Abstract
Background:
Most patients with small-cell lung cancer (SCLC) experience disease progression after first-line chemotherapy. Notably, nab-paclitaxel monotherapy has antitumor activity in relapsed SCLC.
Objective:
This study evaluated the efficacy and safety of combined of nab-paclitaxel and immune checkpoint inhibitors (ICIs) in relapsed SCLC.
Design:
We retrospectively analyzed patients with relapsed SCLC who received nab-paclitaxel or combined nab-paclitaxel and ICIs (anti-programmed death-1, PD-1 or anti-programmed cell death 1 ligand, PD-L1) between February 2017 and September 2021.
Methods:
Efficacy and safety data were collected from electronic health records. Progression-free survival (PFS) and overall survival (OS) were assessed using the Kaplan–Meier method and a standard log-rank test.
Results:
We included 56 patients with relapsed SCLC, of which 29 received nab-paclitaxel alone (Group A), and 27 received combined nab-paclitaxel and ICIs (Group B). Baseline characteristics were similar between the two groups. Group B had a numerically higher objective response rate than Group A (40.7% versus 17.2%; p = 0.052). However, combined nab-paclitaxel and ICIs failed to demonstrate survival superiority over nab-paclitaxel monotherapy [median PFS: 3.2 months versus 2.8 months (p = 0.5225); median OS: 11.0 months versus 9.3 months (p = 0.7298)]. The safety profiles of Groups A and B were both tolerable.
Conclusion:
This study indicated that compared with nab-paclitaxel monotherapy, combined nab-paclitaxel and ICIs failed to improve survival in relapsed SCLC.
Introduction
Small-cell lung cancer (SCLC) is an aggressive tumor comprising approximately 15% of all lung cancers. 1 It is highly sensitive to chemotherapy, with an objective response rate (ORR) of approximately 50–70%. However, most patients experience relapse or disease progression after initial treatment.
In disease progression, the efficacy of the subsequent treatment is influenced by the response to first-line chemotherapy. The recommended chemotherapeutic agents for relapsed SCLC include topotecan, irinotecan, amrubicin, temozolomide, paclitaxel, gemcitabine, and docetaxel.2–4 Topotecan has a modest antitumor activity; it is approved by the FDA and National Medical Products Administration (NMPA) for relapsed SCLC. However, it must be used with caution for hematologic toxicity. Paclitaxel has shown antitumor activity even in refractory relapsed SCLC. 5 However, it has a relatively high risk for hypersensitivity reactions. Recently, immune checkpoint inhibitors (ICIs) have shown promising antitumor activity against several solid tumors, including SCLC. According to the CheckMate-032 study and the basket studies KEYNOTE-028 and KEYNOTE-158, nivolumab and pembrolizumab demonstrate antitumor activity in previously treated SCLC.6,7
The combination of chemotherapy and ICIs presents synergistic efficacy in treating multiple solid tumors. In a phase II multicenter study, patients with SCLC progression after first-line chemotherapy received paclitaxel and pembrolizumab as second-line treatment. The ORR was 23.1%, the median progression-free survival (PFS) was 5.0 months, and median overall survival (OS) was 9.1 months. 8 Compared with solvent-based paclitaxel, nab-paclitaxel exhibits an improved therapeutic index, reduced incidence of hypersensitivity reactions, and more favorable efficacy and safety profiles. 9 In several retrospective studies, nab-paclitaxel exhibits anticancer activity in patients with relapsed SCLC.10–12 The clinical efficacy of combined nab-paclitaxel and ICIs has not been compared with that of nab-paclitaxel alone in relapsed SCLC. We conducted this retrospective study to evaluate the efficacy and safety of these two treatment strategies.
Patients and methods
A total of 239 patients with extensive disease SCLC (ED-SCLC) received first-line therapy in our department at the Cancer Hospital Chinese Academy of Medical Sciences between February 2017 and September 2021, of which 166 patients received later-line therapy. We reviewed the medical records of patients with relapsed SCLC treated. The inclusion criteria were as follows: (1) pathologically proven SCLC, (2) disease progression after first line, platinum-based chemotherapy (cisplatin or carboplatin and etoposide), and (3) receipt of either nab-paclitaxel monotherapy or combined nab-paclitaxel and ICIs (anti PD-1 or anti-PD-L1). Patients extensively treated with more than two lines of regimens were also included. Patient baseline characteristics, clinical responses, and adverse events were collected from electronic health records. This study was reviewed and approved by the Ethics Committee of the Cancer Hospital Chinese Academy of Medical Sciences. Written informed consent was obtained from all patients.
Sensitive relapse was defined as disease relapse more than 90 days after first-line chemotherapy completion. Refractory relapse was defined as primary resistance to initial chemotherapy or disease progression within 90 days after first-line chemotherapy completion.
PFS was measured from the initiation of combined nab-paclitaxel and ICIs therapy to disease progression or death. OS was measured from the first administration of combined nab-paclitaxel and ICIs to death. Clinical response was evaluated using the Response Evaluation Criteria in Solid Tumors Version 1.1. 13 Adverse events were graded using the Common Terminology Criteria for Adverse Events Version 5.0 (U.S. Department of Health and Human Services National Institute of Health National Cancer Institute).
Statistical analysis was performed using SPSS Version 23.0 (IBM Corp., Armonk, NY, USA). Patient baseline characteristics are presented as frequencies. The x2 test was used to compare the ORR between groups. Kaplan–Meier estimation was used to calculate PFS and OS and then compared using a standard log-rank test. A p value < 0.05 was considered statistically significant.
Results
Patient characteristics
We included 56 patients with relapsed SCLC treated with nab-paclitaxel alone or combined with ICIs between February 2017 and September 2021. Group A comprised 29 patients who received nab-paclitaxel alone, and Group B comprised 27 patients who received combined nab-paclitaxel and ICIs. The schema for patient screening was shown in Figure 1. Patient baseline characteristics were similar between groups (Table 1). A total of 10 (34.5%) and 11 (40.7%) patients exhibited refractory relapse in Groups A and B, respectively. Five patients received chemotherapy and atezolizumab as first-line therapy, of which four were from Group B and one was from Group A.

The schema for patient screening.
Patients characteristics.

Heavy smoker defined as patient who had smoked more than 20 pack-years.
ECOG, Eastern Cooperative Oncology Group; ICIs, immune checkpoint inhibitors.
Response and efficacy
Nab-paclitaxel was administered at 130 mg/m2 on Days 1 and 8 of a 21-day cycle. Anti-PD-1 therapy included pembrolizumab, nivolumab, sintilimab, tislelizumab, and camrelizumab, and the last three regimens were approved by the NMPA. The ICIs were administered according to the manufacturer’s instructions. Group B had a numerically higher ORR than Group A (40.7% versus 17.2%; p = 0.052) (Figure 2). The combination therapy failed to demonstrate survival superiority over nab-paclitaxel monotherapy [median PFS: 3.2 months versus 2.8 months (p = 0.5225); median OS: 11.0 months versus 9.3 months (p = 0.7298)] (Figure 3(a) and (b)). The combination therapy did not exhibit improved clinical efficacy across all subgroups (Table 2). Four patients received nab-paclitaxel and atezolizumab after failed first-line chemoimmunotherapy, three of which had a partial response, and one had a stable disease. One patient showed no clinical response to nab-paclitaxel after a failed treatment with etoposide, carboplatin, and atezolizumab (Table 3).

Clinical response by treatment groups.
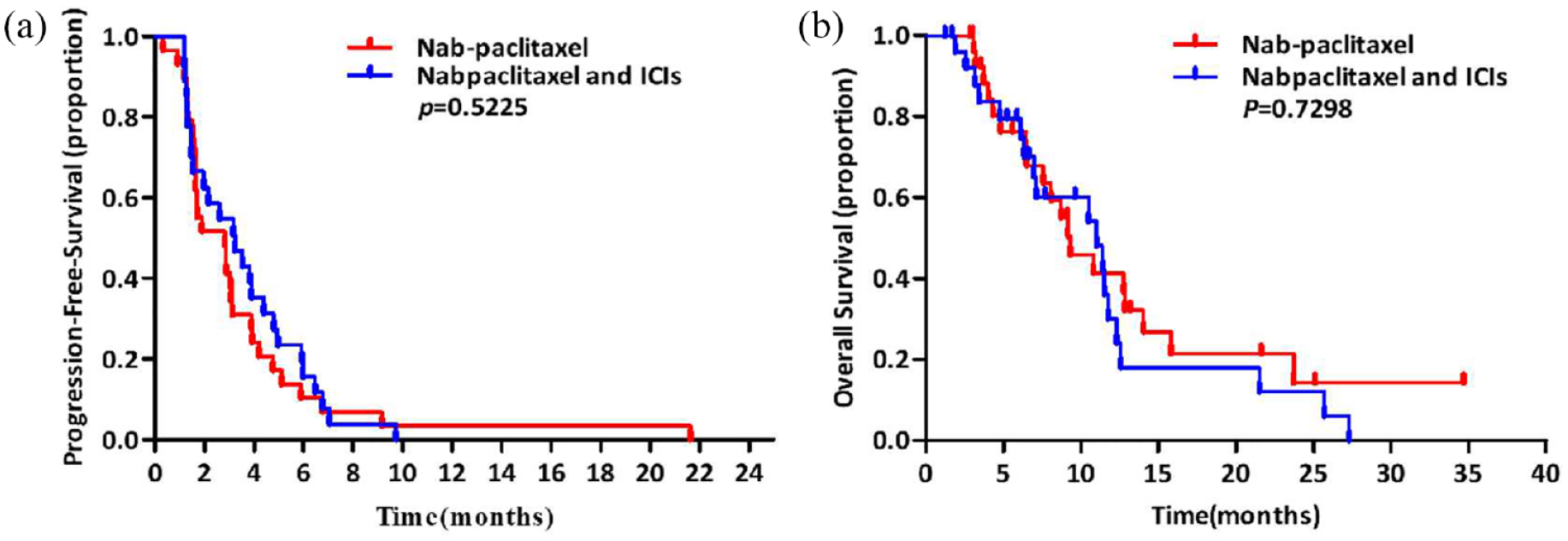
PFS and OS. (a) PFS in nab-paclitaxel monotherapy group and nab-paclitaxel combined with ICIs. (b) OS in nab-paclitaxel monotherapy group and nab-paclitaxel combined with ICIs.
PFS and OS according to baseline characteristics.

ICIs, immune checkpoint inhibitors; OS, overall survival; PFS, progression-free survival.
Patients characteristics and clinical response.

Heavy smoker defined as patient who had smoked more than 20 pack-years.
ICIs, immune checkpoint inhibitors; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.
Safety
The toxicity profiles of Groups A and B were both tolerable (Table 4). The grade 3 or higher adverse events in Group B were leukopenia (7.4%). Treatment-related mortality was not observed in any patient. However, one was diagnosed with grade 2 pneumonia attributed to ICIs.
Toxicity profile.

Discussion
To the best of our knowledge, this is the first study to compare the efficacy and safety of nab-paclitaxel and ICIs combination therapy and nab-paclitaxel monotherapy in relapsed SCLC. The combination therapy demonstrated numerically superior ORR; however, it failed to improve the PFS and OS. Nevertheless, its toxicity was manageable. Furthermore, following failed first-line chemotherapy with anti-PD-L1 therapy, anti-PD-L1 crossover from first- to second-line therapy combined with nab-paclitaxel showed a relatively high clinical response and durable disease control. Further investigation is warranted to confirm this finding.
According to the randomized clinical trials Impower133 and CASPIAN, combined anti-PD-L1 and chemotherapy significantly improve OS in treatment-naïve, extensive-stage SCLC compared with chemotherapy alone.14,15 However, the long-term benefit from first-line chemoimmunotherapy is limited, and most patients still experience disease progression. Topotecan is approved for patients with relapsed SCLC, with an ORR of 18.3–21.9% and a median OS of 33–35 weeks. 2 However, apart from a moderate clinical benefit, bone marrow toxicity must also be considered by oncologists. The clinical need for subsequent therapy following failed standard therapy remains largely unmet in patients with SCLC. Previous studies on paclitaxel, docetaxel, and nab-paclitaxel have reported antitumor activity and a good safety profile in relapsed SCLC.5,12,16 In this retrospective study, we found that combined anti-PD-1 or anti-PD-L1 and chemotherapy did not exhibit improved clinical efficacy compared with nab-paclitaxel alone. However, anti-PD-L1 crossover from first- to second-line therapy showed a relatively high clinical response. Thus, further studies on ICIs crossover therapy in relapsed SCLC must be conducted.
In Group B, approximately 70% of patients received anti-PD-1 combined with nab-paclitaxel after failed standard therapy. Nivolumab and pembrolizumab have been approved by the FDA for relapsed SCLC in 2018 and 2019, respectively. However, the manufacturers of both agents withdrew their indication of SCLC after failed phase III randomized clinical trials in 2020 and 2021.17,18 This study suggested that anti-PD-1 combined with nab-paclitaxel does not exhibit synergistic effects in treating relapsed SCLC. The poor efficacy of anti-PD-1 therapy in relapsed SCLC may be attributed to high tumor heterogeneity. According to the expression levels of key transcriptional regulators, SCLC can be divided into four subtypes: SCLC-A (high expression of ASCL1), SCLC-N (high expression of NEUROD1), SCLC-Y (high expression of YAP1), and SCLC-P (high expression of POU2F3). 19 The SCLC-Y subtype can potentially benefit from ICIs; however, it accounts for only <20% of all SCLC. Further studies on precise treatments based on the new molecular subtypes must be conducted.
Finding appropriate predictive biomarkers of ICIs in SCLC is crucial for improving clinical efficacy. PD-L1 expression is widely used as predictive biomarkers in solid tumors including non-SCLC. However, there was no clear correlation between PD-L1 expression and survival benefit from atezolizumab plus chemotherapy in the IMpower133 trial. 14 KEYNOTE-604 and CASPIAN studies reached similar results.17,20 The results of current clinical trials do not support PD-L1 expression as a predictive biomarker for ICIs response in ED-SCLC. SCLC is characterized by high tumor mutation burden (TMB). High TMB can induce strong T-cell response, which may bring potential benefits for SCLC. 21 Results of CheckMate 032 trial showed ORR, 1-year PFS rate and 1-year OS rate in the high TMB group were significantly longer than those in the low TMB group, regardless of nivolumab monotherapy or combination with ipilimumab. 22 However, in the IMpower133 study, investigators also explored blood TMB (bTMB) as the predictive biomarkers, they found that bTMB was not associated with clinical efficacy. 14 Therefore, further research based on TMB is still needed.
This study had several limitations. Due to the retrospective design of the study, potential bias cannot be ruled out. Furthermore, we used the largest cohort to evaluate the safety and efficacy of combined nab-paclitaxel and ICIs in relapsed SCLC. However, the sample size was still relatively small. Moreover, the different ICIs combinations used may influence immunotherapy efficacy. Lastly, only four patients received anti-PD-L1 crossover from first- to second-line therapy; Thus, ICIs crossover and combination with other treatment strategies must be explored further.
Conclusions
Combined nab-paclitaxel and ICIs demonstrated numerically superior ORR; however, it failed to improve survival. After failed first-line chemotherapy with anti-PD-L1, anti-PD-L1 crossover, and combination with nab-paclitaxel showed a relatively high clinical response. Further studies are warranted to confirm this finding.
