Abstract
Background:
Systemic inflammation has been linked to cancer progression and survival outcomes. The neutrophil-to-lymphocyte ratio (NLR) is a readily available biomarker that may reflect this process in metastatic urothelial carcinoma (mUC).
Objectives:
To evaluate the prognostic impact of baseline NLR on overall survival in mUC patients receiving chemotherapy or immunotherapy.
Design:
A multicenter retrospective cohort study.
Methods:
We retrospectively analyzed 196 patients with advanced, unresectable urothelial carcinoma treated at four centers (Ankara University School of Medicine, Dr. Abdurrahman Yurtaslan Ankara Oncology Training and Research Hospital, Ankara Bilkent City Hospital, and Gazi University School of Medicine) between 2005 and 2023. These patients were stratified into high and low NLR groups using a cutoff of 4.2. Survival outcomes were assessed using Kaplan–Meier curves, and the prognostic significance of NLR was evaluated using univariate (log-rank test) and multivariate (Cox proportional hazards model) analyses.
Results:
The median overall survival (OS) for the entire cohort was 27 months (95% CI: 19.1–34.9). Patients with low NLR had significantly longer OS than those with high NLR (56.6 vs 14.6 months; p < 0.001). In multivariate analysis, NLR remained an independent predictor of OS after adjusting for age, liver metastases, and bone metastases (HR: 2.28, 95% CI: 1.37–3.81; p = 0.002).
Conclusion:
Elevated NLR is an independent prognostic marker in mUC, underscoring the role of systemic inflammation in cancer progression. These findings highlight the potential of NLR as a readily available biomarker for risk stratification in mUC, irrespective of treatment modality.
Keywords
Background
Epidemiology
Urothelial carcinoma is the ninth most common cancer worldwide, with an estimated incidence of 5.6 cases per 100,000 individuals and a mortality rate of 1.8 per 100,000 according to GLOBOCAN 2022 data. 1 Metastatic disease is observed in 5%–10% of patients at the time of diagnosis. Among operable patients, up to 50% develop distant metastases during follow-up. 2 The 5-year survival rate for metastatic urothelial carcinoma (mUC) is approximately 8%. The advent of novel treatments, including antibody-drug conjugates and immune checkpoint inhibitors, has significantly enhanced survival outcomes. However, identifying patients most likely to benefit from these therapies remains a major challenge, highlighting the need for reliable prognostic markers to optimize treatment strategies.3,4
Prognostic models
The Bajorin risk model incorporates Karnofsky performance status and the presence of visceral metastases as independent predictors of survival in mUC patients treated with platinum-based therapy. 5 After that, Bellmunt defined the model, including liver metastases, hemoglobin levels, and Eastern Cooperative Oncology Group (ECOG) performance status. 6 Apolo proposed a four-variable model that included low hemoglobin, low albumin, visceral metastasis, and Karnofsky performance status. 7 Galsky’s model, which investigates the probability of overall survival (OS) at 1, 2, and 5 years, is based on primary tumor site, number of visceral metastatic sites, presence of lymph node metastases, ECOG PS, and leukocyte count. 8 Multiple studies have continued to explore prognostic scores that predict treatment response and survival rates. One approach to predicting risk involves assessing systemic inflammation.
Rationale using the neutrophil-to-lymphocyte ratio
Inflammation is a crucial factor in cancer development and progression. 9 Tumor-associated inflammation facilitates the proliferation, survival, and migration of cancer cells within the tumor microenvironment. 10 As a result, systemic inflammatory markers have been identified as potential prognostic biomarkers.11,12 Among these markers, the neutrophil-to-lymphocyte ratio (NLR) reflects both systemic inflammation and the host immune response. High NLR has been associated with poor prognosis in various malignancies, including breast cancer, lung cancer, colorectal cancer, renal cell cancer, prostate cancer, and others. 13 In patients with early-stage or metastatic urothelial bladder cancer, several studies have identified pretreatment NLR as a prognostic factor associated with poor outcomes.14,15 Although the NLR is a long-established biomarker, its role remains relevant in the modern era of cancer therapy. In particular, NLR may provide additional insights for prognostic assessment in patients treated with immune checkpoint inhibitors and antibody–drug conjugates.
Therefore, this study investigates the prognostic role of baseline NLR in patients with advanced and mUC receiving chemotherapy or immunotherapy. By evaluating the relationship between NLR and OS, we aim to determine its potential as a simple, cost-effective prognostic biomarker in clinical practice.
Methods
Study design and patient selection
We conducted a retrospective cohort study by extracting patient data from the hospital databases of four cancer centers in Turkey (Ankara University School of Medicine, Dr. Abdurrahman Yurtaslan Ankara Oncology Training and Research Hospital, Ankara Bilkent City Hospital, and Gazi University School of Medicine) A total of 220 patients with advanced, unresectable, and mUC who received either chemotherapy or immunotherapy between 2005 and 2023 were screened. Twenty-four patients were excluded because their baseline laboratory data, required for NLR calculation, were unavailable, leaving a final cohort of 196 patients aged 18 years or older. All included patients were treatment-naïve for systemic therapy in the metastatic setting. Of the 196 patients, 84 (42.9%) were de novo metastatic at diagnosis, while 112 (57.1%) were diagnosed at an early stage, treated locally, and subsequently developed metastatic disease during follow-up. None of these early-stage patients had a history of adjuvant or neoadjuvant immunotherapy. All eligible patients were included without additional selection criteria. Baseline demographic, clinical, radiologic, and laboratory data (including neutrophil and lymphocyte counts) were collected by reviewing electronic medical records from each participating center. Each center used standard automated hematology analyzers for complete blood counts as part of their usual clinical practice, which allowed us to compare results across centers. No data imputation was performed; all cases were manually reviewed, and any missing or inconsistent entries were checked. A detailed flow diagram of patient selection and exclusions is provided in Supplemental Figure 1. The reporting of this retrospective cohort study adheres to the STROBE guidelines for cohort studies, and the completed checklist is provided in Supplemental Table 1.
Outcome measures
The primary outcome was OS, defined as the interval from initiation of first-line treatment to death or last follow-up. The baseline NLR was calculated by dividing the neutrophil count by the lymphocyte count, using values obtained before treatment initiation. Each center used standard automated hematology analyzers for complete blood counts as part of their usual clinical practice, which allowed us to compare results across centers. Receiver operating characteristic (ROC) curve analysis, with OS as the endpoint, yielded an area under the curve (AUC) of 0.61 (95% CI: 0.53–0.69, p = 0.006). The optimal cutoff value of 4.2 was selected according to the Youden index, which identifies the point on the ROC curve that maximizes the sum of sensitivity and specificity. Patients were subsequently stratified into a high-NLR group (⩾4.2) and a low-NLR group (<4.2).
Statistical analysis
IBM SPSS version 26.0 was utilized for all statistical analyses. The Mann–Whitney U test evaluated differences in continuous variables. Categorical variables were analyzed using the chi-square test or Fisher’s exact test, depending on data distribution. The study population was determined by the availability of patients meeting the inclusion criteria; therefore, no sample size calculation or power analysis was conducted. Kaplan–Meier survival curves were used for OS estimation, with group comparisons performed using the log-rank test.
The Cox proportional hazards model was used to evaluate independent predictors of OS. Age and sex were included as standard demographic factors. Liver and bone metastases were selected because they have consistently been reported as adverse prognostic indicators in mUC, in line with the risk models proposed by Bajorin and Bellmunt. Lung metastases were also considered, given their frequency in this patient population. A p-value <0.05 was considered statistically significant.
Ethical approval
The study was conducted in accordance with ethical standards as a multicenter retrospective cohort analysis using de-identified data. Primary ethical approval was obtained from the Ankara University School of Medicine Ethics Committee for Clinical Research (approval number 18-1209-18). This approval covered data collection and analysis across all participating centers, including Dr. Abdurrahman Yurtaslan Ankara Oncology Training and Research Hospital, Ankara Bilkent City Hospital, and Gazi University School of Medicine. Permission to access patient records was obtained from the administrations of these hospitals, and separate local Institutional Review Board approvals were not required. Due to the retrospective design and the use of anonymized patient records, the requirement for informed consent was waived by the ethics committee.
Results
Patient characteristics
A total of 196 patients with metastatic or advanced urothelial carcinoma were included in the analysis. The median age was 64 years (range: 30–86), with 169 males (86.2%) and 27 females (13.8%). Most patients had an ECOG performance status of 0 or 1. At baseline, 11 patients (5.6%) had brain metastases, 84 (42.6%) had lung metastases, 42 (24.3%) had liver metastases, and 83 (42.1%) had bone metastases.
Overall survival and NLR
The mean baseline NLR was 5.2 (range: 0.53–45.9), with a median of 4.09. ROC curve analysis yielded an AUC of 0.61 (95% confidence interval: 0.53–0.69, p = 0.006) for NLR. The optimal NLR cutoff value was 4.2, corresponding to a sensitivity of 60% and specificity of 46.1%. Using this threshold, 101 patients were classified as having high NLR (⩾4.2) and 95 as having low NLR (<4.2).
NLR values were assessed across all subgroups. Among patients with lung metastases, 53.6% had NLR values above 4.2 and 46.4% below. For bone metastases, 68.7% were above 4.2 and 31.3% below. In the liver metastases subgroup, 69% were above 4.2 and 31% below.
The median follow-up duration was 17.9 months (95% CI: 14.5–21.3), calculated using the reverse Kaplan–Meier method. The median OS for the entire cohort was 27 months (95% CI: 19.1–34.9). Patients in the NLR-low group had significantly longer OS than those in the NLR-high group (56.6 months (95% CI: 10.1–103.1) vs 14.6 months (95% CI: 10.09–19.2), p < 0.001). In multivariate analysis, adjusting for confounding factors such as age, liver metastases, and bone metastases, high NLR remained an independent predictor of worse OS (HR: 2.28, 95% CI: 1.37–3.81; p = 0.002).
At 20 months, estimated survival was 65% for patients with NLR below 4.2 and 49% for those with NLR of 4.2 or higher, a 16% difference. At 40 months, survival rates were 52% and 25%, respectively, a 27% difference.
Survival outcomes by treatment modality
When survival outcomes were analyzed based on treatment modality, OS was significantly longer in the NLR-low group in both chemotherapy and immunotherapy subgroups. The number of chemotherapy patients was 127, and the number of immunotherapy patients was 36; however, the treatment information was unavailable for 33 patients. Among patients receiving chemotherapy, median OS was 28.7 months in the NLR-low group and 18.3 months in the NLR-high group. In patients receiving immunotherapy, median OS was 71.3 months in the NLR-low group and 18.2 months in the NLR-high group. Kaplan–Meier survival curves illustrating these differences are presented in Figures 1–3.
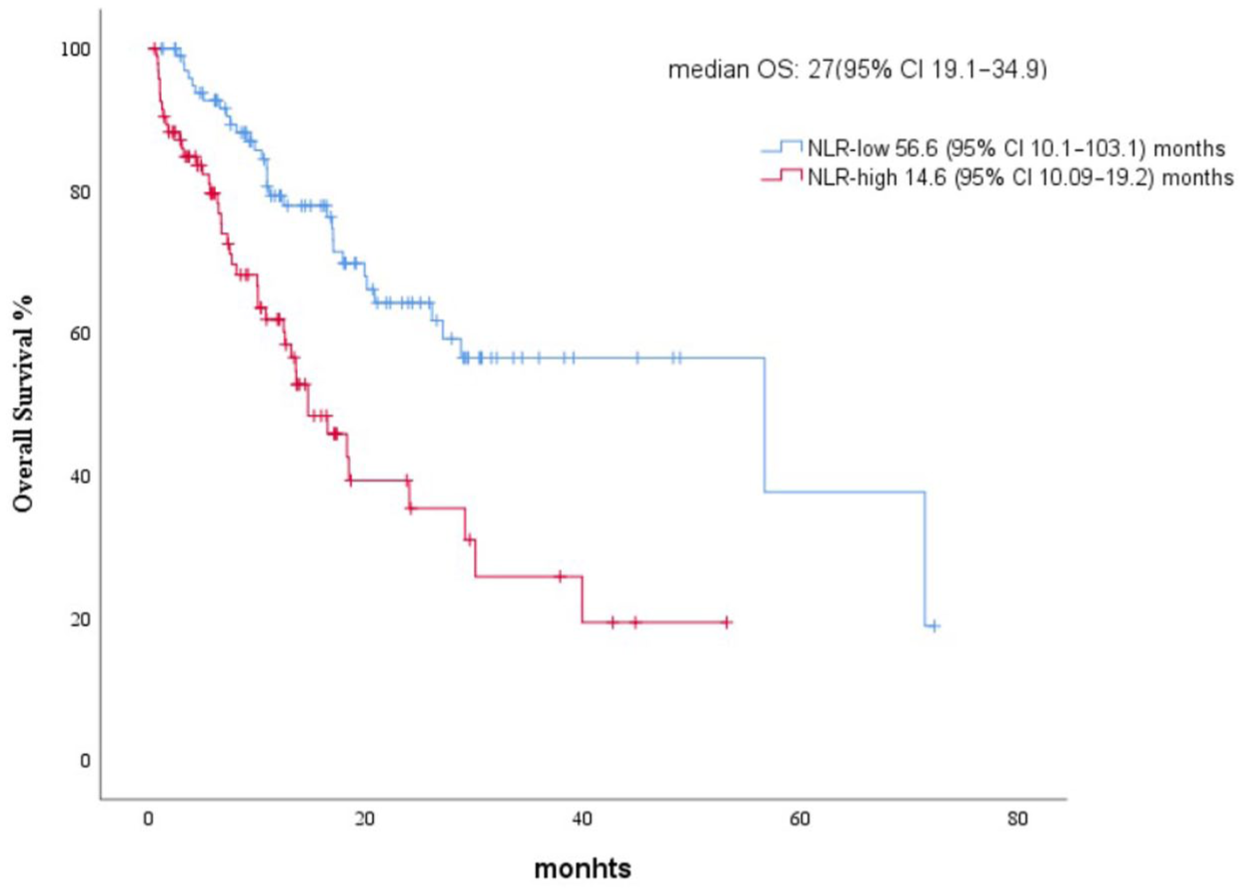
Kaplan–Meier curve for overall survival.
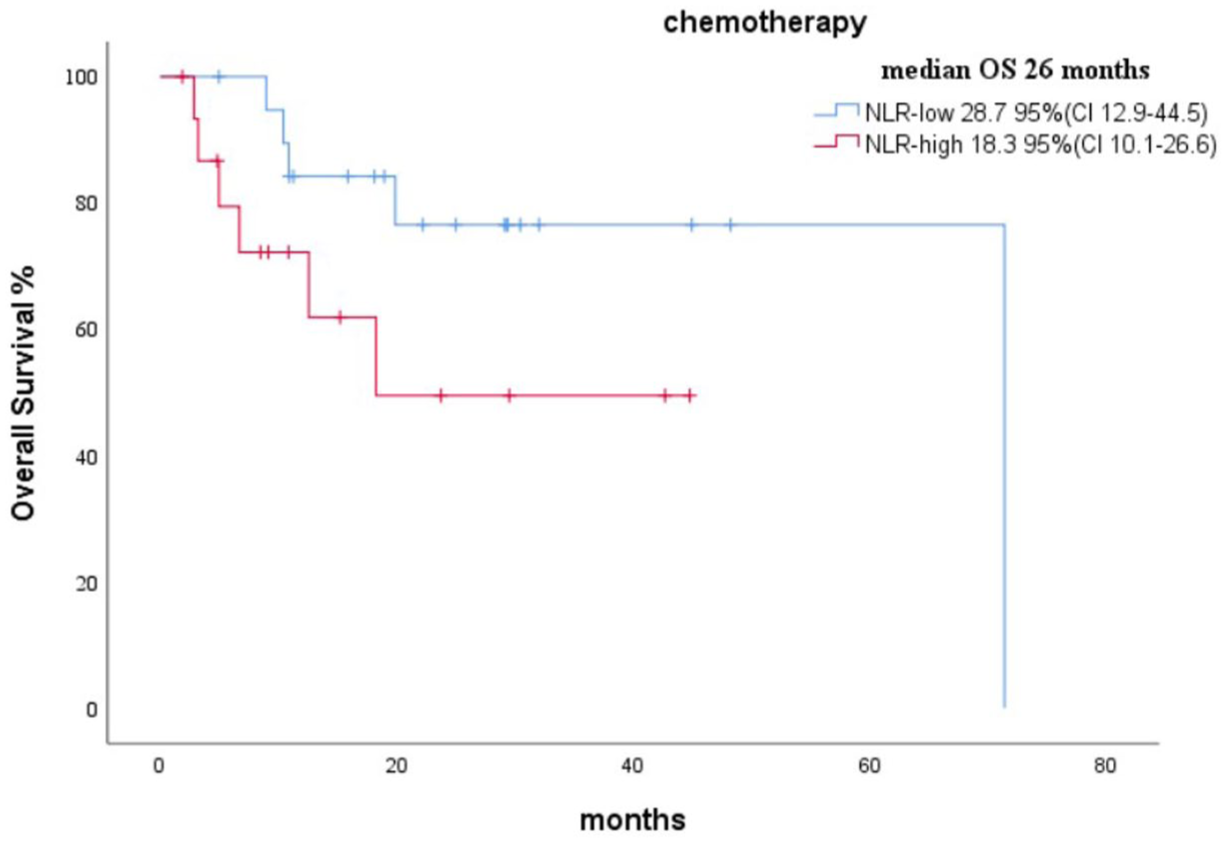
Kaplan–Meier survival curves for OS in patients treated with chemotherapy, stratified by NLR group (low vs high).
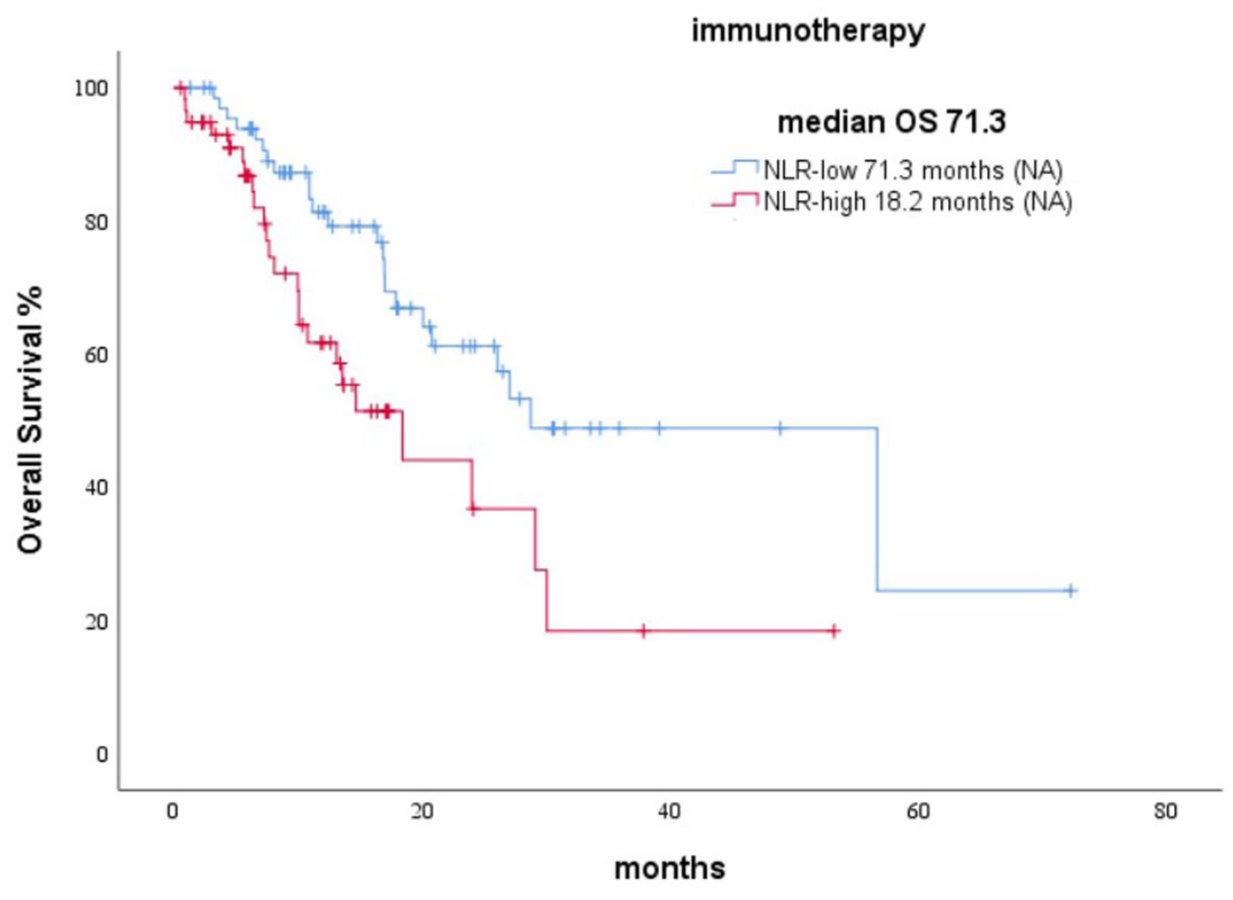
Kaplan–Meier survival curves for OS in patients treated with immunotherapy, stratified by NLR group (low vs high).
Association between NLR and metastatic burden
The association between NLR and metastatic burden was also examined. Bone metastases were significantly more common in the NLR-high group (68.7%) compared to the NLR-low group (33.6%) (p < 0.001). Similarly, liver metastases were more frequent in the NLR-high group (30.5%) than in the NLR-low group (12.9%) (p = 0.003). However, no significant relationship was observed between the rate of lung metastases and NLR. The incidence of lung metastases was 41.7% in the NLR-high group and 58.3% in the NLR-low group (Table 1).
Baseline characteristics and comparative analysis.

Patients with missing baseline NLR values were excluded prior to analysis. Therefore, no missing data were present for NLR in the final cohort. For other variables, the number of patients with available data is indicated in the table. Survival differences were assessed by the log-rank test.
NLR, neutrophil–lymphocyte ratio.
Discussion
Our study confirms that high NLR is an independent prognostic factor for OS in patients with mUC. Patients with low NLR had significantly longer OS than those with high NLR, regardless of treatment modality. Specifically, the median OS was 56.6 months in the NLR-low group compared with 14.6 months in the NLR-high group. Even after adjusting for age, liver metastases, and bone metastases, high NLR remained a strong predictor of poor survival. Furthermore, NLR retained its prognostic value in both chemotherapy- and immunotherapy-treated subgroups. The optimal cutoff value identified in our analysis was 4.2, although this threshold may vary depending on patient cohort characteristics. When survival probabilities were compared over time, patients with NLR <4.2 showed notably higher survival rates at both 20 months (65% vs 49%) and 40 months (52% vs 25%), corresponding to absolute differences of 16% and 27%, respectively. These findings highlight the persistent negative prognostic impact of systemic inflammation throughout the disease course.
The observed high median OS in the group with low NLR who received immunotherapy warrants further investigation. The relatively small sample size may influence this outcome in this subgroup. The wide confidence interval likely resulted from the limited number of patients who received immunotherapy. Increasing access to immunotherapy and repeating this study with more patients could make the results more reliable and widely applicable.
A notable finding in our study is the association between NLR and metastatic burden. Patients in the NLR-high group had a significantly higher incidence of bone and liver metastases. This suggests that systemic inflammation, as reflected by elevated NLR, may be linked to more aggressive disease phenotypes and tumor progression. Finally, our study strengthens the evidence supporting NLR as a robust prognostic biomarker in mUC.
NLR reflects the interplay between innate and adaptive immunity. Elevated NLR indicates persistent inflammation and immune system stress.16,17 It is widely recognized as a reliable and accessible prognostic marker in solid tumors. 18 However, no standardized cutoff value has been established for clinical use. 19
Chronic inflammation contributes to the development and progression of invasive and metastatic bladder cancer. Therefore, NLR may serve as a valuable clinical marker for urothelial cancer prognosis.
A pooled analysis of 13 studies, including 9 studies of operable bladder cancer patients and 4 studies of metastatic or unresectable urothelial carcinoma patients, demonstrated that pretreatment with a high NLR is a poor prognostic factor for progression-free survival and OS. The NLR cutoff values in these studies ranged from 3 to 5. 20 Another multicenter study involved patients treated with chemotherapy with mUC, demonstrating that high NLR was associated with poor prognosis. The investigators assessed NLR by dividing it into tertiles and reported that OS was higher in the lowest tertile (NLR 0.6–2.78) compared to the highest tertile (NLR 4.70–48.9). 15 Another study involving mUC patients treated with immune checkpoint inhibitors found that high NLR had significantly poorer prognoses. This study used a cutoff value of 7.1 and included 355 patients. 21 However, a different study included mUC patients treated with immune checkpoint inhibitors, defined the NLR cutoff as 3, and demonstrated that the higher NLR group has a worse prognosis. 22
Collectively, these data support NLR as a prognostic factor in urothelial carcinoma, though the optimal threshold remains undefined. Therefore, further studies in larger cohorts, particularly including patients treated with new therapies, are warranted.
This study has several limitations. First, its retrospective design may introduce selection bias and limit causal inferences. Second, due to the retrospective design and the relatively small number of patients, a formal power analysis was not feasible. Third, heterogeneity in treatment regimens across patients could affect survival outcomes. Fourth, although we used a well-established NLR cutoff, there is no universally accepted threshold, which may impact reproducibility in different cohorts. Lastly, we did not evaluate longitudinal changes in NLR, which might provide additional prognostic insights. Despite these limitations, our study has notable strengths. It includes a large, multi-center cohort, enhancing generalizability. We used rigorous statistical analyses, including multivariate adjustment, to confirm NLR as an independent prognostic factor. In addition, we evaluated NLR’s prognostic role in both chemotherapy and immunotherapy-treated patients, providing valuable insights for clinical decision-making. In addition, the median follow-up duration (17.9 months) was shorter than the observed median OS, reflecting the censoring rate in this cohort.
Conclusion
High NLR is a simple, inexpensive, and widely available biomarker associated with poor survival in mUC. These findings highlight its potential to aid in risk stratification and treatment decision-making in clinical practice. Although first-line treatment for mUC has evolved with the introduction of enfortumab vedotin plus pembrolizumab, access remains limited, and platinum-based chemotherapy continues to be widely used. Prognostic markers such as NLR remain highly relevant for optimizing treatment strategies in routine care. Future prospective studies should validate the role of NLR in diverse patient populations and evaluate its dynamic changes over time, as well as its predictive value for emerging therapies. Integrating NLR with other inflammatory or molecular biomarkers may further improve prognostic accuracy and guide personalized treatment approaches.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251403390 – Supplemental material for The prognostic role of the neutrophil-to-lymphocyte ratio in metastatic urothelial carcinoma
Supplemental material, sj-docx-1-tam-10.1177_17588359251403390 for The prognostic role of the neutrophil-to-lymphocyte ratio in metastatic urothelial carcinoma by Sevinc Balli, Fatih Yildiz, Mehmet Ali Nahid SSendur, Nuriye Yildirim Ozdemir, Oguzcan Gumuscubuk, Emre Yekeduz and Yuksel Urun in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251403390 – Supplemental material for The prognostic role of the neutrophil-to-lymphocyte ratio in metastatic urothelial carcinoma
Supplemental material, sj-docx-2-tam-10.1177_17588359251403390 for The prognostic role of the neutrophil-to-lymphocyte ratio in metastatic urothelial carcinoma by Sevinc Balli, Fatih Yildiz, Mehmet Ali Nahid SSendur, Nuriye Yildirim Ozdemir, Oguzcan Gumuscubuk, Emre Yekeduz and Yuksel Urun in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
