Abstract
One hallmark of human aging is increased brain inflammation represented by glial activation. With age, there is also diminished function of the adaptive immune system, and modest decreases in circulating B- and T-lymphocytes. Lymphocytes traffic through the human brain and reside there in small numbers, but it is unknown how this changes with age. Thus we investigated whether B- and T-lymphocyte numbers change with age in the normal human brain. We examined 16 human subjects in a pilot study and then 40 human subjects from a single brain bank, ranging in age from 44–96 years old, using rigorous criteria for defining neuropathological changes due to age alone. We immunostained post-mortem cortical tissue for B- and T-lymphocytes using antibodies to CD20 and CD3, respectively. We quantified cell density and made a qualitative assessment of cell location in cortical brain sections, and reviewed prior studies. We report that density and location of both B- and T-lymphocytes do not change with age in the normal human cortex. Solitary B-lymphocytes were found equally in intravascular, perivascular, and parenchymal locations, while T-lymphocytes appeared primarily in perivascular clusters. Thus, any change in number or location of lymphocytes in an aging brain may indicate disease rather than normal aging.
More people are reaching older ages than they did a half century ago, and the average age of our population continues to increase (Beard et al., 2016). Age-related immune changes have been well-documented (Frasca et al., 2008). It is well established that older adults exhibit multiple signs of impaired immune function, including increased incidence and severity of infections, and declines in efficacious immune response to vaccines (Frasca et al., 2011; Nikolich-Žugich, 2018; Yoshikawa, 2000).
One of the immune shifts that occur with age is a decrease in circulating adaptive immune cells (B- and T- lymphocytes). In humans, peripheral B-cells decline by 10-50% during aging (Breitbart et al., 2002; Frasca et al., 2008), with naive B-cells decreasing while memory B-cells increase (Ademokun et al., 2010; Frasca et al., 2011; Scholz et al., 2013). Absolute counts of circulating T-cells also decrease with age in humans (Freitas et al., 2019; Jentsch-Ullrich et al., 2005). These declines in the blood are coupled with decreased proliferation of B- and T-cells in bone marrow (Ogawa et al., 2000). Still, whether adaptive immune cells in humans also decline with age in other tissue spaces including the brain remains unknown. Furthermore, data from mice suggest that the trajectory of B- and T-cell counts with age may vary by tissue and organ type (Ademokun et al., 2010; Ramos et al., 2017).
In addition to cell-intrinsic changes, a number of pro-inflammatory and pro-neurodegenerative factors have been shown to increase in the systemic circulation with human aging (Pluvinage & Wyss-Coray, 2020). Innate immune cell activation, involving both microglia and astrocytes, increases throughout the brain as humans age (Koellhoffer et al., 2017; Palmer & Ousman, 2018; Wong, 2013). This activation appears to be influenced by signaling factors in aging human plasma (Yousef et al., 2019). Furthermore, these activated innate immune cells may secrete chemoattractants that recruit adaptive immune cells (Sokol & Luster, 2015).
Increased innate immune activation with age is consistent with recent findings reporting that T-cells increase in brains of normal aging mice (Dulken et al., 2019; Mrdjen et al., 2018). While past published data have established that B- and T-cells reside in the normal human brain parenchyma (Anthony et al., 2003; Busse et al., 2012; Doyle et al., 2015; Machado-Santos et al., 2018; Merlini et al., 2018; Togo et al., 2002), much less is known about whether lymphocyte numbers change in the aging human brain. Dulken et al. found a strong trend towards roughly twice the number of CD3+ T-cells in periventricular neurogenic regions of 6 old human subjects compared to 5 young subjects (Dulken et al., 2019, Table 2). Whether this potential increase in T-cell density is restricted to this neuroanatomic region has not been studied, and other studies of neurological disease have not examined age.
Subject Characteristics.
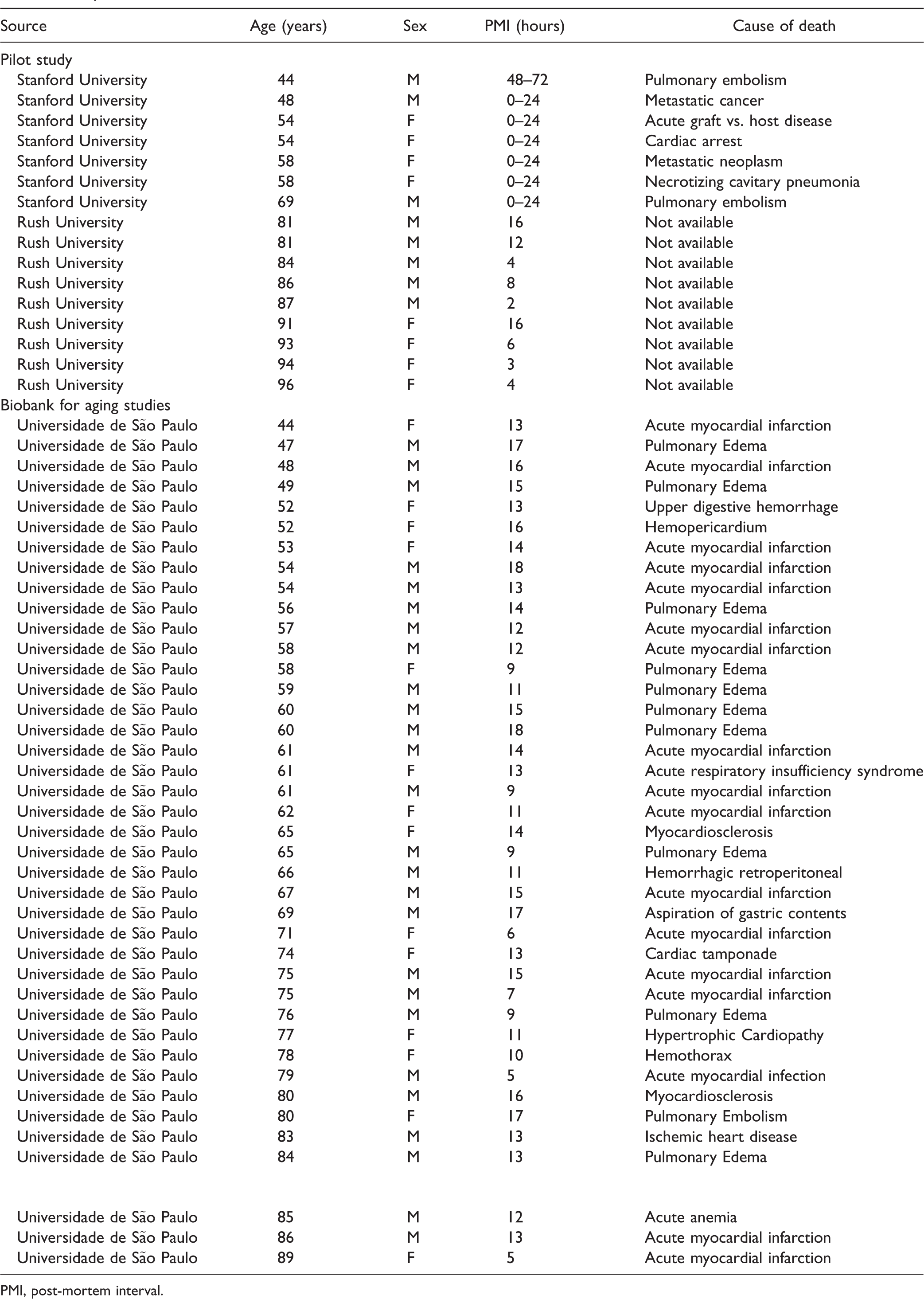
PMI, post-mortem interval.
Previous Studies With Data on B- and T-Lymphocytes in Normal Human Brain Samples.
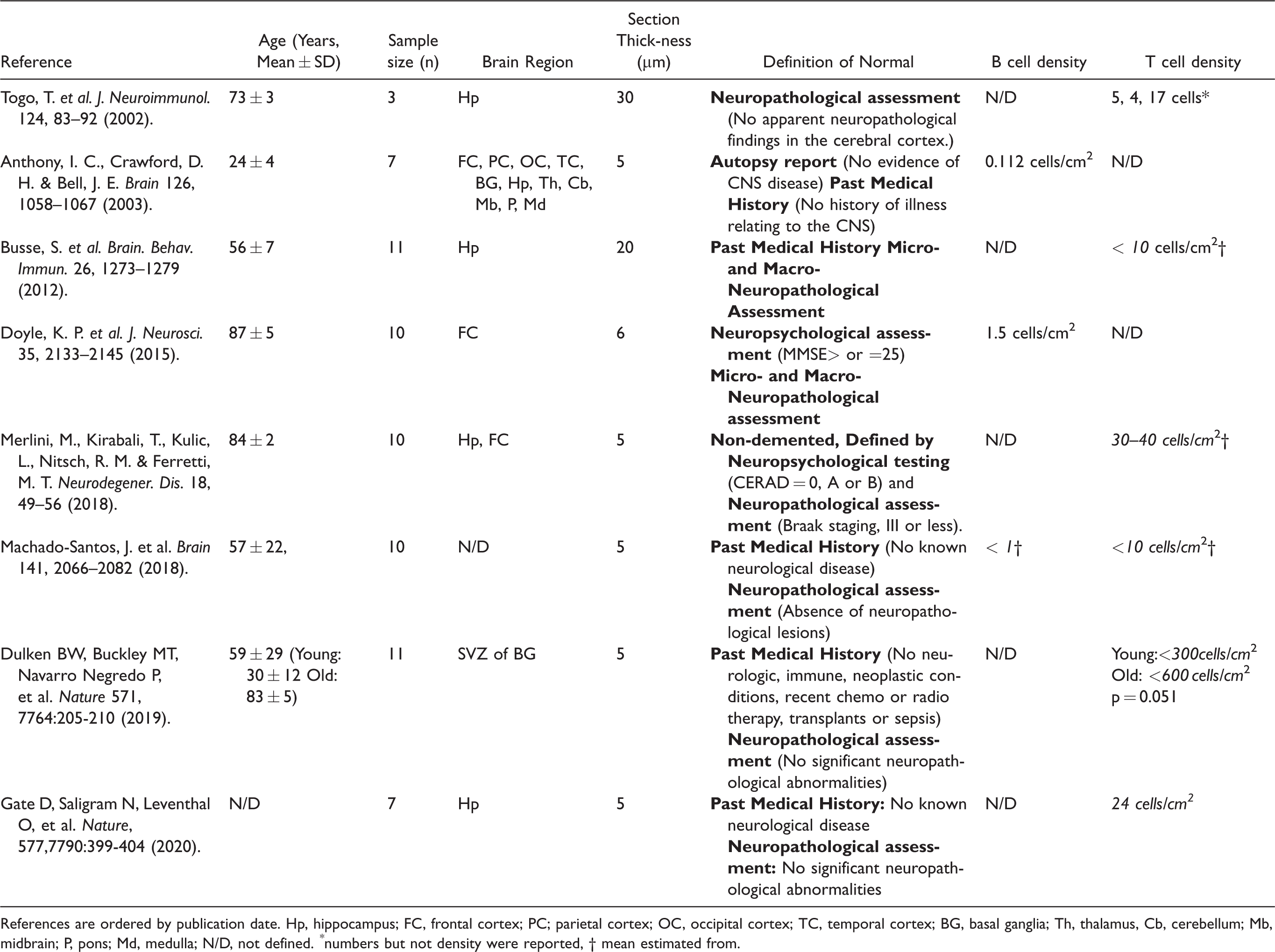
References are ordered by publication date. Hp, hippocampus; FC, frontal cortex; PC; parietal cortex; OC, occipital cortex; TC, temporal cortex; BG, basal ganglia; Th, thalamus, Cb, cerebellum; Mb, midbrain; P, pons; Md, medulla; N/D, not defined. * numbers but not density were reported, † mean estimated from.
To investigate whether the densities of B- and T-lymphocytes in the cortex increases with age in humans, we first performed a small pilot study in normal cortical post-mortem brain tissue and used immunohistochemistry to compare the densities of B- and T-cells in young (44–69 year) and old (81–96 year) subjects. We used this to power a larger study from a single brain bank (Biobank for Aging Studies) (Suemoto et al., 2017). In the larger study, we examined normal cortical brain tissue from 40 individuals, 44–89 years of age. We report these results here, and compare them to prior studies.
Materials and Methods
Subject Selection
For the pilot study, we obtained “normal” cortical tissue from two sources: the Rush Alzheimer’s Disease Center Pathology Core/Rush Memory and Aging Project and Rush Religious Orders Study (Bennett et al., 2012a, 2012b), and the Department of Pathology of Stanford University. Six Stanford subjects were selected from a cohort of 72 participants (ages 44–69) whose brains were examined in Stanford University Dept. of Pathology autopsies between 2014 to 2016. Each subject’s autopsy and brief clinical history reports were reviewed. Exclusion criteria included a history of solid organ or bone marrow transplantation or immunosuppression. The subjects we selected had brains classified as either “no significant gross or microscopic pathology” or “mild to moderate hypertensive vasculopathy,” but no other findings by Stanford University neuropathologists, including no evidence of neurodegenerative disease. While measures were taken to limit the inclusion of participants with immunoproliferative diseases, these participants happened to be relatively common in the Stanford University autopsy collection. They were therefore included when their autopsy showed no significant pathology. Subjects enrolled from Rush University. were longitudinally screened for cognitive impairment before death, and the 9 cases selected from these collections were not demented, had no or low amyloid and tangle pathology, no Lewy bodies, no macro or microinfarcts, and no hippocampal sclerosis.
After generating data for a power calculation, we selected 20 subjects less than 65 years old (44–62 years), and 20 subjects 65 years and older (65–89 years), from a collection of 1,092 brains at the Universidade de São Paulo Biobank for Aging Studies (Suemoto et al., 2017). Clinical data were reviewed, and brain tissue was examined macroscopically and microscopically by at least one pathologist. Inclusion criteria were normal cognition (Clinical Dementia Rating = 0), and without significant neuropathological lesions observed on hematoxylin-eosin stain, and immunostains for beta-amyloid, phosphorylated tau, alpha-synuclein, and TDP-43. Exclusion criteria were macroscopically detectable acute brain infarctions or tumors, hemorrhages or trauma, severe systemic chronic conditions (severe heart failure or chronic kidney failure) that might damage cognitive function prior to death, acidosis due to severe agonal status (cerebrospinal fluid pH < 6.5), and unreliable clinical information. We also excluded subjects with a history of systemic inflammatory disease, and those cases with infections detected by the autopsy exam.
All studies followed local and federal regulations for the study of human specimens from deceased subjects, including the Helsinki Declaration of the World Medical Association. Research protocols were approved by their respective local and federal committees.
Tissue Preparation and Immunostaining
Brains were fixed and processed by each brain bank using routine methods (Bennett et al., 2012a, 2012b; Grinberg et al., 2007), and paraffin-embedded sections were prepared from cortical tissue blocks. Stanford and Rush utilized 6 µm-thick midfrontal cortex sections, whereas Universidade de São Paulo utilized anatomically matched 5 µm-thick tissue sections from prefrontal cortex (Brodmann area 46).
Tissue from Stanford and Rush was immunostained for B- and T-lymphocyte markers in the Buckwalter Lab using the following antibodies: 1:500 CD20 monoclonal mouse anti-human Dako Clone L26 (Agilent Cat# M0755, RRID:AB_2282030); 1:100 CD3 monoclonal mouse anti-human Dako Clone F7238 (Abcam Cat# ab17143, RRID:AB_302587). Tissue from the Biobank for Aging Studies was immunostained using the following antibodies: CD20 monoclonal mouse anti-human, Agilent Dako L26 IR60461-2) (Agilent Cat# M0755, RRID:AB_2282030); CD3 polyclonal rabbit anti-human, Agilent Dako IR503 (Agilent Cat# IS50330-2, RRID:AB_2732001). Normal human tonsil tissue was used as a positive control stain for the pilot study; for the confirmatory study at Universidade de São Paulo, human skin tissue was used for positive control CD3 stain, and human lymph node was used as a positive control CD20 stain.
For the pilot study, tissue was de-paraffinized, rehydrated in stepped washes, boiled in citrate buffer (0.01 M sodium citrate, 0.05% Tween, pH = 6); permeabilized (0.3% H2O2, 0.5% Triton-X in 1x PBS), and blocked overnight at 4° (10% serum in 0.5% Triton-X in PBS). We incubated slides overnight with primary antibody (5% blocking serum in 0.5% Triton-X in PBS), and then with secondary antibody for 4 hours at 25° (1:200 in 5% serum in 1x PBS).
Samples from the Universidade de São Paulo Biobank for Aging Studies were de-paraffinized and rehydrated, and epitope retrieval was performed by placing slides in hot deionized water in PT Link (Agilent Dako) at 97 °C for 40 minutes, followed by four baths of buffer solution (EnVision Flex Wash Buffer 20x, Agilent Dako). Endogenous peroxidase was blocked with H2O2 (EnVision Flex Peroxidase-Blocking Reagent, Agilent Dako) for 10 minutes. Tissue was incubated for 35 minutes with CD20 (Monoclonal mouse anti-human, Clone L26, IR60461-2, Agilent Dako) and for 15 minutes with CD3 (Polyclonal rabbit anti-human ready to use, IR503, Agilent Dako), at room temperature. Secondary antibody was then applied for 20 minutes: EnVision Flex Mouse Link (Agilent Dako) for CD20, and EnVision Flex Rabbit Link (Agilent Dako) for CD3. Afterwards, slides were incubated for 25 minutes with polymer (EnVision Flex HRP system, Agilent Dako). The chromogen (EnVision Flex DAB + Chromogen diluted in EnVision Flex Substrate Buffer, Agilent Dako) was applied for 7 minutes. Finally, we washed the slides in running water for 2 minutes, bathed slides in four baths of ethanol and ten baths of xylene for 15 seconds each, and then coverslipped (Coversleeper, Agilent Dako).
Quantification and Analysis
The number of CD20+ and CD3+ cells were counted by a rater who was blinded to demographics. Tissue from the Stanford and Rush Pathology subjects was counted manually using a microscope under both 20x and 40x magnification. Tissue from the Biobank for Aging Studies was analyzed on scanned slide sections using Pannoramic Viewer software (Pannoramic Viewer, RRID:SCR_014424). Lymphocytes in these digital images were then visualized, also with Pannoramic Viewer software, and counted manually. We counted lymphocytes that met the following criteria; morphology resembling a lymphocyte with round or oval nucleus of at least 4 µm, smooth regular cell membrane borders, a brown hue consistent with a DAB stain, and present in perivascular, or parenchymal, or intravascular regions. In all cases, we counted lymphocytes throughout the entire tissue section with the exception of meninges when present. Lymphocytes found in meninges were excluded to avoid introducing bias given that not all tissue sections contained meninges. Total tissue area was quantified using ImageJ software on a whole slide image; meninges were again excluded. Mean tissue section area was 2.6 cm2 ± 0.85 for pilot test samples, and 2.91 cm2 ± 0.62 for Biobank for Aging Studies samples.
For analyses, lymphocytes were quantified as the number of cells per square centimeter (cells per tissue section/tissue section area (cm2)). Results from samples from Stanford and Rush were analyzed in a pilot study and for the power calculation only; they were excluded from analysis in the large confirmatory study, comprised only of samples from Universidade de São Paulo.
To ensure that the blind rater who counted the cells (KB) had strong intra-rater reliability, we randomly selected 10% of sections to count again after all 40 slides had been counted initially. Sufficient intra-rater reliability was set at 90% similarity between replicate counts. Average intra-rater reliability was determined to be 93% between the two counts and thus sufficient.
Statistical analyses were performed with GraphPad Prism 8 software (GraphPad Prism, RRID:SCR_002798). The means of B- and T-lymphocyte densities between groups were compared using a Kolmogorov-Smirnov test, to allow comparisons across normal and non-normal data distributions. A Spearman’s rank correlation coefficient was utilized to look for associations in non-normal data; Pearson’s was used for data passing normality testing. Grubbs analysis was used to identify statistical outliers.
Results
Pilot Study and Power Calculation
In the pilot study, comprised of tissue samples from Stanford and Rush, B-cell density in younger subjects (mean = 0.9 cells/cm2 ± 1.5.) versus older subjects (mean = 3.0 cells/cm2 ± 2.6) showed a trend (p = 0.066) indicating that B-cells might increase with age. T-lymphocyte density in younger subjects (mean = 58.7 cells/cm2 ± 43.9) did not differ significantly from the T-cell density in older subjects (mean = 45.1 cells/cm2 ± 45.0; p = 0.679). Neither B- nor T-lymphocyte densities varied between males and females (p = 0.622 and 0.660, respectively). We hypothesized that B-lymphocyte density increases in the brain with age, and determined that it would take a sample size of 19 to detect a 1 cell/cm difference or more B-cell densities between the age groups with 80% power.
Subject and Tissue Characteristics
The confirmatory study was comprised of tissue samples from Universidade de São Paulo only. Of the 1,092 brains available from Universidade de São Paulo’s Biobank for Aging Studies collection, roughly 40% fulfilled criteria for a neuropathological diagnosis. Of the remaining 60% that met inclusion criteria for “normal” tissue, we selected 40 subjects for our study. Subjects’ ages ranged from 44–96 (Table 1). Sex ratio did not differ between older and younger brain groups; each contained 13 male and 7 female subjects. There was a small but statistically significant difference between length of post-mortem interval in the younger (13.7 ± 0.6 hr) vs. older (11.6 hr ± 0.8 hr) groups (unpaired t-test, p = 0.048).
Lymphocyte Densities
CD20 staining did not demonstrate a significant difference in average B-cell density: young subjects’ mean = 7.8 cells/cm2 ± 1.4; old subjects’ mean = 6.2 cells/cm2 ± 1.0; p = 0.875 (Figure 1A). Density of CD20 cells demonstrated one statistical outlier that was excluded from further analyses (Subject: male, age 48 years; B lymphocyte density: 40.1 cells/cm2). CD3 staining also did not demonstrate a significant difference in average T-cell densities with age: young subjects’ mean = 145.6cm2 ±13.9; old subjects’=142.3 cells/cm2 ± 15.8; p = 0.560 (Figure 1B). There was no association between post-mortem interval and either B- or T-lymphocyte density (B cells, Spearman r = −0.230, p = 0.158; T cells, Spearman r = −0.0917, p = 0.574). We also found no difference in lymphocyte densities by sex for either B- or T-cells (p = 0.993 and p = 0.660, respectively).
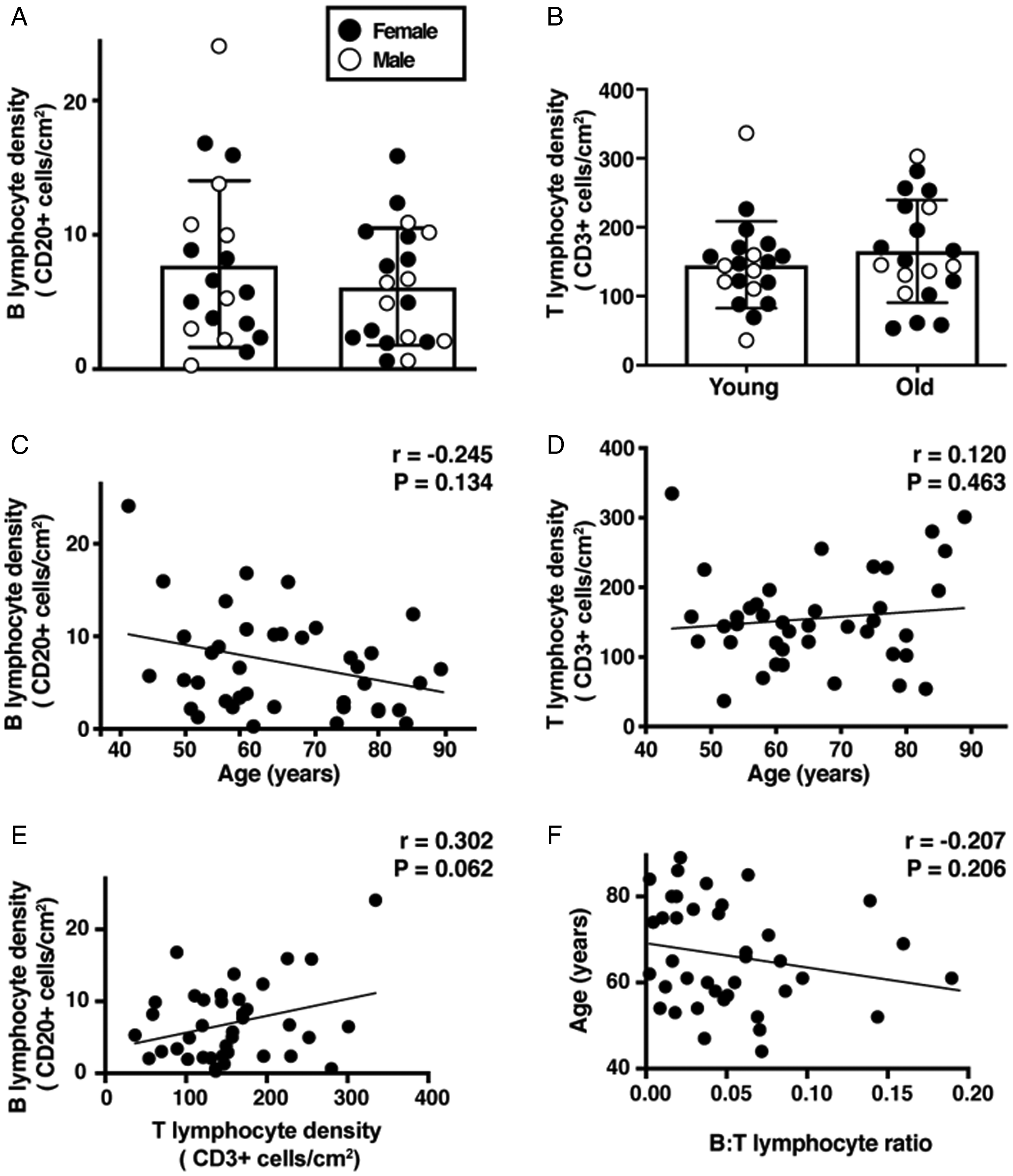
Density of B- and T-Lymphocytes Did Not Change With Age. There was no significant difference in B- and T-cell densities with age in human cortical tissue obtained from Universidade de São Paulo, by Student’s t test or linear regression. A: CD20+ B-lymphocytes per cm2 in young (44–62 years, n = 19) and old (65–89 years, n = 20) subjects: Young subjects’ mean = 7.8 cells/cm2 ± 1.4; old subjects’ mean = 6.2 cells/cm2 ± 1.0; p = 0.875. B: CD3+ T-lymphocytes per cm2 in young (44–62 years, n = 20) and old (65–89 years, n = 20) subjects: young subjects’ mean = 145.6cm2 ±13.9; old subjects’=142.3 cells/cm2 ± 15.8; p = 0.560. C: Spearman linear regression of CD20+ B-lymphocyte density with age. D: Pearson linear regression of CD3+ T-lymphocyte density with age. Bars, mean +/− SEM. E: Linear regression of CD20+ B-lymphocyte density with CD3+ T-lymphocyte density. F: Linear regression of B:T-lymphocyte (CD20+:CD3+) ratio with age.
Finally, we assessed the continuous relationship between age and B- or T-cell density and found no significant association between age and lymphocyte density in linear analysis (Figure 1C and D). Likewise, while there was evidence of a possible trend toward a weak correlation between B- and T-cell densities (r = 0.302, p = 0.062, Figure 1E), there was no evidence of a statistically significant association between B/T cell ratio and age (r = −0.207, p = 0.206, Figure 1F).
Lymphocyte Location
We approximated the location of each positively stained cell in relation to other lymphocytes and to vasculature (Figure 2). B-cells were most often present as individual cells, whereas T-cells typically clustered in clumps of 3–20 cells in the perivascular and intravascular spaces. On average, B-cells localized as follows: 41% parenchymal, 32% perivascular, 27% intravascular. In contrast, the averages for T-cells were 20% parenchymal, 67% perivascular and 14% intravascular. We also found that intravascular lymphocyte densities correlated with parenchymal and perivascular densities. (B-cells, Spearman r = 0.669, p < 0.0001; T-cells, Spearman r = 0.34, p = 0.015).
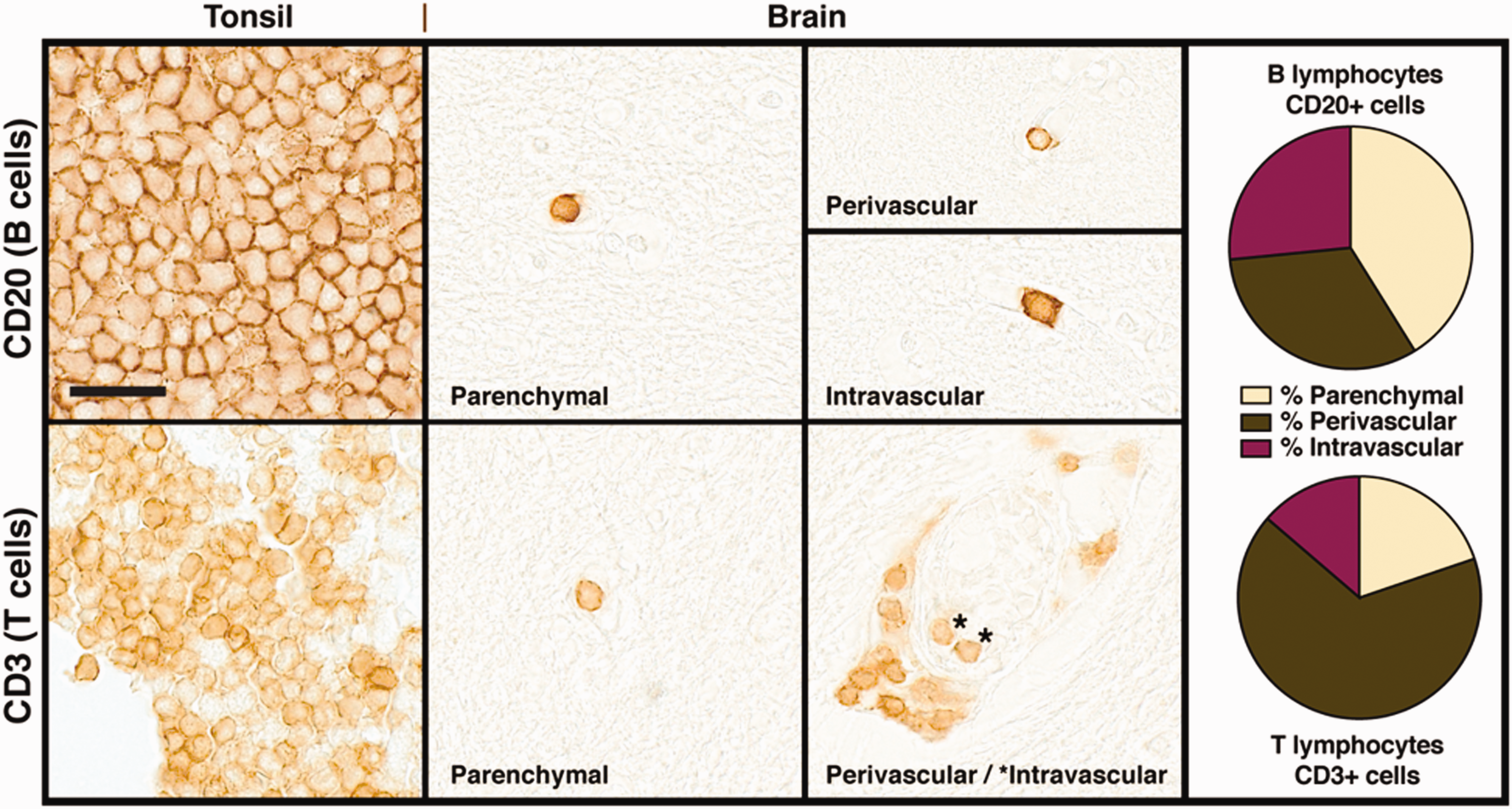
Lymphocyte Distribution Within the Brain. Representative photomicrographs of immunostaining for CD20 (B-lymphocytes, top) and CD3 (T-lymphocytes, bottom) in normal human tonsil (left), and within the brain parenchyma, perivascular and intravascular spaces. On the right are pie charts of the proportion of lymphocytes assigned to each of the three brain locations, representing pooled data from all subjects. All pictures taken at 400x magnification; Scale bar, 20 µm; *intravascular T-cells.
None of our findings changed in statistical significance when we excluded intravascular cells from our analysis, when we considered only parenchymal cells, when we re-included the statistical outlier in B-cell analysis, or when we used parametric statistics (unpaired t-tests).
Discussion
We examined lymphocyte densities in the normal human brain across age. We found no increase in B- or T-lymphocyte densities or distribution with age in the normal human brain. These results are surprising given the robust existing evidence that brain-innate immune cells (microglia and astrocytes) become increasingly activated with age, that activated microglia produce cytokines and chemokines that have been shown to attract lymphocytes to the brain in disease states, and that T-lymphocytes increase in the mouse brain with age (Dulken et al., 2019; Koellhoffer et al., 2017; Mrdjen et al., 2018; Palmer & Ousman, 2018; Wong, 2013)). It may be that such innate activation has no effect on brain-resident lymphocyte numbers, or that it is seen only in disease (Schneider et al., 2009, 2007). Moreover, since our inclusion criteria were designed to identify brains in a normal state, but disease is quite common in the aging brain, our sample may actually represent a picture of “successful” rather than normal aging. Further work is necessary to determine if this is the case.
In our study we utilized a rigorous, systematic selection of aged human tissue, and analyzed a total of 56 subjects (16 subjects in our pilot study, and 40 subjects from the Biobank for Aging Studies). This makes it the largest study of its kind, 4 times larger than previous studies of human lymphocyte counts. There was little prior data on the number of B- and T-cells in the healthy human brain, or how that number changes with age. A handful of studies on neuropathological diseases include data on lymphocyte counts in small numbers of control human brains (Anthony et al., 2003; Busse et al., 2012; Doyle et al., 2015; Machado-Santos et al., 2018; Merlini et al., 2018; Togo et al., 2002) (Table 2). Only two of these previous papers quantified B-cells in the normal human brain (Anthony et al., 2003; Doyle et al., 2015). Several others reported B- and T-cell densities, demonstrating them to be lower than lymphocyte densities in brains of people with various neurological, psychiatric, and neurodegenerative diseases, but without reporting quantification of controls. All sampled fewer than 11 subjects from an age range spanning no more than two decades, had variable inclusion criteria for defining “normal” brains, or reported the presence of lymphocytes using semi-quantitative or qualitative methods only. The differences in reported lymphocyte densities in previously published studies may be due to differences in protocols such as tissue section thickness, total area of tissue analyzed, fixation and antibody staining, and/or quantification methods. Considering differences in section thickness, these prior studies may be consistent with our results.
No previous studies have assessed B-lymphocytes in the aging human brain. Machado-Santos et al. (2018) reported that B-cell counts decline with both age and duration of illness in the brains of people with multiple sclerosis. However, they did not use controls in their examination of age changes (Machado-Santos et al., 2018). and so this may be a disease-specific finding, possibly related to decreased immune activation with aging (Nikolich-Žugich, 2018). Our study demonstrates a non-significant trend towards lower B-lymphocyte density with age, so it is possible that with more subjects we would see a modest decrease in brain density of B-cells. However, because the trend is largely driven by one young subject, we think it is more likely that B-cell density does not decline in healthy brains.
To our knowledge, Dulken et al. (2019) is the only study to date to have investigated T-cell numbers in the human brain with age (Dulken et al., 2019). Note that Gate et al. (2020) reported T-cell numbers in the brains of participants age-matched to those with Alzheimer’s, but they did not directly assess age as a factor (Gate et al., 2020). Dulken et al. (2019) compared T-cell numbers in older and younger subjects, and reported an increase in the peri-ventricular region, an area known to be neurogenic—one of the few known regions where new neurons are generated in the adult brain. In addition to this regional limitation, their study was also limited by small sample size. Given that the numbers of lymphocytes they found in both young and old subjects were much higher than densities reported in other human brain lymphocyte studies (Table 2), these intriguing findings may reflect a feature of T-lymphocyte density in this particular neurogenic brain region rather than more generalized global brain aging.
Our study is strong not merely because of its large sample size and rigorous criteria for defining a “normal” human brain, but also because we included a wide age range of adult subjects, aged 44–96 in the pilot experiment, and then aged 44–89 years in our study. In particular, identifying normal subjects of young ages is challenging. Not all brain banks have large numbers of subjects younger than 70, let alone those with normal cognition and an absence of neuropathological changes. We utilized material from the Biobank for Aging Studies, which has several advantages. In São Paulo, Brazil, an autopsy is mandatory in cases where 1) the death is deemed to be from natural causes, and 2) where the individual either did not have medical assistance at the time of death or when the specific natural cause is otherwise unclear. The Autopsy Service is the largest morgue serving the metropolitan area of São Paulo and performs roughly 14,000 autopsies per year. In addition to providing the brain tissue itself, the donor’s next-of-kin provide socio-demographic information that includes the donor’s years of education, medical history, and lifestyle factors such as physical activity and socioeconomic status. Age, gender and race of the subjects were confirmed using the deceased’s official death certificate.
We also classified lymphocytes according to their location within three compartments: intravascular, perivascular, and intraparenchymal. There was a striking similarity between these distributions in all cohorts and ages, but with marked differences between B-cells and T-cells. B-lymphocytes were typically found as isolated cells, while T-lymphocytes were usually clumped. Furthermore, B-cells were evenly distributed throughout these three locations, while T-cells were largely perivascular. These differences in location and density raise mechanistic questions about the differences between B- and T-cell proliferation and/or trafficking within the brain. It is also interesting that the intravascular counts of both cell types correlated with cell numbers in the perivascular space and in the brain. It may be that the activation state of brain endothelial cells is related to both, as we know that both decline with age in the peripheral blood (Ademokun et al., 2010; Frasca et al., 2011). However, future studies with specific labeling of blood vessels, and/or tracking of lymphocytes, are required to answer these questions definitively.
There are limitations to this study. First, our pilot study had a definition of “normal” that allowed for inclusion of those with mild to moderate hypertensive vasculopathy; an inability to match by exact region beyond “frontal cortex”; and an inability to exclude the possibility that methodological differences between sites might have obscured a true association between age and lymphocyte density (all younger subjects came from Stanford University while older subjects came from Rush University).
We largely overcame these limitations with rigorous anatomic matching from a single study site with uniform methodology for our confirmatory study (Universidade de São Paulo biobank), which we report in detail here. A limitation remains that our study could not address whether lymphocyte densities vary with age in regions outside the cortex, or between gray and white mater. However, two prior studies found no difference in B-cell density across many regions, and no differences between gray and white cortical matter (Anthony et al., 2003; Bø et al., 2003), suggesting it is plausible that our findings, which are from mixed cortical white and gray matter, may generalize to other brain regions. Also, we did not counterstain for vasculature to precisely define the location of lymphocytes but rather examined background tissue density to determine vessel locations. However, our statistical analyses did not change when we included vs. excluded intravascular cells, there was no change in lymphocyte density with age with any combination of compartments included. Finally, we note that lymphocyte density is not a proxy for lymphocyte function, nor do the markers CD20 and CD3 capture the phenotypic diversity of B and T cells that may be present. Future work is needed to investigate whether there might be age-related shifts in the sub-types of B and T cells present in the cortex, even while overall lymphocyte densities remain stable.
We did observe different lymphocyte densities in the cohorts from Stanford, Rsh and the cohort from Universidade de São Paulo. Methodological differences may explain the discrepancy. For example, tissue from the Stanford and Rush cohorts was stained using different antibody clones, and analyzed using a live microscope, while Universidade de São Paulo’s cohort tissue was counted using images from a digital microscope. Biological or environmental differences between the cohorts, such as genetic factors, microbiome differences, diet, and exposure to pollutants or even parasites (Landrith et al., 2015), could influence lymphocyte densities.
In conclusion, in this large immunohistochemical study of human cortical tissue, we found that there was no difference in density or location of B- and T-lymphocytes in the cortex in normal adult individuals with age. Future studies are needed to understand how neurodegenerative diseases and other diseases of the aged influence B- and T-lymphocytes within the brain.
Summary Statement
In this immunohistochemical study of 56 human samples, we found no evidence that B- and T-cell densities change with age in normal cortex, suggesting that any lymphocyte density changes in aged human cortex may suggest disease and not normal aging.
Footnotes
Acknowledgments
We thank the participants who volunteered to donate their brains and/or their family members’ brains for research, and also AE Robinson for cogent manuscript edits.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by UCSF Summer Explore Fellowship (KCB); American Heart Association/Allen Initiative in Brain Health and Cognitive Impairment [9PABHI34580007] (MSB); Leducq Foundation Transatlantic Network of Excellence [Stroke-IMPaCT] (MSB); National Institute of Health [K24Ag053435] (LTG); São Paulo Research Foundation [FAPESP 2017/24066-1] (DSF-I); and the National Institute of Aging [R01AG017917] (JAS).
