Abstract
Background:
Pancreatic ductal adenocarcinoma (PDAC) is characterized by poor prognosis and limited therapeutic options, particularly in the metastatic setting. Novel strategies to overcome treatment resistance are highly anticipated and may potentially include the repurposing of existing pharmacologic agents.
Objectives:
To prove the positive effect of angiotensin II receptor blockers (ARBs) or angiotensin-converting enzyme inhibitors (ACEIs) on survival in PDAC.
Design:
A systematic review and meta-analysis were conducted based on Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.
Data sources and methods:
A systematic search of PubMed, Web of Science, and Cochrane databases was conducted to identify studies evaluating survival outcomes in patients with PDAC receiving concomitant treatment with ARBs or ACEIs. Survival outcomes were analyzed with random-effects models using hazard ratios (HRs) and 95% confidence intervals (CIs) extracted from included trials. Studies without sufficient survival data were reviewed qualitatively only.
Results:
A total of 14 studies, comprising a total of 19,287 patients, met the inclusion criteria. Results indicated that the concurrent treatment with ACEIs or ARBs in patients with PDAC was associated with significantly improved overall survival compared to no use (HR = 0.80; 95% CI: 0.76–0.86; p < 0.001). The survival benefit associated with the concomitant ACEI/ARB treatment was observed both in the curative setting (HR = 0.78, 95% CI 0.65–0.93, p = 0.007) and the non-curative setting (HR = 0.83, 95% CI 0.76–0.90, p < 0.001). Three studies without sufficient survival data for meta-analysis reported other potential benefits associated with the ACEI/ARB use.
Conclusion:
Our meta-analysis supports the efficacy of renin–angiotensin system inhibitors as concomitant therapy in patients with PDAC. Prospective randomized clinical trials are warranted to validate ACEI/ARB as adjunctive therapy in well-defined PDAC patient populations.
Plain language summary
Why was this study done?
Pancreatic cancer is one of the deadliest cancers, and there are very few effective treatments. Some common blood pressure medicines, called ACE inhibitors and ARBs, may help fight cancer by affecting how tumors grow.
What did the researchers do?
Researchers looked at 14 studies with more than 19,000 people who had pancreatic cancer. They compared how long people lived depending on whether they were taking these blood pressure medicines or not.
What did they find?
People who took ACE inhibitors or ARBs lived longer on average than those who didn’t. This was true for both early-stage cancer (when surgery was possible) and advanced cancer (when it was not).
What does this mean?
Blood pressure medicines like ACE inhibitors and ARBs might help people with pancreatic cancer live longer. More studies, especially clinical trials, are needed to confirm these results.
Keywords
Introduction
Pancreatic ductal adenocarcinoma (PDAC) ranks among the most aggressive malignancies. 1 According to GLOBOCAN 2020 data, PDAC is the 12th most common cancer type globally and the 7th leading cause of cancer-related mortality in both men and women. 2 Furthermore, the incidence of PDAC is steadily increasing, and it is projected to become one of the three leading causes of cancer-related deaths within the next few years.2,3 Major risk factors for PDAC include smoking, alcohol consumption, diabetes, obesity, chronic pancreatitis, and genetic factors.4,5 Despite recent advancements in treatment, PDAC remains a malignancy with a notably poor prognosis. 3 The global 5-year overall survival (OS) rate for PDAC is estimated to be approximately 10%.6–8 Surgical resection is the only potentially curative treatment 3 ; however, only 10%–20% of patients are eligible for surgery due to the advanced stage at diagnosis. 9 Despite surgical intervention, the 5-year survival rate remains low at 10%–20%,10,11 primarily due to the high propensity for recurrence. 12 In advanced stages, systemic chemotherapy remains the mainstay of treatment, with commonly used regimens including gemcitabine monotherapy, gemcitabine plus nab-paclitaxel (GN), and FOLFIRINOX (5-fluorouracil, leucovorin, irinotecan, and oxaliplatin).3,13 However, due to their limited efficacy, there remains a significant unmet need to refine therapeutic strategies for patients with PDAC.
The pathogenesis of PDAC is significantly influenced by complex interactions between carcinoma cells and the surrounding stromal microenvironment. Tumor microenvironment (TME) consists of vascular, inflammatory, and neural cells, as well as acellular elements such as collagen and other extracellular matrix proteins.14,15 Under physiological conditions, these components maintain pancreatic homeostasis; however, during carcinogenesis, they may contribute to the initiation of a desmoplastic response, thereby promoting tumor cell proliferation, chemoresistance, and metastasis formation. 16
The renin-angiotensin-aldosterone (RAA) system plays a central role in the regulation of blood pressure and electrolyte homeostasis. 17 Beyond its cardiovascular function, the RAA system is also involved in the modulation of cellular metabolism, proliferation, and inflammatory response. 18 A key component of the RAA system is angiotensin II, which has been shown to enhance the proliferation and aberrant activation of stromal cells, thereby promoting the secretion of tumor growth factors.19–21 Emerging evidence suggests that manipulation of the RAA system may exert antitumor effects in pancreatic cancer.22,23 Angiotensin II receptor blockers (ARBs) and angiotensin-converting enzyme inhibitors (ACEIs), two classes of antihypertensive agents, have attracted increasing attention in clinical research as potential repurposed therapies for the management of PDAC.
In a previous retrospective analysis, we demonstrated that the use of ARBs and ACEIs was associated with a significant improvement in OS among our patient cohort, including both those who received radical treatment and those undergoing palliative care. 24 To further validate and corroborate these findings, we undertook a systematic review and meta-analysis of the existing literature on this topic.
Methods
Search strategy
The systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines; the appropriate checklist of guidelines can be found in Supplemental File 1. 25 The electronic databases PubMed, Web of Science, and Cochrane Library were systematically searched for relevant articles published up to January 15, 2025. The main search strategy is outlined in Table 1 in Supplemental File 2. Duplicate records, conference abstracts, case reports and case series, studies without original data (reviews, systematic reviews, meta-analyses, editorials, comments, clinical trial protocols, letters to the editor), preprints, and non-English articles were excluded. All identified studies were initially screened by title and abstract. The search was further supplemented by manual inspection of articles selected during the primary screening. Two reviewers jointly reviewed and resolved any discrepancies that arose during the search process (A.S./P.C.).
Baseline characteristics of the studies included in the meta-analysis.
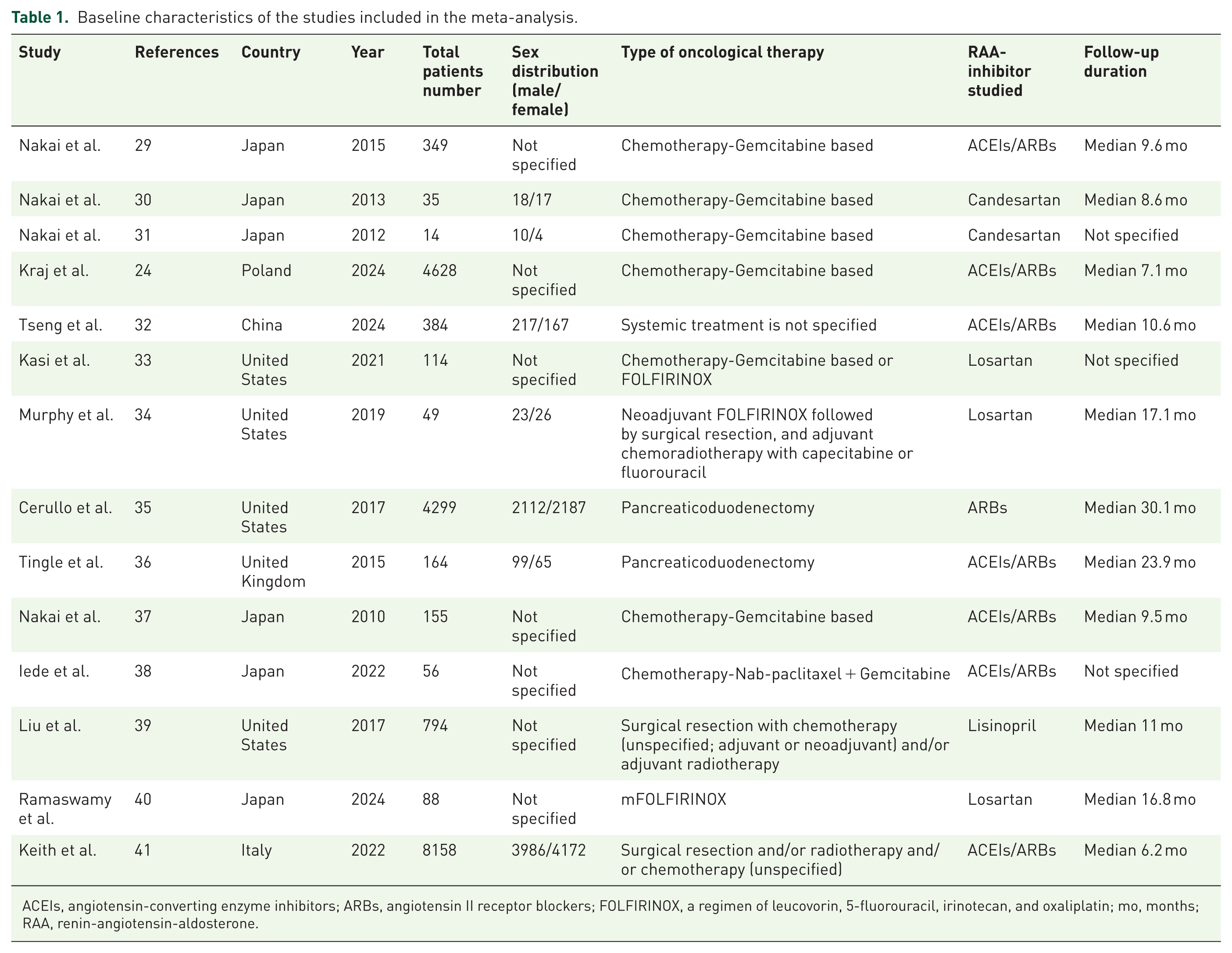
ACEIs, angiotensin-converting enzyme inhibitors; ARBs, angiotensin II receptor blockers; FOLFIRINOX, a regimen of leucovorin, 5-fluorouracil, irinotecan, and oxaliplatin; mo, months; RAA, renin-angiotensin-aldosterone.
Eligibility criteria
The inclusion criteria were as follows: (1) only original studies, (2) studies cover the topic of PDAC patient’s survival in relation to concomitant use of ACEIs or ARBs, (3) reported hazard ratios (HRs) and 95% confidence intervals (CIs) for OS or provided sufficient data to calculate these values, and (4) the subjects were divided into the group receiving concomitant treatment and those not receiving or provided data was sufficient to estimate these groups. Exclusion criteria were: (1) pathological type was not PDAC; (2) not relevant to the prognosis of PDAC; (3) the risk effects and corresponding 95% CI were not provided and available data were not sufficient to calculate them; (4) studies without original clinical data, such as reviews, systematic reviews, meta-analysis, expert opinions, editorial, or comment; (5) clinical trials exclusively evaluating novel strategies among patients with PDAC.
The primary search identified a total of 195 studies (Figure 1). After exclusion of duplicates (n = 39), conference abstracts (n = 26), case reports, and case series (n = 4), studies lacking original data (n = 30), and non-English original articles (n = 3), a total of 90 articles remained for title and abstract screening. Following assessment for eligibility, 14 studies met the inclusion criteria and were included in the final analysis. No additional studies were identified through manual reference list screening.

Flowchart presenting the process of article selection, according to PRISMA guidelines.
Data extraction and quality assessment
Relevant data were extracted from each study, including the author’s name, publication year, country, study design, sample size, details of surgery and chemotherapy, treatment intent, patient survival outcomes (OS, event-free survival, progression-free survival (PFS)), concomitant use of ACEIs/ARBs, tumor characteristics (diameter), and risk factors. Where available, we extracted HRs along with their 95% CI. In cases where HRs were not explicitly provided, we derived them using available study parameters. Quality assessment of observational studies was conducted using the Newcastle–Ottawa Scale (NOS) method, where applicable. 26 For clinical trials, NOS was not utilized; the quality of these trials was considered high based on the standardized criteria typically applied in their design and conduct.
Data analysis
The pooled HR value for OS was calculated using a random-effects model. The employment of a random-effects model as the guiding method directly addresses the needs of our meta-analysis, as studies combined in a meta-analysis exhibit differences in their design and conduct, particularly in the case of PDAC, which can lead to heterogeneous results. Concurrently, it has been demonstrated to be the most effective method for avoiding bias and achieving the most robust results. 27 The meta-analysis was performed based on the generic inverse variance according to each study. The heterogeneity among the included studies was assessed using the Q value and the I2 statistic. A random-effect model was used for all data to reduce the influence of studies’ heterogeneity on the final results. Studies with more than one arm, with the same group of drugs, or in all subgroups, were separately included in the analysis. The sensitivity of each meta-analysis was examined using the leave-one-out method, and the results do not exceed a 10% change in the final HR values. Statistical significance of the pooled treatment effect was considered for the p-values <0.05. Statistical analysis was performed and visualized using Review Manager 5.2 (The Cochrane Collaboration). 28
Results
Characteristics of the included studies
The baseline characteristics of the included studies are summarized in Table 1. We were able to identify a cohort of 19,287 patients, predominantly female (>50%), with a median follow-up duration ranging from 6.2 to 30.1 months. ARBs were analyzed in 13 of 14 studies, with losartan and candesartan being the most common, while ACEIs were assessed in 8 of 14 studies. All of the included studies exhibited high quality, as reflected by the NOS scores of 9 in case of 10 studies and the incorporation of 4 clinical trials. The studies included predominantly European populations. The assessed interventions included curative and palliative therapies; all were analyzed collectively and within subgroups. The HRs for OS were available in 11 of 14 included studies, providing a comprehensive evaluation of the outcomes. The characteristics of the patient groups from the studies included in the analysis are summarized in Table 2. The NOS assessment is summarized in Table 3, with detailed information in Supplemental Table 2.
Characteristics of patient groups in included studies.

, Patients with these diagnoses included in the study.
Newcastle–Ottawa quality assessment scale summary report.
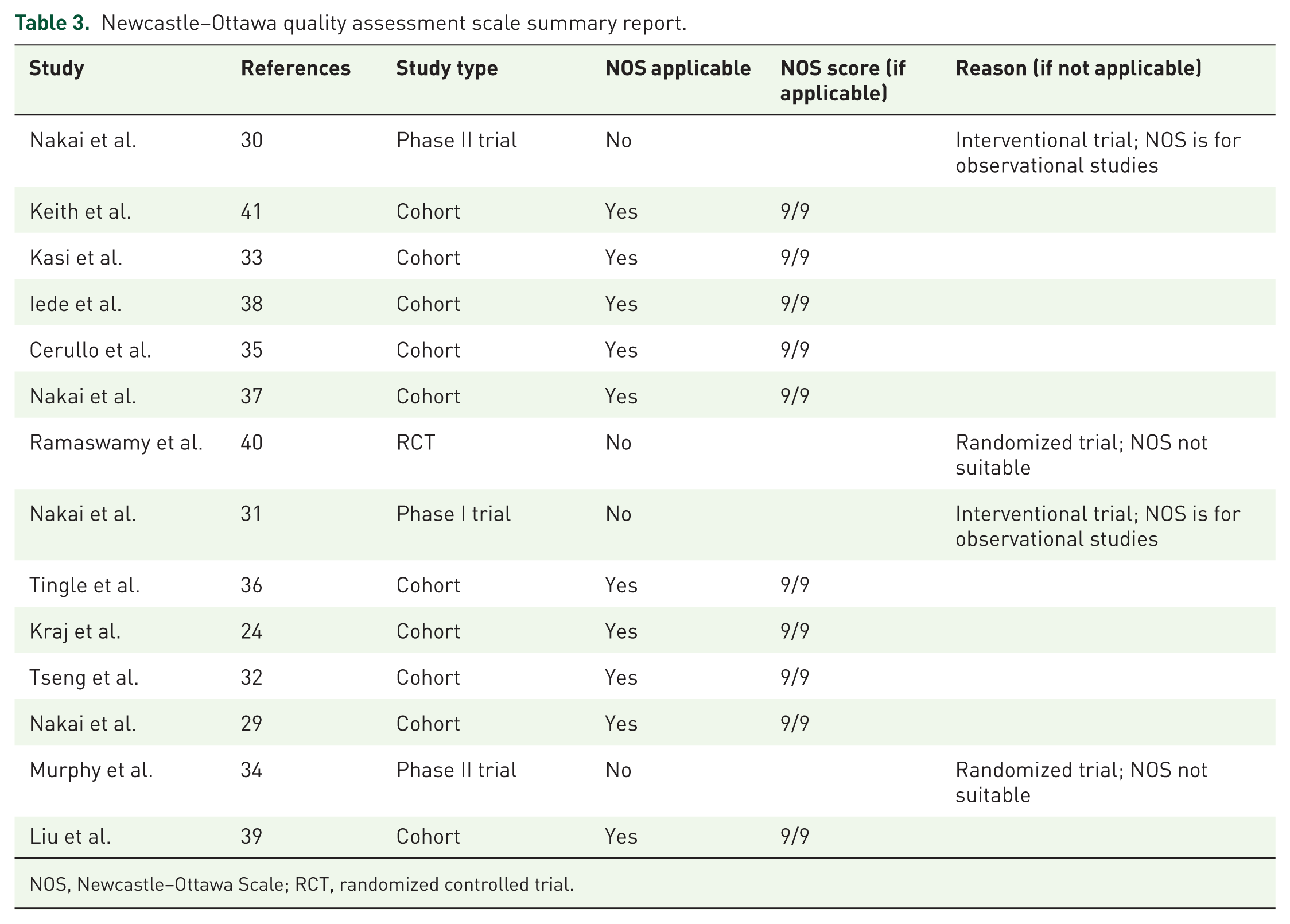
NOS, Newcastle–Ottawa Scale; RCT, randomized controlled trial.
Pooled HR values for all of the studies
By assessing the overall effect, we found that the concomitant use of ACEIs and ARBs in patients with PDAC (including both radically treated patients and those receiving palliative therapy) was significantly associated with improved OS compared to no ACE/ARB treatment (HR = 0.80; 95% CI: 0.76–0.86; p < 0.001). The forest plot illustrating this estimate is presented in Figure 2. The analysis revealed no substantial heterogeneity among the studies (I2 = 31.0%).

Effect of concomitant use of angiotensin-converting enzyme inhibitors and Angiotensin II receptor blockers on overall survival in patients with pancreatic ductal adenocarcinoma.
Pooled HR estimates stratified by treatment intent
Due to substantial prognostic variability in PDAC associated with treatment intent, the studies included in the overall analysis were stratified by treatment type. Curative treatment was defined as radical surgical resection, whereas palliative (non-curative) treatment consisted of systemic therapy without surgery. Separate estimations were conducted for patients treated with curative intent and those receiving palliative care. In the cohort undergoing radical treatment, concomitant use of ACEI/ARB was associated with improved OS (HR = 0.78; 95% CI: 0.65–0.93; p = 0.007; Figure 3). A similar outcome was observed in the palliative care group, where ACEI/ARB treatment was also associated with superior OS (HR = 0.83; 95% CI: 0.76–0.90; p < 0.001; Figure 4).

Pooled HR values for patients treated with curative intent.

Pooled HR values for patients treated with non-curative intent.
Pooled HR values for ARBs in PDAC
To delineate the effect of individual antihypertensive agents on OS in PDAC, additional subgroup analyses were performed. In the cohort receiving only concomitant ARBs, ARB use was associated with a significant improvement in OS compared to no ACEI/ARB treatment (HR = 0.82; 95% CI 0.76–0.88; p < 0.001). The forest plot depicting this estimate is presented in Figure 5.

Pooled HR values for patients treated with concomitant angiotensin receptor blockers.
Pooled HR values for ACEIs in PDAC
Analogous outcomes were observed in patients receiving concomitant treatment with an ACEI. The utilization of ACEI was associated with improvement in OS compared to no ACEI/ARB treatment (HR = 0.77, 95% CI 0.7–0.86, p < 0.001). The forest plot depicting this estimate is presented in Figure 6.

Pooled HR values for patients treated with concomitant angiotensin-converting enzyme inhibitors.
Influence analysis
To further validate our results, we performed a sensitivity analysis. The study by Keith et al. appeared to be the predominant source of the combined HR calculations. Subgroup analysis of the presented calculations led to a reduction in the percentage of patients compared to the entire cohort, resulting in a maximum of 28.0% weight of the effect of the subgroup from the Keith et al. 41 study. Following the exclusion of the Keith et al. 41 study, four of the five analyses retained statistical significance, as illustrated in Figure 7.
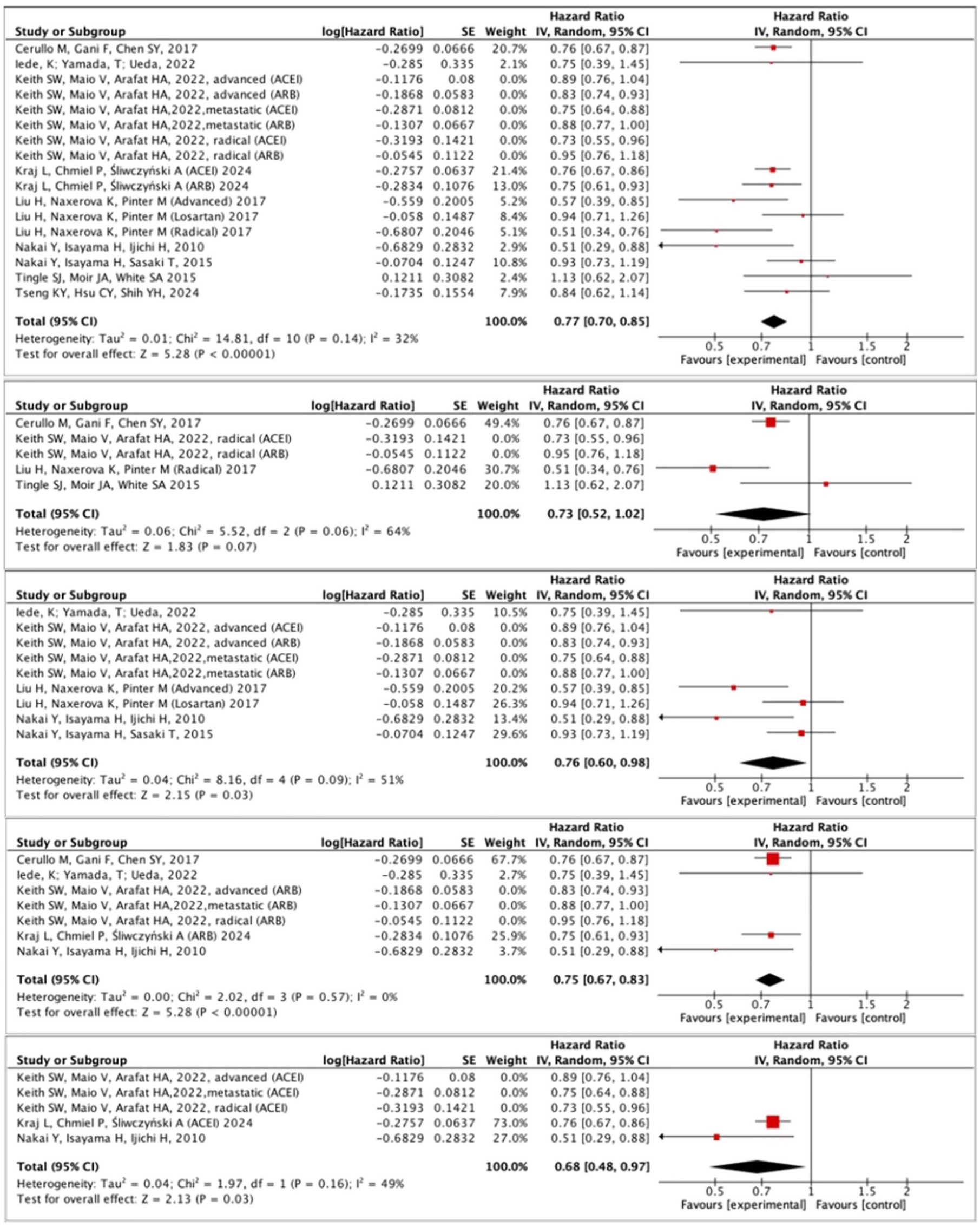
Influence analysis of included studies.
Qualitative analysis of the studies not included in the meta-analysis and additional findings
Several studies met the inclusion criteria for the systematic review but lacked the necessary data for inclusion in the meta-analysis. This primarily pertained to early-phase clinical trials that did not provide comparative measures of the ACEI/ARB efficacy. The phase I GECA1 study demonstrated the safety of the addition of candesartan (16 mg daily) to gemcitabine chemotherapy, reporting a median PFS of 7.9 months and a median OS of 22.9 months, 31 exceeding the historical outcomes for gemcitabine monotherapy. 42 These findings were further supported by the subsequent GECA2 trial, which reported median PFS and OS of 4.3 and 9.1 months, respectively, for the gemcitabine and candesartan combination. 30 In another phase II study involving patients with locally advanced PDAC (i.e., unresectable with conventional surgical techniques), total neoadjuvant FOLFIRINOX combined with losartan (ARB) followed by chemoradiotherapy resulted in the radical resection (R0) rate of 61% among 49 evaluable patients and 88% of patients who underwent resection, substantially surpassing historical R0 resection rates. 34 Conversely, the AFPAC study, which compared modified FOLFIRINOX (mFOLFIRINOX) with and without losartan in locally advanced or metastatic PDAC, did not demonstrate a PFS benefit from losartan (median PFS was 8.44 months in the control group and 7.36 months in the losartan group). In this study, the observed effect was attributed to the study group, as only patients with distant metastases and very advanced disease were included. Furthermore, 22% of patients required discontinuation of losartan at some point due to complications associated with concomitant chemotherapy. 40 In addition, a retrospective analysis failed to show a beneficial effect of losartan on OS or PFS in patients with PDAC, although it lacked detailed outcome measures or statistical data necessary for meta-analysis inclusion, specifically HR and data necessary for its determination. 33
Discussion
PDAC remains one of the most challenging in oncology, with only limited therapeutic progress achieved over the past few decades. 43 Prognosis remains poor: in localized or locoregional disease, long-term survival rates range from 5% to 15%, while in advanced or metastatic cases, 5-year survival rates rarely exceed 5%.6,44,45 Furthermore, it is noteworthy that only a minority of patients (20%–30%) present with localized disease at the time of diagnosis, 46 in whom surgical resection remains the potentially curative treatment. For the majority of patients, systemic chemotherapy—most commonly with classical regimens such as FOLFIRINOX (with conventional or liposomal irinotecan) or GN constitutes the cornerstone of treatment.3,13 Recent advancements in oncology, in particular in immunotherapy and molecularly targeted therapies, have very limited applicability in the therapeutic management of PDAC. Consequently, the mounting demand for novel therapeutic strategies has prompted interest in repurposing existing drugs, many of which were not originally developed for oncological applications. The demographic profile of PDAC patients typically includes older individuals with multiple comorbidities, particularly cardiovascular diseases. Furthermore, certain conditions associated with metabolic syndrome are recognized as risk factors for PDAC. 47 This etiopathogenetic overlap has created an opportunity to investigate the impact of concomitant medications, such as antihypertensive drugs, including ACEIs and ARBs, on treatment outcomes in pancreatic cancer, in patients undergoing either radical or palliative therapy. 48
Our analysis highlights the prognostic significance of ACEIs and ARBs in patients with PDAC. The concomitant use of ACEIs and/or ARBs was associated with a 20% reduction in the mortality risk across the entire patient cohort. A subgroup analysis of five studies involving 2263 patients who underwent radical treatment demonstrated a 22% reduction in mortality associated with the use of these antihypertensive agents. Similarly, in the palliatively treated cohort (comprising 6903 patients), the use of ACEIs and/or ARBs was associated with a 17% decrease in the risk of death. Heterogeneity existed in how these medications were categorized and analyzed. Some studies combined patients receiving either ACEIs or ARBs, while others analyzed them separately based on the specific class of drug administered. To ascertain whether the observed improvement in OS was attributable to a specific class of RAAS-blocking agents, we conducted a stratified analysis based on the type of drug administered. Five studies evaluated ARBs exclusively, including a total of 2159 patients, while three studies focused on ACEIs only, encompassing 3264 patients. The results indicated that both drug classes, when used as monotherapy, were associated with a significant reduction in mortality: 18% for ARBs and 23% for ACEIs. Importantly, the robustness of these findings was supported by the high methodological quality of the included studies, as assessed using the NOS. Heterogeneity across studies was low to moderate, with I² values ranging from 20% to 57%, further validating the consistency and reliability of the observed effects.
Despite clinical evidence, the mechanisms underlying the effects of RAAS-blocking drugs remain incompletely understood and elucidated. Some studies have revealed a significant upregulation of angiotensin type 1 receptor (AT1R) expression in human PDAC specimens in comparison to normal pancreatic tissue, while the expression of angiotensin type 2 receptor (AT2R) was reduced in neoplastic tissue. 49 Furthermore, administration of a selective AT2R agonist has been shown to reduce the growth of murine graft models by inducing apoptosis in PDAC cells. 49 ARBs have been demonstrated to enhance the chemosensitivity of PDAC tumors by suppressing c-Jun expression via inhibition of the Hippo/YAP1/TAZ signaling pathway. 50 Furthermore, ACEIs may enhance the expression of genes associated with T cell and antigen-presenting cell activity, potentially influencing cancer progression. 51 Considering the critical role of desmoplasia within TME in mediating chemoresistance in PDAC, the potential therapeutic benefits of ACEIs and ARBs in this context are noteworthy. 23 Blockade of AT1R has been shown to suppress obesity-induced fibrosis and tumor progression, while enhancing the response to chemotherapy. 52 Furthermore, AT1R inhibition decreases the infiltration of tumor-associated neutrophils and Tregs, while promoting CD8+ T cell infiltration through the suppression of stellate cell activation and downregulation of IL-1β expression. 52 ARBs may also modulate TGF-β activity, resulting in decreased collagen deposition and reduced accumulation of Treg within the TME. 53 In addition, both ACEIs and ARBs may contribute to extracellular matrix remodeling, thereby reducing solid stress and enhancing vascular perfusion. This, in turn, leads to decreased tumor hypoxia and improved distribution and efficacy of anticancer drugs and nanotherapeutics. 51 These mechanisms have been corroborated by evidence from a clinical trial, in which PDAC tumors from patients treated with RAA inhibitors showed a reduction in Tregs, decreased C-FOXP3 levels, and an increase in CD8+ T cells in the TME. 54 In summary, a multitude of direct and indirect biological effects may potentially contribute to improved clinical outcomes in PDAC patients exposed to RAA inhibitors. Further research is warranted to validate these findings.
Notwithstanding the promising therapeutic potential of incorporating ACEIs and ARBs into the treatment of patients with PDAC, our study is not without limitations. A significant limitation is the inconsistency in reported clinical endpoints across the included studies. Of the 14 studies analyzed, 11 provided complete data on OS, two studies lacked sufficient data to derive HR for OS, and one study reported PFS only as its endpoint. In addition, the heterogeneity of the included studies presents a challenge, as the topic has been explored primarily in small, single-center retrospective analyses and early-phase clinical trials. The diversity among studies is also reflected in the stage of disease and treatment intent, with both metastatic and radically treated patients being assessed. However, to mitigate the impact of clinical heterogeneity, we performed additional stratified analyses. In contrast to the aforementioned heterogeneity, the homogenous study area also appears to be a limitation. Of the 14 studies included in the analysis, 13 originated from Europe, the United States, or Asia. The analysis was limited by the absence of objective data from Africa or South America, which resulted in a study population that was constrained in its generalizability. Another notable limitation is the absence of comprehensive clinical data in several studies, particularly regarding tumor location, chemotherapy regimens, and surgical interventions, which hinders the ability to accurately stratify patients. Finally, interpretation of the results is further complicated by the fundamental question of whether the benefits of concomitant use of ACEIs/ARBs arise from their direct cellular effects on the tumor, or rather from improvements in the patients’ overall condition due to better management of comorbidities such as hypertension, diabetes, or heart failure. In summary, to fully elucidate the impact of RAA inhibitors on the prognosis of patients with PDAC, well-designed clinical trials are necessary, specifically recruiting patients without significant comorbidities or prior baseline use of RAA inhibitors, to minimize confounding factors and reduce bias by association. It is, therefore, critical to await the results of studies such as NCT03563248 to validate these findings.
Conclusion
A robust correlation has been observed between the concurrent administration of ACEIs or ARBs and improved survival in patients with PDAC, in both early and advanced stages of the disease. While several potential mechanisms underlying this effect have been proposed, a significant gap remains in mechanistic research exploring how modulation of the RAA system influences the phenotype and pathophysiology of PDAC cells and their TME. Moreover, broader incorporation of these agents into clinical practice requires further refinement of available evidence, ideally through randomized, prospective clinical trials employing carefully designed eligibility criteria and stratification methods to mitigate potential sources of bias. Concurrently, several clinical trials are underway, and the results of these studies are eagerly awaited to further refine our understanding of this complex subject. In summary, the findings of our meta-analysis provide evidence supporting the clinical benefit of ACEIs and ARBs in patients with PDAC and affirm their practical and safe applicability in routine oncologic care.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251404266 – Supplemental material for Impact of concomitant use of renin–angiotensin system inhibitors on survival in patients with pancreatic ductal adenocarcinoma: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251404266 for Impact of concomitant use of renin–angiotensin system inhibitors on survival in patients with pancreatic ductal adenocarcinoma: a systematic review and meta-analysis by Leszek Kraj, Paulina Chmiel, Alicja Skrobucha, Hanna Grabowska, Łukasz Szymański, Piotr T. Wysocki and Michał Peller in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251404266 – Supplemental material for Impact of concomitant use of renin–angiotensin system inhibitors on survival in patients with pancreatic ductal adenocarcinoma: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359251404266 for Impact of concomitant use of renin–angiotensin system inhibitors on survival in patients with pancreatic ductal adenocarcinoma: a systematic review and meta-analysis by Leszek Kraj, Paulina Chmiel, Alicja Skrobucha, Hanna Grabowska, Łukasz Szymański, Piotr T. Wysocki and Michał Peller in Therapeutic Advances in Medical Oncology
