Abstract
Background:
The 5-year overall survival rates in acute lymphoblastic leukemia (ALL) vary depending on the patient’s age group (from 93% to under 30%). However, in the case of relapsed/refractory ALL (R/R ALL), the complete remission (CR) rate in the pediatric patient group is 44% and in the adult population—18%. Therefore, further research on new therapeutic compounds is necessary.
Objectives:
This systematic review aims to analyze the results of clinical studies concerning the use of venetoclax alone or in combination with other drugs in the treatment of ALL.
Design:
The systematic review was conducted in accordance with the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analysis. The Quality Assessment Tool for Before-After (Pre-Post) Studies With No Control Group by the National Heart, Lung, and Blood Institute was used to assess the quality of included studies.
Data sources and methods:
PubMed and Web of Science were used for the literature review. All clinical studies reporting outcomes in patients with ALL treated with venetoclax met the inclusion criteria. As a result of our search process for articles, a total of 989 records were obtained. After excluding records that did not meet the inclusion criteria or met the exclusion criteria, seven articles were finally obtained.
Results:
In seven identified clinical trials, CR rate ranged from 22% to 59.6% in patients with R/R ALL, while in the group of patients with newly diagnosed ALL, it ranged from 90.9% to 96%. The most common adverse events of grade 3 or higher were hematological complications, including neutropenia (71 episodes among 110 patients), anemia (70/141), and thrombocytopenia (70/141).
Conclusion:
Based on the results of phase I clinical trials, further clinical trials should be conducted to assess the therapeutic potential of venetoclax in the treatment of ALL.
Introduction
Acute lymphoblastic leukemia (ALL) is a hematopoietic malignancy that results from the uncontrolled clonal proliferation of immature lymphoid cells. 1 According to the National Cancer Institute, the rate of new cases of ALL in the United States was 1.9 per 100,000 people per year (based on data from 2018 to 2022). 2 Given that over 60% of ALL cases are diagnosed in the pediatric population, the incidence rate among people under 15 years of age is approximately 3.5 per 100,000 people per year.3,4 The 5-year overall survival rates vary depending on the patient’s age group and range from 93% (children aged 0–14 years) to under 30% (patients aged 60+). 5 According to the National Cancer Institute, the 5-year relative survival for the general population in the United States in the period 2015–2021 was 72.6%. 2 However, it is worth emphasizing that in the case of relapsed/refractory ALL (R/R ALL), the complete remission (CR) rate in the pediatric patient group is 44% and in the adult population—18%. 6 Therefore, further research on new therapeutic compounds that precisely target the disrupted signaling pathways of cancer cells is necessary to increase the safety profile of the treatment and further improve patient survival. 7
One of the pathways whose impaired functioning may contribute to carcinogenesis is the process of programmed cell death, regulated, among others, by proteins from the B-cell lymphoma 2 (Bcl-2) family.1,8 These include (i) multidomain antiapoptotic proteins, for example, Bcl-2, (ii) multidomain proapoptotic proteins, for example, Bcl-2-associated X protein, and (iii) Bcl-2 homology 3 (BH3)-only proteins—see Figure 1.8,9 The dysregulation at the transcriptional and post-translational level, leading to the increased expression and stabilization of antiapoptotic proteins—mediated, among others, by phosphorylation or impaired ubiquitination—may lead to resistance to chemotherapy and, consequently, prevent remission or contribute to the relapse of the neoplastic disease.8,10 Moreover, Bcl-2 family proteins, through their involvement in oxidative phosphorylation, may contribute to metabolic dysregulation. 11 Therefore, one of the new therapeutic options in ALL may be Bcl-2 inhibitors. Their mechanism of action is presented in Figure 1.9,12,13

Bcl-2 family members’ structure and mechanism of action. (a) The Bcl-2 protein family is divided into three groups: multidomain antiapoptotic proteins, multidomain proapoptotic proteins, and BH-3-only proteins. The antiapoptotic proteins include Bcl-2, Bcl-xL, Mcl-1, Bcl-w, and Bcl-2A1, while the proapoptotic proteins include BAX, BAK, and BOK. BH-3-only proteins are further divided into activators, which include BIM, BID, and PUMA, and sensitizers, which include BIK, BAD, NOXA, HRK, and BMF, depending on their function. Antiapoptotic proteins contain four BH domains (BH1–BH4), proapoptotic proteins contain three domains (BH1–BH3), while BH-3-only proteins contain one domain. (b) Venetoclax is an inhibitor of the antiapoptotic protein Bcl-2. Overexpression of genes encoding the Bcl-2 protein in cancer cells may lead to the predominance of antiapoptotic over proapoptotic protein activity and resistance to chemotherapy. Venetoclax, through inhibition of Bcl-2, may promote apoptosis of cancer cells. (c) Physiologically, Bcl-2 family proteins participate in the regulation of the intrinsic pathway of apoptosis. Intrinsic lethal stimuli promote activators (A) from the BH3-only group to activate proapoptotic BAX and BAK. At the same time, sensitizers (S) from the BH3-only group inhibit antiapoptotic proteins, for example, Bcl-2, which, by inhibiting activators (A) and proapoptotic proteins, can inhibit the process of programmed cell death. If this does not happen, BAX and BAK proteins, due to interactions with BIM and BID, oligomerize in the outer mitochondrial membrane, initiating the process of MOMP. SMAC and cytochrome C molecules are released into the cytoplasm through pores in the outer mitochondrial membrane. This leads to the formation of apoptosomes, caspase activation, and ultimately to apoptosis.
One of the representatives of Bcl-2 inhibitors is venetoclax (ABT-199), already approved by the Food and Drug Administration (FDA) for the treatment of chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), and acute myeloid leukemia (AML). In 2018, the FDA granted regular approval to venetoclax for patients with CLL and SLL, with or without 17p deletion, who have received at least one prior treatment. 14 A year later, approval was granted for all adult patients with CLL/SLL. 15 In 2018, accelerated approval was also granted for the use of venetoclax in combination with azacitidine/decitabine/low-dose cytarabine in newly diagnosed patients with AML, aged above 75 years. 16 In 2020, it was converted to regular approval. 17 The results of the conducted preclinical studies indicate that venetoclax effectively induces the apoptosis of immature T-cell (T-ALL) and B-cell ALL (B-ALL) cell lines in vivo and in vitro.18–21 This led to the initiation of clinical trials assessing the efficacy and safety of the drug in the treatment of ALL. This systematic review aims to analyze the results of clinical studies concerning the use of venetoclax alone or in combination with other drugs in the treatment of ALL.
Methods
Data sources and search strategy
The systematic review was conducted in accordance with the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA). Two independent authors (A.K. and J. Z. (Julia Zarychta)) conducted the literature review. The following databases were used for this purpose: PubMed and Web of Science. The literature review included articles published up to March 17, 2025. The search strategy was based on searching for combinations of keywords such as “venetoclax,” “acute lymphoblastic leukemia,” “refractory/relapsed acute lymphoblastic leukemia,” “R/R ALL,” “efficacy,” and “safety.” Exact terms used during the search of relevant articles are presented in Supplemental Table 2.
Inclusion and exclusion criteria
All clinical studies reporting outcomes in patients with ALL treated with venetoclax met the inclusion criteria. The exclusion criteria for the consideration of the studies were as follows: (1) review articles, (2) systematic review articles, (3) case reports, (4) meeting abstracts, (5) letters to the editor, (6) correspondence, (7) preclinical studies, and (8) articles in languages other than English.
Data extraction
In the first step, duplicates were removed and then double-checked. After removing duplicates, two authors (A.K. and J.Z (Julia Zarychta)) independently analyzed the articles based on their title, abstract, and keywords to exclude inappropriate articles. Then, the obtained results were assessed for meeting the inclusion and exclusion criteria.
Each identified study was analyzed to extract the following data: authors, title, journal, year of publication, intervention, number of patients, patient characteristics, treatment results, and occurrence of adverse events.
Quality assessment
The Quality Assessment Tool for Before-After (Pre-Post) Studies With No Control Group by the National Heart, Lung, and Blood Institute was used to assess the quality of included studies. 22 The tool contained 12 questions that could be answered with “Yes” or “No.” Based on the answers, the quality of the study was classified as “Good,” “Fair,” or “Poor.” The quality assessment was performed by two authors (A.K. and J.Z (Julia Zarychta)) independently. In the case of differences in the assessment, the quality assessment was performed by the third and fourth authors (M.L. and J.Z. (Joanna Zawitkowska)).
Results
Search and selection of studies
As a result of our search process for articles according to the criteria given earlier, a total of 989 records were obtained. After the initial search, 672 duplicates were removed, which resulted in further evaluation of 317 articles. The most common reasons for excluding a record based on the title/abstract were as follows: meeting abstracts (n = 58), literature review (n = 54), and case reports (n = 52). In the next stage, records for which full access could not be obtained (n = 12) were rejected. In the last stage, 114 articles were evaluated by means of a thorough analysis of the full text. After excluding records that did not meet the inclusion criteria or met the exclusion criteria, seven articles were finally obtained.6,23–28 The process of searching and selecting articles for our systematic review is presented in the form of a PRISMA flow diagram (Figure 2).
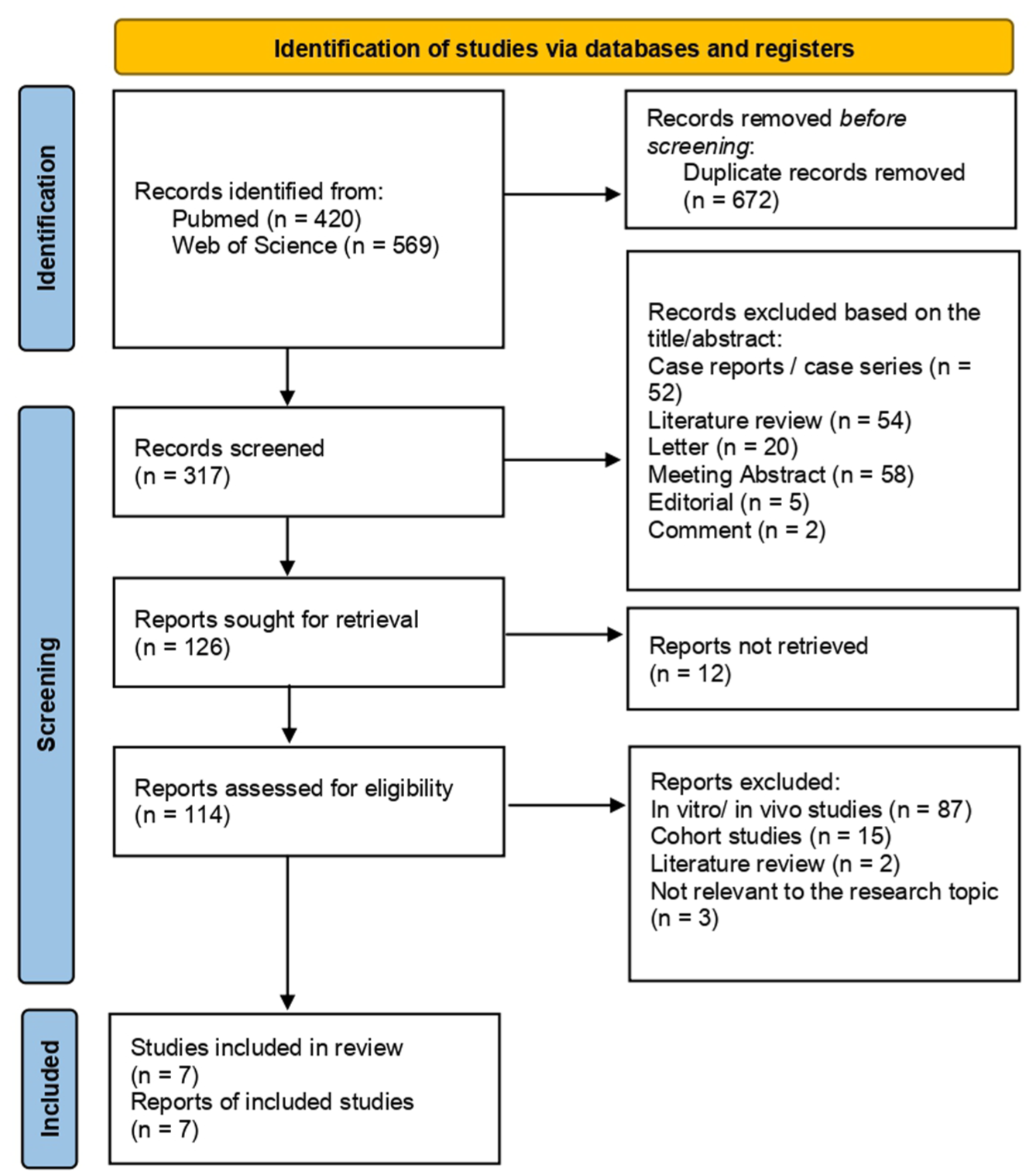
Flow diagram according to PRISMA format.
Characteristics of included studies
As a result of the search and selection process, seven studies were obtained, and these were clinical trials. None of them was a randomized controlled trial. The oldest publication is from 2021, three articles were published in 2024, and the last three in 2025. A total of 182 patients were included in these studies. Almost all studies were phase I studies (one study was a phase I/II study), indicating the early stage of work in this field.
All studies investigated the use of venetoclax in the treatment of patients with ALL. Four studies included only adult patients, one study included children, adolescents, and young adults, and two studies included both children and adults. In most studies, the study group consisted primarily of men. The majority of studies focused on patients with R/R ALL—five studies included only patients with relapsed or refractory disease, and one included both R/R ALL and newly diagnosed patients. One study included only newly diagnosed patients. In three studies, a small proportion of the study group also included patients with lymphoblastic lymphoma (LL) (ranging from 3 to 11 patients).6,24,25
In all of the analyzed clinical trials, venetoclax was administered in combination with chemotherapy. The exception was the study by Place et al., 27 where patients received venetoclax alone or in combination with chemotherapy (vincristine, dexamethasone, and pegasparaginase-based regimen or cytarabine-based regimen). In the remaining studies, different chemotherapy regimens were used (mini-hyper-CVD—cyclophosphamide, vincristine, dexamethasone; cytarabine, aclarubicin, granulocyte colony-stimulating factor, etoposide (CAGE); CALGB 10403). In the study by Pullarkat et al., 6 navitoclax—a B-cell lymphoma-extra large (Bcl-xL)/Bcl-2 inhibitor with chemotherapy—was additionally used in the treatment. In turn, in the work by Shi et al., 24 the synergistic effect of venetoclax and daratumumab (anti-CD38 antibody) was studied. Detailed information on the analyzed clinical trials is found in Table 1.
Characteristics of the included studies.

ALL, acute lymphoblastic leukemia; B-ALL, B-cell acute lymphoblastic leukemia; B-LL, B-cell lymphoblastic lymphoma; CAGE, cytarabine, aclarubicin, granulocyte colony-stimulating factor, etoposide; DL, dose level; LL, lymphoblastic lymphoma; L-VCR, liposomal vincristine; mini-hyper-CVD, cyclophosphamide, vincristine, dexamethasone; NA, not available; R/R, relapsed/refractory; T-ALL, T-cell acute lymphoblastic leukemia; T-LL, T-cell lymphoblastic lymphoma.
Quality assessment
To assess the quality of the included studies, we used The Quality Assessment Tool for Before-After (Pre-Post) Studies with No Control Group by the National Heart, Lung, and Blood Institute (see Supplemental Table 1). As a result, all clinical studies were classified as of satisfactory quality and were included in this systematic review.
Efficacy of venetoclax
The primary outcomes of interest were CR rate and overall survival (OS). Detailed data on the results of venetoclax treatment are presented in Table 2. Of the clinical studies identified, data on CR were obtained in all of them. CR rate ranged from 22% to 59.6% in patients with R/R ALL, while in the group of patients with newly diagnosed ALL, it ranged from 90.9% to 96%. When divided into R/R B- or T-ALL, the CR rate ranged from 33.3% to 64.0%, and from 40.0% to 75.0%, respectively. CR rates also varied between pediatric and adult groups. Among patients with R/R ALL, they ranged from 42% to 75% in children and from 22% to 57% in adults. In two studies involving patients with R/R T-ALL, differences were also observed between patients with R/R ETP-ALL and R/R non-ETP-ALL.6,24 Both studies reported a higher CR rate among patients with R/R ETP-ALL. In the study by Pullarkat et al., the CR rate was 66.7% (8/12 patients) in the group of patients with R/R ETP-ALL and 33.3% (2/6 patients) in the group of patients with R/R non-ETP-ALL. In the study by Shi et al., the CR rate in these groups was 71.4% (5/7 patients) and 35.7% (5/14 patients), respectively. Of all the studies, in four of them, BH3 profiling was performed.6,26–28 We were able to extract data regarding the relationship between BH3 dependency at baseline and the treatment response in two of these studies.6,27 The results are presented in Table 3.
Outcomes of the included studies.

This study included three patients under 18 years of age; however, due to the collective results of the study, detailed results for the pediatric and adult populations cannot be distinguished.
This study included children and young adults; however, due to the collective results of the study, detailed results for specific groups cannot be distinguished.
ALL, acute lymphoblastic leukemia; CAGE, cytarabine, aclarubicin, granulocyte colony-stimulating factor, etoposide; CI, confidence interval; CR, complete remission; IGH NGS, immunoglobulin heavy chain next-generation sequencing; L-VCR, liposomal vincristine; mini-hyper-CVD, cyclophosphamide, vincristine, dexamethasone; MRD, minimal residual disease; NA, not available; OS, overall survival; PD, progressive disease; PR, partial response; R/R, relapsed/refractory; SD, stable disease.
The BH3 dependency at baseline and treatment response.

ALL, acute lymphoblastic leukemia; B-ALL, B-cell acute lymphoblastic leukemia; BCL-2, B-cell lymphoma 2; BCL-xL, B-cell lymphoma-extra large; BH3, Bcl-2 homology 3; CR, complete remission; MCL-1, Myeloid cell leukemia 1; R/R, relapsed/refractory; T-ALL, T-cell acute lymphoblastic leukemia.
As for median OS, it was possible to obtain information on it in three identified studies. All of them were concerned patients with R/R ALL. Median OS ranged from 3.9 to 7.8 months (3.9–9.7 when distinguishing B- and T-ALL subgroups). In four studies, 12-month OS was reported. It ranged from 29% to 35.6% in patients with R/R ALL (in the study by Pullarkat et al., after separating the pediatric population, this rate was 60.8%) and from 91% to 96% in newly diagnosed patients. Data on minimal residual disease negativity were also obtained from the analyzed studies. This rate ranged from 11% to 34% for patients with refractory or relapsed disease (in the study by Pullarkat et al., after separating the pediatric population, this rate was 50.0%). In newly diagnosed patients, the values ranged from 90.9% to 91%. Detailed results are presented in Table 2.
Safety of venetoclax
Data on the adverse events of the therapy were also extracted from the analyzed articles. The most common adverse events of grade 3 or higher were hematological complications, including neutropenia (71 episodes among 110 patients), anemia (70/141), and thrombocytopenia (70/141). The most common non-hematological adverse events were increased alanine aminotransferase (34/163), hypokalemia (25/96), and increased aspartate aminotransferase (23/141). Details of adverse events reported in the analyzed clinical trials are provided in Table 4.
Safety of venetoclax in the included studies.

ALT, alanine aminotransferase; AST, aspartate aminotransferase.
Discussion
The current standards of treatment for ALL vary depending on the age and profile of the patient. For pediatric patients, it is multidrug chemotherapy, usually consisting of three phases: induction, consolidation, and maintenance. In the induction phase, aimed at achieving remission, patients usually receive up to three drugs (L-asparaginase, vincristine, and corticosteroids). In the case of high-risk patients, anthracyclines are additionally used. 30 In the case of adult patients, the basis of ALL treatment is also intensive multidrug chemotherapy regimens (e.g., CALGB or hyper-CVAD). Like in the case of pediatric patients, the following phases are distinguished here: induction, consolidation, and maintenance therapy. The main components of treatment regimens in the induction phase for adults are vincristine, corticosteroids, and anthracyclines. 31
In the analyzed clinical studies, venetoclax was used in combination with the aforementioned standard chemotherapy regimens. In addition, in two studies, navitoclax 6 and daratumumab 24 were added to the therapy. The simultaneous use of venetoclax and daratumumab is an example of a synergistic therapy, in which each drug is directed at a different target point. While venetoclax acts by inhibiting antiapoptotic protein—Bcl-2, daratumumab is an antibody directed at the CD38 antigen. Its task is, therefore, to stimulate the immune response against cancer cells expressing it. 32 There have been reports of good efficacy of such a therapy in patients with ALL and AML.33–35 In the study by Shi et al., 24 such a combination of drugs in patients with R/R T-ALL/LL allowed for a CR of 47.6%. In the study by Pullarkat et al., a combination of venetoclax and navitoclax with standard chemotherapy was used. Navitoclax is a Bcl-xL/Bcl-2 inhibitor, so the simultaneous use of these drugs allows targeting two proteins from the Bcl-2 family. In addition, the authors indicate that such a solution reduces the need for high doses of navitoclax and thus decreases the risk of thrombocytopenia associated with the use of navitoclax, which could contribute to the need to lower the drug dose during treatment. Such a therapy in patients with R/R ALL/LL allowed for achieving CR at the level of 59.6%. 6
There are currently 24 ongoing clinical trials registered on clinicaltrials.gov evaluating the use of venetoclax in ALL, the details of which are provided in Table 5.
Currently recruiting clinical trials testing the use of venetoclax in the treatment of acute lymphoblastic leukemia.

ALL, acute lymphoblastic leukemia; alloHSCT, allogenic hematopoietic stem cell transplantation; B-ALL, B-cell acute lymphoblastic leukemia; CAR-T, chimeric antigen receptor T cell; ETP, early T-cell precursor; G-CSF, granulocyte colony-stimulating factor; HSCT, hematopoietic stem cell transplantation; L-ASP, L-asparaginase; Ph−/+, chromosome Philadelphia −/+; R/R, relapsed/refractory; T-ALL, T-cell acute lymphoblastic leukemia; T-LBL, T-cell lymphoblastic lymphoma.
In currently recruiting clinical trials, venetoclax was most often combined (in addition to commonly used drugs in ALL chemotherapy) with blinatumomab, tyrosine kinase inhibitors, inotozumab ozogamicin, and chimeric antigen receptor T-cell therapy.
It is noteworthy that the efficacy of venetoclax treatment varied depending on the disease subtype. This is consistent with preclinical studies demonstrating that specific ALL subtypes, including Ph-like B-ALL and B-ALL with KMT2A gene rearrangements, as well as ETP-ALL, are characterized by elevated BCL-2 expression. Consequently, patients with these disease subtypes may potentially gain the greatest clinical benefit from venetoclax therapy. By contrast, mature T-ALL subtypes demonstrate increased BCL-xL expression, suggesting that the use of venetoclax as the sole BCL-2 inhibitor may not be sufficient to achieve optimal clinical response in this patient group. 36 Of the three studies included in the systematic review that identified subgroups of patients with R/R B-ALL and R/R T-ALL, one reported a higher CR rate for patients with R/R B-ALL (64.0%; 16/25 patients) than for patients with R/R T-ALL (52.6%; 10/19 patients). 6 In the remaining two studies, the efficacy of venetoclax in patients with R/R B-ALL versus R/R T-ALL was 50.0% (9/18 patients) versus 75.0% (3/4 patients) and 33.3% (1/3 patients) versus 40.0% (2/5 patients), respectively.23,26 However, it is worth noting the low number of patients with R/R T-ALL in the latter two studies, which may have contributed to the higher CR rate in this group. Differences were also observed between patients with R/R ETP-ALL and R/R non-ETP-ALL. CR rates for these patients ranged from 66.7% (8/12 patients) to 71.4% (5/7 patients), and from 33.3% (2/6 patients) to 35.7% (5/14 patients), respectively.6,24 However, the small number of patients and the heterogeneity of the study groups do not allow for the identification of the relationship between ALL subtype and response to venetoclax treatment. On the other hand, due to the diverse expression profile of BCL-2 family proteins in individual ALL subtypes, conducting studies assessing the expression of these proteins in patients qualified for BCL-2 inhibitor therapy may allow for the selection of personalized therapeutic combinations aimed at blocking various antiapoptotic proteins.
As mentioned earlier, venetoclax is not the only drug from the Bcl-2 inhibitor group. The Bcl-2 protein family, which includes antiapoptotic proteins, apart from Bcl-2 itself, also includes myeloid cell leukemia 1 (Mcl-1), Bcl-xL, B-cell lymphoma-w (Bcl-w), and Bcl-2-related protein A1 (Bcl-2A1). 37 The previously mentioned navitoclax is a Bcl-2/Bcl-xL inhibitor, intensively tested in preclinical and clinical studies. 38 Its advantage over venetoclax is certainly the ability to simultaneously inhibit two proteins from the Bcl-2 family, which may translate into greater efficacy in ALL.38,39 However, a significant limitation of navitoclax is the increased thrombocytopenia induced by its action, which may force a reduction in the drug dose and thus reduce the efficacy of the therapy.38,40 It has been shown that Bcl-xL mediates the aging process of platelets, similar to apoptosis.40,41 Hence, the use of navitoclax may cause acute thrombocytopenia. Another dual Bcl-2/Bcl-xL inhibitor is AZD4320, tested in preclinical studies. Its greatest advantage is the occurrence of significantly less severe thrombocytopenia compared to navitoclax, which allows the use of this drug without the need to reduce the dose.42,43 Another dual Bcl-2/Bcl-xL inhibitor is APG-1252, tested in the treatment of, among others, hepatocellular carcinoma, gastric cancer, nasopharyngeal carcinoma, as well as myeloproliferative neoplasm.44–47 Inhibitors of Mcl-1 (including S64315, AZD5991, S63845, AMG-176), Bcl-xL (including A1331852, A1155463, WEHI-539), and Bcl-2A1 are also in early development.48–55
Due to the heterogeneity of ALL and the development of resistance mechanisms, it is necessary to identify subgroups of patients in the case of which the use of venetoclax in the therapy of leukemia will allow for achieving clinical benefits. Zhang et al. 56 showed that mutations in the WT1 and FLT3 genes are associated with increased sensitivity to venetoclax, while KRAS and PTPN11 mutations were associated with resistance to Blc-2 inhibitor. Although the study analyzed the data from patients with AML, the discussed mutations, although less frequently, may also occur in patients with ALL.56–60 Potential clinical benefits from the use of venetoclax, in addition to patients with WT1 and FLT3 mutations, may also be achieved by patients with treatment-resistant subtype of B-ALL [t(17;19) TCF3::HLF] and [t(4;11) MLL:: AF4]. 12 The TCF3/HLF and MLL/AF4 fusion genes contribute to the increased expression of the BCL-2 gene. 12 Cancer cells with a higher amount of Bcl-2 actively sequestering proapoptotic proteins (strongly primed) will be more sensitive to the use of the Bcl-2 inhibitor compared to cells with a lower amount of active Bcl-2. 61 In addition, MLL/AF4 does not affect the expression of genes encoding other antiapoptotic proteins, for example, Mcl-1. 62 Hypodiploid B-ALL also predisposes to the increased expression of Bcl-2 and the decreased expression of the antiapoptotic protein Bcl-xL. 12 This is beneficial in the case of treatment with venetoclax, because the excessive expression of genes encoding antiapoptotic proteins may lead to the sequestration of proapoptotic effector proteins, even despite the inhibition of Bcl-2 function by venetoclax.12,63 However, further studies on the mechanisms of venetoclax resistance in ALL are necessary. Enzenmüller et al. showed that ALL cancer cells insensitive to venetoclax were characterized by increased levels of Mcl-1, which, by binding proapoptotic Bcl2-interacting mediator of cell death, inhibited the action of the drug. It is worth emphasizing, however, that Mcl-1 inhibition did not restore full sensitivity to venetoclax, and Bcl-xL expression remained unchanged in these cells; therefore, resistance to venetoclax may also be conditioned by additional mechanisms. 64 In addition to (i) the increased expression of genes encoding antiapoptotic proteins, (ii) the development of loss-of-function mutations in genes encoding proapoptotic proteins or proapoptotic regulatory molecules, (iii) mutations leading to an alteration of the venetoclax binding site to the BH3 domain (e.g., F101 mutation), another mechanism of venetoclax resistance may be (iv) impaired cytokine signaling.64–66 In the study of Shah et al., 65 long-term exposure to venetoclax led to the increased phosphorylation of signal transducer and activator of transcription 5 (STAT5) and 3 (STAT3) in drug-resistant T-ALL cell lines, probably via cytokine pathways related to interleukin 3, interleukin 12, and interleukin 23. A study conducted in an AML cell line showed that gilteritinib (tyrosine kinase inhibitor), by inactivating STAT5, reduced the expression of the gene encoding the antiapoptotic protein Bcl-2A1, thereby reducing venetoclax resistance. 67 However, further work is needed on the molecular mechanisms through which STAT3 and STAT5 signaling contributes to the development of venetoclax resistance in ALL.65,67
Another mechanism of venetoclax resistance is metabolic reprogramming. In the study by Enzenmüller et al., Blc-2 inhibitor-resistant ALL cell lines were characterized by (i) increased oxidative metabolism (higher oxygen consumption, higher adenosine triphosphate (ATP) production rate), (ii) the increased expression of mitochondrial deoxyribonucleic acid, and (iii) the lower expression of the gene encoding dynamin-related protein 1 (Drp1), involved in mitochondrial fission. Reduced Drp1 levels most likely correlate with the altered mitochondrial morphology found in venetoclax-resistant leukemic cells. Their organelles were larger and longer, compared to mitochondria present in venetoclax-sensitive cells, which may be due to impaired mitochondrial fission processes. The use of oligomycin, a compound that inhibits oxidative phosphorylation, led to re-sensitization of resistant ALL cell lines to venetoclax; however, side effects resulting from the blockade of mitochondrial ATP synthase limit the use of oligomycin in clinical practice. 64
Due to primary but also developing secondary resistance to venetoclax, the challenge in clinical practice may be an adequate optimization of the therapy with inhibitors of antiapoptotic proteins from the Bcl-2 family and personalization of the treatment regimen before its implementation, as well as changes of the therapeutic regimen in response to the development of resistance. 37 A functional test including dynamic BH3 (DBP) profiling can be used to predict the response to venetoclax therapy in patients with ALL.37,61 The technique allows for the measurement of the permeability of the outer mitochondrial membrane after exposure of cancer cells to drugs inducing apoptosis. 61 By correlating DBP results with the response to treatment with a given compound, it is possible to develop an effective, personalized drug combination and its modification during the course of treatment.12,37 This may lead to improved therapy outcomes in patients with cancers resistant to some Bcl-2 inhibitors while reducing the side effects of multi-drug anticancer therapy in patients whose cancer is sensitive to single-agent treatment. 37
The limitation of this systematic review is the small number of available clinical trials (n = 7), with a small number of patients qualified for the studies, assessing the efficacy and safety of venetoclax in the treatment of ALL. In addition, a lack of randomized studies in this area prevents carrying out a meta-analysis that would allow for the formulation of general conclusions. Due to the lack of randomization, a possible bias cannot be excluded. It is also worth emphasizing the heterogeneity of the analyzed studies. Venetoclax was administered in different therapeutic regimens in a diverse patient population (pediatric patients vs adults, patients with newly diagnosed ALL vs R/R ALL). In addition, depending on the study, the dosage of venetoclax varied. Also, to more accurately analyze the efficacy of venetoclax in ALL, it is important to stratify patients enrolled in future clinical trials in terms of ALL subtypes, BH3 profiling, and cytogenetic abnormalities routinely used in prognostic and therapeutic algorithms.
Conclusion
To improve the safety profile of the therapy and improve the survival of patients with ALL, studies on new therapeutic compounds that precisely target the disrupted signaling pathways of cancer cells are needed. One of the new therapeutic options in ALL may be Bcl-2 inhibitors. This systematic review analyzed the results of seven clinical trials of venetoclax in the treatment of ALL. CR rate ranged from 22% to 59.6% in patients with R/R ALL, while in the group of patients with newly diagnosed ALL, it ranged from 90.9% to 96%. The most common adverse events of grade 3 or higher were hematological complications, including neutropenia, anemia, and thrombocytopenia. Based on the results of phase I clinical trials, further clinical trials should be conducted to assess the therapeutic potential of venetoclax in the treatment of ALL.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251403912 – Supplemental material for The use of venetoclax in the treatment of acute lymphoblastic leukemia—a systematic review
Supplemental material, sj-docx-1-tam-10.1177_17588359251403912 for The use of venetoclax in the treatment of acute lymphoblastic leukemia—a systematic review by Adrian Kowalczyk, Julia Zarychta, Joanna Zawitkowska and Monika Lejman in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251403912 – Supplemental material for The use of venetoclax in the treatment of acute lymphoblastic leukemia—a systematic review
Supplemental material, sj-docx-2-tam-10.1177_17588359251403912 for The use of venetoclax in the treatment of acute lymphoblastic leukemia—a systematic review by Adrian Kowalczyk, Julia Zarychta, Joanna Zawitkowska and Monika Lejman in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251403912 – Supplemental material for The use of venetoclax in the treatment of acute lymphoblastic leukemia—a systematic review
Supplemental material, sj-docx-3-tam-10.1177_17588359251403912 for The use of venetoclax in the treatment of acute lymphoblastic leukemia—a systematic review by Adrian Kowalczyk, Julia Zarychta, Joanna Zawitkowska and Monika Lejman in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
