Abstract
Background:
Sacituzumab govitecan (SG), an anti-Trop-2 antibody-drug conjugate (ADC), was approved for metastatic triple-negative breast cancer (mTNBC) with ⩾1 prior therapy and hormone receptor-positive (HoR+) breast cancer progressing on cyclin-dependent kinase 4/6 inhibitor and chemotherapy. However, little real-world evidence is available in China, and the potential predictive biomarker of SG has not been fully investigated.
Objectives:
Our study aimed to evaluate the effectiveness, safety, and biomarker of SG in Chinese metastatic breast cancer (MBC) patients.
Design:
A total of 165 MBC patients treated with SG between June 2023 and December 2024 in 4 institutions nationwide were included in this study. SG was administered intravenously at a dosage of 10 mg/kg on days 1 and 8 of a 21-day treatment cycle.
Methods:
Demographic and clinical data were retrospectively collected and analyzed. Clinical outcomes included real-world progression-free survival (rwPFS), overall survival (OS), objective response rate (ORR), disease control rate, and toxicity.
Results:
In 103 mTNBC patients, median rwPFS was 5.1 months (95% confidence interval (CI): 3.8–7.4) and median rwOS was 19.7 months (95% CI: 17.3–not reached); in 62 HoR+HER2− patients, median rwPFS was 5.8 months (95% CI: 4.7–7.9), and OS data were immature. Prior PD-1/PD-L1 inhibitors exposure in TNBC showed significantly shorter rwPFS (p = 0.025) in multivariate analysis. Longer rwPFS was observed in patients receiving SG as front line (line 1–2) compared to later line (line ⩾3) in both populations. mTNBC patients harboring PIK3CA mutation were associated with reduced PFS of SG (p = 0.034). No new safety signal was observed in this study.
Conclusion:
In conclusion, SG showed similar effectiveness with ASCENT and TROPiCS-02 study in heavily pretreated Chinese MBC patients. Given the compromised effectiveness in patients with prior PD-1/PD-L1 inhibitors or PIK3CA mutation, future investigations are warranted to explore SG-based combination regimens or novel therapeutic strategies in the above population.
Plain language summary
Keywords
Introduction
Breast cancer (BC) is the most frequently diagnosed cancer in women, and also a significant cause of female mortality in China.1,2 Although the early diagnosis and (neo)adjuvant therapies have significantly improved the cure rates of early BC, 20%–40% of patients ultimately develop recurrent or metastatic disease. 3 The 5-year survival rate for metastatic breast cancer (MBC) remains approximately 20%, with a median overall survival of 2–3 years. 4 First-line therapies—such as cyclin-dependent kinase (CDK) 4/6 inhibitors for hormone receptor-positive (HoR+) disease and immunotherapy for triple-negative breast cancer (TNBC)—have demonstrated promising clinical efficacy.5–7 However, effective treatment options remain limited for heavily pretreated, drug-resistant HER2-negative (HER2−) mBC patients.
Sacituzumab govitecan (SG), a trophoblast cell surface antigen 2 (Trop-2)-targeted antibody-drug conjugate (ADC), combines a humanized anti-Trop-2 antibody with the topoisomerase I inhibitor SN-38 via a hydrolyzable linker.8–11 Approximately 50% of ER-positive and 93% of TNBCs highly express Trop-2.12,13 The phase III ASCENT study demonstrated that SG significantly prolonged progression-free survival (PFS) and overall survival (OS) compared with single-agent chemotherapy in patients with metastatic TNBC (mTNBC) who had previously received at least two prior lines of chemotherapy. 14 A statistically significant improvement of PFS was also observed in patients receiving SG with HoR+HER2− MBC progressed on prior CDK4/6 inhibitors and chemotherapy in phase III TROPiCS-02 study. 15 These findings established SG as an effective therapeutic option for later lines for triple-negative (TN) and HoR+HER2− MBC.
In China, SG was approved for patients with mTNBC who have received at least two prior systemic therapies on June 10, 2022 and metastatic HoR+HER2− BC who have received prior endocrine therapy, a CDK4/6 inhibitor, and at least two lines of chemotherapy, at least one of which was in the metastatic setting on March 21, 2025. Considering the recent approval, there are few studies of SG treatment in Chinese patients in the real-world setting, most of which only included the mTNBC population.14,16,17
Real-world studies assess effectiveness in routine clinical practice by including populations often excluded from clinical trials—such as the elderly, those with significant comorbidities, or poor performance status. This approach yields data that better reflects treatment outcomes across a more diverse and representative patient cohort. Through long-term evaluation grounded in actual clinical practice and emphasis on clinically meaningful endpoints, real-world studies provide a robust assessment of the effectiveness and safety profile of SG. Furthermore, it enhances our understanding of potential characteristics and biological factors influencing the effectiveness of SG. Thus, we conducted this multicenter, retrospective study to evaluate the effectiveness, safety, and biomarkers of SG in patients with HER2− MBC in China and to explore potential predictive factors for treatment outcomes.
Materials and methods
Patients
The study included MBC patients treated with SG between June 2023 and December 2024 across 4 institutions, including Fudan University Shanghai Cancer Center (FUSCC), Shanghai Minhang Cancer Hospital, Tumor Hospital of the Chinese Academy of Medical Sciences, and Sanhuan Cancer Hospital. Patients who fulfilled the following criteria were enrolled in this study: (1) age ⩾18 years; (2) prior treatment with at least one dose of SG; (3) histologically confirmed recurrent or MBC, including those with de novo stage IV disease; (4) complete medical history and adverse event (AE) documentation. Patients were excluded if they had incomplete medical records, were pregnant, or breastfeeding.
Baseline demographic features, treatment history, and survival outcomes were retrospectively collected from medical records. SG was administered intravenously at a dosage of 10 mg/kg on days 1 and 8 of a 21-day treatment cycle. Treatment discontinued due to disease progression, intolerable toxicity, or the patient’s decision. The study protocol and all amendments were initially approved by the Institutional Review Board of Fudan University Shanghai Cancer Center (Approval No. 1812195-6). This approval was subsequently acknowledged, and the study was conducted in accordance with its provisions at all other participating sites. The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. The reporting of this study conforms to the STROBE guideline (Supplemental File 2).
Clinical outcomes
The assessment variables included real-world progression-free survival (rwPFS), real-world overall survival (rwOS), objective response rate (ORR), disease control rate (DCR), safety profile, and potential biomarkers predictive of PFS. rwPFS was defined as the time from the first SG treatment to disease progression or death due to various causes, according to the Response Evaluation Criteria in Solid Tumors (RECIST) 1.1. OS was defined as the time from the first SG treatment to death from various causes or the last follow-up visit. ORR was defined as the percentage of evaluable patients at baseline who had either a complete response (CR) or partial response (PR) as the best objective tumor response. DCR was defined as the percentage of evaluable patients at baseline who had CR, PR, or stable disease as the best objective tumor response. Intracranial ORR (icORR) was defined as the proportion of patients achieving intracranial CR and PR in patients with brain metastasis as the best overall response. AE was evaluated according to the National Cancer Institute-Common Terminology Criteria for Adverse Events (NCI-CTCAE) version 5.0. Tumor response, safety assessments, and pathological and genetic testing were performed locally by the investigators at each center.
Post hoc biomarker assessment
Tumor biopsy and blood DNA in FUSCC were isolated from fresh samples using QIAamp DNA Mini Kit (Cat. No. 51306; Qiagen, Hilden, Germany) or QIAamp DNA Blood Mini Kit (Cat. No. 51106; Qiagen, Hilden, Germany). The purity and quantity of the total DNA were assessed by OD260/OD280 with NanoDrop2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). FUSCC next-generation sequencing (NGS) 523-gene panel 18 sequencing was used to detect somatic and germline mutations in both biopsy (n = 12) and circulating tumor DNA samples (n = 28). Gene alteration tested by external institutions (n = 29) were obtained from clinical records.
Statistics
Clinicopathologic characteristics were analyzed using descriptive statistics. Missing data were minimized by using complete medical records. They were handled as follows: for time-to-event endpoints (rwPFS and rwOS), patients were censored at their last known follow-up date; for response assessments (ORR, DCR), only patients with at least one measurable lesion at baseline and at least one post-baseline tumor assessment were included in the evaluable population, while patients missing response assessments were considered not evaluable; for exploratory biomarker analysis, samples with insufficient sequencing quality or coverage were excluded, and analyses were performed on the available dataset. No statistical imputation methods were applied for other missing baseline or clinical variables.
The Kaplan–Meier analysis and 95% confidence intervals (CIs) were used to estimate median rwPFS and median rwOS. Cox proportional hazards model was used to estimate hazard ratios (HRs) and corresponding 95% CIs. Independent prognostic factors for rwPFS were obtained by univariate and multivariate Cox regression analysis. Significant variables in the univariate analysis entered multivariate model. The enter limit and remove limit was p = 0.10. All statistical analyses were performed using SPSS software (SPSS version 26.0; SPSS Inc., Chicago, IL, USA). All statistical tests were two-sided, with a p-value less than 0.05 considered statistically significant. As the subgroup and biomarker analyses were exploratory and conducted post hoc in a retrospective study, we did not adjust for multiple testing.
Results
Characteristics of patients
From June 13, 2023 to December 16, 2024, a total of 165 patients were enrolled in 4 institutions in China with 103 (62.4%) TN and 62 (37.6%) HoR+HER2−. Demographics and clinical characteristics of TN and HoR+HER2− patients are summarized in Table 1.
Demographic and baseline clinical characteristics of TN patients and HoR+HER2− patients.

ADC, antibody-drug conjugate; HER−, HER-negative; HoR+, hormone receptor-positive; TN, triple-negative; TNBC, triple-negative breast cancer.
Among TN and HoR+HER2− patients, median age was 51 and 58 years; 10.7% and 12.7% patients had ECOG scores of 2; most patients had visceral metastases (72.8% in TN and 85.5% in HoR+HER2−); 13.6% and 16.1% of patients had brain metastasis; median number of lines of therapy (LOT) for metastatic setting was 2 and 3 lines, respectively. In TN patients, 34.0% of patients didn’t have an initial TNBC diagnosis, 47.6% of patients received prior PD-1/PD-L1 inhibitors. In HoR+HER2− patients, 87.1% of patients received prior CDK4/6 inhibitors (43.5% for >12 months). In total, 15.5% TN patients and 30.6% HoR+HER2− patients received prior ADCs, and the detailed treatment patterns were shown in Supplemental Table 1. All patients received a median of 4 cycles of SG (range 1–25 for TN and 1–21 for HoR+HER2−). The majority of patients were treated with SG monotherapy, while approximately 10% patients were treated with SG-based combination therapy including bevacizumab, immune checkpoint inhibitors, or other chemotherapy in both subtypes.
Effectiveness
At the cutoff date (January 21, 2025), the median follow-up was 9.4 months (interquartile range (IQR) 3.4–13.6 months) and 7.1 months (IQR 2.1–12.3 months) in TN and HoR+HER2− patients, respectively. In TN patients, 59 (57.2%) patients experienced disease progression and 23 (22.3%) patients had died; the median rwPFS was 5.1 months (95% CI: 3.8–7.4 months), and the median rwOS was 19.7 months (95% CI: 17.3–not reached, Figure 1(a) and (b)); the ORR was 17.5% (18/103) while the DCR was 47.6% (49/103; Table 2). In HoR+HER2− patients, 33 (53.2%) patients experienced disease progression and 8 (12.9%) patients had died; the median rwPFS was 5.8 months (95% CI: 4.7–7.9 months), and the median rwOS data were immature (Figure 1(c) and (d)); the ORR was 9.7% (6/62) while the DCR was 40.3% (25/62; Table 2).
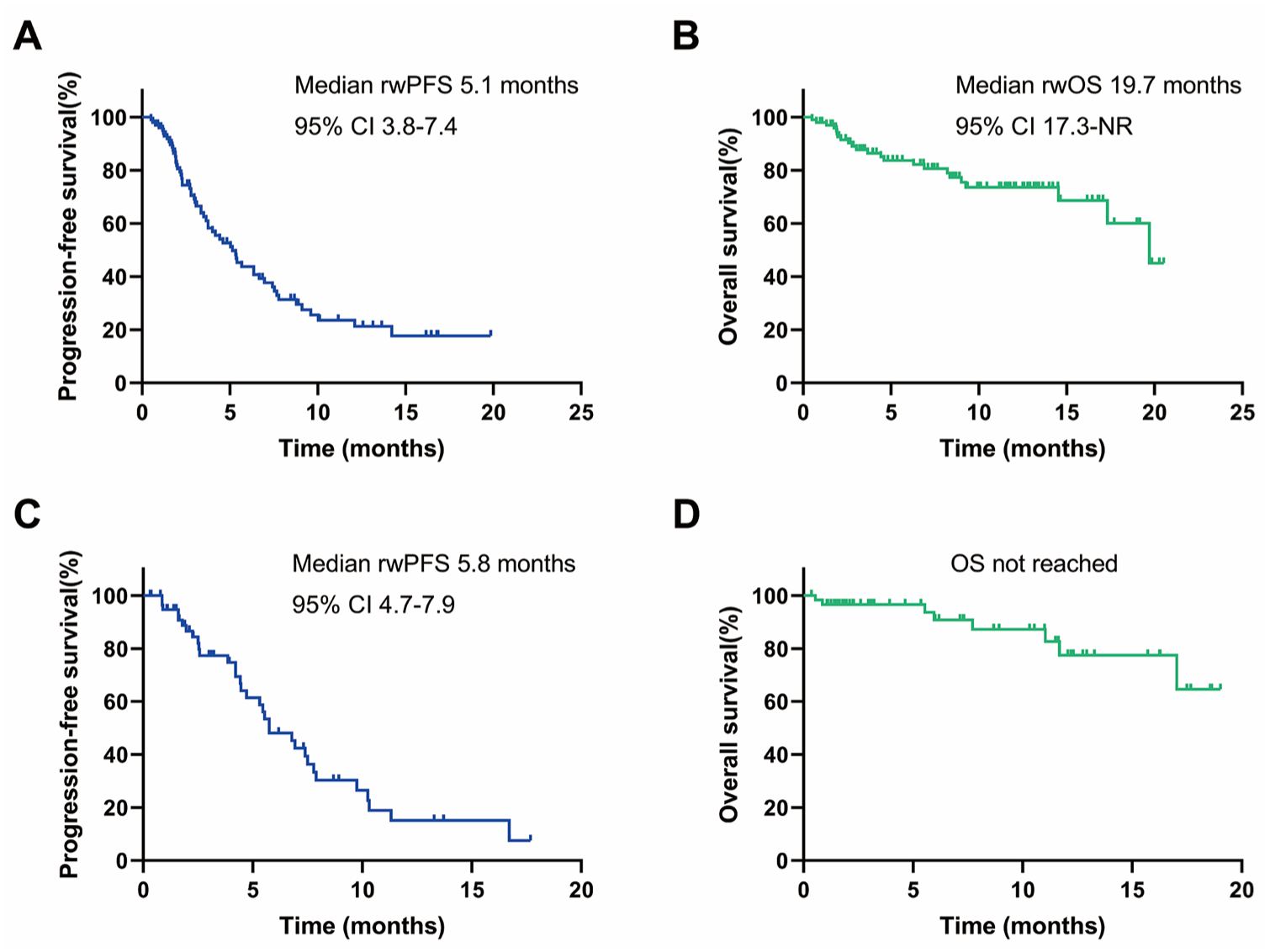
Kaplan–Meier plot for rwPFS and rwOS in patients treated with SG. (a) Kaplan–Meier plot for rwPFS in TN patients. (b) Kaplan–Meier plot for rwOS in TN patients. (c) Kaplan–Meier plot for rwPFS in HoR+HER2− patients. (d) Kaplan–Meier plot for rwOS in HoR+HER2− patients.
Tumor response.
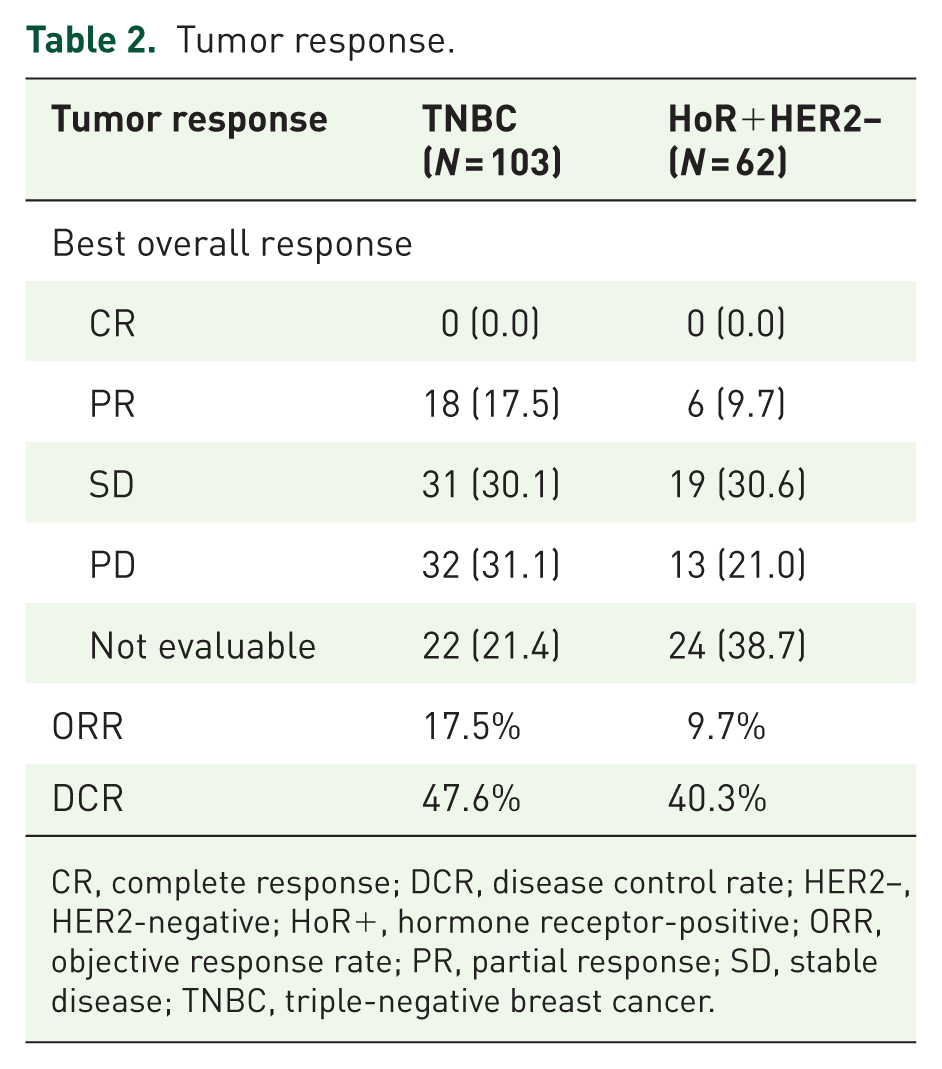
CR, complete response; DCR, disease control rate; HER2−, HER2-negative; HoR+, hormone receptor-positive; ORR, objective response rate; PR, partial response; SD, stable disease; TNBC, triple-negative breast cancer.
Univariate analysis (Supplemental Table 2) indicated that in TN patients, number of metastatic sites (p = 0.002), liver metastasis (p = 0.025), number of prior LOT in metastatic setting (p = 0.017), and previous PD-1/PD-L1 inhibitor exposure (p = 0.004) were significantly related to PFS. Multivariate analysis (Supplemental Tables 2 and 3) further confirmed that mTNBC patients with previous exposure of PD-1/PD-L1 inhibitors significantly reduced rwPFS compared with those without (3.6 vs 7.8 months, HR = 1.91, 95% CI: 1.08–3.37, p = 0.025; Figure 2(a)). Among HoR+HER2− patients, liver metastasis independently predicted shorter rwPFS (5.5 vs 7.9 months, HR = 2.96, 95% CI: 1.18–7.43, p = 0.021), identified by univariate and multivariate analysis (Figure 2(b)). Brain metastasis (p = 0.094 in TN patients and p = 0.485 in HoR+HER2− patients) and prior use of ADCs (p = 0.506 in TN patients and p = 0.151 in HoR+HER2− patients) did not impact the rwPFS of SG in both groups. Response rate was also observed in patients with brain metastases. The systemic ORR was 14.3% (2/14) in TNBC patients and 10.0% in HoR+/HER2− patients. Intracranial responses were less frequent, with an icORR of 7.1% in TNBC and no observed intracranial objective responses (0%) in HoR+HER2− patients (Supplemental Table 4).

Kaplan–Meier curves for PFS according to potential predictive factors. (a) Previous PD-1/PD-L1 exposure in TN patients. (b) Liver metastasis in HoR+HER2− patients.
It’s worth noting that longer rwPFS was observed in patients receiving SG as front line (line 1–2) compared to those as later line (line ⩾3) both in TN (7.8 vs 3.8 months, HR = 1.94, p = 0.017) and HoR+HER2− patients (7.9 vs 5.8 months, HR = 1.32, p = 0.652; Supplemental Figure 1). Furthermore, we explored SG-based combination therapy in a subset of patients (n = 13 in TN patients, n = 5 in HoR+HER2− patients). In the TNBC cohort, SG was combined with PD-1/PD-L1 inhibitors alone (n = 7), bevacizumab alone (n = 2), both PD-1/PD-L1 inhibitor and bevacizumab (n = 3), or capecitabine plus bevacizumab (n = 1); in the HoR+HER2− cohort, combinations included both PD-1/PD-L1 inhibitor and bevacizumab (n = 1) or bevacizumab alone (n = 4). SG-based combination therapy showed a trend to improved PFS compared to monotherapy, both in TNBC (14.2 vs 4.6 months, HR = 0.51, p = 0.115) and HoR+HER2− patients (11.3 vs 5.8 months, HR = 0.35, p = 0.159; Supplemental Figure 2). In this exploratory combination cohort, ORR was 15.4% (2/13) with DCR of 69.2% (9/13) for TN patients, and 0.0% (0/5) with DCR of 40.0% (2/5) for HoR+HER2− patients (Supplemental Table 5). Given the small sample size and heterogeneity of regimens, these findings are descriptive and hypothesis-generating.
Additionally, exploratory analyses were conducted in patient subgroups typically underrepresented in clinical trials, including elderly adults and those with poor performance status. Among patients aged ⩾60 years (n = 21 in TN patients, n = 28 in HoR+HER2− patients), SG demonstrated clinical activity. In the TN subgroup, the median rwPFS was 5.3 months (95% CI: 1.1–9.6), with ORR of 14.3% (3/21) and DCR of 52.4% (11/21). In the HoR+HER2− subgroup, the median rwPFS was 5.8 months (95% CI: 5.4–6.1), with ORR of 3.6% (1/28) and DCR of 35.7% (10/28; Supplemental Table 6). Univariate analysis showed that in both populations, age had no significant impact on rwPFS (p = 0.393 in TN patients and p = 0.998 in HoR+HER2− patients; Supplemental Tables 2 and 3).
Of particular note, clinical benefit was also observed in patients with an ECOG performance status of 2 (n = 11 in TN patients, n = 8 in HoR+HER2− patients). In this frail subgroup, the median rwPFS was 3.4 months (95% CI: 1.5–5.2) in patients with TNBC and 5.5 months (95% CI: 3.9–7.2) in the HoR+HER2− cohort. The ORR and DCR were 9.1% (1/11) and 27.3% (3/11) for TNBC patients, and 0.0% (0/8) and 25.0% (2/8) for HoR+HER2− patients, respectively (Supplemental Table 6). Univariate analysis showed that in both populations, ECOG had no significant impact on rwPFS (p = 0.975 in TN patients and p = 0.485 in HoR+HER2− patients; Supplemental Tables 2 and 3). While the observed response rates in these frail patients (ECOG 2) were modest, the achievement of disease control in this subgroup suggests that SG may still offer a meaningful therapeutic option for a population with limited alternatives and a high unmet need. Taken together, these results suggest that SG may retain therapeutic potential in clinically challenging Chinese patient populations, such as the elderly and those with poor performance status.
Exploratory outcomes
To identify potential biomarkers predictive of PFS and further elucidate the molecular features of patients receiving SG, targeted exome NGS of baseline specimens was performed in 57 patients, with 40 TN and 17 HoR+HER2− patients (Supplemental Table 7, Figure 3(a)).

Genomic characteristics in the study cohort. (a) Oncoplot of somatic and germline mutations assessed by NGS in the study cohort. (b) Kaplan–Meier plots of PFS and PIK3CA mutation status in TN patients.
In total, 798 gene mutation events were detected, and the predominant mutation type observed was a missense mutation (87.2%). The most frequent somatic mutation genes were TP53 gene (31.6%), followed by PIK3CA (29.8%) and ESR1 (10.5%); the most frequent germline mutation genes were BRCA2 gene (49.1%), followed by ATM (47.4%), APC (47.4%), PSM2 (47.4%; Supplemental Table 7). The most frequent pathogenic somatic mutation genes were PIK3CA (28.1%), TP53 (19.3%) and FGFR1 (7.0%); the most frequent pathogenic germline mutation genes were BRCA1 (3.5%), BRCA2 (1.8%), and PALB2 (1.8%). TN patients with pathogenic PIK3CA mutation had significantly worse rwPFS of SG compared to those without (2.3 vs 4.4 months, HR = 2.60, p = 0.034; Figure 3(b)). No difference in PFS was observed among patients with pathogenic gBRCA mutation (p = 0.982, Supplemental Table 8). A total of 25 patients were tested for TMB, and the median value of TMB was 4.20 mutations per megabase (mut/Mb; range 0–48.6). However, no correlation was observed between TMB and PFS (p = 0.572, Supplemental Table 8).
Safety
Table 3 shows the summary of AEs. Overall, 87 patients (52.7%) reported any-grade AEs and grade ⩾3 AEs was 20.0%. Anemia (26.1%), neutropenia (20.0%), leukopenia (17.0%), and lymphocytopenia (10.3%) were the most common treatment-emergent AEs of any grade (Supplemental Table 9). Overall, 16 patients (9.7%) experienced serious AEs (SAEs), which resulted in prolonged hospitalization. No fatal SAE was reported. Neutropenia was the most common SAE (nine patients, 5.5%), followed by leukopenia (six patients, 3.6%) and thrombocytopenia (two patients, 1.2%). All cases of neutropenia, including febrile neutropenia, were managed by institutional standards, which included dose delays, dose reductions, and the use of supportive care such as granulocyte colony-stimulating factors where clinically indicated. AEs leading to dose reduction of SG occurred in 15 patients (9.1%), with leukopenia (n = 7, 4.2%) and neutropenia (n = 4, 2.4%) emerging as the most common factors (Supplemental Table 10). Permanent discontinuation of SG occurred in 14 patients (8.5%), with the leading causes of anemia (n = 2, 1.2%), lung infection (n = 2, 1.2%), and alopecia (n = 2, 1.2%; Supplemental Table 10).
Summary of AEs.
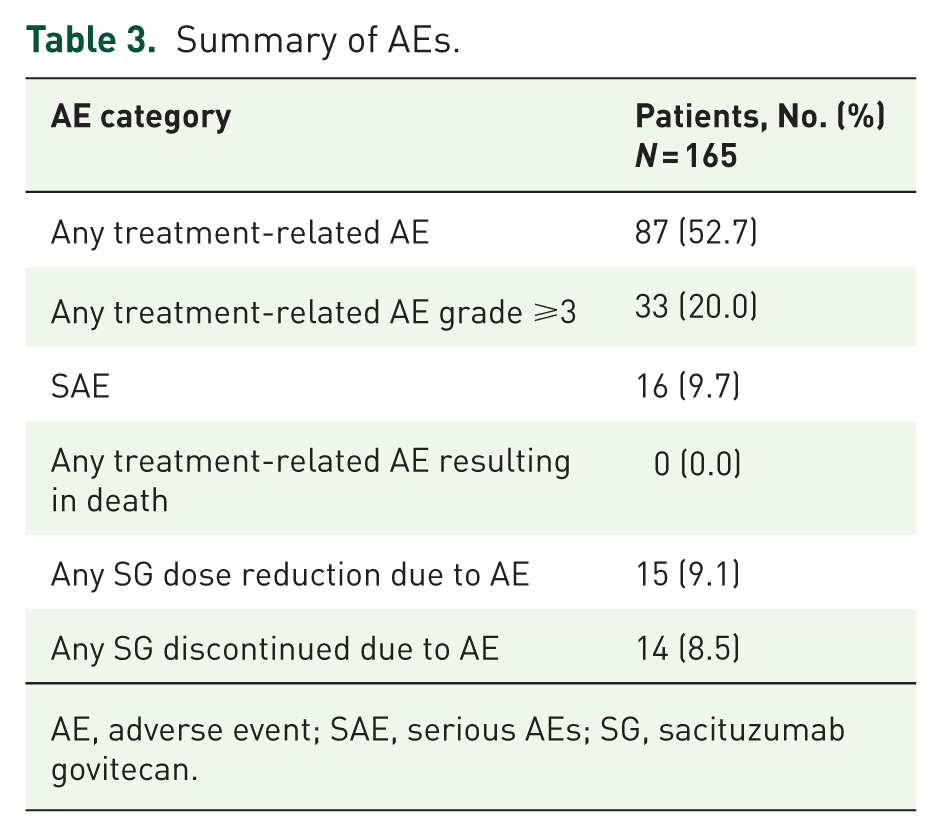
AE, adverse event; SAE, serious AEs; SG, sacituzumab govitecan.
Discussion
This multicenter, retrospective study found that in 103 TN and 62 HoR+HER2− Chinese patients, SG showed a median PFS of 5.1 and 5.8 months, respectively. The median OS of TN patients was 19.7 months, while the OS data of HoR+HER2− patients were immature. Prior PD-1/PD-L1 inhibitors exposure and liver metastasis were independent predictors of the effectiveness of SG in TNBC and HoR+HER2− patients in multivariate analysis, respectively. Biomarker analysis suggests PIK3CA mutation in TN patients is significantly associated with shorter PFS. The toxicity of SG was tolerable, and hematologic toxicities were the most common AEs observed in this study. This study provides first-hand data of effectiveness and safety profile and explores predictive biomarkers of SG in Chinese MBC patients, addressing critical gaps in real-world evidence for SG in Chinese populations.
Our real-world outcomes demonstrated comparable effectiveness to pivotal trials. The median rwPFS of TN patients in our study was 5.1 months, similar to those in IMMU-132-01 study (5.5 months) and ASCENT study (5.6 months).14,19 For HoR+HER2− patients, the median rwPFS was 5.8 months, consistent with TROPiCS-02 study (5.5 months) and numerically longer than EVER-132-002 study (4.3 months), which mainly enrolled Asian populations.15,20 Notably, we included 11 patients (10.7%) with ECOG performance status 2, who were frail or had more comorbidity, and were usually excluded from clinical trials. In this context, the effectiveness and safety of SG appeared consistent with its established profile.
Several real-world studies conducted across multiple countries have documented the use of SG in patients with mTNBC, which have primarily focused on evaluating treatment outcomes in patients receiving SG as a ⩾2 line therapy in a metastatic setting.21–25 Among these studies, United States real-world study showed an improvement of rwOS in patients receiving SG as second line therapy compared with later lines (median rwOS 10.0 vs 13.9 months). 25 In our TN patient group, a significantly longer rwPFS was also observed in patients receiving SG as front line (line 1–2) compared to those as later line (line ⩾3; 7.8 vs 3.8 months, HR = 1.94, p = 0.017). This suggests that early administration of SG may optimize its anti-tumor activity. In our real-world study, a subset of TNBC patients (n = 12) received SG as first-line therapy in a metastatic setting as off-label use, due to the rapid disease progression (six of the patients had a disease-free interval (DFI) <12 months), heavy tumor burden (three of the patients had ⩾3 metastatic sites), or intolerance to previous therapies. Emerging evidence from ASCENT-04 26 and ASCENT-03 27 confirms that SG shows favorable efficacy compared with chemotherapy in PD-L1 positive mTNBC patients when combined with pembrolizumab and in those who are not candidates for PD-(L)1 inhibitors, both as first-line therapy. In addition, patients with DFI of 6–12 months gained a longer PFS treated with SG in the subgroup analysis of ASCENT-03. The results of these studies are consistent with our findings. This collective evidence strengthens the rationale for optimizing first-line application of SG and helps refine treatment strategies. However, this benefit does not appear to extend uniformly across all BC subtypes. The ASCENT-07 trial 28 in HoR+/HER2− MBC progressing on endocrine therapy failed to meet its primary endpoint of improved PFS for SG versus chemotherapy (HR = 0.85). This finding highlights a differential efficacy profile for SG monotherapy between BC subtypes in the post-endocrine setting.
In this real-world study, we found that patients with mTNBC who had previously received PD-1/PD-L1 inhibitors exhibited shorter rwPFS when treated with SG compared to those without prior PD-1/PD-L1 inhibitor exposure (3.6 vs 7.8 months, HR = 1.91, p = 0.025), which was in line with the trend shown in ASCENT study (mPFS 4.2 vs 6.2 months). 14 This observation may be attributed to several hypothetical mechanisms. First, use of PD-1/PD-L1 inhibitors may induce T-cell exhaustion or promote immune escape mechanisms, such as upregulation of alternative immune checkpoints or secretion of immunosuppressive cytokines, thereby diminishing the effectiveness of subsequent SG therapy through suppression of antibody-dependent cellular cytotoxicity effector functions.29–31 Compared with anti-tubulin, ADC with topoisomerase I inhibitors as payload resulted in less immune activation and decreased tumor control when combined with ionizing radiation. 32 Moreover, PD-1/PD-L1 inhibitor treatment may drive clonal evolution, enriching multidrug-resistant subclones that are less sensitive to SG.33,34 Additionally, changes in biomarkers, such as reduced Trop-2 expression or altered immune cell infiltration, may further compromise the effectiveness of SG. 9 Although these hypotheses offer biologically plausible mechanisms, additional studies are required to fully characterize the interaction between PD-1/PD-L1 blockade and activity of SG.
Analysis of 24 patients with brain metastasis in our cohort (18 stable and 6 active disease) demonstrated that brain metastasis did not compromise the effectiveness of SG. This was evidenced by comparable rwPFS in both groups (p = 0.094 in TN patients and p = 0.485 in HoR+HER2− patients). SG has demonstrated its penetration and antitumor activity in the brain from several preclinical studies using a brain tumor xenograft model.35,36 Subgroup analysis of patients with brain metastases from the ASCENT study showed a median PFS of 2.8 months (95% CI: 1.5–3.9) in patients receiving SG, 37 confirming its potential in treating brain metastases in mTNBC patients. Our finding aligned with a French real-world study. 23 The French cohort, which included 31 brain metastasis patients (65% with active disease), reported intracranial responses in 50% (3/6) of evaluable patients receiving SG monotherapy. French patients with brain metastasis had a median rwPFS of 3.7 months, and this is numerically comparable with our TN cohort (median rwPFS 3.4 months). These findings demonstrated that SG is an active regimen and should be considered as an alternative for brain metastasis, in the context of post-local therapy. Further study will provide more evidence of SG-based combination, especially in active brain metastasis (NCT06238921).
Notably, prior use of ADCs (n = 35, 16 TN and 19 HoR+HER2− patients) had no significant impact on the effectiveness of SG (p = 0.506 in TN patients and p = 0.151 in HoR+HER2− patients). T-DXd was the predominant prior ADC in our cohort (74.3%, 26/35), and 14/26 (53.8%) of whom received SG immediately following T-DXd treatment. Previous study suggested the potential cross-resistance between ADCs carrying topoisomerase I inhibitor as payload.38,39 It should be noted that among patients who had received T-DXd, approximately half (12/26) were administered bridging therapy before switching to SG. The use of bridging chemotherapy between ADC treatments preserved antitumor efficacy. In some cohorts, it was even associated with a favorable efficacy compared to the direct sequencing of ADC with the same payload. 38 Thus, a deeper understanding of ADC mechanisms is critical to realizing their full therapeutic activity. circRNA-mediated resistance mechanism in HER2-low BC is novel and provides a promising therapeutic strategy to overcome T-DXd resistance. 40 However, the small sample size in this subgroup analysis limited the reliability of the conclusions, and half of our subgroup did not have the sequential switch from T-DXd to SG. Therefore, these findings should be considered preliminary, and larger, prospective studies are warranted for further validation in ADCs’ cross-resistance.
In biomarker exploratory analysis, we found that PIK3CA mutation may serve as a predictive biomarker for SG resistance. Biologically, pathogenic PIK3CA mutation may lead to hyperactivated PI3K/AKT/mTOR signaling pathway and is associated with resistance to endocrine therapy, chemotherapy, and targeted agents in BC.41–44 These may compromise anti-tumor activity of SG through enhanced DNA damage repair, anti-apoptotic reprogramming, and metabolic adaptation. 41 The role of PIK3CA mutation in the efficacy of ADCs remains unclear. ADCs targeting HER2, including T-DM1 and T-DXd, appear to be effective in both PIK3CA-mutated and wild-type tumors.45,46 However, our observations indicated that mTNBC patients harboring PIK3CA mutations exhibited diminished response to SG, leading us to speculate that constitutive activation of the PI3K signaling pathway may confer resistance through downregulation of Trop-2 expression. Validation in larger real-world cohorts and investigation into the impact of PIK3CA mutation clonality 47 on SG resistance are warranted. Interestingly, our real-world analysis reveals no significant association between gBRCA mutation and PFS, aligning with biomarker analyses in the phase III ASCENT study of SG in mTNBC, 48 where mPFS was numerically similar between gBRCA-mutant and wild-type patients treated with SG (4.6 vs 4.9 months). This suggests that gBRCA mutation does not substantially affect clinical benefit from SG in advanced setting. In contrast, the NeoSTAR trial in early-stage BC reported a notably high pathologic CR (pCR) rate of 66.7% with SG monotherapy in patients carrying gBRCA mutations compared with the overall population (pCR 30%). 49 Mechanistically, in treatment-naïve settings, gBRCA-mutated tumors retain their hallmark homologous recombination deficiency, conferring high sensitivity to DNA-damaging agents like SG.50,51 However, in the advanced, pretreated setting, clonal evolution through therapy-induced selection pressure often leads to genomic reinstatement and restored HR proficiency,52–54 diminishing the predictive value of gBRCA status. Therefore, the reliance on DNA-damage response pathways differs between treatment-naïve and heavily pretreated BRCA-mutated BCs. Based on these observations, further large-scale studies are warranted to elucidate the impact of BRCA status on SG effectiveness in different treatment contexts. In addition, on-target alterations have also been linked to ADC resistance. Acquired TOP1 mutations have been implicated in resistance to the payloads of both T-DXd and SG. 55 However, no TOP1 mutations were detected in our cohort, as they occur at low prevalence and will increase under the selective pressure from SG treatment.
The real-world safety profile of SG demonstrated an overall lower incidence of AEs compared to previous clinical trial data, though key toxicity profiles remained consistent. The proportion of patients reporting hematologic toxicity (i.e., anemia, leukopenia, lymphocytopenia, thrombocytopenia, and febrile neutropenia) and investigations (ALT/AST increase) was consistent with what was observed in the ASCENT study and TROPiCS-02 study14,15; however, in the real-world study, the incidence of the AEs of interest—including neutropenia, diarrhea, nausea and vomiting, and alopecia—were generally lower than those reported in clinical trials. This discrepancy may be attributed to the greater flexibility in drug dosing and treatment scheduling in real-world practice, as well as the recall bias when patients cannot accurately remember their symptomatic AEs, which can contribute to underreporting in retrospective observational analyses. SG is associated with a significant risk of neutropenia and FN. The management of neutropenia in our cohort warrants careful consideration of G-CSF prophylaxis, as current guidelines recommend its use in patients at high risk for FN. We usually give peg-filgrastim after day 8 for patients with high-risk conditions, such as those with old age.
This study has several limitations. First, the sample size was limited, with only 165 patients enrolled over 18 months across multiple institutions, particularly in the HoR+HER2− subgroup. This relatively slow accrual rate may be attributed to the fact that SG was not covered by the national insurance. Patients prefer other drugs due to economic considerations. This may reduce the statistical power for comparisons. Consequently, the results of these exploratory analyses should be interpreted as descriptive and hypothesis-generating and cannot be considered definitive. Second, the choice of SG may have been influenced by physician preference, drug availability, reimbursement policies, and performance status, potentially introducing selection bias. As SG is not included in China’s National Reimbursement Drug List. Some financially constrained patients treated with SG initially may prefer to reduce doses or discontinue treatment prematurely, potentially decreasing treatment intensity and duration. Meanwhile, other patients with adequate financial resources may have better access to innovative drugs, such as T-DXd and other ADCs, may compromise the effectiveness of SG. Third, key assessments, including tumor response, safety, and pathological/genetic testing, were performed locally rather than by a central review board. Although this reflects real-world clinical practice, it raises the possibility of inter-center heterogeneity in assessment criteria and interpretation. To minimize this, we used standardized criteria (RECIST 1.1 and CTCAE 5.0) across all participating centers. Fourth, the median follow-up time was relatively short (9.4 months for TN and 7.1 months for HoR+HER2− patients), which influenced the assessment of long-term outcomes such as OS. Fifth, as a real-world study with an exploratory design involving multiple comparisons, we did not apply adjustments for multiple testing to the biomarker or subgroup analyses. Consequently, significant associations (e.g., with PIK3CA mutation) should be considered hypothesis-generating and require prospective validation. Finally, data on Trop-2 expression were not tested due to the inability to collect patients’ specimens retrospectively. Consequently, we could not analyze how TROP-2 expression correlates with SG effectiveness in our study, which represents a key gap in understanding TROP-2 as a biomarker for treatment response.
To our knowledge, this multicenter study represents the largest real-world cohort to date evaluating SG in Chinese patients with MBC, providing the first comprehensive analysis of its real-world effectiveness and long-term safety. Importantly, we identified PIK3CA mutation as a novel biomarker associated with reduced clinical benefit from SG in MBC. Our findings reinforce the clinical value of SG as an effective therapeutic option for heavily pretreated MBC patients, while also revealing that prior exposure to PD-1/PD-L1 inhibitors may influence PFS. Based on these results, future research should prioritize prospective validation of PIK3CA as a predictive biomarker for SG efficacy. Furthermore, prospective studies evaluating SG as the first-line setting, particularly in combination with PD-1/PD-L1 inhibitors rather than sequential therapy, are warranted to optimize patient selection and therapeutic strategy.
Conclusion
This multicenter study provides the largest real-world evidence to date on the clinical effectiveness and safety of SG in Chinese MBC patients. In real-world practice, SG exhibited similar effectiveness observed in the ASCENT and TROPiCS-02 study and showed anti-tumor activity in heavily pretreated Chinese MBC patients. Notably, the effectiveness of SG was reduced in patients with prior PD-1/PD-L1 inhibitor exposure or those harboring PIK3CA mutations. Future research should focus on identifying patient populations benefiting from SG therapy, while further exploring SG-based combination regimens or novel therapeutic agents to overcome the above resistance mechanisms.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261430556 – Supplemental material for Effectiveness, safety, and biomarker analysis of sacituzumab govitecan in Chinese metastatic breast cancer: a multicenter real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359261430556 for Effectiveness, safety, and biomarker analysis of sacituzumab govitecan in Chinese metastatic breast cancer: a multicenter real-world study by Mu Li, Die Sang, Jin-feng Zhang, Peng Yuan, Gang Li, Mei Yang, Cheng-Cheng Gong, Yi-zhao Xie, Yi-fan Chen, Shu-ting Li, Qi-xuan Zhao, Yan-nan Zhao and Bi-yun Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359261430556 – Supplemental material for Effectiveness, safety, and biomarker analysis of sacituzumab govitecan in Chinese metastatic breast cancer: a multicenter real-world study
Supplemental material, sj-docx-3-tam-10.1177_17588359261430556 for Effectiveness, safety, and biomarker analysis of sacituzumab govitecan in Chinese metastatic breast cancer: a multicenter real-world study by Mu Li, Die Sang, Jin-feng Zhang, Peng Yuan, Gang Li, Mei Yang, Cheng-Cheng Gong, Yi-zhao Xie, Yi-fan Chen, Shu-ting Li, Qi-xuan Zhao, Yan-nan Zhao and Bi-yun Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359261430556 – Supplemental material for Effectiveness, safety, and biomarker analysis of sacituzumab govitecan in Chinese metastatic breast cancer: a multicenter real-world study
Supplemental material, sj-pdf-2-tam-10.1177_17588359261430556 for Effectiveness, safety, and biomarker analysis of sacituzumab govitecan in Chinese metastatic breast cancer: a multicenter real-world study by Mu Li, Die Sang, Jin-feng Zhang, Peng Yuan, Gang Li, Mei Yang, Cheng-Cheng Gong, Yi-zhao Xie, Yi-fan Chen, Shu-ting Li, Qi-xuan Zhao, Yan-nan Zhao and Bi-yun Wang in Therapeutic Advances in Medical Oncology
