in resting state) and FLA (in resting state and reaching behavior) from all control (left) and SCI rats (right). Red indicates greater power and blue indicates less power (B). Note. LFP = local field potential; HLA = hindlimb area; FLA = forelimb area; SCI = spinal cord injury
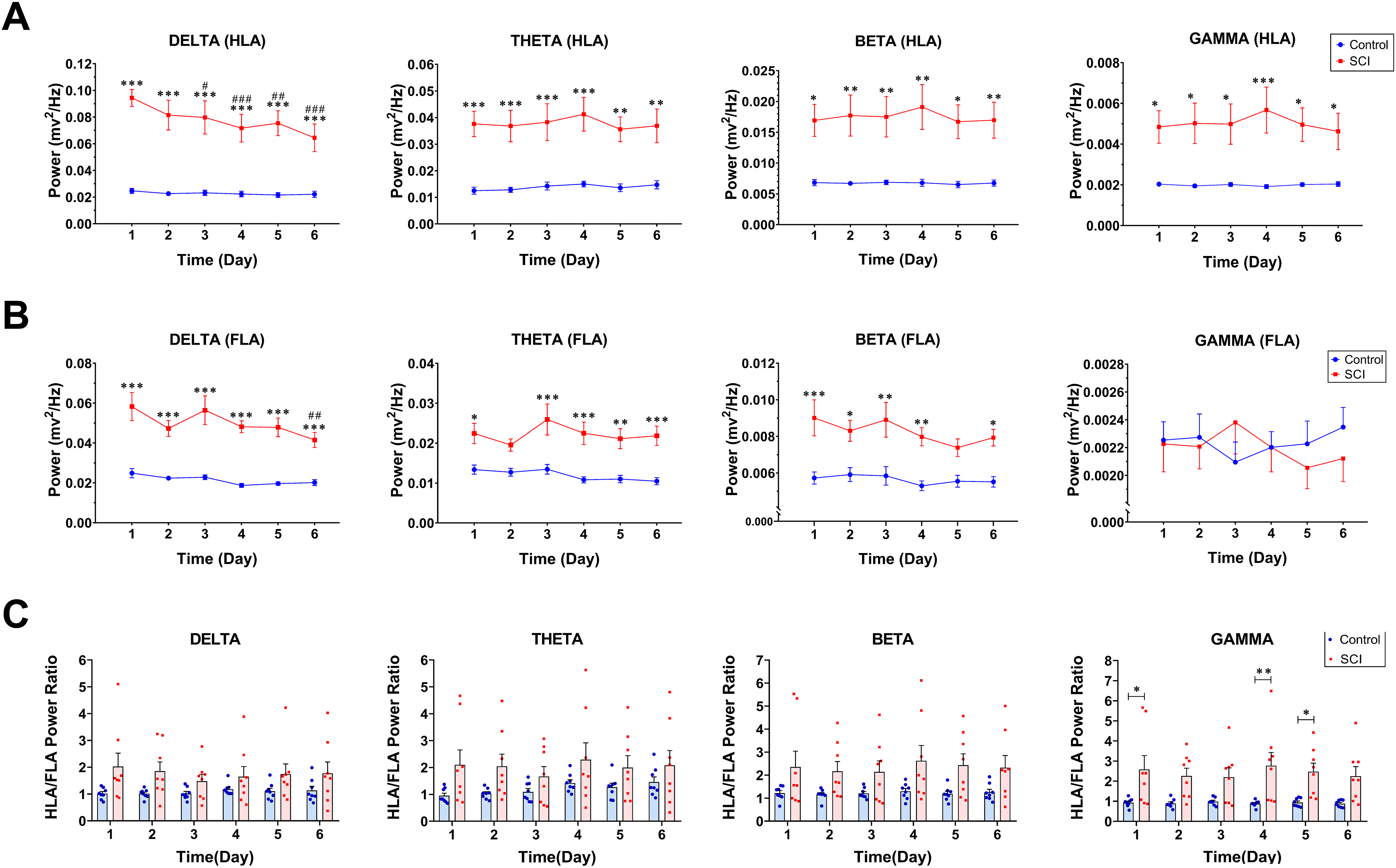
Thoracic SCI enhances oscillatory powers of HLA and FLA in the resting state. Demonstration of delta, theta, beta, and gamma LFP power throughout six days in the resting state (A: HLA; B: FLA). Demonstration of the HLA/FLA power ratio for delta, theta, beta, and gamma throughout six days (C). Data are presented as means ± SEM (n = 8 per group). Denotation of significant differences between control and SCI groups (*P <.05, **P <.01, ***P <.001). Denotation of significant differences within-group comparison with respect to first day (#P <.05, ##P <.01, ###P <.001). Note. HLA = hindlimb area; FLA = forelimb area; SCI = spinal cord injury.
LFP activity was recorded in FLA contralateral to preferred forelimb from control and SCI rats (n = 8 in both groups) throughout six days during the skilled forelimb reaching task (Figure 3A). The time–frequency spectrogram of recorded signals in both groups represented notable enhancement of LFP power during the reaching task (Figure 3B). Two-way repeated-measures ANOVA followed by Sidak's multiple comparisons test for each frequency band demonstrated that the LFP power of FLA in task condition was significantly higher at delta (group effect: F(1, 14) = 53.38, P <.0001), theta (group effect: F(1, 14) = 74.45, P <.0001), beta (group effect: F(1, 14) = 79.95, P <.0001) and gamma frequency bands (group effect: F(1, 14) = 41.93, P <.0001) overall six training days in SCI rats compared with controls (Figure 5A). Furthermore, we compared the LFP power at all frequency bands under resting state and subsequent reaching task, in both the Control and SCI groups (Figure 5B and C, respectively). The SCI rats showed a significant enhanced power at all frequencies in the task condition compared with resting state (delta: F(1, 7) = 95.66, P <.0001; theta: F(1, 7) = 108.8, P <.0001; beta: F(1, 7) = 70.24, P <.0001; gamma: F(1, 7) = 95.60, P <.0001) over all training days, while in Control rats, the LFP power was significantly higher in task condition at delta (F(1, 7) = 134.5, P <.0001; throughout six days), theta (F(1, 7) = 180.4, P <.0001; throughout six days), and beta (F(1, 7) = 26.49, P = .0013; on days 4–6) when compared with resting state. Furthermore, post hoc analysis in the SCI group did not show any difference over the days in either condition. As such, in the Control group, there was no difference over the days in the resting state, whereas the LFP power of FLA at all frequency bands tended to increase over the training days in the task condition that it reached significance on the last training days (time effect, delta: F(5, 35) = 4.33, P = .0036; theta: F(5, 35) = 5.21, P = .0011; beta: F(5, 35) = 6.23, P = .0003; gamma: F(5, 35) = 5.00, P = .0015).
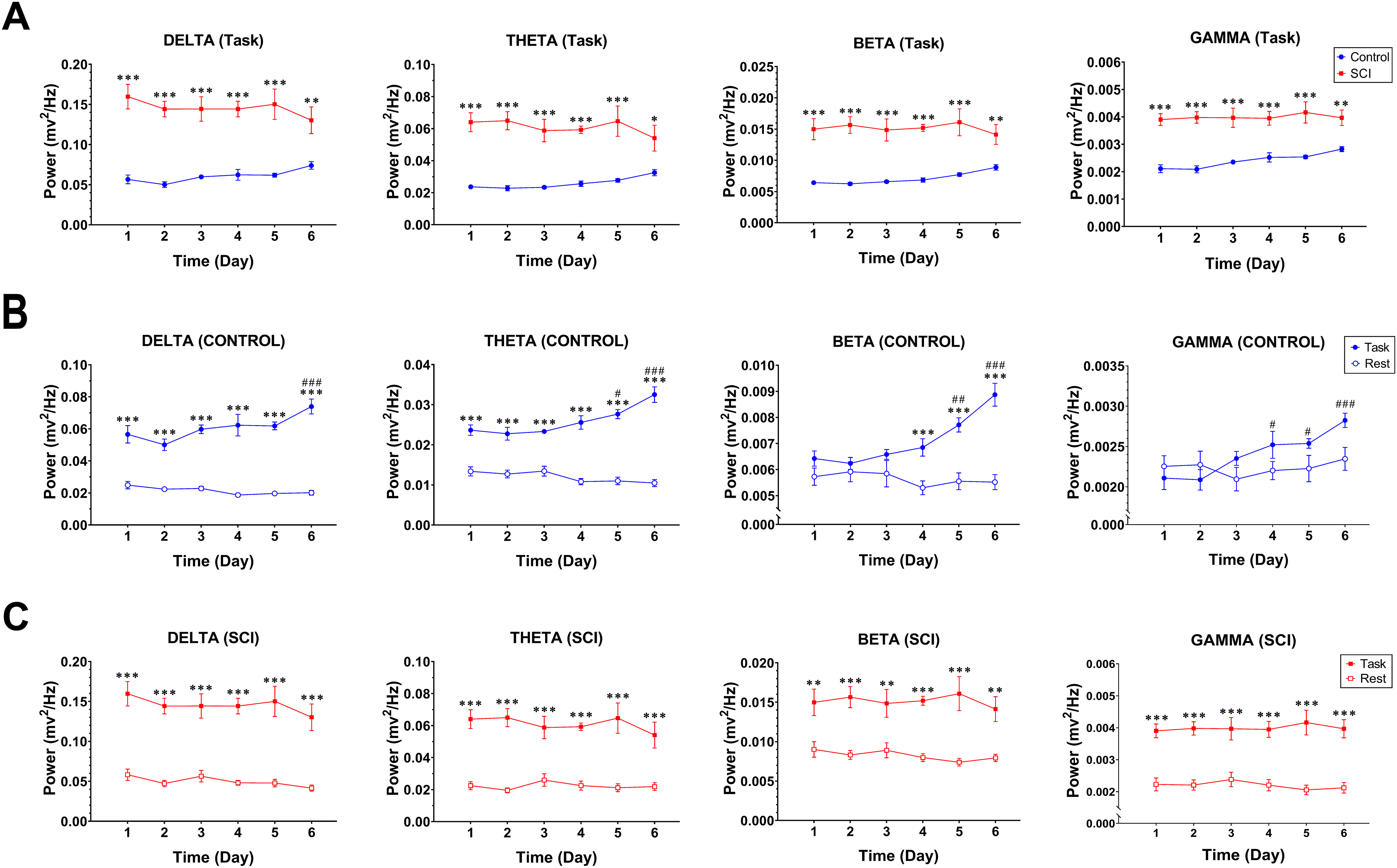
Thoracic SCI enhances oscillatory powers of FLA during reaching behavior. Between-groups comparison for delta, theta, beta, and gamma LFP power throughout six days during reaching behavior (A). Within-groups comparison for delta, theta, beta, and gamma throughout six days (B; Control, C; SCI). Data are presented as means ± SEM (n = 8 per group). Denotation of significant differences between control and SCI groups or between rest and task conditions in each group (*P <.05, **P <.01, ***P <.001). Denotation of significant differences with respect to first day (#P <.05, ##P <.01, ###P <.001). Note. SCI = spinal cord injury; FLA = forelimb area; LFP = local field potential.
DA concentrations in the Ipsi-FLA, Cont-FLA, and Cont-HLA were determined in both the trained Control (n = 5) and SCI (n = 5) groups (Figure 6A), at the conclusion of the last training day. An untrained control subgroup (UT-Control; n = 5) was considered to assess baseline levels of DAergic markers. Two-way mixed ANOVA for DA concentrations showed significant effects of group (F(2, 12) = 4.546, P = .034) and cortical area (F(2, 24) = 16.23, P <.0001). In between-groups comparisons, we found only significantly lower DA levels in the Cont-HLA of SCI rats in contrast to uninjured rats (UT-Control, P = .009; Control, P = .013). Based on within-group comparisons, there was no significant difference between the three cortical areas of M1 in either Control subgroups. However, in the SCI group, the DA concentrations in the Cont-HLA were significantly lower than that in the Cont-FLA (P = .0001) and Ipsi-FLA (P = .005).
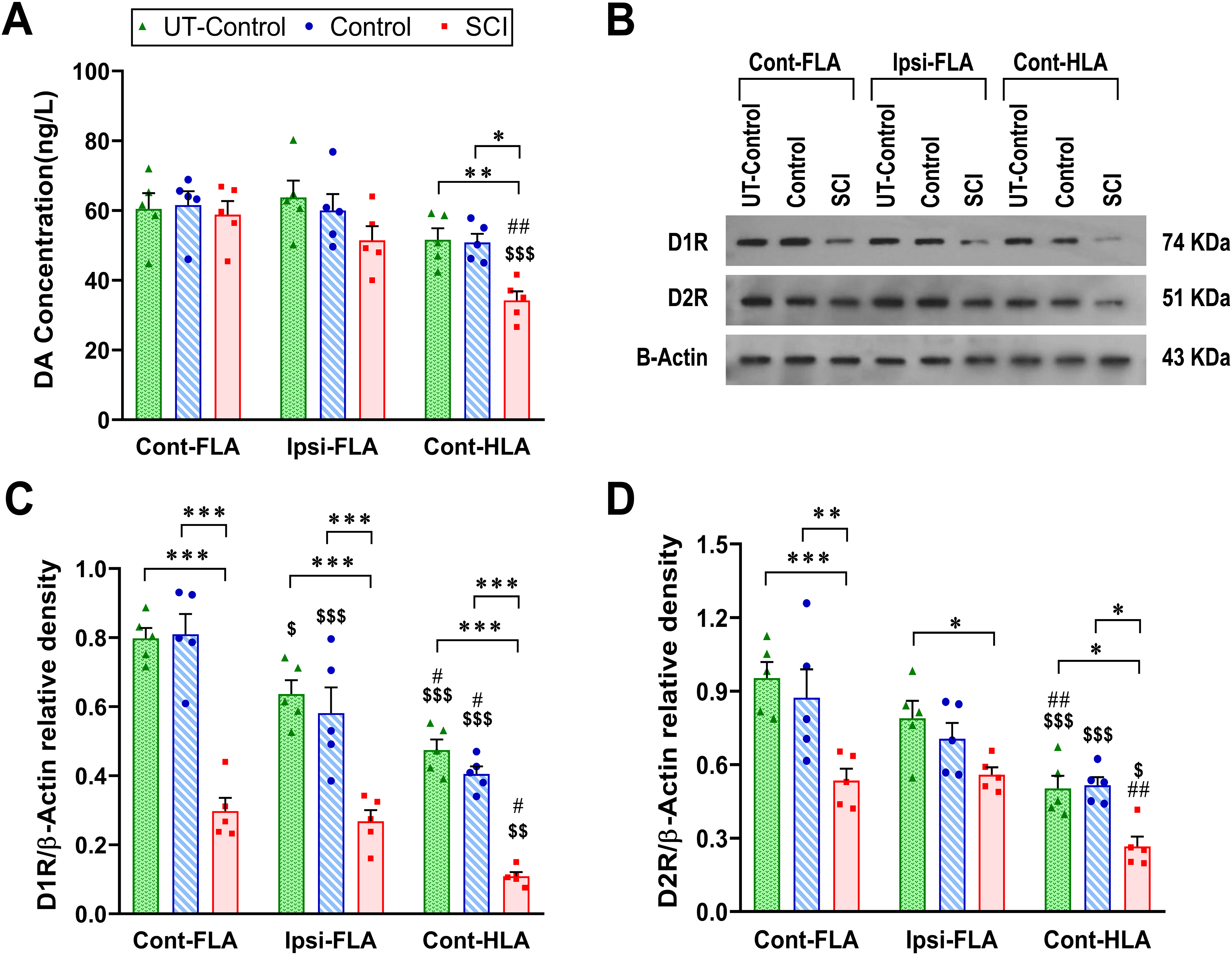
Thoracic SCI reduces DA levels, D1R and D2R expression in M1. DA levels and expression of dopamine receptors (D1R and D2R) were determined in contralateral FLA (Cont-FLA), ipsilateral FLA (Ipsi-FLA), and contralateral HLA (Cont-HLA) of the untrained Control (UT-Control) subgroup, and both trained Control and SCI main groups. Bar diagram demonstrates DA levels of cortical areas in all groups (A). A representative immunoblotting bands of D1R and D2R (B). Bar diagrams show D1R (C) and D2R (D) levels (normalized against β actin) of cortical areas in all groups. Data are presented as means ± SEM (n = 5 per group). Denotation of significant differences between all three groups (*P <.05, **P <.01, ***P <.001). Denotation of significant differences within-group comparison with respect to Cont-FLA ($P <.05, $$P <.01, $$$P <.001) and Ipsi-FLA (#P <.05, ##P <.01). Note. SCI = spinal cord injury; DA = dopamine; HLA = hindlimb area; FLA = forelimb area.
The expression of D1R and D2R were determined in the Ipsi-FLA, Cont-FLA and Cont-HLA of Control (Control, n = 5; UT-Control, n = 5) and SCI (n = 5) groups, at the conclusion of the last training day (Figure 6B). Two-way mixed ANOVA for expression of D1R showed significant effects of group (F(2, 12) = 69.96, P <.0001) and cortical area (F(2, 24) = 47.42, P <.0001) (Figure 6C). Between-groups comparisons demonstrated that the expression of D1R was significantly lower in the Cont-FLA, Ipsi-FLA, and Cont-HLA of SCI rats compared with both trained and untrained control rats (P <.0001 for all comparisons). Based on within-group comparisons for each group, D1R expression in Cont-HLA was statistically lower with respect to Cont-FLA (UT-Control: P <.0001; Control: P <.0001; SCI: P = .006) and Ipsi-FLA (UT-Control: P = .019; Control: P = .01; SCI: P = .022). Moreover, uninjured rats showed significantly higher D1R expression in Cont-FLA compared to Ipsi-FLA (UT-Control: P = .02; Control: P = .001).
There were significant effects of group (F(2, 12) = 16.68, P = .0003) and cortical area (F(2, 24) = 28.12, P <.0001) for expression of D2R (Figure 6D). Between-groups comparisons demonstrated that the D2R expression was significantly lower in the Cont-FLA (UT-Control vs. SCI: P = .0001; Control vs. SCI: P = .0015), Ipsi-FLA (UT-Control vs. SCI: P = .039), and Cont-HLA (UT-Control vs. SCI: P = .033; Control vs. SCI: P = .023) of SCI rats compared with control rats. Within-group comparisons for each group showed that the expression of D2R in Cont-HLA was statistically lower than that in Cont-FLA (UT-Control: P <.0001; Control: P = .001; SCI: P = .013) and Ipsi-FLA (UT-Control: P = .007; SCI: P = .006). However, there was no significant difference between the Cont-FLA and Ipsi-FLA in either groups.
Correlation Between LFP Activity in FLA and the Performance of Skilled Forelimb Reaching Task
Considering the fundamental role of brain activity in motor and cognitive performance, we investigated the hypothesis that success rate in skilled reaching behavior throughout six training days would be correlated with simultaneous LFP power of Cont-FLA in each rat during task condition. Significant positive day-by-day correlations were found between the first reach success rate (%) and the power of delta (Rat No. 4: r = 0.86, P = .03; Rat No. 5: r = 0.92, P = .009; Rat No. 8: r = 0.98, P = .0002), theta (Rat No. 1: r = 0.81, P = .048; Rat No. 5: r = 0.96, P = .003), beta (Rat No. 1: r = 0.90, P = .014; Rat No. 4: r = 0.80, P = .05; Rat No. 5: r = 0.90, P = .013; Rat No. 8: r = 0.86, P = .028), and gamma (Rat No. 1: r = 0.88, P = .021; Rat No. 2: r = 0.90, P = .013; Rat No. 4: r = 0.88, P = .019; Rat No. 5: r = 0.95, P = .004; Rat No. 6: r = 0.93, P = .006; Rat No. 7: r = 0.92, P = .009 Rat No. 8: r = 0.96, P = .002) frequency bands in the Control group. In contrast, we found no significant correlations between the first reach success rate (%) and LFP powers in none of the SCI rats (Figure 7A).
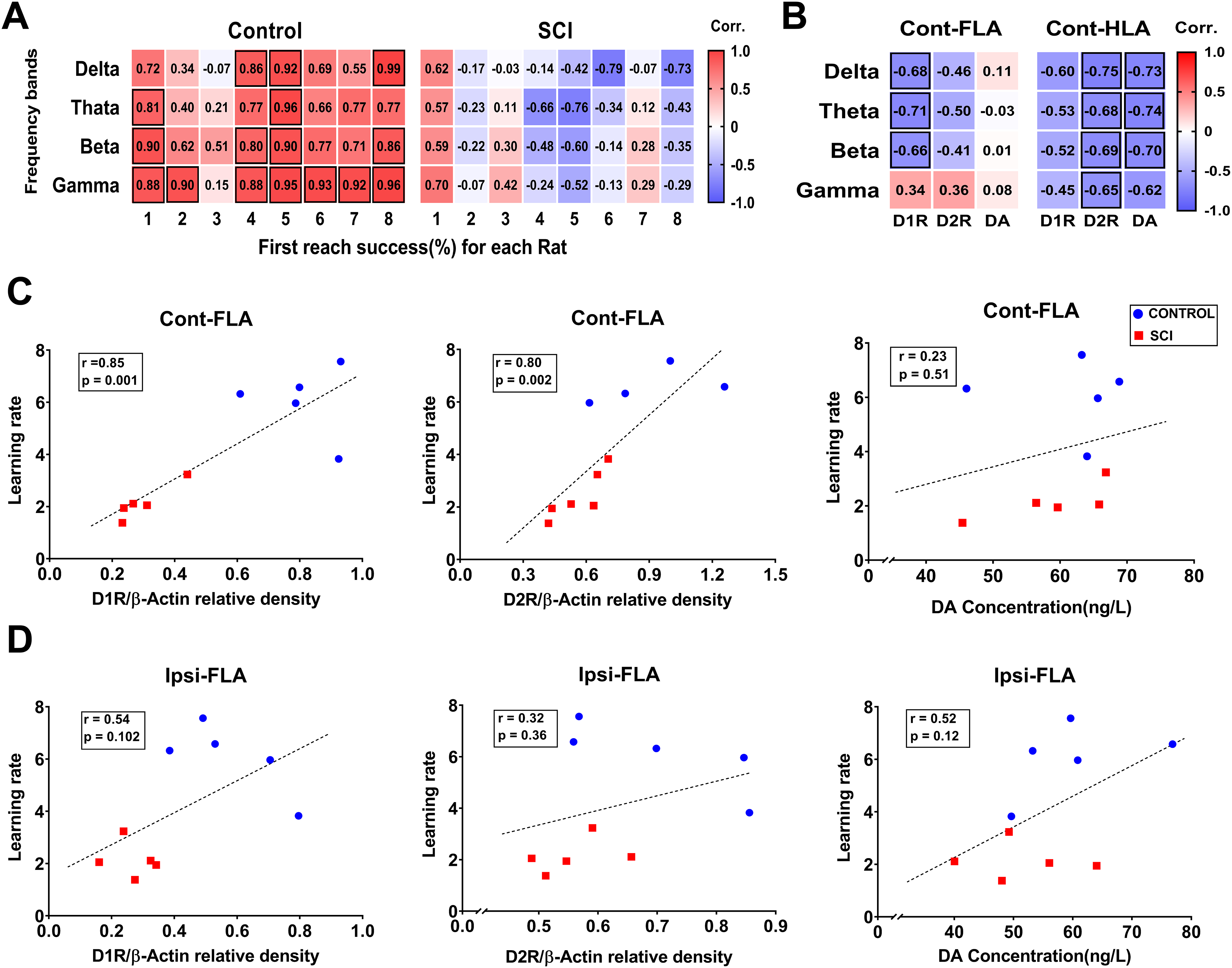
Multiple correlations among motor skills, DAergic markers and oscillatory powers. Day-by-day correlations between Cont-FLA oscillatory powers during task condition and the first reach success rate for each rat (A). Animal-by-animal correlations between resting state oscillatory powers (day 6) and DA/D1R/D2R levels in the Cont-FLA and Cont-HLA (B). Note: Bordered squares indicate significant correlation. The color bar is ranged from blue (strong negative correlation) to red (strong positive correlation). Animal-by-animal correlations between learning rate for first reach success and DA/ D1R/ D2R levels in Cont-FLA (C) and Ipsi-FLA (D). Note. DA = dopamine; FLA = forelimb area; HLA = hindlimb area.
To determine whether changes of LFP activity observed in HLA and FLA following SCI were correlated with DAergic markers, the animal-by-animal correlations between the LFP power in resting state (day 6) and DA, D1R, and D2R levels were assessed in Cont-FLA and Cont-HLA (Figure 7B). In FLA, the LFP power at all frequency bands except gamma was correlated negatively with expression of D1R (delta: r = −0.68, P = .029; theta: r = −0.71, P = .020; beta: r = −0.66, P = .039), but not in relationship with the DA and D2R levels. In HLA, there were significant negative correlations between all frequency bands and the DA levels (delta: r = −0.73, P = .017; theta: r = −0.74, P = .015; beta: r = −0.70, P = .024; gamma: r = −0.62, P = .05), and expression of D2R (delta: r = −0.75, P = .012; theta: r = −0.68, P = .029; beta: r = −0.69, P = .029; gamma: r = −0.65, P = .041). However, there was no significant correlation in relationship with expression of D1R.
Correlation Between Levels of DAergic Markers and Learning Rate of Skilled Forelimb Reaching Task
In order to determine whether the deficit in motor skills learning following SCI is associated with the levels of DAergic markers, we examined the animal-by-animal correlations between the learning rate for first reach success and DA, D1R, and D2R levels in Cont-FLA (Figure 7C) and Ipsi-FLA (Figure 7D). We found strong positive correlations between the learning rate and expressions of D1R (r = 0.85, P = .001), and D2R (r = 0.80, P = .002) in Cont-FLA but not in Ipsi-FLA. There was no significant correlation in relationship with DA levels in bilateral FLA.
Discussion
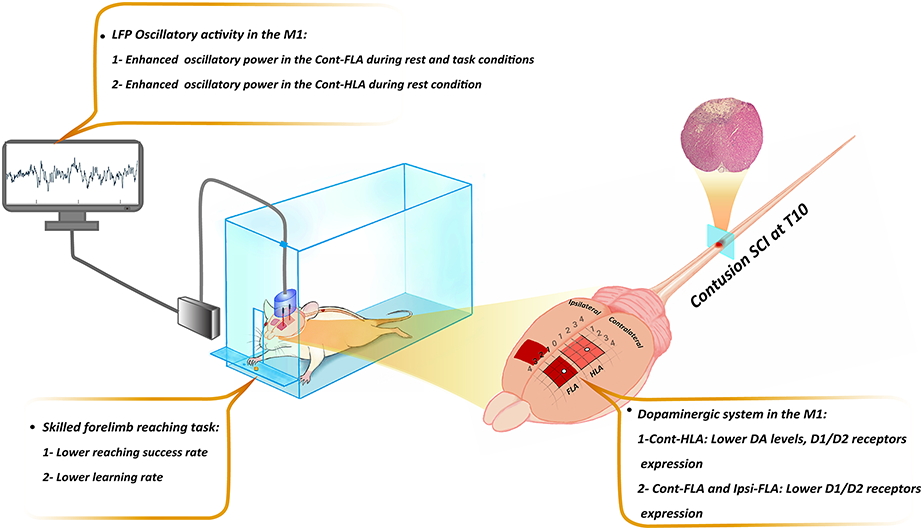
This study demonstrated that the thoracic spinal contusion along with the sustained increase of LFP power in the HLA and FLA, and decreased levels of DAergic markers in these areas, coincide with a deficit in motor skills learning. This evidence proves that the functional, molecular, and neural deficits, following thoracic SCI, are not only limited to the HLA, but also the FLA is touched by that.
The present study, for the first time, showed that dysfunctional effects of thoracic SCI could extend far beyond the hindlimb sensorimotor paralysis, and cause deficits in forelimb motor skills learning. Despite similar success rates of both control and SCI groups on the first training day of the skilled forelimb reaching task, the injured animals could not achieve an acceptable performance over the subsequent training days compared to control ones. Furthermore, the poor performance of the injured rats in the first reach success may indicate a lack in the tuning of forelimb motor skills execution. Motor skills learning, as a cognitive-motor function, like other types of cognitive functions, requires the formation and consolidation of acquired memories (Luft & Buitrago, 2005). Within-day learning, as an indicator of fast learning, was well developed during the first training day in the SCI group, denoting the intact primary formation of motor memory (Censor et al., 2012). Although acquired memory was not maintained over the first and second 24 h intervals, therefore the first session of subsequent training days was started with a lower success rate. These findings suggest that the overnight consolidation of motor skills might be disrupted following SCI, and consequently, the running acquired memory cannot be stabilized to organize slow learning over training days. Despite the low dependance of skilled forelimb reaching task to hindlimb motor activity, It could be raised that the less success rate of injured rats may not reflect pure motor skill deficit, but may be confounded by loss of the hindlimb role in body-weight-bearing, and increase of weight shift to the forelimbs. To rule out this interfering factor, the PRE–POST group was defined as a secondary group, that had been acquired the reaching skill before the SCI and recalled it post-SCI. Thoracic SCI did not affect the total success rate in recalling days, while the first reach success rate was significantly lowered when recalled in the post-SCI training days. On the other hand, this success rate was considerably higher than the main SCI group, who had learned the task as a new one. These findings confirm that the decreased postural stability and increased weight-bearing on forelimbs after thoracic SCI, to some extent, affect forelimb motor skills performance by reducing the accuracy of movement. However, the present study suggests that a possible major reason for the poor performance of injured rats in the reaching skill task is due to the cognitive dysfunction associated with motor skills learning.
A series of experimental studies have reported that thoracic SCI initiates progressive neurodegeneration in the hippocampus and cortex, which results in impairment of spatial learning and memory (Wu et al., 2014a, b, 2016). It seems that motor skills learning, independent of hippocampal circuits, requires appropriate neuroplasticity in the M1 (Georgopoulos, 2000; Luft et al., 2004; Mutha & Haaland, 2014); therefore, deficits in this type of learning following thoracic SCI, as shown in the present study, may be due to retrograde pathological changes in M1.
After thoracic SCI, the HLA and FLA showed an increase in the LFP power at all frequency bands in the resting state that was more notable at low frequencies. In contrast to control rats, the ratio of LFP power in HLA to FLA was tended to increase after thoracic SCI, reflecting the fact that injury to the thoracic spinal cord has a greater effect on the HLA neural activity, which directly innervates the paralyzed hindlimb. The present results are along with most previous clinical imaging-electrophysiological studies that reported, the activity of M1 increases in thoracic SCI subjects (Kokotilo et al., 2009). Nevertheless, electroencephalography (EEG) findings show contradictory results in terms of theta, alpha, and beta frequencies power in SCI patients, as an index of functional connectivity in the M1(Athanasiou et al., 2018; Castro et al., 2007; Fallani et al., 2007; Gourab & Schmit, 2010; Mattia et al., 2006). The variety of results in these studies might be partly attributed to the heterogeneity of the post-injury time, severity or level of the lesion (Kokotilo et al., 2009).
Animal models of SCI, despite the neuroanatomical differences with humans, provide more homogeneous results to investigate the underlying mechanisms involving in the alteration of brain activity following SCI. In this study, using the locomotion and histological assessments, we confirmed that the severity of contusion-induced SCI was identical in all injured animals. Given the minimization of confounding factors, it can be concluded that the wide range of LFP powers among SCI rats can be due to inter-individual differences in pathophysiological responses to SCI. Regarding the present study, Frost et al. (2015) reported that, spontaneous activity increased in the HLA and, interestingly FLA after thoracic contusion SCI in the rat (Frost et al., 2015). Considering the functional connectivity between the forelimb and hindlimb cortical areas, these results might be derived from the enhanced glutamatergic excitation and reduced GABAergic inhibition (Ghosh et al., 2010). In animal models, similar to clinical findings, the loss of spinal cord pathways induce a retrograde degeneration in the M1, which possibly alters the dynamic activity of the brain following SCI (Nardone et al., 2013; Wrigley et al., 2009; Zhu et al., 2015).
Herein, we demonstrated that thoracic SCI retrogradely attenuates DA levels in the Cont-HLA, as well as the D1R and D2R expression in both Cont-HLA and FLA of rats. The loss of DA, Parallel with the D1R and D2R in HLA, could represent the possible effect of SCI on the DAergic system at both presynaptic and postsynaptic levels (Volkow et al., 1998). Disruption of DA transmission in M1 occurs by various neurodegenerative disorders such as PD, multiple sclerosis, and TBI (Gaspar et al., 1991). According to pre-clinical and clinical findings of PD studies, the reduction of DAergic projections from VTA to M1 is accompanied by the appearance of exaggerated spectral power of theta, beta, and gamma frequency bands in M1(Nimmrich et al., 2015; Ozkan et al., 2017; Wang et al., 2015). In line with these studies, our findings showed that, the reduction of D1R and D2R levels in FLA and HLA is correlated with increased LFP power after SCI. The negative correlation of LFP power with the simultaneous decrease of D1R and D2R expressions may reflect the synergistic effects of both receptors on the modulation of M1 neural activity (Awenowicz & Porter, 2002). These effects may be mediated directly via the expression of D2R in glutamatergic pyramidal neurons or indirectly through an expression of D1Rs on GABAergic interneurons (Ozkan et al., 2017; Vitrac et al., 2014). It is noteworthy that the current study was designed to assess the effect of SCI on oscillatory activity and the DAergic system of M1, rather than to determine the underlying mechanism of their interactions. Therefore, it is not specified how exactly the reduction of DAergic markers after SCI correlate with enhanced LFP power in M1. Evaluation of the glutamatergic and GABAergic systems besides the dopaminergic system could help to focus on this hypothesis.
Previous studies have shown that, the intact DA signaling in M1 is an essential component for the acquisition and storage of new motor skills at the cellular and synaptic levels (Hosp & Luft, 2013; Ozkan et al., 2017). Confirming these theoretical notions, we found that deficit in motor skills learning following SCI is correlated with the reduced expression of D1R and D2R in Cont-FLA. Furthermore, we showed that the expression of D1R and D2R are typically higher in the FLA compared with HLA in all groups, supporting the finding of previous study, that mesocortical DA projections to M1 specifically innervate the FLA (Vitrac et al., 2014). Otherwise, herein, both uninjured groups showed significantly higher D1R expression in Cont-FLA compared to Ipsi-FLA; however, there was no significant difference between the trained and untrained control rats. This finding may indicate that, the expression of this receptor is naturally dominant in this area which corresponds to the preferred forepaw, so that the skilled forelimb reaching task at least in the defined training period, has a minimal effect on D1R expression.
Finally, it has been found that the LFP power of FLA at all frequency bands was significantly higher in SCI rats in contrast to controls during the skilled forelimb reaching task. It is in line with human investigations that showed greater activation volume in the contralateral M1-upper limb region among thoracolumbar SCI patients compared with healthy subjects during hand movements (Curt et al., 2002; Sabre et al., 2016). In addition to discussed interpretations for resting state, another possible reason for exaggerated LFP power of FLA in the injured rats might be partly due to an increased weight-bearing role of forelimb during task condition (Ghosh et al., 2010).
Compared to the resting state, a steady enhanced power was observed at all frequency bands during six training days in SCI rats; however, the raised oscillatory power in control rats was evident in the last days of motor learning. It is also worth mentioning that LFP powers especially at beta and gamma frequency bands were positively correlated with the success rate achieved by control rats. Improved motor skills learning accompanied by a functional enhancement of FLA oscillatory activity in uninjured rats, mentioning the pivotal role of motor cortex oscillations, especially beta and gamma bands in successful encoding and storage of motor skills memory in FLA (Babiloni et al., 2004). In contrast, the aberrant exaggerated oscillations in FLA of SCI rats over the training sessions may indicate a disturbance in underlying mechanisms of motor skills learning. The overall evidence suggests that enhancement of brain oscillatory power is linked to a wide range of motor and cognitive processes, including voluntary movements control and memory operations (Ghosh et al., 2010; Rosenkranz et al., 2007). On the flip side, it has been recognized that horizontal and intralaminar connections in M1, and the density of dendritic spines in pyramidal neurons are strengthened during successful motor skills learning, which is manifested as increased excitability of M1 and evoked LFP in cortical slices (Li et al., 2017).
Conclusion
The present study provides a new insight that the functional and molecular reorganization of the M1 following thoracic SCI is not restricted to the HLA, and interestingly extends to the FLA, which appears as the dysfunctional change in LFP activity and DAergic system, associated with a deficit in motor skills learning of non-affected forelimb.
 However, complementary mechanistic studies are needed to confirm these observations and to better understanding the effect of SCI on brain oscillations, DAergic function, and their interaction in motor skills learning.
However, complementary mechanistic studies are needed to confirm these observations and to better understanding the effect of SCI on brain oscillations, DAergic function, and their interaction in motor skills learning.
Supplemental Material
sj-docx-1-asn-10.1177_17590914211044000 - Supplemental material for Forelimb Motor Skills Deficits Following Thoracic Spinal Cord Injury: Underlying Dopaminergic and Neural Oscillatory Changes in Rat Primary Motor Cortex
Supplemental material, sj-docx-1-asn-10.1177_17590914211044000 for Forelimb Motor Skills Deficits Following Thoracic Spinal Cord Injury: Underlying Dopaminergic and Neural Oscillatory Changes in Rat Primary Motor Cortex by Omid Salimi, Hamid Soltani Zangbar, Soheila Hajizadeh Shadiabad, Meysam Ghorbani, Tahereh Ghadiri, Abbas Ebrahimi Kalan, Hasan Kheyrkhah and Parviz Shahabi in ASN Neuro
Footnotes
Acknowledgments
The authors would like to express their gratitude to Sajjad Badalkhani for his assistance in electrophysiological analysis.
Author Contribution
O.S., H.S.Z. and P.S. designed the study; O.S.., H.S.Z. and M.G. performed data collection; P.S., A.E.K., and O.S. analyzed behavioral and biochemical data; S.H.S., H.S.Z., and O.S. analyzed electrophysiological data. P.S., H.S.Z., and O.S. wrote the paper, and T.G. edited the paper.
Declaration of Conflicting Interests
The authors claim that there are no conflicts of interest.
Ethical Approval
All procedure and handling techniques were approved by the Ethical Committee for Animal Research of Tabriz University of Medical Sciences (IR.TBZMED.REC.1396.874), and surgical procedures were conducted under aseptic conditions.
Funding
This study was supported by grants (No. 58802) from Tabriz University of medical Science.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
