Abstract
Background:
Utidelone (UTD1), a genetically engineered epothilone derivative, has been approved in China for use in combination with capecitabine in treating metastatic breast cancer (MBC) patients previously treated with anthracyclines or taxanes.
Objectives:
To evaluate the real-world efficacy and safety of UTD1 in Chinese patients with MBC and to explore potential predictors of therapeutic effectiveness.
Design:
A multicenter, retrospective, real-world study.
Methods:
MBC patients who received UTD1 between March 2021 and August 2023 were identified using an electronic database. Outcome variables included progression-free survival (PFS), overall survival (OS), time to treatment failure (TTF), objective response rate (ORR), clinical benefit rate (CBR), and adverse events (AEs).
Results:
A total of 270 MBC patients were included, with 81.1% presenting with visceral metastasis and 23.7% with brain metastasis. The median number of treatment lines for UTD1 was 3. UTD1 showed a median PFS of 3.97 months (95% confidence interval (CI) 3.33–4.61) and a median OS of 20.63 months (95% CI 16.72–24.54). Among the patients, 17.4% received UTD1 monotherapy, and 82.6% received UTD1-based combination therapy. The median TTF was 2.80 months (95% CI 2.31–3.29). The ORR was 8.4%, and the CBR was 33.5%. The most common AE was peripheral neuropathy (PN, 55.2%). Patients with unresolved PN from previous therapy or receiving UTD1 through intravenous infusion on days 1–5 were more likely to develop ⩾grade 3 PN.
Conclusion:
UTD1 is a new option for patients who have previously received taxanes and anthracyclines, with its clinical toxicity controllable.
Plain language summary
UTD1 is a new chemotherapy drug approved in China for patients with metastatic breast cancer (MBC) who have previously been treated with anthracyclines and taxanes. While clinical trials demonstrate the effectiveness of treatments in highly controlled environments, real-world patients are more diverse and face different challenges. Real-world studies help determine whether treatments are effective in everyday practice, include a broader range of patients, and offer insights into long-term outcomes and side effects, ultimately supporting more informed treatment decisions in clinical care.
This multicenter, retrospective, real-world study analyzed medical records from 270 Chinese patients with MBC treated with UTD1. Most patients (72.2%) received UTD1 as third-line or later therapy. The study population had a high disease burden, with 81.1% of patients having visceral metastases and 23.7% presenting with brain metastases.
The real-world efficacy of UTD1 showed consistent results with other studies in similarly pretreated populations. Peripheral neuropathy was the most common adverse event, affecting 55.2% of patients, with 14.1% experiencing grade 3 or higher symptoms. In clinical practice, UTD1 was often used in combination with other agents such as bevacizumab, capecitabine, and platinum. Three administration methods were used in real-world settings. The intravenous days 1–5 schedule was associated with improved outcomes but carried a higher risk of severe peripheral neuropathy.
These findings indicate that UTD1 could be a valuable treatment option for pretreated MBC patients with limited choices. However, the results also highlight the difficulties of managing a diverse patient population in real-world settings and balancing treatment effectiveness with the risk of side effects. Careful consideration of administration methods and personalized treatment planning are crucial for optimizing outcomes.
Introduction
Breast cancer is the second most prevalent cancer globally, with 2.3 million new diagnoses, representing 11.6% of all new cancer cases. Among women, it is the most frequently diagnosed cancer and the leading cause of cancer-related death. 1 Approximately 10% of women are newly diagnosed with de novo stage IV breast cancer, and roughly 30% of those with early-stage disease will eventually experience relapse and/or metastasis.2,3 Chemotherapy remains the cornerstone of treatment for patients with metastatic triple-negative (TN), human epidermal growth factor receptor 2 (HER2)-positive, or hormone receptor (HoR)-positive breast cancer that is resistant to endocrine therapy. 4 Anthracyclines and taxanes are standard therapy for (neo)adjuvant therapy in early breast cancer. 4 This trend has resulted in a higher number of patients with heavily pretreated metastatic breast cancer (MBC) and an increased risk of cumulative toxicity. 5 Limited options exist for MBC patients previously treated with anthracyclines and taxanes.
Epothilones are a category of naturally occurring microtubule inhibitors derived from the cellulose-degrading myxobacterium Sorangium cellulosum SoCe90. 6 They promote microtubule polymerization and induce apoptosis, with a mechanism similar to taxanes but a distinct molecular structure. 6 Consequently, patients resistant to taxanes may remain sensitive to epothilone. 7 Since epothilones were discovered, various derivatives have been synthesized, but most have failed in phase II and III clinical trials. 6 Ixabepilone, a semi-synthetic analog, is the only epothilone approved by the US Food and Drug Administration (FDA) for MBC treatment. 6 In two phase III studies, ixabepilone combined with capecitabine significantly prolonged median progression-free survival (PFS) compared to capecitabine monotherapy, although no clinically significant improvement was observed in overall survival (OS).8,9 Results from phase II studies on ixabepilone monotherapy indicated a median PFS of 2.2–3.1 months in taxane-resistant MBC patients.10,11 However, adverse events (AEs) such as myelosuppression, peripheral neuropathy (PN), and fatigue often led to treatment discontinuation. 12 Ixabepilone is not available in China. Therefore, it is necessary to develop a more effective and safer epothilone analog.
Utidelone (UTD1) is an epothilone derivative developed through genetic engineering of the epothilone gene cluster. 6 It has shown potent activity against paclitaxel-sensitive tumors in vitro and in vivo, including breast tumors, leukemia, and multidrug-resistant human colon cancer. 13 A phase I study demonstrated that UTD1 monotherapy was effective against tumors typically resistant to taxanes. 14 UTD1 has also shown synergistic therapeutic activity with capecitabine.15,16 The phase III clinical study (BG01-1323L study) showed that patients receiving UTD1 combined with capecitabine had a significantly improved median PFS compared to those receiving capecitabine alone (8.44 vs 4.27 months, hazard ratio (HR) 0.46, 95% confidence interval (CI) 0.36–0.59, p < 0.0001). 15 The final analysis of OS, updated in 2020, also showed a significant improvement with the addition of UTD1 (19.8 vs 16.0 months, HR 0.75, 95% CI 0.59–0.94, p = 0.0142).15,16 The BG01-1323L study confirmed that the UTD1 combined with capecitabine regimen was the only non-paclitaxel chemotherapy regimen that could achieve a dual PFS/OS benefit in breast cancer treatment.15,16 Based on the results of the BG01-1323L study, UTD1 was approved by the National Medical Products Administration (NMPA) in China in 2021 for use in combination with capecitabine in patients with locally recurrent or MBC previously treated with anthracyclines or taxanes.15,16 However, further research is needed to evaluate its real-world effectiveness and identify potential determinants of its efficacy.
Hematological toxicity is a common AE of chemotherapy drugs, leading to dose reduction or treatment delay, increasing the cost of treatment, and reducing chemotherapy efficacy and patients’ quality of life. While many chemotherapy regimens cause varying degrees of myelosuppression, UTD1 stands out as the only microtubule-dynamics inhibitor so far demonstrating no significant myelosuppression toxicity. 15 The BG01-1323L study reported a relatively low incidence of grade 3/4 neutropenia (11.6%) in the UTD1 combined with capecitabine group.15,16 By contrast, studies of paclitaxel, ixabepilone, and eribulin have reported grade 3/4 neutropenia and leukopenia rates of up to 70%.17 –20 In terms of hematological toxicity, UTD1 was superior to ixabepilone and eribulin.
A phase II clinical study (UTOBIA-BM study) of UTD1 combined with bevacizumab and etoposide for treating HER2-negative breast cancer patients with brain metastases presented preliminary data at the 2023 ESMO conference. 21 The results showed that the objective response rate of central nervous system lesions (CNS-ORR) and the clinical benefit rate of central nervous system lesions (CNS-CBR) reached 73% and 91%, respectively. 21 This was the first clinical trial to demonstrate the anti-tumor activity of UTD1 in breast cancer patients with brain metastases. The FDA has granted orphan drug designation to UTD1 as a potential treatment option for patients with breast cancer brain metastases.
Given the lack of real-world clinical data on UTD1 in China, we conducted this multicenter retrospective study to evaluate its efficacy and safety in heavily pretreated MBC patients and to identify potential predictors of its therapeutic effectiveness.
Patients and methods
Study design and population
This multicenter, retrospective, observational study involved patients diagnosed with MBC who received UTD1 treatment between March 2021 and August 2023 at five institutions: Fudan University Shanghai Cancer Center, Tongji Hospital, Hunan Cancer Hospital, Sun Yat-Sen University Cancer Center, and Nanchang People’s Hospital. MBC was defined as either de novo stage IV or recurrent breast cancer confirmed by clinical, imaging, histological, or cytological methods. Clinical data were collected retrospectively from electronic medical records. The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement (Supplemental File 1). 22
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the Fudan University Shanghai Cancer Center (Approval No. 1812195-6). Because of its retrospective design, the Ethics Committee waived the need for individual informed consent. The participating hospitals did not obtain separate ethics approvals because the study was retrospective.
Evaluation
Baseline clinicopathological characteristics included age, menopausal status, disease characteristics, molecular subtypes, Ki67 index, number and sites of metastases, treatment line of UTD1, prior treatments, agents used in combination, and administration method. Visceral metastasis was defined as involvement of internal organs, including the lung, liver, peritoneum, pleura, or central nervous system. Taxane resistance was defined as disease progression during taxane treatment or within 3 months after the last dose in the metastatic setting, or recurrence within 6 months in the (neo)adjuvant setting. 8 The duration of prior (taxane, capecitabine, eribulin) treatment was defined as the time from initial treatment for MBC to disease progression.
The primary endpoint was PFS, while the secondary endpoints included OS, time to failure (TTF), ORR, and clinical benefit rate (CBR). PFS was defined as the time from the first UTD1 administration to disease progression or death from any cause, based on the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. OS was defined as the time from the first UTD1 administration to death from any cause or last follow-up visit. TTF was defined as the time from the first UTD1 administration until treatment discontinuation for any reason. The reasons for discontinuation included disease progression, intolerable toxicity, the treating physician’s decision, or the patient’s personal choice. ORR was defined as the proportion of evaluable patients achieving a complete response (CR) or partial response (PR) as their best objective tumor response. CBR was defined as the proportion of evaluable patients achieving CR, PR, or stable disease lasting at least 24 weeks.
Safety analyses included all patients who received at least one dose of UTD1. AEs were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAEs) version 5.0 and managed based on clinical severity. For PN, management strategies included dose reductions, interval prolongation, treatment interruption, or symptomatic care such as analgesics, vitamin B supplementation, or physical therapy. As this was an observational real-world study, specific treatment decisions and AE management were determined by physicians at each center based on routine clinical practice, and these were documented as faithfully as possible in our data collection. The onset time of PN was defined as the time from the first UTD1 administration to the first occurrence of PN. Recovery duration was defined as the time from the onset of ⩾grade 3 PN to recovery to grade 0–1.
Statistical analysis
Categorical variables were summarized as frequencies (n) and percentages (%), while continuous variables were presented as medians and ranges. Baseline characteristics between patients with and without ⩾grade 3 PN were compared using the chi-square or Fisher’s exact test for categorical variables, and the Wilcoxon rank-sum test for continuous variables. Kaplan–Meier plots were used to calculate median PFS, OS, and TTF, with corresponding 95% CIs. Stepwise multivariate Cox proportional hazards models were used to evaluate potential predictors of treatment efficacy, with results presented as HRs with 95% CIs and p-values. Variables with a p-value <0.1 in univariate analysis were included in the multivariate analysis. Missing data were handled using complete case analysis. Statistical evaluation was conducted using SPSS software (version 21.0; IBM Corp., Armonk, NY, USA). p < 0.05 was considered statistically significant.
Results
Patient characteristics
This retrospective study included 270 MBC patients treated with UTD1 at five hospitals between March 2021 and August 2023. Baseline characteristics are detailed in Table 1. The median age at initiation of UTD1 therapy was 52.5 years, ranging from 28 to 76 years. A majority (55.9%) were older than 50 years. Postmenopausal patients accounted for 61.9%. Most patients (83.3%) had recurrent disease, while 16.7% were diagnosed with de novo stage IV disease.
Patient baseline characteristics.
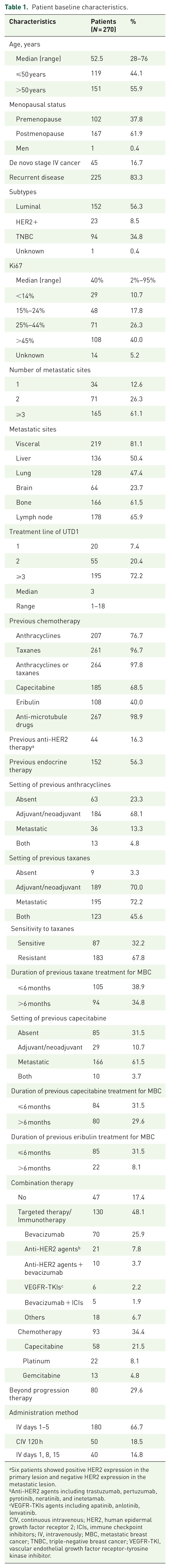
Six patients showed positive HER2 expression in the primary lesion and negative HER2 expression in the metastatic lesion.
Anti-HER2 agents including trastuzumab, pertuzumab, pyrotinib, neratinib, and inetetamab.
VEGFR-TKIs agents including apatinib, anlotinib, lenvatinib.
CIV, continuous intravenous; HER2, human epidermal growth factor receptor 2; ICIs, immune checkpoint inhibitors; IV, intravenously; MBC, metastatic breast cancer; TNBC, triple-negative breast cancer; VEGFR-TKI, vascular endothelial growth factor receptor-tyrosine kinase inhibitor.
Regarding molecular subtypes, 56.3% were luminal, 34.8% triple-negative breast cancer (TNBC), and 8.5% HER2-positive. Most patients (61.1%) had ⩾3 metastatic sites. 81.1% had visceral metastases, with liver (50.4%) and lung (47.4%) being the most common sites of metastasis. The median number of UTD1 treatment lines was 3, with 72.2% of patients receiving UTD1 as third-line or later treatment. In adjuvant or metastatic settings, nearly all patients (96.7%, 261/270) had received taxanes. In addition, 76.7% (207/270) had received anthracyclines, 68.5% (185/270) had received capecitabine, and 40.0% (108/270) had received eribulin. 67.8% of patients were resistant to taxanes.
17.4% of patients (47/270) received UTD1 monotherapy, while 82.6% (223/270) received combination regimens with targeted agents or other chemotherapy. Bevacizumab was the most commonly used combination therapy (25.9%, 70/270). Among the 36 HER2-positive patients (HR+/HER2+: 13; HR−/HER2+: 23), most (11.5%, 31/270) received UTD1 combined with anti-HER2 therapy, while 4 received UTD1 monotherapy and 1 received UTD1 with capecitabine. A total of 93 patients (34.4%) received UTD1 combined with chemotherapy: 21.5% (58/270) with capecitabine, 8.1% (22/270) with platinum, and 4.8% (13/270) with gemcitabine. The median duration of UTD1 treatment cycles was 3, ranging from 1 to 21 cycles. Administration methods in the real-world included the following: (1) 30 mg/m2 intravenously (IV) on days 1–5 every 21 days, (2) 65 mg/m2 intravenously on days 1, 8, and 15 every 28 days, (3) 200 mg/m2 continuous intravenous (CIV) pump infusion over 120 h every 21 days.
Efficacy
As of the cutoff date in August 2023, the median follow-up was 19.5 months (interquartile range (IQR) 11.4–24.4). PFS events occurred in 198 patients (73.3%), with a median PFS of 3.97 months (95% CI 3.33–4.61) (Figure 1(a)). OS events were observed in 76 patients (28.1%), with a median OS of 20.63 months (95% CI 16.72–24.54) (Figure 1(b)). TTF events occurred in 224 patients (83.0%), with a median TTF of 2.80 months (95% CI 2.31–3.29) (Figure 1(c)). The CBR was 33.5%, and the ORR was 8.4% (Table 2).

Kaplan–Meier curves for (a) progression-free survival (PFS), (b) overall survival (OS), and (c) time to treatment failure (TTF) in patients treated with utidelone (UTD1).
Evaluation of efficacy.

CBR, clinical benefit rate; CI, confidence interval; CNS, central nervous system; ORR, objective response rate.
Univariate analysis (Table 3) showed that age, liver metastasis, taxane sensitivity, number of treatment lines of UTD1, and administration method were significantly associated with PFS. Multivariate analysis revealed that the administration method was an independent predictor of PFS. Patients treated with IV on days 1–5 had significantly longer PFS than those treated IV on days 1, 8, and 15 (median PFS 4.77 vs 3.13 months, HR 1.58, 95% CI 1.06–2.36, p = 0.028) (Figure 2).
Univariate and multivariate Cox regression analyses of factors predicting PFS in patients treated with UTD1.
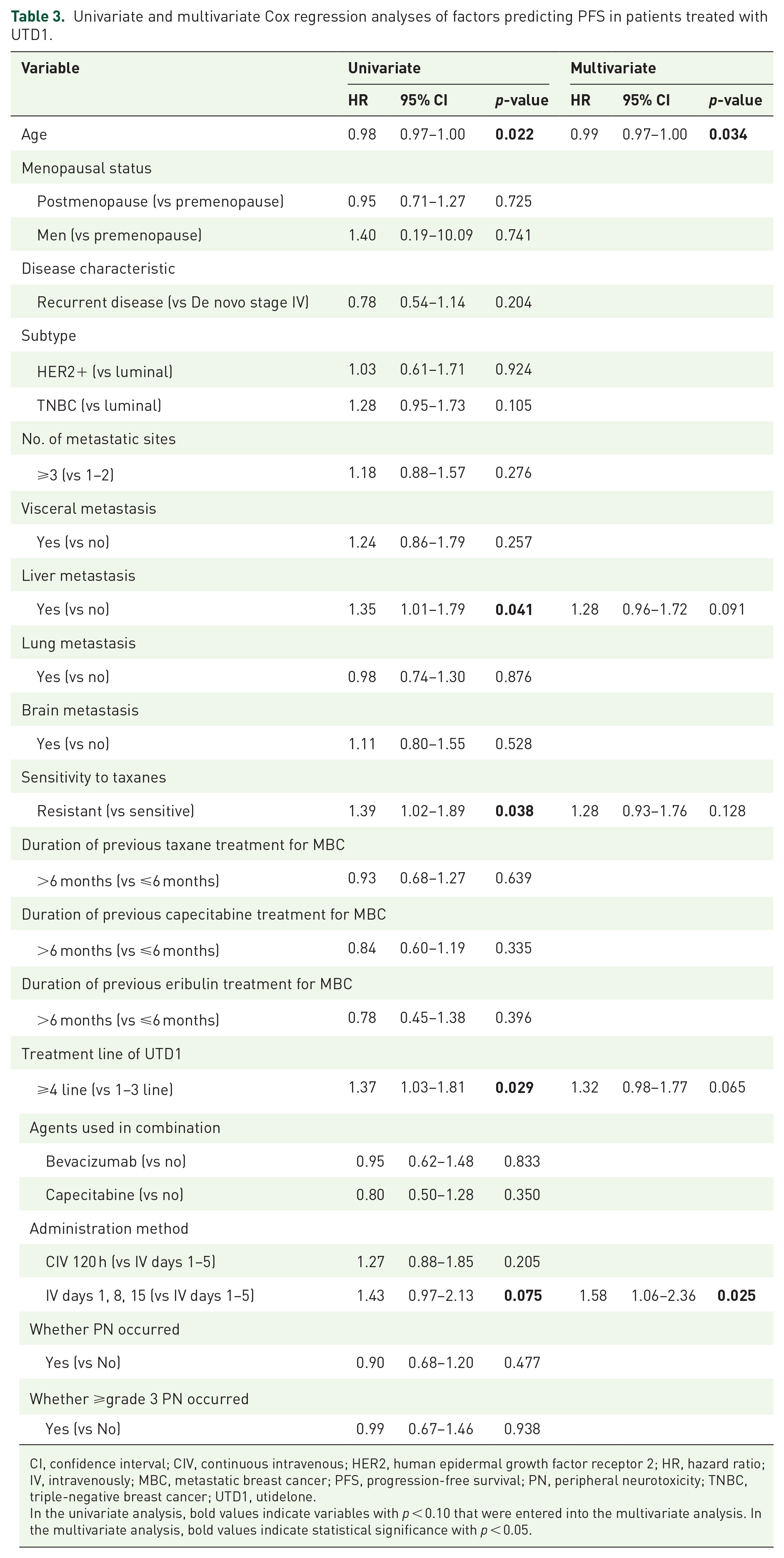
CI, confidence interval; CIV, continuous intravenous; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; IV, intravenously; MBC, metastatic breast cancer; PFS, progression-free survival; PN, peripheral neurotoxicity; TNBC, triple-negative breast cancer; UTD1, utidelone.
In the univariate analysis, bold values indicate variables with p < 0.10 that were entered into the multivariate analysis. In the multivariate analysis, bold values indicate statistical significance with p < 0.05.

Kaplan–Meier curves for progression-free survival (PFS) according to administration method of utidelone (UTD1).
Secondary analyses were conducted in subgroups receiving UTD1 monotherapy, combination therapy, TNBC patients, and those with brain metastases. For those receiving UTD1 monotherapy, the median PFS was 3.33 months (95% CI 1.55–5.11) (Tables S1 and S2). Patients receiving combination therapy had a median PFS of 4.17 months (95% CI 3.21–5.13) (Tables S3 and S4). Among TNBC patients, the median PFS was 3.50 months (95% CI 2.97–4.03) (Tables S5 and S6). For patients with brain metastases, the median PFS was 3.13 months (95% CI 2.43–3.83), with CNS-CBR and CNS-ORR of 28.6% and 10.7%, respectively (Tables S7 and S8).
Safety
PN was the most common AE, affecting 55.2% of patients: 41.1% with grade 1–2, 13.7% with grade 3, and one patient with grade 4 (Table 4). The median onset time of ⩾grade 3 PN was 3.0 weeks (Table 5). Among patients with ⩾grade 3 PN, 50.0% recovered to grade 0–1, with a median recovery duration of 17.4 weeks (Table 5, Figure 3). Other AEs included aspartate transaminase/alanine aminotransferase increases (21.4%), leukopenia (7.8%), and neutropenia (7.1%) (Table 4).
Adverse events.
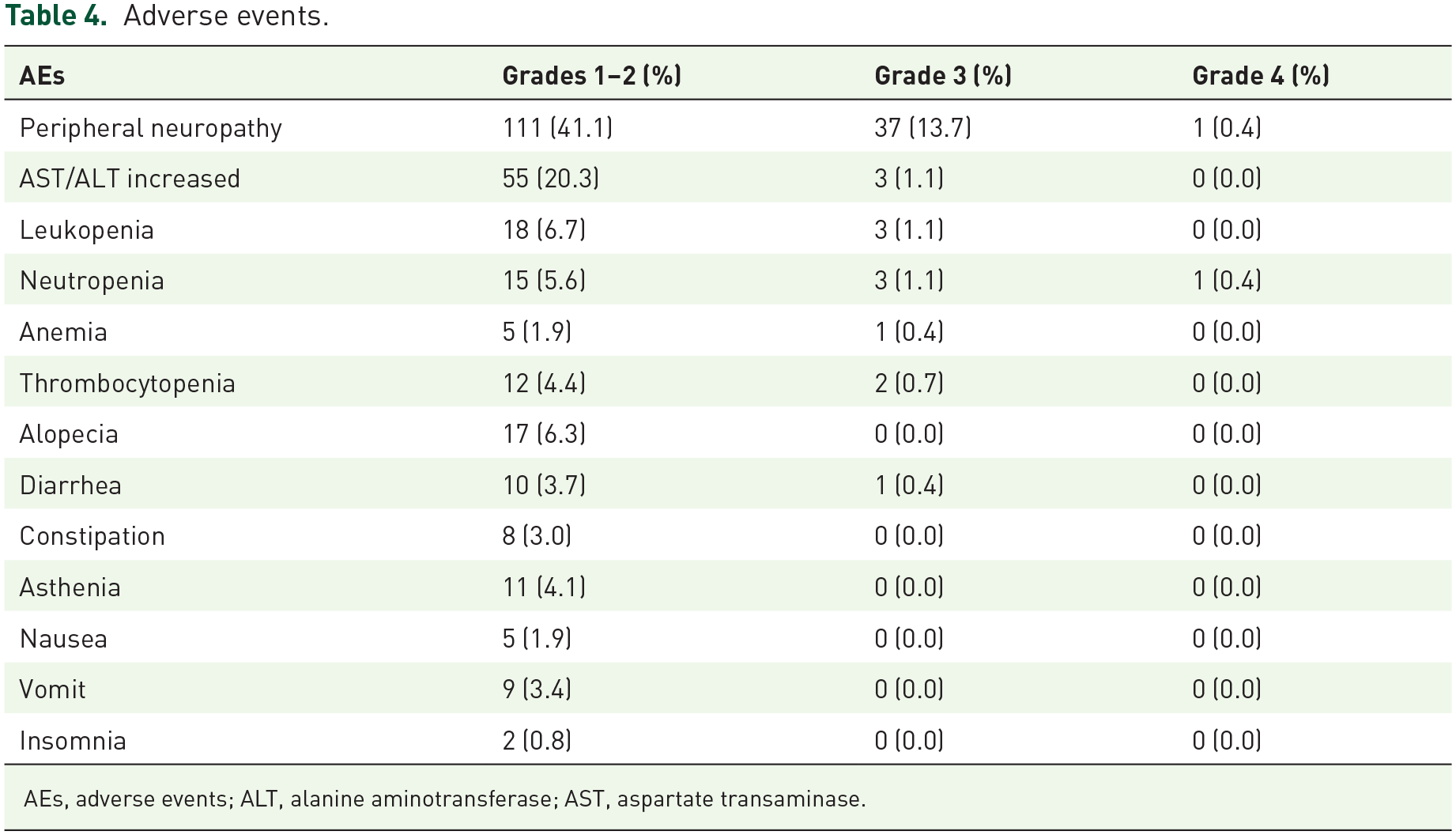
AEs, adverse events; ALT, alanine aminotransferase; AST, aspartate transaminase.
Grade 3 and above peripheral neurotoxicity.

PN, peripheral neurotoxicity.

Kaplan–Meier curve for time to resolution of grade ≥3 peripheral neuropathy (PN).
Table 6 compares the baseline clinicopathological characteristics of patients who developed ⩾grade 3 PN during UTD1 treatment (n = 38) and those who did not (n = 232). Patients with ⩾grade 3 PN were more likely to be older than 50 years (73.7% vs 53.0%, p = 0.017), postmenopausal (71.1% vs 60.3%, p = 0.045), have recurrent disease (94.7% vs 81.5%, p = 0.042), have a history of PN (50.0% vs 25.0%, p = 0.002), or present with baseline unrecovered PN (34.2% vs 12.1%, p < 0.0001). Univariate and multivariate logistic regression analyses further identified IV days 1–5 administration (p = 0.012) and baseline unrecovered PN (p = 0.013) as independent risk factors for developing ⩾grade 3 PN (Table 7).
Comparison of baseline characteristics between patients with ⩾grade 3 PN and those without.

HER2, human epidermal growth factor receptor 2; ICIs, immune checkpoint inhibitors; IV, intravenously; MBC, metastatic breast cancer; PN, peripheral neurotoxicity; TNBC, triple-negative breast cancer; UTD1, utidelone.
Bold values indicate statistical significance with p < 0.05.
Univariate and multivariate logistic regression analyses of factors predicting ⩾grade 3 PN in patients treated with UTD1.

ICIs, immune checkpoint inhibitors; IV, intravenously; PN, peripheral neurotoxicity; UTD1, utidelone.
Bold values indicate statistical significance with p < 0.05.
By the data cutoff in August 2023, 224 patients had discontinued UTD1 treatment. Reasons for discontinuation included disease progression (58.5%), intolerable toxicity (14.4%), and physician decision (10.0%) (Table S9). Among those who discontinued due to AEs, most cases were attributed to PN (11.1%). After stopping UTD1, 157 patients (58.1%) received subsequent anticancer treatments, including chemotherapy (33.3%), endocrine therapy (11.5%), and targeted therapy (31.9%) (Table S10).
Discussion
This multicenter retrospective study explored the efficacy and safety of UTD1 in 270 heavily pretreated Chinese women with MBC. The median PFS and OS of UTD1 were 3.97 and 20.63 months, respectively. Bevacizumab, capecitabine, and platinum were commonly used regimens in combination with UTD1. The administration method of UTD1 was identified as an independent predictor of its efficacy. PN was the most common AE of UTD1.
To our knowledge, this study represents the first large-scale, multicenter, real-world study in China to explore the efficacy and safety of UTD1 in a real clinical setting. Comparing our study with its phase III randomized controlled trial (RCT), both were multicenter studies and included 270 Chinese patients treated with UTD1. While RCTs are considered the gold standard for evaluating medical interventions, they have limitations as they often study subjects under ideal conditions that may not reflect real-world clinical practice. By contrast, real-world studies (RWS) like ours use data and conditions from everyday clinical practice, offering a more realistic view of treatment outcomes. 23
Comparing the patient characteristics of the two populations, we can find that the RWS represents a population with a higher disease burden (>2 metastatic sites: 61.1% vs 50%) and heavier treatment exposure (median treatment lines: 3 vs 2). In addition, elderly patients (⩾60 years old, 22.6% vs 12%) and those with brain metastases (23.7% vs 2%) were more common in the real world. The median PFS of UTD1 in the RWS was 3.97 months, shorter than the 8.44 months reported in the BG01-1323L study. 15 The heterogeneous population and patients’ compliance with treatment might contribute to this difference.
Moreover, our real-world outcomes are broadly comparable to other published RWS of heavily pretreated MBC. This study demonstrated a median PFS of 3.97 months and an ORR of 8.4% for UTD1, similar to our previous multicenter study of eribulin in China, which reported a median PFS of 3.5 months and an ORR of 9.0%. 24 Similarly, Feng et al. conducted a real-world comparative study of eribulin versus UTD1 plus capecitabine, reporting a median PFS of 5.2 months for eribulin and 7.7 months for UTD1 plus capecitabine. 25 However, it is essential to note that the Feng et al. cohort primarily consisted of patients receiving first- or second-line therapy, whereas 72.2% of patients in our study received UTD1 as a third-line or later treatment. In addition, our cohort showed a higher disease burden, with 81.1% of patients having visceral metastases and nearly 24% having brain metastases. These variations in treatment lines, disease severity, and metastatic patterns emphasize the challenges of attaining significant clinical benefits in such a refractory and diverse real-world population. Another study evaluated the efficacy of UTD1 and capecitabine in 43 patients with MBC, 26 but its sample size was too small to demonstrate the complexity of the real-world situation.
In the RCTs, UTD1 was used in combination with capecitabine.13,15,16,26 In the BG01-1323L study, patients who had previously been treated with capecitabine were only allowed to participate if they discontinued capecitabine due to economic problems rather than resistance, and 28 (10%) patients met the criteria. 15 In the RWS, however, the situation is entirely different. 61.5% of patients had received capecitabine treatment after metastasis. Therefore, multiple regimens were used to combine with UTD1 in clinical practice. In patients with HER2-positive MBC, 21 patients received UTD1 combined with anti-HER2 treatment. Among them, one patient received UTD1 combined with inetetamab and pyrotinib, while another received UTD1 combined with inetetamab and neratinib. The IPU study enrolled 47 HER2-positive MBC patients who were treated with UTD1 combined with inetetamab and pyrotinib. 27 Preliminary results of the IPU study presented at the 2023 ESMO meeting demonstrated an ORR of 79.3% and a disease control rate (DCR) of 93.1% for this triplet. Another phase II clinical study (ICU study) reported at the 2023 ASCO meeting, exploring the efficacy of UTD1 in combination with inetetamab and camrelizumab in HER2-positive MBC patients, showed a median PFS of 5.59 months.28,29 In terms of safety, both IPU and ICU studies did not observe any AEs above grade 3.27 –29 A single-arm, multicenter, open-label phase II study of UTD1 combined with pyrotinib for HER2-positive MBC with previous trastuzumab failure is also ongoing. In HER2-negative MBC, a head-to-head study of UTD1 versus docetaxel as first-line chemotherapy (NCT05430399); in TNBC, a phase III study of UTD1 plus carboplatin versus gemcitabine plus carboplatin as first-line chemotherapy is also ongoing. These combinational regimens can also be seen in RWS, indicating that RWSs can genuinely reflect the oncologists’ treatment preferences. In addition, significant unmet medical needs can be seen through RWS, providing important clues for future explorations. Results from these ongoing clinical trials would further validate the effectiveness of these novel combinational regimens.
In 64 MBC patients with brain metastases in our RWS, the CNS-CBR and CNS-ORR of UTD1-based therapy were 28.6% and 10.7%, respectively. Previous studies have shown that the intracranial drug concentration of UTD1 was significantly higher than that of taxane. 30 A phase II clinical study, the UTOBIA-BM study, investigated the efficacy of the combination of UTD1, bevacizumab, and etoposide in treating HER2-negative MBC patients with brain metastasis. In all, 17 patients were enrolled in this study, and the CNS-CBR and CNS-ORR were 91% and 73%, respectively. 21 Compared with patients in the UTOBIA-BM study, those in our study were younger (⩾60 years old, 15.6% vs 21%) and had a higher proportion of previous local therapy (67.2% vs 9%). In the RWS, UTD1 was mainly combined with capecitabine for patients with brain metastases (28.1%), followed by bevacizumab (18.8%). The results of the UTOBIA-BM study show that UTD1 has anti-tumor activity and manageable toxicity in MBC patients with brain metastases. 21 However, since only 11 patients were evaluable for response in that study, its effectiveness and safety still need further validation.
Due to the different survival among different biological subgroups, 31 previous studies have investigated the efficacy of UTD1 across various molecular subtypes. The result indicated that UTD1 combined with capecitabine had significant benefits for PFS and OS in HER2-negative/HR-positive breast cancer patients (compared with capecitabine alone). 16 In this study, there were no statistically significant differences in PFS and OS among patients with HR+, HER2+, and TN subtypes, suggesting that UTD1 had similar anti-tumor activity in different breast cancer subtypes. In addition, in our study, the presence or absence of visceral or liver metastasis did not affect PFS, indicating that UTD1 was effective even for patients with visceral or liver metastasis.
PN was the dose-limiting toxicity of UTD1 and a major concern for oncologists. 16 Our study observed that 55.2% of patients experienced PN, with 14.1% developing ⩾grade 3 PN, lower than the rates reported in the BG01-1323L study (85.4% and 25.1%, respectively). 16 The real-world clinical setting may partly explain this difference. In our study, adverse event data, including PN, were primarily collected through medical records, laboratory results, and patient-reported symptoms during outpatient visits and telephone follow-ups. Although these methods align with routine clinical practice, they are inherently less standardized and less frequent than the structured, protocol-driven monitoring in RCTs. For example, in outpatient settings, neurologic symptoms may be underreported due to variability in patient communication, limited visit time, or differences in clinician documentation. These limitations in real-world monitoring may also help explain the differences observed in the timing of onset and recovery from severe PN. In our study, the median onset time of ⩾grade 3 PN was 3.0 weeks, earlier than the 10.29 weeks reported in BG01-1323L. 16 In addition, 50.0% (19/38) of patients recovered to grade 0–1 PN with a median recovery time of 17.4 weeks, compared to 95.5% and 3.14 weeks in the trial. Patients in real-world practice are typically more heterogeneous and heavily pretreated, with prior cumulative neurotoxic exposure that may lower their threshold for developing severe PN. Less frequent follow-up may also delay the recognition and management of early symptoms, allowing neuropathy to progress more quickly to higher grades and prolong recovery. These factors underscore the challenges of accurately capturing and managing non-hematological toxicities such as PN in retrospective real-world analyses.
In clinical practice, three main methods of administering UTD1 were employed: continuous intravenous infusion over 120 h (CIV 120 h), intravenous administration on days 1–5 (IV days 1–5), and administration on days 1, 8, and 15 (IV days 1, 8, 15). Our study found that the IV days 1–5 method was associated with significantly better PFS compared to the weekly regimen, suggesting a potential pharmacodynamic advantage. However, this method also led to a higher occurrence of ⩾grade 3 PN. The CIV 120 h and IV days 1, 8, and 15 regimens demonstrated a more favorable safety profile, especially for patients with baseline PN or poor tolerance. Therefore, clinical decision-making should consider the patient’s overall condition, disease features, and neurotoxicity risk when selecting an administration method.
This study has several important limitations inherent to its retrospective, real-world design. Data loss and recall bias were inevitable, and adverse event evaluation, especially for non-hematological toxicities such as neurotoxicity, fatigue, and muscle pain, relied on subjective patient reporting, likely leading to underestimation of their incidence. Differences in evaluation frequency across centers may also have affected PFS assessments. Selection bias is unavoidable, as treatment decisions are based on physician judgment, patient preferences, and local practice patterns, resulting in heterogeneous baseline characteristics. The use of various combination regimens and administration schedules further complicates direct comparisons of efficacy and safety outcomes. While this large multicenter study offers valuable real-world insights into UTD1 use in heavily pretreated MBC patients in China, these findings should be interpreted with caution. Future prospective multicenter studies with standardized treatment protocols and systematic follow-up are needed to validate these observations and better guide clinical decision-making.
Conclusion
In summary, UTD1 represents a promising treatment option for heavily pretreated MBC patients, demonstrating satisfactory anti-tumor efficacy, mild myelosuppression, and manageable PN. However, patients with unresolved PN from prior therapies or those receiving UTD1 through IV days 1–5 were more likely to develop severe PN. These findings underscore the importance of individualized treatment planning and vigilant monitoring of neurotoxicity in clinical practice.
Supplemental Material
sj-doc-2-tam-10.1177_17588359251368684 – Supplemental material for Efficacy and safety of utidelone in pretreated patients with metastatic breast cancer in China: a multicenter, real-world study
Supplemental material, sj-doc-2-tam-10.1177_17588359251368684 for Efficacy and safety of utidelone in pretreated patients with metastatic breast cancer in China: a multicenter, real-world study by Shihui Hu, Tengfei Chao, Ning Xie, Quchang Ouyang, Fei Xu, Hong Wang, Yannan Zhao, Chengcheng Gong and Biyun Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-1-tam-10.1177_17588359251368684 – Supplemental material for Efficacy and safety of utidelone in pretreated patients with metastatic breast cancer in China: a multicenter, real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359251368684 for Efficacy and safety of utidelone in pretreated patients with metastatic breast cancer in China: a multicenter, real-world study by Shihui Hu, Tengfei Chao, Ning Xie, Quchang Ouyang, Fei Xu, Hong Wang, Yannan Zhao, Chengcheng Gong and Biyun Wang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
