Abstract
Background:
The aging population presents significant challenges to healthcare worldwide. Evidence concerning the safety and efficacy of neoadjuvant immunochemotherapy in patients with non-small-cell lung cancer (NSCLC) aged 70 years or older remains limited.
Objectives:
To investigate the safety and efficacy of neoadjuvant immunochemotherapy in NSCLC patients stratified by age into three groups, and to identify factors associated with overall survival (OS) and disease-free survival (DFS).
Design:
We performed a retrospective cohort study including 171 NSCLC patients with NSCLC who underwent neoadjuvant immunochemotherapy followed by surgical resection. The patients were categorized by age into three groups: ⩾70 years, 60–69 years, and <60 years.
Methods:
The safety and efficacy of neoadjuvant immunochemotherapy were comprehensively evaluated. Safety was assessed based on the incidence of treatment-related adverse events (AEs) and complications. Efficacy was determined through analyses of tumor response and survival outcomes. OS and DFS were analyzed using the Kaplan–Meier method, and independent prognostic factors were identified through the Cox proportional hazards model.
Results:
The study cohort comprised 24 patients aged ⩾70 years, 73 patients aged 60–69 years, and 74 patients under 60 years. OS and DFS did not differ significantly among the three age groups following neoadjuvant immunochemotherapy. Multivariate analysis identified major pathological response (MPR) as a significant independent predictor of OS (hazard ratio (HR): 0.232, 95% confidence interval (CI): 0.079–0.678, p = 0.008). For DFS, both MPR (HR: 0.342, 95% CI: 0.184–0.638, p = 0.001) and the occurrence of postoperative complications (HR: 2.115, 95% CI: 1.208–3.705, p = 0.009) were independent predictors. Overall, patients across all age groups exhibited acceptable tolerance to neoadjuvant immunochemotherapy.
Conclusion:
Neoadjuvant immunochemotherapy demonstrated consistent safety and efficacy across all age groups in this cohort of NSCLC patients. Achieving MPR was associated with improved OS and DFS, whereas the occurrence of postoperative complications was associated with diminished DFS.
Introduction
Non-small-cell lung cancer (NSCLC) is the primary cause of cancer-related deaths globally. Approximately 30%–55% of patients would experience relapse, metastasis, or mortality following surgery.1,2 Recently, neoadjuvant immunochemotherapy has emerged as a promising strategy to downstage tumors, improve resectability, and target micrometastatic disease.3,4 The phase III CheckMate 816 trial demonstrated that preoperative immunochemotherapy significantly enhanced pathological complete response (pCR) rates and event-free survival compared to chemotherapy alone. 5 Despite the promising results, concerns remain unresolved regarding the effectiveness and safety of neoadjuvant immunochemotherapy, especially in elderly patients.
Elderly individuals, who comprise the majority of NSCLC, face unique challenges in cancer treatment. Age-related comorbidities, physiologic changes at the organ level, disabilities, and geriatric syndromes may contribute to treatment-related side effects and poorer outcomes, which make the employment of chemotherapies circumspect. 6 In addition, aging is associated with a decline in immune surveillance and antigen presentation, a rise in memory lymphocytes and immunosuppressive regulatory T cells, and a reduction in lymphocytes capable of mounting anti-tumor responses.7,8 These alterations lead to a paradoxical state in which basal immune activity is heightened, yet responsiveness to immunogenic stimuli, such as immune checkpoint inhibitors (ICIs), is diminished.
The efficacy of neoadjuvant immunochemotherapy in elderly cohorts remains controversial. The KEYNOTE-189 study reported that elderly patients derived less benefit from immunochemotherapy compared to younger patients after immunochemotherapy, while the IMPOWER-130 study revealed similar benefits across age groups.9,10 Furthermore, most studies have employed 65 years as the cutoff for defining elderly patients, which has failed to reflect the increasingly aging global population and may not accurately represent contemporary clinical demographics.9–11 Given that over two-thirds of lung cancer cases occur in patients aged 65 years or older, coupled with a median NSCLC diagnosis age of approximately 70 years,12,13 there is an urgent need for comprehensive evaluation of neoadjuvant immunochemotherapy efficacy and safety specifically in the population aged greater than 70 years.
The study aimed to evaluate the safety and effectiveness of neoadjuvant immunochemotherapy in NSCLC patients across three age groups: ⩾70, 60–69, and <60 years. The factors related to overall survival (OS) and disease-free survival (DFS) in NSCLC patients receiving neoadjuvant immunochemotherapy were further identified.
Methods
We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline 14 (Supplemental File 1).
Patients
Patients with resectable NSCLC who underwent neoadjuvant immunochemotherapy at Wuhan Union Hospital from September 2019 to February 2023 were included. The inclusion criteria were as follows: (1) NSCLC patients aged 18 years or older with stage IIA–IIIB; (2) patients who underwent at least two cycles of immunochemotherapy, with no other concomitant treatments administered; and (3) patients with an Eastern Cooperative Oncology Group performance status of 0–1. The exclusion criteria were as follows: (1) patients with incomplete clinical and pathological data and (2) patients with other malignancies or distant metastases. Then, the patients were categorized into three groups based on age: the elderly NSCLC group (E-NSCLC, aged 70 years and above), the middle-aged NSCLC group (M-NSCLC, aged 60–70 years), and the young NSCLC group (Y-NSCLC, under 60 years).
Treatment protocol
Before surgery, all patients were treated with at least two cycles of neoadjuvant immunochemotherapy based on multidisciplinary team evaluation and in accordance with contemporaneous clinical guidelines and consensus recommendations.15,16 The ICIs consisted of sintilimab (200 mg), camrelizumab (200 mg), pembrolizumab (200 mg), durvalumab (200 mg), or tislelizumab (200 mg). The paclitaxel–platinum (TP) regimen consisted of paclitaxel (175 mg/m2) and platinum-based drugs, such as carboplatin (AUC of 5 mg/ml/min), cisplatin (75 mg/m²), or nedaplatin (75 mg/m²). Surgical approaches were composed of thoracotomy and thoracoscopy, accompanied by routine lymph node dissection.
Follow-up and efficacy assessment
Follow-up CT examinations were performed at baseline and before surgery to assess tumor response. The images were independently assessed by two radiologists using a blinded approach to identify tumor features and response. According to the response evaluation criteria in solid tumor version 1.1 (RECIST 1.1), 17 tumor responses were classified into progressive disease (PD), stable disease (SD), partial response (PR), or complete response (CR). The calculation of the objective response rate (ORR) involved summing the rates of CR and PR. Pathological response included major pathological response (MPR), where 10% or fewer viable tumor cells were present, and pCR, where no viable tumor cells existed in the tumor bed and lymph nodes. OS referred to the duration from operation until mortality or the end of follow-up. DFS was determined as the time between operation and disease progression, death, or the end of follow-up. Follow-up was performed via telephone, and patients were followed up until October 18, 2024. To explore potential predictors of OS and DFS, factors including age, gender, smoking history, comorbidities, tumor size, location, tumor-node-metastasis (TNM) stage, 18 treatment time, surgical approach, surgical procedure, histology, epidermal growth factor receptor (EGFR) status, anaplastic lymphoma kinase (ALK), programmed cell death ligand 1 (PD-L1) expression level, and resection margin were recorded.
Assessment of safety
Adverse events (AEs) after neoadjuvant immunochemotherapy, as well as postoperative complications, were documented to evaluate safety. AEs were assessed according to the Common Terminology Criteria for Adverse Events, version 5.0. 19 The pulmonary postoperative complications included hydropneumothorax, pneumothorax, respiratory failure, pneumonia, atelectasis, bronchospasm, chylothorax, and empyema. 20
Statistical analysis
Statistical analyses were conducted using GraphPad Prism (version 8.0.2) and SPSS Statistics 26 (IBM Inc., Armonk, NY, USA). Continuous data were shown as median (interquartile range), and categorical data were reported as frequencies (percentages). The Kruskal–Wallis H test and Fisher’s Exact test were applied for statistical analysis. Kaplan–Meier methods were generated and compared using the log-rank tests. The Cox proportional hazards model was applied for univariate and multivariate analyses of OS and DFS. Variables with p values of ⩽0.2 from the univariate analysis were incorporated into the multivariate model to determine independent prognostic factors. The hazard ratio (HR) and 95% confidence intervals (CIs) were estimated. A statistically significant difference was regarded as a two-tailed p value <0.05.
Results
Patient characteristics
The study included 171 NSCLC patients in total. Of these patients, 24 (14.0%) were aged 70 or older (E-NSCLC), 73 (42.7%) were aged between 60 and 70 years (M-NSCLC), and 74 (43.3%) were younger than 60 years (Y-NSCLC). Elderly patients were more likely to have cardiovascular diseases (p = 0.015) and chronic respiratory diseases (p = 0.003). While lobectomy was the predominant procedure (88.9%), the E-NSCLC group showed higher rates of wedge resection or segmentectomy compared to M-NSCLC and Y-NSCLC groups (8.3% vs 1.4% vs 0%, respectively). No statistical differences were observed in tumor size, location, TNM stage, treatment time, surgical approach, histology, and resection margin (all p > 0.05; Table 1). Among the study population, EGFR mutation testing was performed in 26 patients, all of whom were wild type. ALK rearrangement was assessed in 32 patients, with all results negative. PD-L1 expression was conducted in 26 patients, with 8 showing <1%, 12 between 1% and 49%, and 6 ⩾50%.
Demographic and clinical characteristics of patients between the different groups.

CAVDs, cardiovascular diseases; CEVDs, cerebrovascular diseases; CRDs, chronic respiratory diseases; E-NSCLC, elderly NSCLC; M-NSCLC, middle-aged NSCLC; NSCLC, non-small-cell lung cancer; Y-NSCLC, young NSCLC.
Follow-up and tumor response
The median follow-up time was 29.47 months for OS and 25.33 months for DFS. The OS rates were 16.7%, 12.3%, and 16.2% in E-NSCLC, M-NSCLC, and Y-NSCLC groups, respectively, and the DFS rates were 25.0%, 34.2%, and 35.1%, respectively. Within the E-NSCLC group, 3 patients reached CR, 15 patients reached PR, 4 patients obtained SD, and 2 patients obtained PD, resulting in an ORR of 75% (18/24). In the M-NSCLC group, 3 patients attained CR, 49 patients attained PR, 19 patients experienced SD, and 2 patients experienced PD, resulting in an ORR of 71.2% (52/73). In the Y-NSCLC group, 2 patients reached CR, 46 patients reached PR, 24 patients experienced SD, and 2 patients experienced PD, with an ORR of 64.9% (48/74). Regarding pathological response, the rates of MPR in E-NSCLC, M-NSCLC, and Y-NSCLC groups were 45.8% (11/24), 46.6% (34/73), and 44.6% (33/74), respectively. The rate of pCR in the E-NSCLC group was 20.8% (5/24), 30.1% (22/73) in M-NSCLC, and 27% (20/74) in the Y-NSCLC group (Figure 1). Overall, no significant differences were found in treatment response and survival outcomes among the three groups (all p > 0.05; Table 2).

The distribution condition of tumor response, including OS (a), DFS (b), ORR (c), MPR (d), pCR (e), and PD/SD/PR/CR (f).
Tumor response and survival outcomes after neoadjuvant immunochemotherapy.

CR, complete response; DFS, disease-free survival; E-NSCLC, elderly NSCLC; M-NSCLC, middle-aged NSCLC; MPR, major pathological response; NSCLC, non-small-cell lung cancer; ORR, objective response rate; OS, overall survival; pCR, pathological complete response; PD, progressive disease; PR, partial response; RECIST 1.1, response evaluation criteria in solid tumor version 1.1; SD, stable disease; Y-NSCLC, young NSCLC.
Safety
Among the 171 patients, three patients developed AEs of grade ⩾3, including anemia and thrombocytopenia, all of whom belonged to the Y-NSCLC group. AEs, such as leukopenia, agranulocytosis, anemia, thrombocytopenia, nausea or emesis, and hepatic injury, were observed in all groups, with no significant differences among them. All aforementioned AEs were relieved after symptomatic treatment, and no patients experienced delays in surgery. In addition, two patients died during the perioperative period due to respiratory failure. Hydropneumothorax and pneumothorax occurred in all groups, while respiratory failure and pneumonia were observed only in M-NSCLC and Y-NSCLC groups. No significant differences were observed (Table 3).
AEs after neoadjuvant immunochemotherapy and postoperative complications.
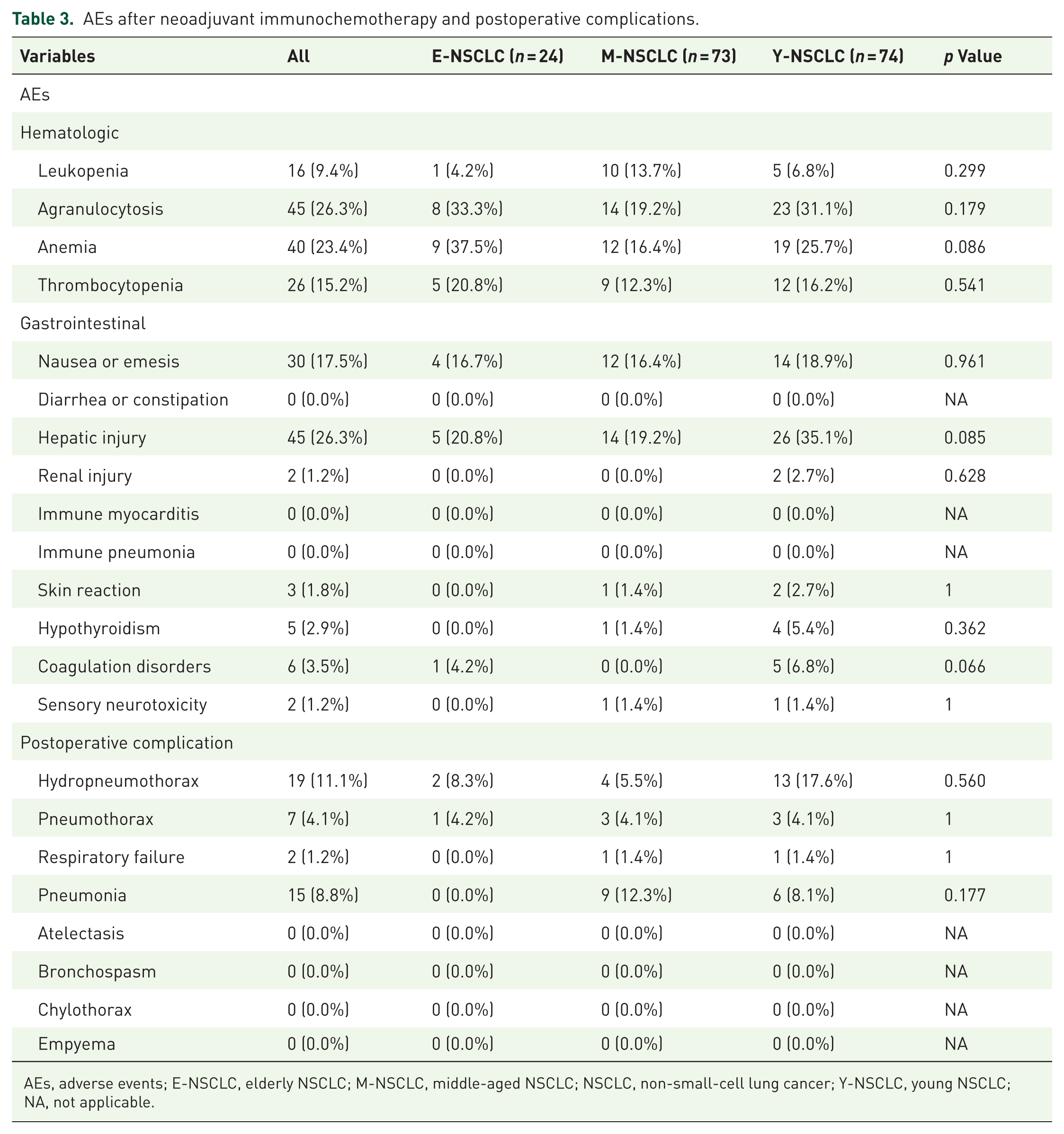
AEs, adverse events; E-NSCLC, elderly NSCLC; M-NSCLC, middle-aged NSCLC; NSCLC, non-small-cell lung cancer; Y-NSCLC, young NSCLC; NA, not applicable.
Survival analysis
In the univariate analysis, tumor location (p = 0.181), surgical approach (p = 0.129), and MPR (p = 0.008) were significantly associated with OS. In the multivariate analysis, only MPR (HR: 0.232, 95% CI: 0.079–0.678, p = 0.008) continued to be significant (Table 4). Similarly, univariate analysis identified tumor location (p = 0.153), MPR (p = 0.001), and postoperative complication (p = 0.003) as factors significantly related to DFS. Multivariate Cox analysis further confirmed that MPR (HR: 0.342, 95% CI: 0.184–0.638, p = 0.001) and postoperative complication (HR: 2.115, 95% CI: 1.208–3.705, p = 0.009) were independent prognostic factors for DFS in NSCLC patients receiving neoadjuvant chemoimmunotherapy (Table 5).
Results of univariate and multivariate analyses of factors associated with OS.

ADC, adenocarcinoma; CI, confidence interval; E-NSCLC, elderly NSCLC; HR, hazard ratio; M-NSCLC, middle-aged NSCLC; MPR, major pathological response; NSCLC, non-small-cell lung cancer; ORR, objective response rate; OS, overall survival; SCC, squamous cell carcinoma; TNM, tumor-node-metastasis; Y-NSCLC, young NSCLC.
Results of univariate and multivariate analyses of factors associated with DFS.

ADC, adenocarcinoma; CI, confidence interval; DFS, disease-free survival; E-NSCLC, elderly NSCLC; HR, hazard ratio; M-NSCLC, middle-aged NSCLC; MPR, major pathological response; NSCLC, non-small-cell lung cancer; ORR, objective response rate; SCC, squamous cell carcinoma; TNM, tumor-node-metastasis; Y-NSCLC, young NSCLC.
Kaplan–Meier curves for OS and DFS were assessed according to MPR and postoperative complications. The differences in survival times among the groups were statistically significant (Figure 2). Moreover, Kaplan–Meier curves for OS and DFS in the E-NSCLC, M-NSCLC, and Y-NSCLC groups are shown in Figure 2, although no statistical significances were observed.

Kaplan–Meier analysis of OS (a–c) and DFS (d–f) based on major pathological response (a, d), postoperative complication (b, e), and age (c, f).
Discussion
With the growing prevalence of NSCLC and the aging population at diagnosis, it remains crucial to determine the optimal treatment strategy for NSCLC patients aged 70 years and older. In this study, we observed that advanced age did not independently contribute to contraindications for neoadjuvant immunochemotherapy, nor was it associated with shorter OS or DFS, AEs, or postoperative complications. MPR and postoperative complications were recognized as prognostic factors for survival outcomes.
Although elderly patients were not considered contraindicated, they were often excluded from clinical trials or practice due to higher rates of comorbidities, which could raise neoadjuvant immunochemotherapy-related complications. 21 We found that the E-NSCLC group had a higher percentage of cardiovascular diseases and chronic respiratory diseases than the M-NSCLC and Y-NSCLC groups. Wedge resection or segmentectomy was more common in the E-NSCLC group, while sleeve resections were more frequent in the Y-NSCLC group. This difference may be due to patient age and tumor characteristics. Elderly patients prefer less invasive surgery to preserve lung function. Sleeve resection is typically used for tumors that infiltrate the central airway or blood vessels to achieve local control and preserve lung function. 22 We noticed that the proportion of central NSCLC in the Y-NSCLC group in the study was higher compared to other age groups, although the difference was not statistically significant. Considering that younger patients typically present with fewer comorbidities and better overall health status, they are more eligible for sleeve resection. To note, for patients with resectable stage IIA–IIIB lung adenocarcinoma receiving neoadjuvant immunochemotherapy, surgery is generally considered when the disease achieves remission or remains stable. In PD, surgical decisions require careful multidisciplinary evaluation.15,16,23
The research revealed that there was no significant difference in treatment efficacy and survival outcomes among the three groups, in line with prior studies on the efficacy of neoadjuvant immunochemotherapy in elderly groups with varying age thresholds. Liu et al. 11 reported comparable OS and DFS rates between elderly and young NSCLC patients, with a cutoff age of 65 years receiving neoadjuvant immunochemotherapy. Zheng et al. 24 found that NSCLC patients over 65 years had similar pCR rates with patients under 65 years after neoadjuvant immunochemotherapy. These results were further corroborated by our investigation, which demonstrated that elderly patients over 70 years receiving neoadjuvant immunochemotherapy exhibit comparable tumor response to those aged under 70 years. These findings offer additional support that age has no significant impact on the effectiveness of neoadjuvant immunochemotherapy.
Safety was generally comparable across the three age groups in our study. Although the incidence of specific AEs did not differ significantly across the groups, age-related patterns emerged. In elderly patients, agranulocytosis and anemia were the most frequently observed AEs, likely due to age-related factors, such as reduced bone marrow reserve and poor treatment tolerance. 25 Younger and middle-aged patients more commonly experienced hepatic injury, which may be attributed to the use of more intensive treatment regimens and a more robust immune response. In addition, all grade ⩾3 AEs occurred exclusively in the Y-NSCLC group. The incidence of postoperative complications also did not differ significantly among the three age groups. The most common complication in both elderly and younger patients was hydropneumothorax, possibly due to age-related pleural fragility in the elderly and higher postoperative activity in younger individuals. In middle-aged patients, pulmonary infections were more frequent, which may be attributed to suboptimal postoperative pulmonary care or less aggressive infection prophylactic measures.
Recently, the MPR has been recognized as a crucial short-term endpoint capable of serving as a surrogate for long-term survival outcomes in neoadjuvant immunotherapy. 26 Similarly, our study also confirmed that MPR was significantly associated with both improved OS and DFS. In addition, DFS after neoadjuvant immunochemotherapy was influenced by postoperative complications, in line with previous studies.27,28 Other prognostic factors did not show significant differences in OS and DFS among three cohorts.
Our study had several limitations. First, the retrospective nature of the study led to inherent selection biases. Second, heterogeneity in patient selection and treatment regimens may affect the generalizability of our results; thus, larger-scale randomized controlled trials and prospective studies are warranted. Finally, not all patients underwent comprehensive biomarker testing (including EGFR, ALK, and PD-L1), which may limit the interpretability of differential treatment responses and outcomes.
Conclusion
In summary, the study indicated that advanced age had no negative effect on selecting neoadjuvant immunochemotherapy for NSCLC patients. The safety and prognosis of elderly NSCLC patients aged 70 years and above undergoing neoadjuvant immunochemotherapy resembled those of younger patients. In addition, MPR and postoperative complications were recognized as prognostic factors for survival outcome in NSCLC patients receiving neoadjuvant immunochemotherapy.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251399035 – Supplemental material for Safety and efficacy of neoadjuvant immunochemotherapy in non-small-cell lung cancer: a comparative study of patients above and below 70 years
Supplemental material, sj-docx-1-tam-10.1177_17588359251399035 for Safety and efficacy of neoadjuvant immunochemotherapy in non-small-cell lung cancer: a comparative study of patients above and below 70 years by Yuting Zheng, Mengting Huang, Qinyue Luo, Yimeng He, Hanting Li, Xiaoyu Han, Lijie Zhang and Heshui Shi in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
