Abstract
Protein aggregation and accumulation are common pathological hallmarks in neurodegenerative diseases. To efficiently clear and eliminate such aggregation becomes an important cellular strategy for cell survival. Lewy bodies inclusion and aggregation of α-Synuclein (α-Syn) during the pathogenesis of Parkinson’s disease (PD) serve as a good example and are potentially linked to other pathological PD features such as progressive dopaminergic neuron cell death, behavioral defects, and nonmotor symptoms like anosmia, cognitive impairment, and depression. Years of research have revealed a variety of mechanisms underlying α-Syn aggregation, clearance, and spread. Particularly, vesicular routes associated with the trafficking of α-Syn, leading to its aggregation and accumulation, have been shown to play vital roles in PD pathogenesis. How α-Syn proteins propagate among cells in a prion-like manner, either from or to neurons and glia, via means of uptake or secretion, are questions under active investigation and have been of central interest in the field of PD study. This review covers components and pathways of possible vesicular routes involved in α-Syn trafficking. Events including but not limited to exocytosis and endocytosis will be discussed within the context of an overall cellular trafficking theme. Recent advances on α-Syn trafficking mechanisms and their significance in mediating PD pathogenesis will be thoroughly reviewed, ending with a discussion on the advantages and limitations of different animal PD models.
Introduction
History of Parkinson’s disease (PD) began with the very first description appeared in a monograph titled An Essay on the Shaking Palsy around 200 years ago, by the English surgeon James Parkinson (Parkinson, 2002). At the time, it was merely clinical observations on patients described to exhibit involuntary tremulous motion and lessened muscular power, with shaking of the limbs in particular. Over the past decades, efforts bridging translational and basic research greatly expand our knowledge on the cause, development, and the overall process of this neurological disorder. These discoveries elaborate on the initial clinical observations and give us a better understanding on the neuropathological underpinnings of this disease. Despite so, a complete picture of the disease progression and issues on finding a probable cure have not been fully resolved.
As the most common movement disorder in the world, PD is clinically characterized with symptoms such as resting tremor, bradykinesia, postural instability, accompanying nonmotor symptoms like cognitive impairment and autonomic dysfunction. In the brain, a series of neuropathological changes appears throughout the course of PD development, ultimately leading to the diagnosis hallmark: the aggregation of intracellular inclusions named Lewy bodies (LBs) and Lewy neurites (LNs), and the loss of dopaminergic (DA) neurons in the substantia nigra pars compacta (SNpc). This progressive brain pathology can be staged by the LB appearance in different regions of the brain (Braak et al., 2003; Braak et al., 2006), with initial detection of LB in the periphery such as dorsal motor nucleus of the glossopharyngeal and vagal nerves or the olfactory bulb (Lewy, 1912), followed by the appearance in the SNpc DA neurons in mid-stage, finally other parts of the brain. Interestingly, this topographic spread of LB pathology correlates with the progression of PD clinical symptoms (Braak et al., 2006), raising the interest to search for spread pattern of LB pathology and correlative relationship leading to PD progression.
Whereas most of the PD cases are sporadic, studies on rare familial cases identified possible genetic causes for PD. The central component for LB and LN, α-Synuclein (α-Syn), is the gene product from SNCA (PARK1), the first PARK gene identified in the studies of rare familial PD cases. α-Syn contains an amphipathic N-terminal region, followed by a non-Aβ component (NAC) of AD plaques domain responsible for aggregation, and a C-terminal acidic tail (Figure 1). Physiologically, α-Syn is a 140 amino acid protein enriched in presynaptic nerve terminals (Iwai et al., 1995). As a vesicular protein, α-Syn was shown to directly interact with VAMP2 and promotes SNARE complex assembly (Burre et al., 2010) and interacts with CSPα (Chandra et al., 2005; Ninkina et al., 2012). Significant reduction in excitatory synapse size and synaptic function were detected in mice lacking all three synuclein homologues (α, β, and γ) (Burre et al., 2010; Greten-Harrison et al., 2010). These findings first implicate a role for α-Syn in synaptic vesicle trafficking.
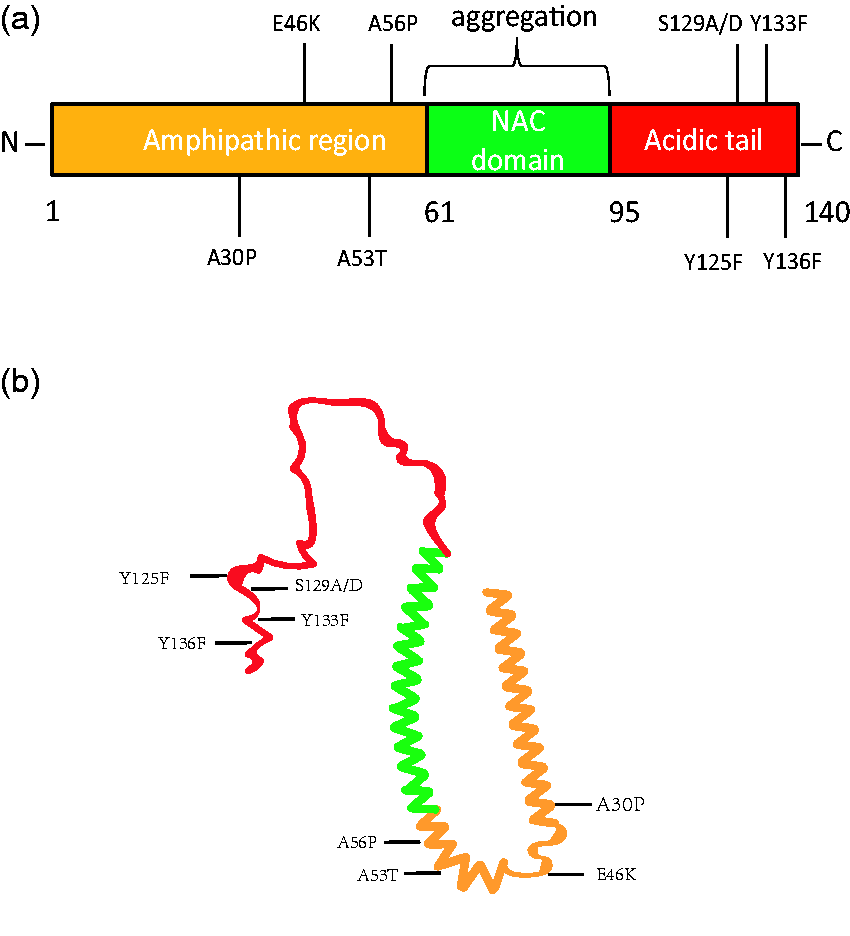
α-Syn protein structure. α-Syn protein containing the N-terminal amphipathic region (yellow), the NAC domain (green), and the C-terminal acidic tail (red) were illustrated in a linear diagram (a) or a representative monomeric structure (b). Note that the locations of point mutations listed in Table 1 were labeled. NAC = non-Aβ component.
Not only genome-wide association (GWAS) studies have identified SNCA SNPs as risk variants for sporadic PD (Satake et al., 2009; Simon-Sanchez et al., 2009), the missense SNCA mutation A53T (Polymeropoulos et al., 1997), along with many others and duplications in the SNCA locus, have all been shown to associate with PD (Kruger et al., 1998; Singleton et al., 2003; Chartier-Harlin et al., 2004; Zarranz et al., 2004; Appel-Cresswell et al., 2013; Lesage et al., 2013; Pasanen et al., 2014). Interestingly, PD-associated mutations of α-Syn confer differential self-aggregation properties. The A53T, H50Q, and E46K mutations tend to increase the rate of α-Syn self-aggregation (Conway et al., 1998; Greenbaum et al., 2005; Ghosh et al., 2013), whereas the A30P, G51D, and A53E mutations decrease the rate (Choi et al., 2004; Fares et al., 2014; Ghosh et al., 2014). These findings implicate that mutant α-Syn with altered propensities are potentially toxic and more prone for aggregation in disease conditions.
As the central component for LB and LN, the properties of α-Syn assembling into different forms, particularly the fibrils and aggregates, have raised substantial interest in the PD research community. Physiologically, α-Syn forms helically folded tetramers that resist aggregation (Bartels et al., 2011; Wang et al., 2011). In pathological conditions, however, oligomeric α-Syn species and the fibrillary aggregates have been shown to be the toxic culprits for PD pathology (Cremades et al., 2012; Bengoa-Vergniory et al., 2017). Moreover, α-Syn assembles into distinct strains displaying differential seeding capacities and strain-specific pathological consequences (Bousset et al., 2013; Peelaerts et al., 2015). On the other hand, α-Syn aggregation was also detected in LB of sporadic PD patients (Spillantini et al., 1997, 1998b), suggesting that abnormal clearance and degradation of α-Syn might occur. These findings altogether reinforce α-Syn as a necessary component in LB pathology and PD, and both its intrinsic aggregation propensities and extrinsic regulation on its protein levels matter.
Clues on the intrinsic assembling properties of α-Syn and the conversion of α-Syn from a normal to toxic form (oligomers, fibrils, ribbons, etc.) that seed α-Syn aggregation have uncovered similarity between α-Syn and the prion protein, leading to the proposed prion-like propagation mode for α-Syn spread in PD pathology. In this mode, converted toxic α-Syn has a higher tendency to aggregate and propagate as part of the LB components throughout different areas of the brain (Kordower et al., 2008; Li et al., 2008; Desplats et al., 2009; H. J. Lee et al., 2010; Hansen et al., 2011; Kordower et al., 2011; Angot et al., 2012; Luk et al., 2012; Mougenot et al., 2012; Masuda-Suzukake et al., 2013). At the cellular level, the idea of spread comes from two significant observations: (a) α-Syn has been detected extracellularly, suggesting that it is secreted by cells in a mobile way (Ueda et al., 1993; Jakes et al., 1994). Years later, the presence of α-Syn at nanomolar concentration in the cerebrospinal fluid and plasma of PD patients was described, further supporting the existence of extracellular α-Syn (Borghi et al., 2000; El-Agnaf et al., 2003). (b) α-Syn filamentous deposits in oligodendrocytes, the glial cytoplasmic inclusions, are prominent in the atypical Parkinsonism multiple system atrophy (Spillantini et al., 1998a, 1998b; Tu et al., 1998; Watts et al., 2013; Reyes et al., 2014; Prusiner et al., 2015). Due to the dominant expression of α-Syn in neurons, these glial α-Syn aggregates are considered exogenous and potentially delivered from nearby neurons. Thus, knowing how α-Syn propagates among different cells and their means of transfer is crucial for understanding the mechanism in the spread of LB pathology and PD progression. Echoing the Braak model, α-Syn spread from neurons to neurons have been suggested to act in a way of trans-synaptic release mediated by Hsp70 and the cochaperone DnaJ (Danzer et al., 2011; Fontaine et al., 2016). The modes and mechanisms of transmission from neurons to glia or vice versa, however, are far less clear and await further investigation. Despite sparse hints from the literature, mechanisms of α-Syn intercellular propagation, their routes of secretion and uptake operated by different cell types, and their differential dictation in Parkinson-related pathology are all within areas of interest and remain to be further explored.
This review provides a comprehensive overview on the vesicular mechanisms of α-Syn trafficking during LB pathology and PD progression. Insights from fly and mouse PD models will be covered, and specific cell-type mechanisms for α-Syn secretion and uptake will be discussed and compared. α-Syn trafficking, rather than its clearance and degradation, is emphasized as the latter has been a subject for a number of excellent reviews recently (Kinghorn et al., 2017; Pitcairn et al., 2018). Of note, impaired trafficking and defects in other trafficking components, in addition to α-Syn, also play significant roles in PD pathology.
Transgenic α-Syn overexpression models
Traditional approaches that model PD use toxins such as 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine to kill DA neurons and recapitulate PD symptomatic phenotypes so that potential drug targets are identified to ameliorate these symptoms. These toxin-based models, which have been used both in flies and mice, are usually acute, rapid, and with a drawback that does not model the molecular pathology of PD. Virus vector-based rat or primate models are also available to manipulate α-Syn expression. On the other hand, transgenic PD models built by our knowledge on potential genetic PD risk factors recapitulate the disease lesions and exhibit more strength in understanding the underlying molecular mechanism of PD. Most of the mouse transgenic models overexpress the human wild-type or mutant α-Syn, to model different dosage of SNCA gene expression. Nonetheless, only a few of these rodent models recapitulate the cardinal features of PD. Lack of neurodegeneration, α-Syn aggregation, or locomotor deficits suggests that these mice do not perfectly model the PD pathology, raising the concern that additional animal models might be needed to investigate the basic pathogenic mechanisms.
The diverse behaviors and sophisticated genetic tools have made Drosophila melanogaster a powerful in vivo system to examine PD pathology. Fly α-Syn transgenic models are mainly based on overexpression of human wild-type or mutant α-Syn due to the lack of endogenous fly α-Syn. Such strategy implements elevated α-Syn levels in the system and mimics the increased SNCA gene expression in the PD patients carrying α-Syn mutation or duplication/triplication, making the transgenic flies well suited for modeling PD. Recently, a number of reviews have summarized different types of PD animal models (Vanhauwaert and Verstreken, 2015; Hewitt and Whitworth, 2017; Koprich et al., 2017; Xiong and Yu, 2018). Here, we selectively discuss fly and mouse transgenic models based on α-Syn overexpression, with an attempt to lay the groundwork for subsequent discussion on α-Syn vesicular mechanisms in PD (Table 1).
Fly and mouse α-Synuclein transgenic models.
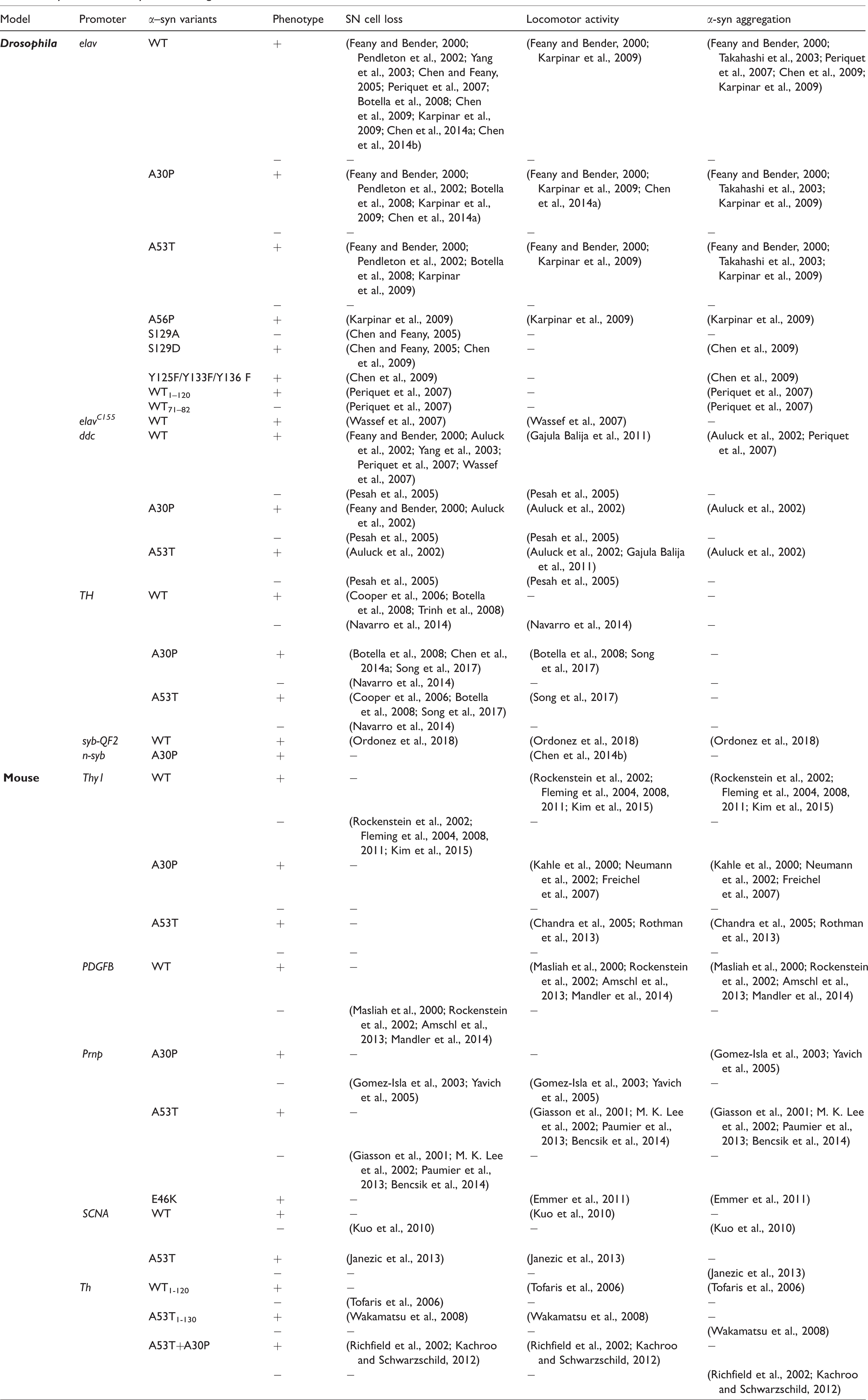
Most of the transgenic mouse models were established based on expression of different α-Syn forms under a variety of promoters. Human wild-type α-Syn, pathogenic α-SynA53T, and α-SynA30P (hα-Syn, hα-SynA53T, and hα-SynA30P) are frequently used for overexpression, and in some cases mouse α-Syn (Chandra et al., 2005; Rieker et al., 2011) or C-terminal truncated aSyn (Tofaris et al., 2006; Wakamatsu et al., 2008; Daher et al., 2009) are also used. Mice overexpressing hα-Syn under control of the human platelet-derived growth factor subunit B (PDGFB) promoter exhibit progressive development of α-Syn and ubiquitin inclusions starting as early as 3 months, with signs of DA neuron loss and locomotor defects (Masliah et al., 2000; Rockenstein et al., 2002; Amschl et al., 2013). On the other hand, mice overexpressing hα-Syn under the mouse thymus cell antigen 1 (Thy1) promoter show early and progressive sensorimotor anomalies and olfactory deficits (Fleming et al., 2004, 2008). Some degree of motor deficits and α-Syn aggregation were also detected in mice overexpressing hα-SynA53T or hα-SynA30P under the Thy1 (Kahle et al., 2000; Neumann et al., 2002; Freichel et al., 2007; Rothman et al., 2013) or the gene encoding prion protein (prnp) promoter (Giasson et al., 2001; M. K. Lee et al., 2002; Gispert et al., 2003; Gomez-Isla et al., 2003; Yavich et al., 2005; Paumier et al., 2013; Bencsik et al., 2014). Interestingly, mice overexpressing hα-Syn under the tyrosine hydroxylase (Th) or SNCA promoter tend to lack α-Syn inclusions and other neurodegenerative features (Richfield et al., 2002; Tofaris et al., 2006; Wakamatsu et al., 2008; Kuo et al., 2010; Janezic et al., 2013). Moreover, α-Syn aggregation sometimes is absent in some of the models accompanying motor and degenerative deficits (Matsuoka et al., 2001; Gispert et al., 2003; Yavich et al., 2005), suggesting that α-Syn aggregation does not necessarily correlate with the occurrence of other PD symptomatic phenotypes as the Braak model proposed. Taken together, these observations suggest a greater variation in phenotypes recapitulated by different mouse models, leading to a pressing need for establishing other animal models to help with further investigation.
Studies in flies provide significant insights and advance our understanding on the basic pathogenic mechanism of PD. Fly α-Syn models were first established when hα-Syn and its related pathogenic forms were overexpressed in all or specific DA neurons (Feany and Bender, 2000). The authors first demonstrated that hα-Syn overexpression leads to proteinaceous inclusions, locomotor deficits, and DA neuron degeneration in fly adult brains in an age-dependent manner. Subsequently, a series of studies revealed the importance of chaperone HSP70, proteasome-dependent degradation, and phosphorylation on regulating α-Syn toxicity and aggregation (Auluck et al., 2002; Chen and Feany, 2005; Chen et al., 2009; F. K. Lee et al., 2009). Taking advantage of the fly PD models, hα-Syn-overexpressing flies were also treated with pharmaceutical agents to search for potential therapeutic drugs that ameliorate PD symptoms (Auluck and Bonini, 2002; Pendleton et al., 2002; Auluck et al., 2005). Genetically, interacting factors of autophagy, lysosome, and ubiquitin-mediated degradation were also identified to cooperatively work with hα-Syn during PD pathology (Davies et al., 2014; Miura et al., 2014; Alexopoulou et al., 2016; M’Angale and Staveley, 2016).
Fly and mouse models have distinct advantages and limitations in elucidating the mechanism of PD. Experimentally, flies grow faster and have a greater sampling number, whereas mice are recognized for their longer life span, and the hassle in handling them has limited the potential to acquire more data in supporting hypothesis of PD. Anatomically, mice have higher similarity in brain structure and PD-affected regions to human, whereas Drosophila adult brains have conserved DA neuron clusters that also undergo degeneration. Motor activity is much easier to access for flies than mice, whereas cognitive symptoms associated with PD are harder to evaluate in flies, and easier to assess in mice due to the knowledge in circuits controlling these behaviors.
Findings from flies and mice, however, do not favor one or another in modeling PD. Interestingly, analyses of most mouse PD models do not recapitulate the cardinal features of PD, whereas transgenic flies modeling PD, despite its invertebrate origin, consistently show signs in some or all PD-associated features such as DA neurodegeneration, loss of motor activity and life span, and α-Syn aggregation. Some degree of consistency has been detected in observing DA neuron loss or motor deficits in these transgenic fly models. However, discrepancies arise over the strength of α-Syn-induced phenotypes. It is reported that different experimental approaches in mounting the fly brains might lead to different results on hα-Syn-mediated DA neuron loss; these findings are thoroughly discussed elsewhere (Pesah et al., 2005; Whitworth et al., 2006; Navarro et al., 2014). Complementary approaches utilizing these two animal models will probably strengthen and gain greater insights in PD pathology. For example, to look for genetic modifiers of PD, one could easily take on a high-throughput screening approach using flies. Once a target gene is identified, its function can be tested in transgenic mouse models so that more physiological-relevant evidence is acquired.
α -Syn in trafficking: Interaction with Rab proteins
Accumulating evidence has suggested that impairment in vesicular trafficking, such as the blockade by PD-associated mutations in lysosomal genes ATP13A2 (Ramirez et al., 2006) or glucocerebrosidase (GBA) (Sidransky et al., 2009; Schondorf et al., 2014), is a possible cause for PD (Abeliovich and Gitler, 2016). Defects in these trafficking pathways often couple with α-Syn accumulation and aggregation, suggesting that these routes are major means for α-Syn transmission (Gitler et al., 2009; Mazzulli et al., 2011). To investigate further, genetic and biochemical screens have uncovered functional interaction between α-Syn and the central trafficking proteins small GTPase Rabs (Dalfo et al., 2004; Cooper et al., 2006), and α-Syn was also shown to disrupt cellular Rab homeostasis (Gitler et al., 2008). These Rab proteins are relevant to α-Syn-induced defects in trafficking and regulate α-Syn aggregation/toxicity (Gao et al., 2018). For instance, Rab1, Rab7, Rab8, and Rab11 have all been shown to ameliorate α-Syn toxicity in Drosophila (Cooper et al., 2006; Yin et al., 2014; Breda et al., 2015; Dinter et al., 2016). Rab1, a regulator of ER-to-Golgi trafficking, rescues α-Syn-mediated DA neuron loss, compromised autophagy (Cooper et al., 2006; Winslow et al., 2010), and Golgi fragmentation upon α-Syn overexpression in nigral DA neurons (Coune et al., 2011; Rendon et al., 2013). Rab11, the endosomal recycling factor, has been implicated in α-Syn release and secretion (Liu et al., 2009; Hasegawa et al., 2011). In addition to colocalize with α-Syn in intracellular inclusions (Chutna et al., 2014), Rab11 modulates synaptic vesicle size, decreases α-Syn aggregation, and ameliorates several α-Syn-dependent phenotypes in Drosophila (Breda et al., 2015). Taken together, these findings suggest that α-Syn functions in vesicular trafficking via interaction with various Rab proteins, and at the same time serves as a possible target for trafficking via these pathways (Figure 1(a)).
α -Syn in trafficking: Uptake mechanisms by neurons
α-Syn has been found to localize within endosomes and aggregate preferentially in endocytic vesicles (H. J. Lee et al., 2005; Konno et al., 2012; Boassa et al., 2013), supporting the notion that α-Syn is trafficked in vesicles and its aggregation is intimately associated with these trafficking pathways. Nonetheless, how α-Syn is trafficked to and from different subcellular compartments or between cells remains largely elusive. Whereas α-Syn monomers pass directly through the plasma membrane (H. J. Lee et al., 2008b), internalization of oligomeric and fibrillary α-Syn species depends on endocytosis, as treatment with canonical endocytosis inhibitors consistently reduces α-Syn uptake (Desplats et al., 2009; Hansen et al., 2011). A number of studies have shown that α-Syn is internalized by neurons and glial cells via clathrin-mediated endocytosis (CME) (H. J. Lee et al., 2008a, 2008b; Desplats et al., 2009; Konno et al., 2012). Factors involved in CME, such as Rab5, have also been described to mediate α-Syn internalization, leading to subsequent intracellular LB inclusions (Sung et al., 2001). Recently, the homolog of a GWAS risk factor Cyclin-G-associated Kinase (GAK), auxilin, was first identified in flies to regulate life span, locomotor activity, and progressive DA neuron death (Song et al., 2017). Reducing auxilin expression in the presence of hα-Syn overexpression causes premature and enhanced DA neuron loss, suggesting that GAK/auxilin, a clathrin-uncoating factor, might either directly regulate α-Syn endocytosis or participate in other ways to regulate trafficking of α-Syn. Furthermore, lymphocyte activating gene-3 (LAG-3), a leukocyte immunoglobulin protein expressed exclusively on neuronal surface, was found to facilitate the entry of fibrillar α-Syn via CME (Mao et al., 2016). Taken together, these findings strengthen the importance of CME in α-Syn uptake and provide insights on how α-Syn is trafficked to form LB inclusions in PD.
Despite the fact that CME is currently considered to be the major route for α-Syn uptake in neurons and glia, inhibition of this process does not completely block α-Syn uptake (H. J. Lee et al., 2008b; Desplats et al., 2009; Konno et al., 2012), suggesting that alternative routes exist. Interestingly, neurons also utilize a specialized form of endocytosis, macropinocytosis, to uptake α-Syn. Macropinocytosis-mediated actin ruffles mediate α-Syn entry, and this process requires heparan sulfate proteoglycans expressing on the cell surface (Holmes et al., 2013). Overall, these findings indicate that multiple types of mechanisms exist for α-Syn entry into neurons. Novel endocytic sites or receptors await to be identified to allow for a better understanding of the uptake mechanism, and these players serve as good candidates for therapeutic interventions to selectively block the uptake during disease progression (Figure 1(a)).
α -Syn in trafficking: Uptake mechanism by glia
Similar to neurons, CME is also the major route for glia to uptake α-Syn (H. J. Lee et al., 2008a; Kisos et al., 2012; Valdinocci et al., 2017). Glial α-Syn inclusions such as the oligodendrocyte glial cytoplasmic inclusions in multiple system atrophy patients, together with the glial neuroinflammatory responses triggered by α-Syn (H. J. Lee et al., 2010; Hoffmann et al., 2016; Lim et al., 2016), implicate the significance of studying α-Syn trafficking into glial cells. Besides CME, different types of glial cells utilize distinct routes for uptake. Human astrocytes alternatively transfer aggregated α-Syn to healthy astrocytes via tunneling nanotubes (TNTs) to remove pathological inclusions instead of degrading them by lysosomes (Rostami et al., 2017). Astrocytic TNTs do so by utilizing F-actin-based thin protrusions to establish connections with neighboring cells for intercellular exchange and communication (Gousset et al., 2009; Agnati and Fuxe, 2014; Abounit et al., 2016). Furthermore, TNTs are not restricted to facilitating transport between cells of the same type, as they are detected in both neurons and astrocytes (Rustom et al., 2004; Zhu et al., 2005; Sun et al., 2012), thereby serving as an alternative means for α-Syn transmission.
Microglia commonly utilize the specialized form of endocytosis, phagocytosis, to uptake α-Syn (H. J. Lee et al., 2008a; Bliederhaeuser et al., 2016). Some of the receptors functioning in microglial phagocytosis and activation, like the Toll-like receptors (TLRs) TLR2 and TLR4, have been implicated in α-Syn uptake and α-Syn-mediated activation (Fellner et al., 2013; Kim et al., 2013; Valdinocci et al., 2017). Moreover, microglia have been observed in vitro to uptake α-Syn-containing exosomes released by oligodendrocytes via macropinocytosis (Fitzner et al., 2011). In addition to phagocytosis and macropinocytosis, other clathrin-independent routes such as monosialoganglioside (GM1)-dependent lipid rafts have also been shown to mediate microglial uptake of α-Syn (Park et al., 2009). Reduced expression of DJ-1, another PD risk factor, reduces cell surface lipid raft expression in microglia and impairs their ability to uptake soluble α-Syn (Nash et al., 2017). It is worth mentioning that lipid rafts also mediate α-Syn localization at the synapses (Fortin et al., 2004; Kubo et al., 2005), supporting the notion that lipid rafts participate in the trans-synaptic release and neuronal propagation of α-Syn. Thus, lipid rafts function in both neurons and glia to mediate α-Syn entry. Notably, little is known about microglia using TNTs to transfer or uptake α-Syn despite that macrophages, the functional equivalent of microglia in the immune system, use TNTs to mediate phagocytic clearance of foreign materials (Onfelt et al., 2004; Onfelt et al., 2006; Figure 1(b)).
α -Syn in trafficking: Release mechanism
α-Syn has been detected extracellularly, suggesting its release from cells. In addition to leakage from cell death or apoptosis, other types of secretory mechanism come into play such as cargo release in the form of exosomes. Exosomes are 50 to 100 nm vesicles that facilitate intercellular exchange by transporting specific proteins or RNAs (Valadi et al., 2007; Gibbings et al., 2009). Interestingly, exosomes have been utilized as one means of cargo transfer between neurons and glia. Oligodendrocytes release neuroprotective exosomes to support neuronal metabolism (Fitzner et al., 2011; Fruhbeis et al., 2013a, 2013b), whereas astrocytes and neurons facilitate bidirectional transfer of mitochondria via exosomes to support neuronal homeostasis (Davis et al., 2014; Hayakawa et al., 2016). Moreover, microglia were shown to release inflammatory factors in exosomes. Taken together, these findings indicated that exosomes as a type of important secretory organelle for intercellular communication.
It has been shown that exosomes obtained from the plasma of PD patients display higher levels of α-Syn than those from control groups (Schneider and Simons, 2013; Shi et al., 2014). Not only exosomal lipids enhance the aggregation propensity of α-Syn (Grey et al., 2015), oligomeric α-Syn stored in the exosomes are also released by neurons and preferentially taken up via endocytosis than α-Syn fibrils (Danzer et al., 2012). Furthermore, increased calcium levels and dysfunction of autophagy lead to increased release of exosomes containing α-Syn oligomers (Emmanouilidou et al., 2010; Alvarez-Erviti et al., 2011), suggesting that exosomal release of α-Syn is highly regulated. Most of the studies describe exosomal release of α-Syn from neurons, as neuronal release of α-Syn-containing exosomes are often targets for glial uptake, serving a more efficient means of transfer than free α-Syn (Chistiakov and Chistiakov, 2017). Limited evidence, however, has been provided for glial exosomal release of α-Syn.
In addition to exosomes, other unconventional ways for neurons to release α-Syn have been described. For instance, a portion of newly synthesized α-Syn is rapidly secreted from cells via endoplasmic reticulum/Golgi-independent exocytosis in both normal and stress-induced conditions (H. J. Lee et al., 2005; Jang et al., 2010). By this route, intravesicular aggregated α-Syn prone to localize in vesicles are secreted out of cells (H. J. Lee et al., 2005; Jang et al., 2010). Furthermore, tubulin polymerization-promoting protein (TPPP/p25α), a factor that mediates autophagosome-lysosome fusion, is involved in the unconventional secretion of α-Syn through exophagy. The same study also identified Rab27A, a late endosome regulator for exocytosis, that regulates α-Syn secretion in the absence of TPPP/p25α (Ejlerskov et al., 2013). Finally, as discussed, Rab11 regulates α-Syn resecretion in a recycling pathway that differ from classical ER/Golgi-dependent exocytosis (Liu et al., 2009; Hasegawa et al., 2011). In toto, these findings suggest that there is great diversity in the mechanism of α-Syn release, and most of the routes for α-Syn release have been studied more extensively in neurons than in glia (Figure 2).
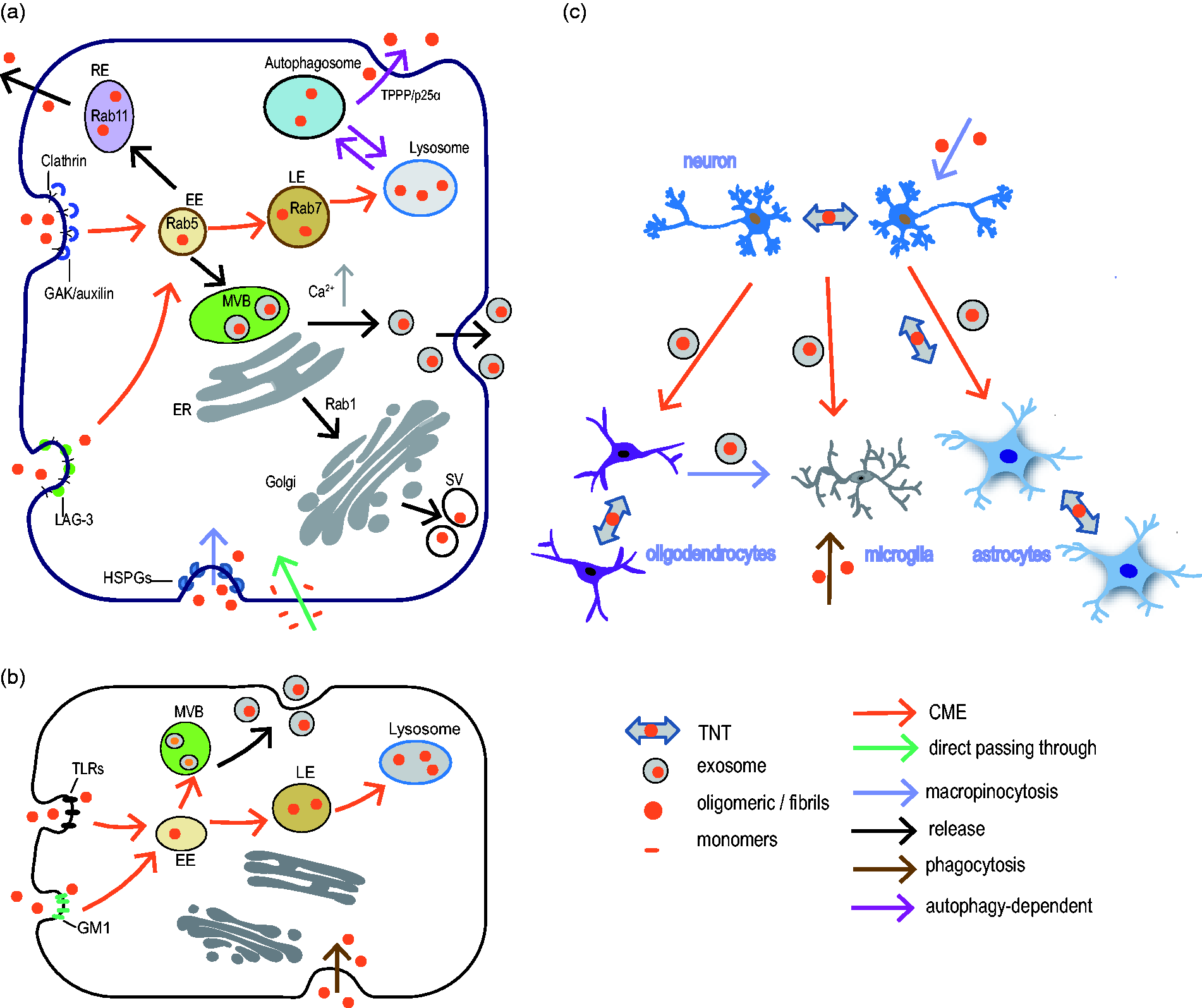
α-Syn trafficking mechanisms in PD. Oligomeric and/or fibrillary α-Syn aggregates are trafficked intracellularly in vesicles and interact with a number of Rab proteins for their function. (a) α-Syn trafficking in neurons: whereas α-Syn monomers pass directly through the plasma membrane, oligomeric or fibrillary α-Syn uptake is mediated by CME and heparan sulfate proteoglycans-dependent macropinocytosis. LAG-3 is implicated as the receptor for uptake. Genetic evidence from Drosophila also suggests that the clathrin-uncoating factor GAK/auxilin is a potential mediator for α-Syn uptake. On the other hand, α-Syn is released from neurons via exosomes, a process regulated by intracellular calcium levels and autophagy. Other unconventional ways of α-Syn release includes ER/Golgi-independent exocytosis, TPPP/p25α-dependent exophagy, and Rab11-mediated resecretion. (b) α-Syn trafficking in glia: α-Syn uptake is potentially mediated by TLRs-dependent phagocytosis and GM1-dependent lipid rafts in microglia, whereas TNTs are one of the major means for α-Syn transfer between astrocytes. α-Syn release via exosomes has been observed in oligodendrocytes. and (c) An overview on α-Syn trafficking among neurons, astrocytes, microglia, and oligodendrocytes. Routes discussed earlier that depend on TNTs, exosomes, or forms of endocytosis/exocytosis are designated. Note that other routes and mechanisms might be involved. TNT = tunneling nanotube; TLR = Toll-like receptor; CME = clathrin-mediated endocytosis.
α -Syn trafficking and degradation
Accumulating evidence has suggested that α-Syn trafficking is closely linked to its aggregation. As discussed earlier, α-Syn interacts with various Rab proteins and regulate their homeostasis. Altered expression of these trafficking genes such as Rab8b, Rab11a, Rab13, and Slp5 also regulates α-Syn secretion and aggregation (Goncalves and Outeiro, 2017). These results suggest that α-Syn secretion might be the most crucial step related to its aggregation as its secretion constitutes the key step for propagation. Thus, an agent targeting α-Syn trafficking, particularly its secretion, might have therapeutic potential by exerting an effect by either facilitating or preventing its aggregation.
Studies from flies and mice have reached a conclusion that silencing the expression of these trafficking genes increases α-Syn aggregation, and their overexpression reduces α-Syn aggregation, thus relieving the induced toxicity in cellular models (Liu et al., 2009; Hasegawa et al., 2011; Yin et al., 2014). These trafficking proteins have also been detected in the α-Syn inclusions, suggesting that α-Syn is being recruited together with the trafficking proteins to the inclusion site. These findings suggest that α-Syn trafficking regulates its aggregation and the clearance of aggregation is favored for toxicity to be ameliorated. Recently, the emerging idea that facilitating protein aggregation might help to relieve the toxicity by stopping protein trafficking and retain the toxicity in local has posed a new and interesting perspective. It is possible that facilitating α-Syn aggregation might allow the construction of a potentially more concentrative milieu for the degradation machineries to proceed (autophagy and proteasomal degradation), increasing the efficiency of α-Syn degradation and disappearance. In this case, α-Syn trafficking might be less favorable. Furthermore, a blocking in the trafficking route might help to accumulate local α-Syn aggregates. However, studies from flies and mice do not support a positive role for facilitating α-Syn aggregation. Toxicity is ameliorated only when trafficking genes are overexpressed and aggregations are reduced, suggesting an overall reduction in α-Syn aggregation is needed for lowering the induced toxicity.
Another plausible explanation for how facilitating α-Syn aggregation might help with the toxicity is that large cytoplasmic inclusions/aggregations are formed by reducing the toxic oligomeric α-Syn species. It has been shown that oligomeric α-Syn species are the most toxic culprits for pathogenic spread and enhance neurotoxicity in Drosophila (Karpinar et al., 2009). Moreover, histone deacetylase 6 (HDAC6) has been shown to promote the formation of large α-Syn inclusions by reducing α-Syn oligomers. In this means, HDAC6 suppresses α-Syn-induced DA neuron loss and locomotor dysfunction in flies (Du et al., 2010). Based on these findings, it is speculated that large cytoplasmic inclusions are formed by reducing α-Syn oligomers, thus reducing toxicity. To this end, these results support the idea that facilitating α-Syn aggregation (i.e., forming large cytoplasmic inclusions) might help to relieve the toxicity, as the toxic oligomeric α-Syn species are reduced by doing so.
α -Syn equilibrium in trafficking
Previous studies have indicated that α-Syn propagates in all forms, via different routes. It has been shown that monomeric α-Syn passes through the membrane directly, whereas oligomeric and fibrillary α-Syn enter and leave cells in a variety of ways. These different routes include CME, lipid raft-dependent macropinocytosis, exosomal release, and classical and nonclassical exocytosis. These diverse routes render α-Syn mobile and diverge into different paths. Different modulators along the way also exert differential effects in regulating the trafficking. Interestingly, no discrete evidence suggested a preference for cells to take up or release a specific form of α-Syn, despite the fact that oligomeric and fibrillary forms are more pathogenic. Therefore, the equilibrium among these different α-Syn forms will probably become significant when it comes to which of the available routes is used for their uptake or release, but not to the choice of what kind of α-Syn is being trafficked. Nonetheless, there are still factors that regulate the α-Syn equilibrium worth to be considered for possible effects on α-Syn trafficking. For instance, it has been shown that α-Syn oligomerization within specific cellular compartments alters the distribution of functional forms of monomeric α-Syn, sequestering the monomer into nonfunctional oligomeric forms (Colla et al., 2012). This piece of evidence suggests that the distribution of functional/nonfunctional monomeric/oligomeric α-Syn might be a factor regulating its own trafficking. On the other hand, α-Syn trafficking is possibly modulated via its structural changes caused by mutation. For example, mutation of α-Syn N-terminal region like A30P and A53T might alter its secondary structure and affect α-Syn trafficking via an increase in the shuttling between the cytoplasm and nucleus (Emamzadeh, 2016) or an alteration in the rate of self-aggregation.
Concluding Remarks
Therapeutic solutions that help to ameliorate symptoms of PD have mainly focused on strategies targeting the elimination and clearance of α-Syn aggregation, a process closely linked to α-Syn trafficking. To understand how α-Syn is trafficked, subsequently the temporal and spatial control of α-Syn aggregation, is therefore an important issue to be addressed. This review summarizes recent updates on the routes and mechanisms of α-Syn trafficking, discusses specific cell-type mechanisms in these routes, and how contributions from fly and mouse models differ or complement each other (Figure 2). These insights help with the investigation on discovering new means of α-Syn transfer and refining the known ones by identifying new players involved. Drug candidates designed by targeting components in these pathways will potentially be helpful in alleviating functional symptoms, at the same time advance our understanding of this mysterious disease over centuries.
Footnotes
Acknowledgments
My sincere apologies to colleagues in the field whose work I was not able to mention because of space limitations. We would like to thank all members of the M.H. Laboratory for comments and suggestions on the manuscript.
Author Contributions
J. C., Q. L., and L. S. wrote the manuscript. J. C. designed the illustration in Figure 1. Q. L. created ![]() . All authors read and approved the manuscript.
. All authors read and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M. S. H., J. C., and Q. L. are all supported by the National Natural Science Foundation of China and the National Basic Research Program of China. Work in the M.H. Laboratory is supported by grants from the National Natural Science Foundation of China (31871039) and ShanghaiTech University.
