Abstract
Background:
Neoadjuvant trastuzumab and pertuzumab combined with carboplatin and taxane (TCbHP) is the standard treatment for human epidermal growth factor receptor 2 (HER2)-positive breast cancer. However, limited clinical data and efficacy biomarkers for TCbHP have been reported in Chinese HER2-positive breast cancer patients.
Objectives:
This study aimed to observe the pathological complete response (pCR) rate in the cohort, with an exploratory analysis of efficacy-related biomarkers in a subset of patients.
Design:
This was a prospective, observational, non-interventional cohort study.
Methods:
Patients with HER2-positive breast cancer treated with TCbHP neoadjuvant therapy were prospectively collected. Exploratory genomic and transcriptomic biomarker analyses were performed in a subset of patients with available baseline tumour specimens retrospectively collected.
Results:
A total of 252 patients with a median age of 48 years were enrolled. Patients with stage III were 69.4% (175/252), and clinical N3 patients accounted for 24.6% (62/252). Patients with hormone receptor (HR) positive were 62.7% (158/252). Total pCR rate was 55.2% (139/252). HR-negative and HR-positive rates were 72.3% (68/94) and 44.9% (71/158), respectively. Among neoadjuvant taxanes, including paclitaxel, docetaxel, and nab-paclitaxel, the pCR rates were 50.0% (57/114), 50.0% (41/82), and 73.2% (41/56), respectively. Multivariate logistic regression analyses showed that HR negativity, receiving nab-paclitaxel, HER2 3+, and cT1–2 were independent predictive factors of high pCR. Genomic and transcriptomic analyses were performed on baseline tumour specimens from 40 patients. Genomic analysis revealed lower pCR rates in patients with PIK3CA mutations (odds ratio = 13.47, p = 0.025) and SPOP amplification (p = 0.047) than in wild types. Transcriptomic analysis revealed that higher pCR rates were associated with elevated ERBB2 (p = 0.004) and CDK12 (p < 0.001) mRNA.
Conclusion:
Neoadjuvant trastuzumab and pertuzumab with carboplatin-based chemotherapy is the recommended regimen for Chinese patients with HER2-positive breast cancer, and nab-paclitaxel may be an optimal alternative taxane for TCbHP regimens. PIK3CA mutations may be predictive biomarkers for poor efficacy.
Introduction
Breast cancer is the most common cancer and the leading cause of cancer-related deaths among women worldwide. 1 Human epidermal growth factor receptor 2 (HER2)-positive breast cancer is a phenotype accounting for 15%–20% of breast cancer cases and is aggressive and linked to disease recurrence and unfavourable prognoses. 2 Over the last decades, the treatment landscape for patients with HER2-positive breast cancer has evolved with the advent of targeted agents. 3
Trastuzumab and pertuzumab, in combination with chemotherapy, have emerged as standard regimens of neoadjuvant therapy, which has improved the pathological complete response (pCR) rate and survival for early HER2-positive breast cancers. In the Neosphere study, the pCR rate was significantly higher in the trastuzumab + pertuzumab + docetaxel (THP) group than in the trastuzumab + docetaxel (TH) group (45.8% vs 29%, p = 0.0141), which was established as the cornerstone of trastuzumab and pertuzumab treatment in neoadjuvant therapy. 4 The PEONY study further validated the efficacy and safety of the THP regimen as neoadjuvant therapy for HER2-positive breast cancer in the Asian population. 5 The TRAIN-2 and TRYPHAENA studies demonstrated that the combination of trastuzumab and pertuzumab with platinum-containing chemotherapy was comparable to the combination with anthracycline-containing chemotherapy.6,7 Given the cardiotoxicity and risk of secondary leukaemia associated with anthracyclines, a combination regimen of taxane, carboplatin, trastuzumab, and pertuzumab (TCbHP) has emerged as the preferred and most commonly used neoadjuvant regimen for HER2-positive breast cancer. Although international trials have established its efficacy, real-world data from China are limited. Since pertuzumab entered the Chinese market in 2019, a large sample of studies has been lacking.
Moreover, not all patients with HER2-positive breast cancer benefit from neoadjuvant therapy, with nearly half failing to achieve pCR. Previous research has indicated that patients with elevated HER2 expression levels (HER2 immunohistochemistry (IHC) 3+), as well as those who are oestrogen receptor (ER)- and progesterone receptor (PR)-negative before treatment, are more likely to have a higher rate of pCR. 8 Recent studies have consistently demonstrated that higher levels of tumour-infiltrating lymphocytes (TILs) and other immune-related gene expression signatures are independently associated with improved prognosis in patients with early-stage HER2-positive breast cancer receiving anti-HER2 therapy.9–12 However, there is a lack of effective biomarkers at the genomic and transcriptomic levels to assess the efficacy of the TCbHP neoadjuvant regimen. The exploration of biomarkers to evaluate the efficacy of neoadjuvant therapy is of utmost importance, as it will facilitate the search for novel strategies to overcome resistance and have a substantial impact on the individualised treatment of HER2-positive breast cancer.
This study aimed to observe the efficacy of neoadjuvant treatment with TCbHP in Chinese patients with HER2-positive breast cancer and identify potential predictive biomarkers of treatment response.
Methods
Patients
This was a prospective, observational, non-interventional cohort study. Patients with HER2-positive breast cancer who underwent neoadjuvant therapy between August 2019 and October 2022 at three hospitals (Cancer Hospital of the Chinese Academy of Medical Sciences, Huanxing Cancer Hospital in Beijing, and Sanhuan Cancer Hospital in Beijing) were included. The inclusion criteria were as follows: (1) pathologically diagnosed HER2-positive (HER2 3+ or HER2 2+ fluorescence in situ hybridisation (FISH)+) breast cancer, (2) clinical stage I–III, (3) received at least four cycles of neoadjuvant TCbHP, and (4) available postoperative pathological results. The exclusion criteria were as follows: (1) stage IV disease at initial diagnosis, (2) concurrent bilateral breast cancer with different subtypes, and (3) other malignant tumours.
Baseline tumour specimens were retrospectively collected for biomarker analysis when available.
Neoadjuvant regimens and data collection
All patients received neoadjuvant chemotherapy with TCbHP administered every 3 weeks for six cycles. The regimen consisted of trastuzumab (loading dose 8 mg/kg, then 6 mg/kg), pertuzumab (loading dose 840 mg, then 420 mg), taxane (paclitaxel 175 mg/m2, docetaxel 75 mg/m2, or nab-paclitaxel 220 mg/m2), and carboplatin (AUC 5). PEG-recombinant human granulocyte colony-stimulating factor support was administered to the patients. The clinical response was evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST 1.1) every two cycles. After the last neoadjuvant chemotherapy session, patients underwent breast and axillary surgery. Adjuvant radiotherapy was administered according to the specialist physicians. Endocrine therapy was permitted in patients with hormone receptor (HR) positivity.
Clinical and pathological data, including age, menstrual status, preoperative clinical TNM stage, histological grade, ER status, PR status, HER2 expression, Ki67 index, neoadjuvant treatment regimen, adverse events, breast and axillary surgery method, postoperative radiotherapy, and postoperative endocrine therapy, were collected. Patients were followed up via telephone or medical records. Missing data were excluded from the final analyses.
ER and PR expression was determined by IHC staining. ER and/or PR ⩾1% were defined as HR positive.13,14 HER2 status was assessed using IHC or FISH. HER2 IHC 3+ or IHC 2+ FISH+ was defined as HER2 positive. 15
Biomarker profiling
Tumour specimens from core needle biopsies were retrospectively collected as formalin-fixed, paraffin-embedded (FFPE) tissue from patients at baseline. Biomarker analysis was an exploratory hypothesis-generating investigation.
Genomic DNA was extracted from FFPE tissue using a QIAamp DNA FFPE Tissue Kit (Qiagen, Hilden, Germany). A Qubit dsDNA HS (High Sensitivity) Assay Kit (Invitrogen, Carlsbad, CA, USA) was used to assess the extracted DNA concentration. DNA samples were quality-checked by agarose gel electrophoresis to ensure they were high-molecular-weight and non-degraded. Only samples with a DNA concentration ⩾10 ng/µL, an OD 260/280 ratio between 1.8 and 2.0, and minimal degradation (DNA Integrity Number ⩾ 7.0) were used for library preparation. A Covaris M220 Ultrasonicator (Covaris, Wobum, MA, USA) was used to cut DNA concentrations to 200–250bp in size. Subsequently, probe capture, magnetic bead isolation and purification, and Polymerase Chain Reaction amplification were performed. Sequencing was performed using the OncoScreen Plus (Illumina, Hayward, CA, USA) high-throughput sequencing platform, which contained 520 genes highly relevant to the development, biology, and personalised treatment of solid tumours.
Samples were extracted and separated to obtain total RNA. The purity, quantity, and integrity number of the extracted RNA were determined. RNA quality was rigorously evaluated using an Agilent Bioanalyzer. Only samples with an RNA Integrity Number (RIN) ⩾ 7.0 were considered suitable for RNA sequencing. We sequenced each library to a minimum depth of 30 million paired-end reads per sample. We used FastQC (v0.12.1) to assess raw read quality. Reads with adapters and low-quality bases (Phred score < 20) were trimmed using Trimmomatic (v0.39). Ribosomal RNA (rRNA) was removed using the KARA RiboErase HMR kit (Roche). RNA libraries were prepared using the KAPA Stranded mRNA-Seq Library Preparation Kit (Roche, Basel, Switzerland). The purified mRNA was fragmented using a fragmentation reagent. Using random hexamer primer and reverse transcriptase, the first strand of cDNA was synthesised from the mRNA template. The second strand of cDNA was then synthesised using DNA polymerase and RNase H. The second strand synthesises and deletes the mRNA to produce double-stranded cDNA. After the double-stranded cDNA was purified by the AMPureXP system, the 3′ end was added with the A tail. The double-stranded cDNA with a joint was purified. The library was then amplified. Computer sequencing was performed after quality inspection.
Multiple testing corrections were applied to the genomic and transcriptomic analyses.
Data processing
Quality control was carried out on the captured sequencing data of the target sequence. After removing low-quality data, Burrows-Wheeler Aligner 0.7.10 (BroadInstitute, Cambridge, MA, USA) was used to compare the sequencing data with the human genome (hg19). 16 Genomic analysis Toolkit 3.2 and VarScan 2.4.3 were used for local alignment optimisation, repeat labelling, and variant invocation.17,18 Tissue samples were compared with their own paracancerous tissue controls to identify somatic variation. DNA translocation analysis was performed using Factera 1.4.3. 19 For whole transcriptome sequencing data, Cutadapt software v1.9.3 was used to decalculate the original reads. Decalculated trimmed reads from each sample were compared to the human pre-mRNAs database. The transcripts per million (TPM) method was used for standardisation.
Study endpoint
The primary objective was to observe the pCR rate with the TCbHP regimen. pCR was defined as ypT0N0 or ypTisN0, with no residual invasive carcinoma in the breast or lymph nodes. The secondary objective was to identify potential predictive biomarkers of treatment response.
This study was conducted according to the STROBE guidelines statement 20 (Table S1).
Statistical analysis
A logistic regression model was used for univariate analysis. Variates with p < 0.05 in the univariate analysis were considered for inclusion in the multivariate model. R package ‘limma’ was used for the analysis of gene expression differences. A fold change ⩾2 or ⩽1/2 was used as screening criteria for differential genes. R package ‘fgsea’ was used for pathway enrichment analysis. Pathways including KEGG and HALLMARK databases were used as annotation pathways. The ESTIMATE algorithm was used to calculate the Immuno Score, Stromal Score, and ESTIMATE Score. 21 The X-cell algorithm was used to analyse 64 types of tumour microenvironment infiltrating cells. 22 All tests were two-tailed, and p < 0.05 was considered a statistically significant difference. All statistical analyses were conducted using IBM SPSS (version 25.0, Armonk, NY, USA) and R (version 4.0.3, Vienna, Austria). GraphPad Prism (version 8.0, San Diego, CA, USA) was used to draw visual graphs.
Results
Patient characteristics
A total of 252 HER2-positive breast cancer patients receiving TCbHP neoadjuvant therapy at stages I–III between August 2019 and October 2022 at three hospitals were enrolled. The flowchart is shown in Figure 1. Among 325 patients with stage I–III HER2-positive breast cancer who received neoadjuvant therapy, 287 were treated with the TCbHP regimen, whereas the remaining 38 received other regimens. A total of 252 patients were included in the final analysis, after excluding 35 patients for the following reasons: 7 did not undergo surgery, 15 had incomplete clinical or pathological records, 6 were lost to follow-up, 4 had bilateral breast cancer, and 3 had other malignancies.
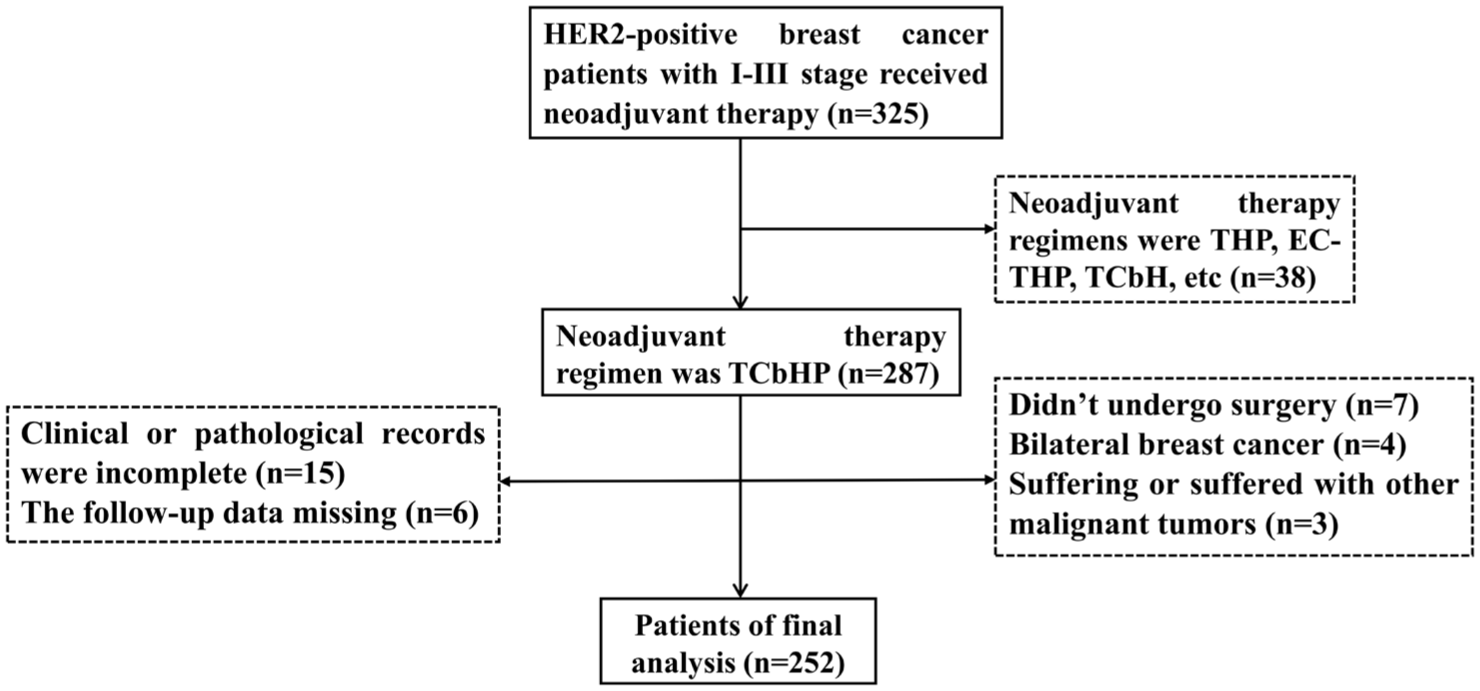
Flowchart of patient screening.
The baseline characteristics of the 252 patients are shown in Table 1. The median age of the patients was 48 years (range, 20–72 years). Two hundred eleven (83.7%) patients had lymph node metastasis (cN+), and clinical N3 patients accounted for 24.6% (62/252). Patients with stage III were 69.4% (175/252). The HR was positive in 158 patients (62.7%). The taxane regimens of paclitaxel, docetaxel, and nab-paclitaxel were administered to 114, 82, and 56 patients, respectively.
Patient characteristics.
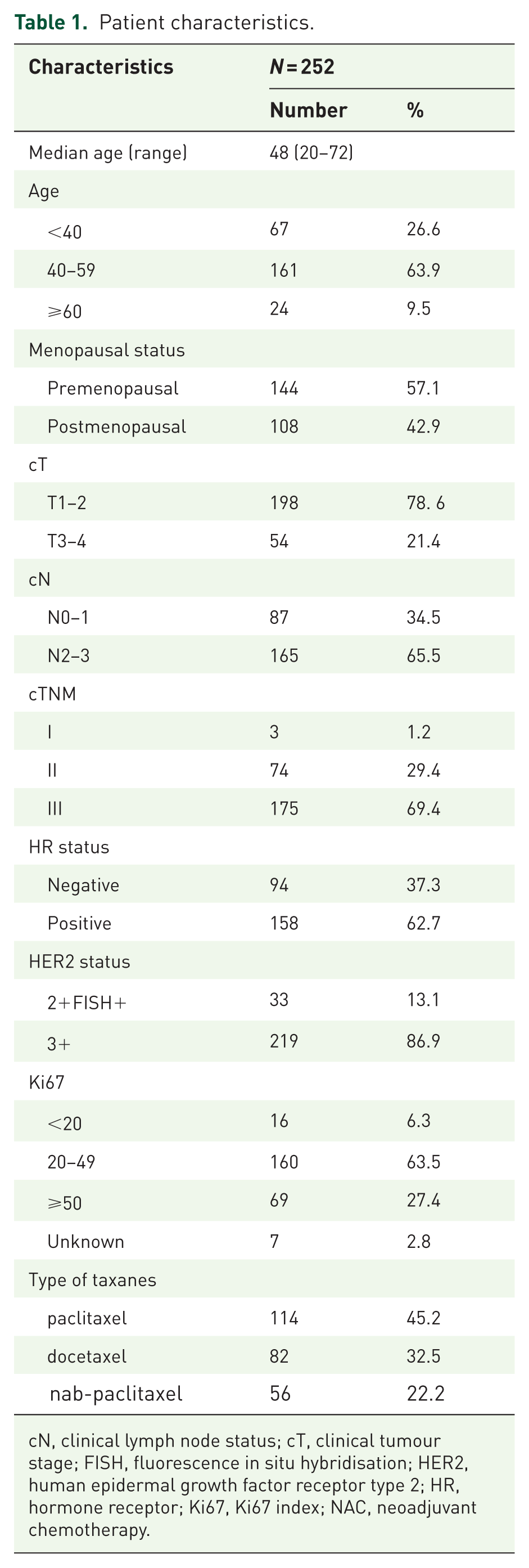
cN, clinical lymph node status; cT, clinical tumour stage; FISH, fluorescence in situ hybridisation; HER2, human epidermal growth factor receptor type 2; HR, hormone receptor; Ki67, Ki67 index; NAC, neoadjuvant chemotherapy.
A total of 252 patients completed at least four cycles of neoadjuvant therapy, and 242 patients completed six cycles of neoadjuvant therapy.
Pathological efficacy
As shown in Figure 2(a), 139 (55.2%) of 252 patients achieved pCR. Among 94 patients with HR-negativity, 68 (72.3%) achieved pCR. Of 158 patients with HR-positivity, 71 (44.9%) achieved pCR. The pCR rate in the HR-negative group was significantly higher than that in the HR-positive group (χ2 = 17.894, p < 0.001, Figure 2(b)). Of 219 patients with HER2 3+, 131 (59.8%) achieved pCR, whereas of the 33 patients with HER2 2+FISH+, only 8 (24.2%) achieved pCR (χ2 = 14.674, p < 0.001).

pCR rates: (a) overall, (b) by HR status, and (c) by taxane type.
Among the 82 patients who received docetaxel, 41 (50.0%) achieved pCR. Similarly, 57 of the 114 patients (50.0%) treated with paclitaxel achieved pCR. In contrast, 41 of 56 patients (73.2%) treated with nab-paclitaxel achieved pCR, which was significantly higher than that achieved with paclitaxel or docetaxel (χ2 = 8.289, p = 0.004 and χ2 = 7.437, p = 0.006, respectively, Figure 2(c)), while no significant difference was observed between paclitaxel and docetaxel (χ2 = 0, p = 1.000).
Univariate analysis showed that cT1–2, HR-negativity, HER2 3+, and nab-paclitaxel treatment were associated with higher pCR rates (Figure 3). Factors such as age, menstrual status, cN stage, and Ki67 expression were not significantly correlated with pCR (p > 0.05). Multivariate logistic regression confirmed that cT1–2, HR-negativity, HER2 3+, and nab-paclitaxel treatment were significant predictors of pCR (Table 2).

Univariate analysis of clinical predictors of pCR.
Multivariate analysis of predictive factors for pathological complete response.

CI, confidence interval; cN, clinical lymph node status; cT, clinical tumour stage; FISH, fluorescence in situ hybridisation; HER2, human epidermal growth factor receptor type 2; HR, hormone receptor; OR, odds ratio.
With a median follow-up of 16.4 months (range 2.9–38.9 months), six recurrences were documented: one local breast relapse and five distant metastases (two bone, one brain, one lung, and one mediastinal lymph node). The 2-year disease-free survival rate in the cohort was 96.4%.
Safety
The most common haematological adverse events were anaemia (36.1%), neutropenia (21.8%), and leukopenia (19.4%). Alopecia (53.2%), nausea (34.9%), elevated alanine aminotransferase/aspartate aminotransferase levels (23.8%), and fatigue (22.2%) were the most frequent non-haematological adverse events. The most common grade 3 or 4 adverse event was neutropenia (9.9%), followed by leukopenia (6.3%) and thrombocytopenia (3.2%). The dose of taxanes was reduced in 22 patients (8.7%), and 2 patients (0.8%) discontinued because of poor tolerance. Carboplatin dose was reduced in 42 patients (16.6%), and 6 patients (2.4%) discontinued because of poor tolerance.
In terms of cardiac safety, only 2.4% of the patients had a left ventricular ejection fraction (LVEF) decrease ranging from 10% to 15%. No patients exhibited an LVEF decrease of ⩾15% or developed symptomatic heart failure.
Genomic analysis
High-throughput genomic sequencing of baseline tissue specimens was performed for 40 patients with early HER2-positive breast cancer. The high-frequency variation genes are as follows: ERBB2 92.5%, TP53 87.5%, CDK12 70.0%, PIK3CA 30.0%, RARA 27.5%, PPM1D 25%, TOP2A 20.0%, KMT2C 20.0%, GNAS 20.0%, ZNF217 20.0%, KMT2D 17.5%, ROS1 17.5%, HOXB13 17.5%, SPOP 15.0%, and GATA3 15.0% (Figure S1). Of the 39 patients who underwent surgery, 19 achieved pCR and 20 non-pCR. No significant differences were observed in clinical and pathological features between the two groups (Table S2).
Patients with PIK3CA mutations had a lower pCR rate than those with the PIK3CA wild type (p = 0.048; after adjustment with HR, p = 0.025, odds ratio (OR) = 13.47, 95% confidence interval (CI): 1.39–130.91). Patients with SPOP amplification exhibit a lower pCR rate than those with the SPOP wild type (p = 0.047, Figure 4).

Association between baseline gene mutations and pCR.
Homologous recombination deficiency (HRD) was calculated using the BRIDGEscore. There were three patients with an HRD score ⩾42 (positive), with two in the pCR group and one in the non-pCR group. The median HRD scores of the pCR group and the non-pCR group were 20 and 23, respectively, and were not significantly different (p = 0.983, Figure S2). The median tumour mutation burden was 4.0 muts/Mb in the pCR group and 6.0 muts/Mb in the non-pCR group, with no statistically significant difference between the two groups (p = 0.506, Figure S2).
Transcriptomics analysis
RNA was extracted from all 40 samples. A total of 28 patients with qualified RNA quality were successfully sequenced using the full transcriptome, and 1 patient had not yet undergone surgery. Twenty-seven patients were included in the analysis: 15 in the pCR group and 12 in the non-pCR group. As shown in Figure 5, there were 20 downregulated and 36 upregulated genes in the pCR group compared with those in the non-pCR group. The downregulated genes were SFRP4, ACTG2, KRT17, C7, KRT14, KRT5, etc., while the upregulated genes were CDK12, PSMB3, S100P, CWC25, LASP1, RPL23, etc.

Heat map of baseline gene expression profiles and pCR.
Pathway enrichment analysis
Pathway enrichment analysis of the differential genes between the pCR and non-pCR groups is shown in Figure S3. Using the pathways included in the KEGG database as annotation pathways, the results showed that the Toll-like receptor signalling pathway and antigen presentation responses were active in the pCR group. The Wnt signalling pathway was active in the non-pCR group. The pathways included in the HALLMARK database were used as annotation pathways, and the results showed that cell proliferation-related signalling pathways (such as G2M checkpoint and MYC targets V1) and interferon-γ were active in the pCR group. The wnt/β-catenin signalling pathway and epithelial-mesenchymal transition (EMT) were active in the non-pCR group.
Analysis of mRNA expression and immune infiltration
We analysed the mRNA expression of high-frequency mutated genes, and the results are shown in Figure S4. High expression of ERBB2 mRNA (p = 0.004) and CDK12 mRNA (p < 0.001) in high-frequency mutated genes correlated with a high pCR rate.
The ESTIMATE algorithm was used to calculate the immune microenvironment score for immune infiltration assessment. The Immune Score was defined as the proportion of immune cell components in the tumour tissue, and the Stromal Score was defined as the proportion of stromal components in the tumour tissue. The ESTIMATE Score was defined as the sum of the Immune Score and Stromal Score. There were no significant differences between the non-pCR and pCR groups in the ESTIMATE Score (p = 0.943), Immune Score (p = 0.943), and Stromal Score (p = 0.905) (Figure S5).
Tumour-infiltrating cells were assessed using the X-cell algorithm. The X-cell algorithm includes 64 cellular features related to the tumour immune microenvironment, including innate and adaptive immune cells, haematopoietic progenitor cells, epithelial cells, and extracellular matrix cells. The results are shown in Figure 6. High expression of CD8 effector memory T cells (CD8Tem) (p = 0.008), plasmacytoid dendritic cells (pDC) (p = 0.012) and T helper 2 cells (Th2) (p = 0.022) was more likely to achieve pCR after neoadjuvant therapy, while low expression of fibroblasts (p = 0.014) and hematopoietic stem cells (HSC, p = 0.034) was associated with pCR.

Association between immune infiltrating cells and pCR. (A) CD8 effector memory T cells and pCR, (B) Fibroblasts and pCR, (C) Hematopoietic stem cells and pCR, (D) Plasmacytoid dendritic cells and pCR, (E) T helper 2 cells and pCR, (F) B cells and pCR, (G) Macrophages and pCR, (H) Natural Killer T cells and pCR, and (I) T helper 1 cells and pCR.
Discussion
Our findings support that neoadjuvant trastuzumab and pertuzumab combined with carboplatin-based chemotherapy is the recommended regimen for Chinese patients with HER2-positive breast cancer, with nab-paclitaxel representing the preferred taxane option within the TCbHP regimens. PIK3CA mutations may serve as predictive biomarkers of diminished therapeutic efficacy.
Previous studies have indicated that the pCR rate for the combination of trastuzumab and pertuzumab with single or double chemotherapy ranges from 39.3% to 68%.5–7 In our study, the pCR rate for the combination of trastuzumab and pertuzumab along with taxanes and carboplatin was 55.2%, which was comparable to that of the TCbHP group in the KRISTINE study (56%) and TRYPHAENA study (51.9%).7,23 Notably, the pCR rate in our study was slightly higher than that of the THP group in the NeoSphere study (45.8%) and PEONY study (39.3%). In addition, the taxane of the NeoSphere, TRYPHAENA, KRISTINE, and PEONY studies all adopted docetaxel, whereas our study incorporated three different taxanes (paclitaxel, docetaxel, and nab-paclitaxel).
Taxanes represent a diverse range of therapeutic options in the clinical setting. In this study, we observed that nab-paclitaxel achieved a pCR rate of 73.2%, which was significantly higher than those achieved with docetaxel (50.0%) and paclitaxel (50.0%). The GeparSepto-GBG 69 study, which focused on neoadjuvant treatment for early breast cancer, showed that nab-paclitaxel increased the pCR rate (38% vs 29%, p = 0.00065) and the 4-year invasive disease-free survival rate (84.0% vs 76.3%, p = 0.002) compared with paclitaxel. 24 However, no significant differences were observed in the HER2-positive subgroup. In the WSG-ADAPT HR+/HER2- study, for high-risk HR+/HER2- early breast cancer, the weekly nab-paclitaxel regimen was compared with dose-dense solvent-based paclitaxel (followed by sequential dose-dense epirubicin plus cyclophosphamide), showing a significant increase in pCR rates (20.8% vs 12.9%, p = 0.002) and improving 5-year survival (84.9% vs 81.7%, p = 0.035). 25 In the HELEN-006 trial, neoadjuvant nab-paclitaxel combined with trastuzumab and pertuzumab achieved a significantly higher pCR rate than standard TCbHP (66.3% vs 57.6%, p = 0.011), further underscoring the superior efficacy of nab-paclitaxel in HER2-positive early breast cancer. 26 The results of the aforementioned studies are consistent with our findings, suggesting that nab-paclitaxel might be the optimal choice among taxanes for HER2-positive breast cancer. However, our study was a non-randomised comparison, and the findings merit further investigation in a randomised controlled trial.
Our study found that HER2 2+ with FISH+ had a poorer pCR rate than HER2 3+ (HER2 3+ vs HER2 2+ FISH+, OR = 4.35, 95% CI: 1.75–10.0, p = 0.001). In the RNA data analysis, we also found that low expression of ERBB2 mRNA was correlated with a decreased pCR rate (p = 0.004). These results indicate that HER2 expression is an important predictor of the response to neoadjuvant therapy in HER2-positive breast cancer. Patients with high HER2 expression levels respond better to HER2-targeted therapy. Results from the TRYPHAENA and KRISTINE trials also indicate that the mRNA and protein expression levels of ERBB2 serve as predictive biomarkers for treatment efficacy in neoadjuvant therapy for HER2-positive breast cancer.27,28
In our study, PIK3CA mutations were identified in 30% of the patients. Compared with wild-type patients, those with PIK3CA mutations showed a significantly lower likelihood of achieving pCR (OR = 13.47, 95% CI: 1.39–130.91, p = 0.025). This finding aligned with those of previous studies, including pooled analysis 29 and clinical trials (CALGB 40601, NeoSphere, KRISTINE),28,30,31 consistently showing lower pCR rates in mutation carriers across various anti-HER2 regimens, although some differences were not statistically significant. Collectively, this evidence suggests that PIK3CA mutations may predict the reduced efficacy of neoadjuvant trastuzumab-based therapy in early HER2-positive breast cancer. Mechanistically, activating PIK3CA mutations confer ligand-independent, constitutive activation of the PI3K–AKT–mTOR axis downstream of HER2. 32 On the other hand, previous study has demonstrated that mutations in PIK3CA can potentiate HER2-mediated oncogenic transformation by upregulating heregulin production and activating HER3. 33 Preclinical studies have shown that PIK3CA mutations accelerate HER2-driven transformation of breast epithelial cells and promote metastatic progression. 34 Furthermore, these mutations alter the intrinsic phenotype of HER2-overexpressing tumours and confer resistance to anti-HER2 therapies. HER2 and mutant PIK3CA thus act synergistically to facilitate mammary tumour initiation and metastasis. Consequently, despite effective HER2 blockade such as with trastuzumab and pertuzumab, persistent downstream signalling sustains proliferative and pro-survival programs. 35
In this study, we found that SPOP amplification correlated with a lower pCR rate. Previous studies have shown that SPOP is an E3 ubiquitin ligase that mediates the ubiquitination and degradation of targeted proteins through proteasomal pathways, and plays an important role in a variety of cancers.36,37 However, current studies on SPOP amplification have mainly focused on other cancers, such as prostate cancer, and are relatively limited in breast cancer.38,39 Preclinical study has shown that SPOP acts as a negative regulator of breast cancer cell motility by promoting K63- and K48-linked ubiquitination of TWIST1, predominantly at lysine 73. Silencing SPOP markedly enhances the EMT program driven by this master regulator, thereby accelerating the migration and invasiveness of breast cancer cells in vitro. 40 However, biomarker findings for PIK3CA and SPOP are exploratory and have not yet been validated. Further studies are needed to further elucidate the underlying mechanism and explore its potential as a prognostic marker and therapeutic target.
Research indicates that the degree of immune cell infiltration and expression levels of immune-related molecules are closely linked to efficacy and survival outcomes.41,42 Our analysis revealed the role of immune-related factors in mediating the treatment response to neoadjuvant therapy in HER2-positive breast cancer. High expression of CD8Tem, pDC, and Th2 cells was positively associated with increased pCR rates, whereas elevated levels of HSC were associated with reduced pCR rates. E Muraro performed extensive immunomonitoring of patients with locally advanced breast cancer patients undergoing neoadjuvant chemotherapy to identify the immunological correlates of pCR induction. The results indicate that the maintenance of functional T cell responses against selected antigens and improvement of natural killer cell proficiency during neoadjuvant chemotherapy are probably critical requirements for pCR induction, especially in HER2-positive breast cancer patients. 43 A total of 230 pretreatment tumours in the CALGB 40601 trial and 138 pretreatment tumours in the PAMELA trial were assessed using immune-related gene expression profiling. These results suggested that multiple B cell-related signatures were more strongly associated with pCR and event-free survival than TILs, which largely represent T cells. When both TILs and gene expression are available, the prognostic value of immune-related signatures appears to be superior. 12 Pathway enrichment analysis also showed that active toll-like receptor signalling pathways were associated with an increased pCR rate. Toll-like receptor signalling pathway is mainly involved in non-specific immunity, activating immune cell response and enhancing anti-tumour immune response. This also indicates that the molecular biological characteristics of HER2-positive breast cancer are diverse, and that the tumour microenvironment is an important factor affecting the efficacy of neoadjuvant therapy. The immune expression analysis performed in this study revealed that the immune function of the tumour microenvironment may be a valid source of suitable predictive biomarkers in the neoadjuvant setting. However, biomarker exploration was hypothesis-generating and had a limited sample size. Therefore, these findings should be interpreted with extreme caution and warrant targeted investigation in larger, validation cohorts.
Our study had several limitations. First, this study was an observational cohort without randomisation, and treatment choices were clinician-driven, concentrating unmeasured confounders in subgroups (such as HR status and type of taxanes) and biasing the results. Second, the follow-up time was short, and the number of events was small; therefore, it was not possible to explore prognostic markers. Third, the exploratory biomarker analysis was limited by a small patient sample, imprecise effect estimates, and extremely wide confidence intervals. The biomarker analyses were restricted to patients for whom adequate residual tissue was available after routine diagnostics, thereby introducing a potential selection bias. Consequently, these data are hypothesis-generating only and need to be validated in larger, adequately powered cohorts. Despite these limitations, this study, based on data from China’s largest dedicated cancer hospital, reflects real-world outcomes of TCbHP neoadjuvant therapy in the contemporary Chinese HER2-positive breast cancer population and can therefore inform preoperative treatment decisions nationwide. However, multicentre prospective studies are still required for definitive validation.
Conclusion
This study provides preliminary evidence supporting the use of trastuzumab and pertuzumab combined with platinum-based chemotherapy as a preferred neoadjuvant regimen for Chinese patients with HER2-positive breast cancer. The potential role of nab-paclitaxel as an optimal taxane within the TCbHP regimen is suggested but requires further validation. Similarly, although PIK3CA mutations were associated with reduced treatment efficacy, these findings are exploratory and warrant confirmation in larger cohorts. Additionally, the possible influence of immune-related tumour microenvironment factors on treatment response merits further investigation.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251403929 – Supplemental material for Trastuzumab and pertuzumab plus carboplatin-based neoadjuvant therapy for HER2-positive breast cancer: a prospective cohort study with integrated genomic and transcriptomic biomarker analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251403929 for Trastuzumab and pertuzumab plus carboplatin-based neoadjuvant therapy for HER2-positive breast cancer: a prospective cohort study with integrated genomic and transcriptomic biomarker analysis by Xiaoyan Qian, Xi Chen, Min Xiao, Lei Ji, Qiao Li, Jiayu Wang, Ying Fan, Yang Luo, Ruigang Cai, Bo Lan, Shanshan Chen, Fei Ma, Binghe Xu and Pin Zhang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
