Abstract
Introduction
Early mobilisation protocols after repair of extensor tendons in zone V and VI provide better outcomes than immobilisation protocols. This systematic review investigated different early active mobilisation protocols used after extensor tendon repair in zone V and VI. The purpose was to determine whether any one early active mobilisation protocol provides superior results.
Methods
An extensive literature search was conducted to identify articles investigating the outcomes of early active mobilisation protocols after extensor tendon repair in zone V and VI. Databases searched were AMED, Embase, Medline, Cochrane and CINAHL. Studies were included if they involved participants with extensor tendon repairs in zone V and VI in digits 2–5 and described a post-operative rehabilitation protocol which allowed early active metacarpophalangeal joint extension. Study designs included were randomised controlled trials, observational studies, cohort studies and case series. The Structured Effectiveness Quality Evaluation Scale was used to evaluate the methodological quality of the included studies.
Results
Twelve articles met the inclusion criteria. Two types of early active mobilisation protocols were identified: controlled active motion protocols and relative motion extension splinting protocols. Articles describing relative motion extension splinting protocols were more recent but of lower methodological quality than those describing controlled active motion protocols. Participants treated with controlled active motion and relative motion extension splinting protocols had similar range of motion outcomes, but those in relative motion extension splinting groups returned to work earlier.
Discussion
The evidence reviewed suggested that relative motion extension splinting protocols may allow an earlier return to function than controlled active motion protocols without a greater risk of complication.
Keywords
Introduction
Recent systematic reviews have found strong evidence that early mobilisation after hand/wrist extensor tendon repair provided better range of motion (ROM) outcomes compared to immobilisation protocols.1–4 Early mobilisation needs to be in a controlled manner to optimise the benefits of mobilisation while avoiding the risks related to unrestricted motion.5–7
The requirement to balance motion with protection has led to the development of early controlled mobilisation protocols where, during the early post-operative period, motion of the injured digit is allowed while being controlled by a splint.3,7–11 Early mobilisation protocols for extensor tendon repairs in zones V and VI include those allowing early passive mobilisation (EPM) and those allowing early active mobilisation (EAM) of the repaired tendon.3,4,12
Three systematic reviews on extensor tendon repair2–4 concluded that there was insufficient evidence to determine whether EAM or EPM protocols provided superior outcomes. However, recent systematic reviews on this subject have recommended EAM protocols over EPM protocols1,4,12 because they provide similar outcomes and the low-profile static splints (usually employed in EAM protocols) are said to be cheaper, quicker to make, and easier for the patient and therapist to manage.1,4,12–15 Furthermore, EAM protocols may have lower complication rates. 12
Various EAM protocols have been described for the management of extensor tendon repairs in zone V and VI with differences in splint design and exercise programmes. However, no published trials have investigated the relative benefits of different types of EAM protocols for extensor tendon repairs in zone V and VI, to determine whether any one provides superior outcomes to any other. Although previous systematic reviews have reviewed EPM and EAM protocols,1–4,12 they have not attempted to specifically examine EAM protocols to identify an optimal approach. Therefore, the objective of this systematic review was to investigate the different types of EAM protocols used after extensor tendon repairs in zone V and VI and to determine whether any EAM protocol provided superior outcomes. This review was structured according to the PRISMA guidelines. 16
Methods
Search strategy
A search strategy was constructed using keywords and search terms related to EAM protocols for extensor tendon repair. These keywords and search terms were expanded through the use of truncation. The search terms used were: ‘extensor tendon injur$’ OR ‘extensor tendon repair$’ OR ‘extensor tenorrhaphy’ AND ‘early motion’ OR ‘relative motion’ OR ‘active motion’ OR ‘splint$’ OR ‘orthos$’ OR ‘rehabilitation.’
Utilising the search strategy, a comprehensive search was conducted using the following databases: AMED (Allied and Complementary Medicine) via Ovid, Embase via Ovid, Medline (R) In- Process & Other Non-Indexed Citations, Medline (R) Daily and Medline (R) via Ovid, Cochrane via Wiley, Cochrane via Ovid and CINAHL. Date limitations were set depending on the relative limitation of the database up to search completion on 5 June 2017.
Inclusion and exclusion criteria for articles for review.

Data extraction
A data extraction form was used with the following data extracted from each study by the primary author (SC): author and date of publication or presentation; type of study; inclusion criteria; zones of injury; intervention groups; sample size; baseline characteristics of participants; results including timing of mobilisation, joint range of motion (ROM), grip strength, time to return to work (RTW), complications, subjective outcomes and amount of hand therapy input.
Assessment of methodological quality
MacDermid’s Evaluation Guidelines for Rating the Quality of an Intervention Study 17 was used to assess the quality of the included studies. This tool, also known as the SEQES (Structured Effectiveness Quality Evaluation Scale) has been used widely in the assessment of hand therapy and musculoskeletal literature18–22 and has been shown to have high inter-rater reliability with regard to scoring of studies. 20 The SEQES was designed to evaluate a variety of intervention study designs including randomised controlled trials (RCTs), cohort and retrospective studies 17 and it provides a numerical score that allows comparison of research quality across included studies.
The SEQES tool consists of 24 items divided into seven sections: Study question (item 1), Study design (items 2–8), Subjects (items 9–12), Intervention (items 13–15), Outcomes (items 16–18), Analysis (items 19–23) and Recommendations (item 24). Items were scored 2 if completely fulfilled, 1 if partially fulfilled and 0 if not fulfilled or not addressed at all. The maximum score obtainable was 48, and the minimum was 0. 17
The SEQES was not provided with a classification from the scoring to attribute to methodological quality. 17 From several studies that have used the SEQES, it is apparent that the classification for quantitative interpretation of scores has varied. For example, previous studies reporting the SEQES tool have classified articles as being of ‘low’ quality if they scored 1–1621 or 1–20, 22 ‘moderate’ if scores were between 17 and 32 21 or 21 and 34 22 and ‘high’ if they were between 33 and 48 21 or 35 and 48. 22 In light of these discrepancies, a decision was made for this systematic review that studies scoring 0–20 were regarded as being of ‘low’, 21–32 as ‘moderate’ and 33–48 as ‘high’ methodological quality. Scoring was carried out independently by two authors (SC and EK). Recommendations for multiple reviewers provided in the original description of the tool were applied. 17 Differences in scoring were discussed; consensus was obtained to within one point of difference in all cases. In the small number of cases where one point of difference remained, the lower score was assigned.
Levels of evidence
The level of evidence of the current systematic review was considered following the evaluation of the included studies. The level of evidence was derived from The Oxford Centre for Evidence-based Medicine 2009 Levels of Evidence 1. 23
Results
A total of 166 articles were identified through the database search; an additional five articles were identified through review of reference lists of included articles. After inclusion and exclusion criteria had been applied, 12 full text articles were selected for the review (Figure 1). A meta-analysis of the results was not possible due to heterogeneity for EAM protocol design and outcomes used.
PRISMA flow diagram.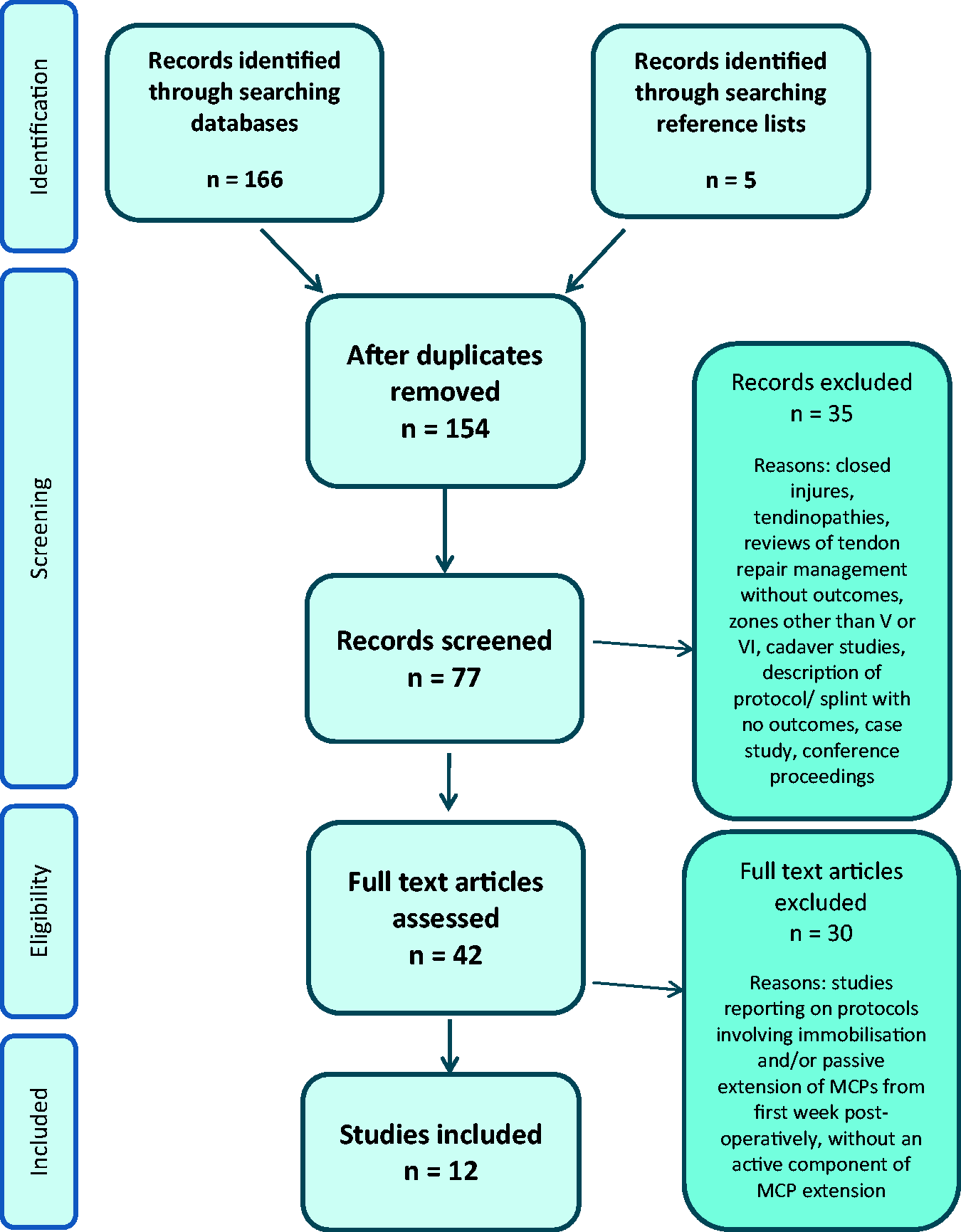
Characteristics of included studies
Study characteristics.
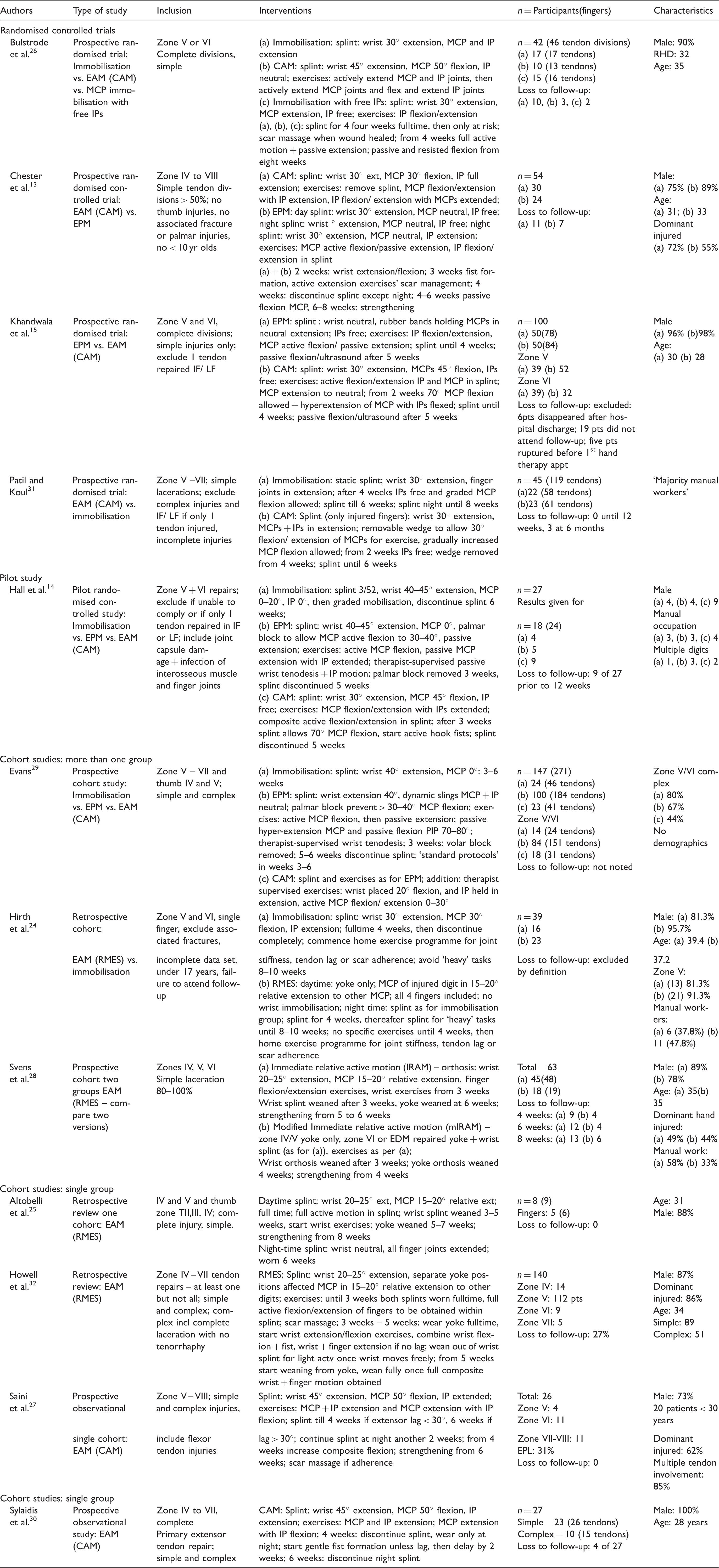
actv: Activities; CAM: controlled active mobilisation protocol; EAM: early active mobilisation protocol; EPL: extensor pollicus longus; EPM: early passive mobilisation protocol; excl: exclude; IF: index finger; incl: include; IP: interphalangeal joint; LF: little finger; MCP: metacarpophalangeal joint; n: number; RMES: relative motion extension splinting protocol.
Study outcomes.
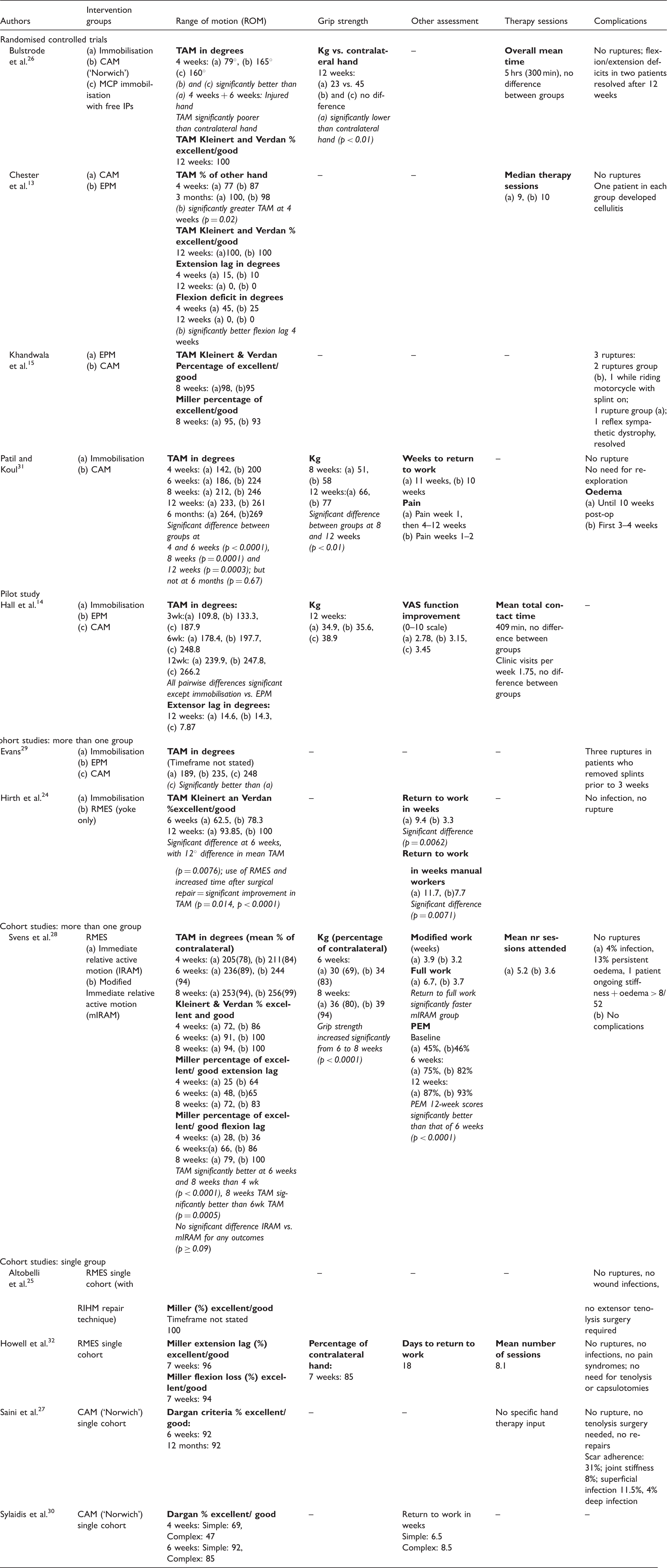
CAM: controlled active motion protocol; EPM: early passive motion protocol; hrs: hours; IPs:interphalangeal joints; kg = kilograms; MCP: metacarpophalangeal joint; min: minutes; PEM: patient evaluation measure; RIHM: running interlocking horizontal mattress technique; RMES: relative motion extension splinting; TAM: total active motion; VAS: visual analogue scale.
Although all protocols used in the studies were classified as EAM, on closer review these could be divided into two groups: ‘controlled active motion’ (CAM) protocols13–15,26,27,29–31 and ‘relative motion extension splinting’ (RMES) protocols.24,25,28,32 None of the included studies directly compared the outcomes of participants treated with CAM and RMES protocols.
The most important difference between the different EAM protocols was the more restrictive splint design used in the CAM protocols. The CAM protocols made use of a forearm-based splint which included the wrist and all the injured MCP joints, preventing full MCP joint flexion.13–15,26,27,29–31 In contrast, the RMES protocols used a small ‘yoke’ splint which included only the MCP joints of the injured digit(s), in relatively more extension than the other digits; the uninjured digits were left free, allowing functional use.24,25,28,32 This difference between the two types of protocols was even greater in some instances where RMES protocols left the wrist free24,28 and some CAM protocols additionally included the interphalangeal (IP) joints.13,27,29–31
Furthermore, participants treated with CAM protocols were advised to commence light activities at four or six weeks after daytime splinting was discontinued,13–15,31 with return to work (RTW) at 6–10 weeks13,14 and full heavy duties from 12 weeks.13,15 In contrast, participants treated with RMES protocols were encouraged to commence light functional activities immediately from the time of splint application, and were advised to return to heavier tasks earlier. RMES participants were advised to RTW on light duties within the first three weeks 28 and return to heavy tasks whilst wearing the splint by three weeks. 32
Generally those studies reporting RMES protocols were more contemporary with none published before 2005, in contrast to the CAM studies where half of the studies were published prior to 2005.13,15,29,30
Outcome measures reported
Range of motion outcomes for included studies.
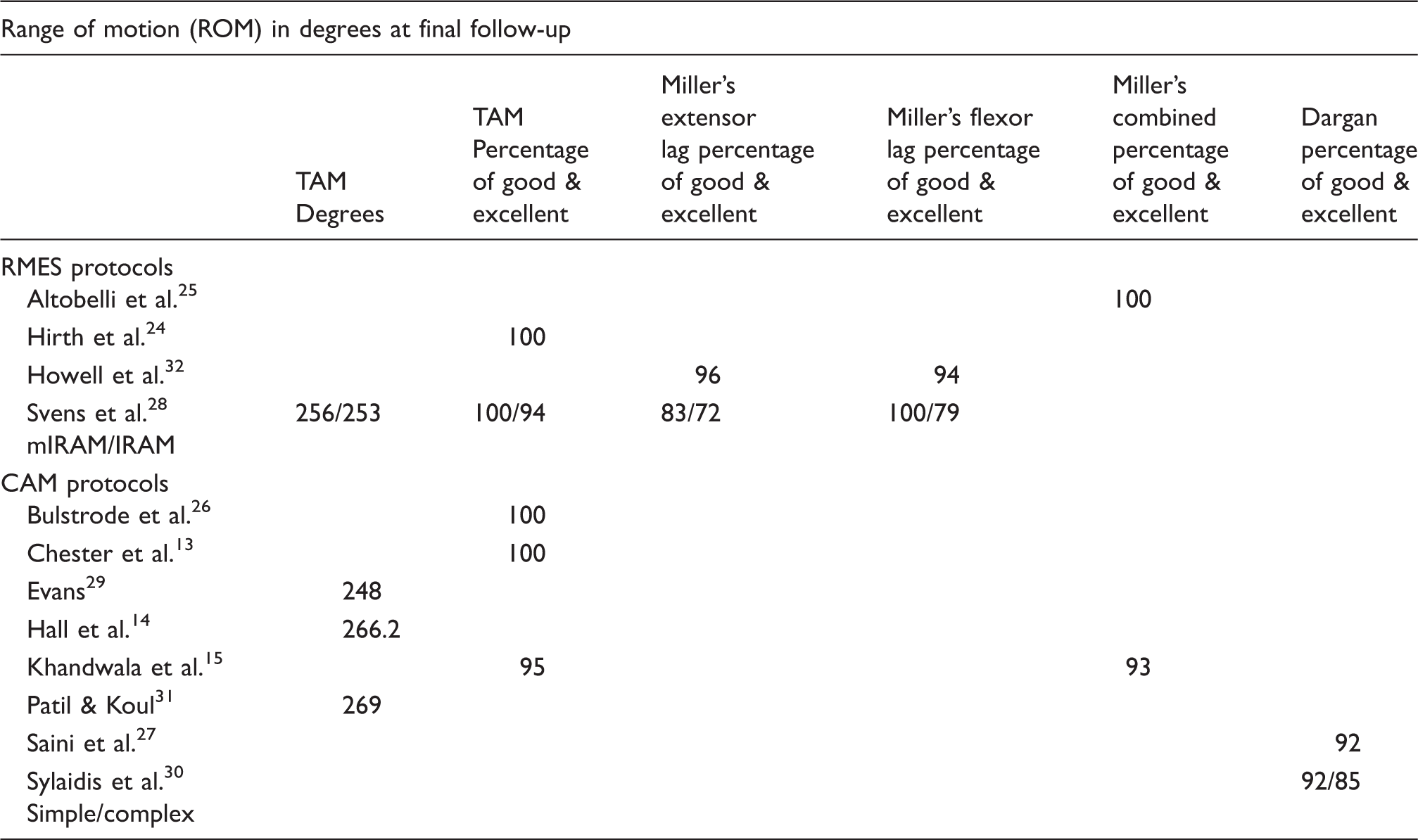
IRAM: immediate relative active motion; mIRAM: modified immediate relative active motion.
Grip strength was the second-equal most frequently reported outcome measure. Mean grip strength at final follow-up was similar for participants treated with a CAM protocol compared to those treated with an RMES protocol. For example, mean grip strength (dynamometer) was 38.9 kg for one CAM group14 and 36–39 kg for RMES groups. 28
The time taken for participants to RTW was the second-equal most frequently measured outcome. Participants treated with CAM protocols had a RTW between 6.5 and 10 weeks30,31 while those treated with RMES protocols had a RTW between 2.6 and 6.7 weeks.24,28,32
Subjective outcomes were infrequently reported. In a study reporting on outcomes of RMES protocols, Svens et al. 28 employed a validated subjective outcome measure, the ‘hand health’ section of the Patient Evaluation Measure (PEM), 33 where mean scores at 12 weeks were from 87% to 93% (100% indicates no problems with hand health). Hall et al. 14 used a non-validated visual analogue scale to report on perceived function in a study which included a CAM group. In another study which included a CAM group, Patil and Koul 31 assessed pain subjectively using a numeric analogue scale. Interestingly, none of the included studies recorded participant adherence, although lack of adherence was recognised as a potential issue for patients who undergo extensor tendon repair in zone V and VI.15,24
Hand therapy intervention was reported as number of sessions or in total therapy time in five of the included studies.13,14,26,28,32 Where hand therapy intervention was described, therapy time ranged from 300 min 26 to 409 min 14 in studies including CAM protocols; the number of hand therapy sessions was 9 in a CAM group13 and 3.6 to 8.1 in two studies of RMES groups.28,32 One study which included a CAM group27 reported that no hand therapy input was required, although their rehabilitation plan involved the use of a plaster splint and exercises.
Complications
Tendon rupture is a potential risk of early motion protocols, however eight of the included studies13,24–28,31,32 reported that there were no ruptures in their populations. Two ruptures occurred in the CAM group in the study by Khandwala et al. 15 Evans 29 reported on three ruptures but did not specify in which group they occurred; all three occurred in participants who removed their splints prior to three weeks. No ruptures were reported in any RMES groups. Two studies14,30 did not report on whether their participants had any ruptures.
Six of the twelve included studies13,24,25,27,28,32 reported on infection rate. Infection rates ranged from 3% to 11.5% in two studies that included CAM groups,13,27 0% in three studies that included RMES groups24,25,32 and 4% in another RMES study. 28 One study that included a CAM group27 and two studies that included an RMES group25,32 reported on the need for tenolysis; no participants in these three studies required tenolysis. One study 15 reported the development of reflex sympathetic dystrophy in one participant.
Methodological quality of included studies
Quality assessment of included articles using the Structured Effectiveness Quality Evaluation Scale (SEQES).
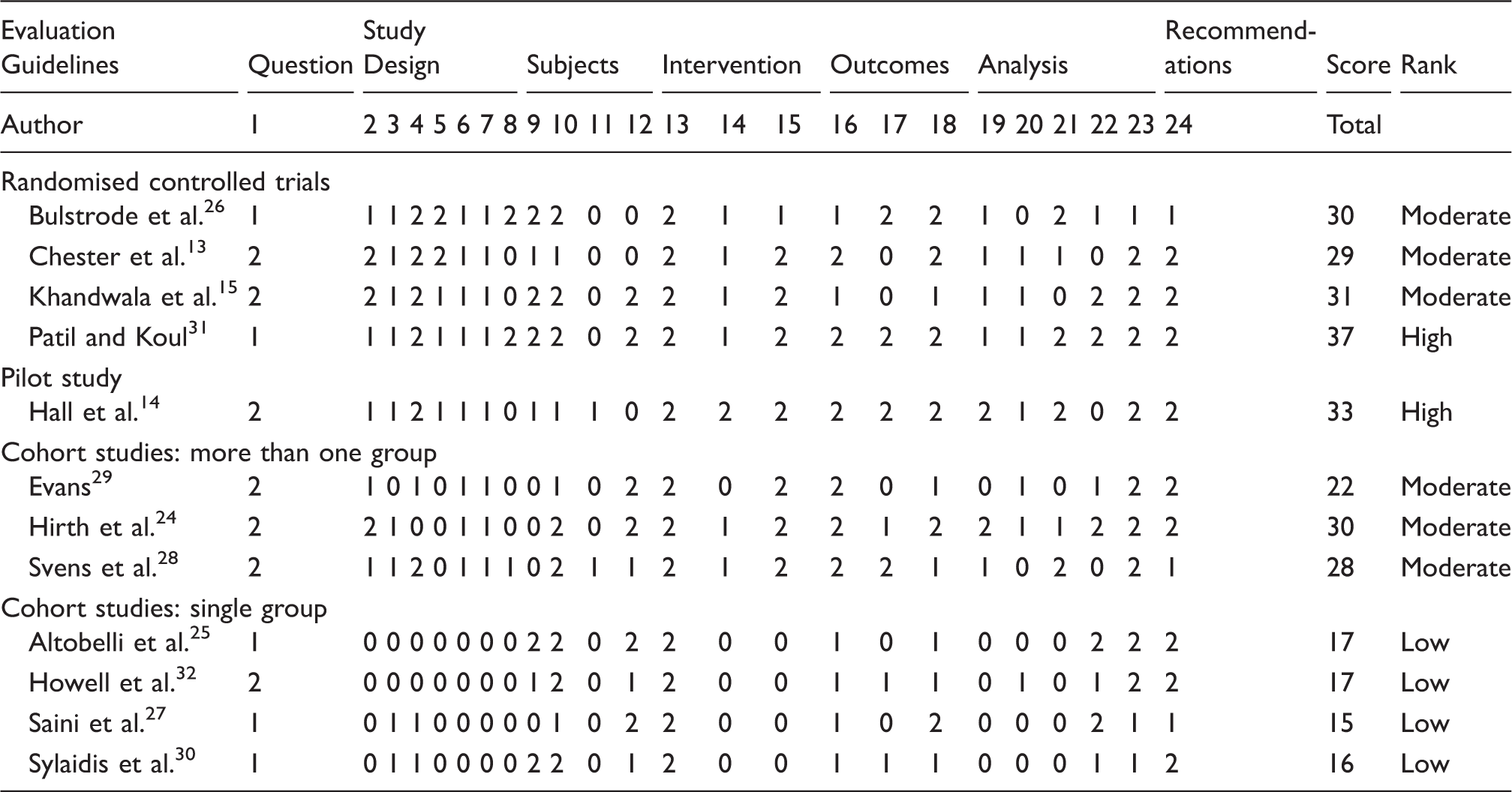
When reviewed according to the type of EAM protocol, the CAM studies included all the RCTs13,15,26,31 and the pilot study with randomised design. 14 None of the RMES studies were randomised. The CAM studies included two14,31 of ‘high’14,31 and four13,15,26,29 of ‘moderate’ methodological quality, while those reporting on RMES protocols included two studies24,28 of ‘moderate’ methodological quality.
Level of evidence
As a systematic review which examines the efficacy of treatment protocols, where over half of the included studies are either RCTs or cohort studies, this systematic review represents level 2a evidence. 23
Discussion
This systematic review was undertaken to investigate the different EAM protocols used after extensor tendon repairs in zone V and VI. The aim was to identify whether any one EAM protocol provided superior outcomes. Only full text, English articles were included which may have led to some bias in the results obtained. From a total of 166 articles identified, 12 studies were selected which met the inclusion and exclusion criteria.
A mix of study designs were represented in 12 included studies: four RCTs, one pilot study, three cohort studies including more than one cohort, and four studies reporting the outcomes of one cohort. With this point in mind, it is potentially a limitation of this current review that a mix of study designs was included. The evidence must be interpreted in the light of the high proportion of non-randomised study designs included.
Following the rating of methodological quality, via the SEQES, two studies achieved a score of ‘high’, six a score of ‘moderate’ and four a score of ‘low’ methodological quality. Common limitations in the 12 included studies were high loss to follow-up, poor statistical analysis and/or reporting, and in many cases, risk of bias due to non-blinding of assessors.
The included studies revealed two main protocol types, CAM and RMES. Studies describing CAM protocols were older and demonstrated a higher level of methodological quality than those describing RMES protocols. No studies compared a CAM to an RMES protocol. RMES protocols had a less restrictive splint design and participants in these studies were advised to return to functional use of the injured hand earlier than those treated with the CAM protocols.
Similar satisfactory ROM and grip strength outcomes were reported for participants treated with CAM and RMES protocols. However, there was a notable difference with regard to reported time to RTW post-operatively: participants treated with an RMES protocol returned to work earlier than those treated with a CAM protocol. This earlier RTW in RMES groups may have been influenced by the less-restrictive splint design and the advice provided to participants regarding functional use of their hand.
The main concern relating to any tendon rehabilitation protocol is the risk of rupture of the repaired tendon. The combination of EAM with less-restrictive splinting and advice to return to functional use of the hand earlier may theoretically have increased the risk of tendon rupture in participants treated with RMES protocols. However, no ruptures were reported in any participants treated with an RMES protocol while small numbers of ruptures were reported in participants treated with a CAM protocol. Factors that may have influenced the difference in rupture rate reported for the CAM and RMES groups are splint design and the strength of the repair.
In the study by Khandwala et al., 15 one participant ruptured the tendon repair when riding a motorbike, while wearing the CAM splint. The design of the splints used in the RMES groups may have reduced the risk of rupture by splinting the affected MCP joint/s in relative extension to the other digits which may harness the supportive effect of the juncturae tendinae connection. 32 Allowing the wrist to be free, as in some RMES protocols, promotes a tenodesis action which reduces tension on the repaired tendon during active digital extension11,34 and may further reduce the risk of rupture.
In the study by Evans, 29 rupture occurred when participants removed their splints for activity. It is possible that ruptures did not occur in the RMES studies because participants were able to use their hands easily while wearing RMES splints and were therefore less tempted to remove the splint for activity.
Studies included in this review reporting on RMES protocols were more recent than those reporting on CAM protocols. The ability to allow more tendon excursion and active motion without increased risk of rupture may additionally be due to recent improvements in suture technique and materials for tendon repair. 35
Heterogeneity of outcome measures used in the included studies meant that it was not possible to perform a meta-analysis. Future studies would be more comparable if they reported ROM in degrees and percentages of TAM and reported on grip strength in kilograms and as a percentage of the contralateral side. There was minimal use of subjective patient-rated outcomes in the studies reviewed. As a number of validated standardised tools now exist to measure subjective outcomes, future studies should employ these tools. Adherence is an important parameter which should be recorded in future studies.
Conclusion
This systematic review has investigated the different EAM protocols used after extensor tendon repair in zones V and VI. Two subcategories of EAM protocols were identified: CAM and RMES. The evidence reviewed suggests that there may be some benefits of RMES protocols over CAM protocols with regard to earlier return to work and decreased incidence of tendon rupture. It is possible that the RMES protocols safely allow easier, earlier functional use of the hand. However, CAM and RMES protocols have not been directly compared. Studies describing RMES protocols are of a lower level of evidence and poorer methodological quality than those describing CAM protocols. The splint used in RMES protocols is low-profile and appears to be minimally restrictive to tendon glide and function of the hand, while providing sufficient protection for the repaired tendon. In light of the possibly superior outcomes of participants treated with the RMES protocols, and the absence of high-level, good quality research comparing RMES and CAM protocols, it may be appropriate to conduct a well-designed prospective trial comparing the two protocols.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable
Guarantor
SC
Contributorship
SC and EK reviewed and scored the studies. SC wrote the initial review. RE reviewed and provided input into the manuscript and prepared it for publication. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
