Abstract
Background:
Metastatic urothelial carcinoma (mUC) remains a challenging malignancy with limited treatment options. The combination of RC48 (Disitamab Vedotin) plus programmed cell death protein 1 (PD-1) inhibitor represents a promising therapeutic strategy.
Objectives:
To evaluate the efficacy and safety of RC48 plus PD-1 inhibitors as first-line therapy for mUC in real-world practice.
Design:
A multicenter, retrospective cohort study.
Methods:
We retrospectively collected data from mUC patients who received RC48 plus PD-1 inhibitors as first-line therapy between July 2021 and February 2024 from three medical centers. Overall survival (OS), progression-free survival (PFS), objective response rate (ORR), and disease control rate (DCR) were calculated to evaluate the clinical efficacy. Treatment-related adverse events (TRAEs) were analyzed to assess the safety profiles.
Results:
A total of 63 patients with mUC were included, with a mean age of 67 years, and 71.4% were male. The most frequent metastatic sites were lymph nodes (77.8%), followed by the lung (31.7%), bone (23.8%), liver (19.0%), and others. The treatment response rates were as follows: 12 patients (19.0%) achieved a complete response, 33 patients (52.4%) had a partial response, and 10 patients (15.9%) experienced stable disease. The ORR was 71.4%, and the DCR was 87.3%. The median PFS was 10.5 months (95% confidence interval: 8.8–14.6 months), and the median OS was not reached. The most common TRAEs included fatigue (36.5%), anemia (34.9%), pruritus (33.3%), peripheral sensory neuropathy (28.6%), and nausea (28.6%). Grade III TRAEs occurred in seven patients (11.1%), and no Grade IV or V TRAEs were observed.
Conclusion:
The combination of RC48 and PD-1 inhibitors administered in 3-week cycles demonstrates efficacy and manageable safety as first-line therapy for mUC patients in a real-world setting.
Introduction
Urothelial carcinoma (UC) is a common urological malignancy with a rising incidence rate. 1 According to GLOBOCAN 2022, bladder cancer ranks as the ninth most common cancer globally, with approximately 614,000 new cases and 220,000 deaths. In China, there were about 90,000 new cases and 430,000 deaths from bladder cancer.2,3 About 5%–10% of patients are diagnosed with metastatic disease at initial diagnosis. Even after surgical treatment, around 30% of patients experience local recurrence, and up to 50% develop distant metastases. 4 Platinum-based chemotherapy remains a first-line treatment for metastatic urothelial carcinoma (mUC). However, about half of the patients cannot tolerate these regimens due to impaired renal function, poor physical condition, or treatment-related complications. Moreover, the efficacy of platinum-based chemotherapy is considered to be limited, with unsatisfactory long-term survival rates.5–7
Immune checkpoint inhibitors (ICIs) and antibody-drug conjugates (ADCs) have revolutionized the treatment of advanced urothelial cancer. ADCs, which consist of an anticancer drug, a linker molecule, and an antibody, have demonstrated antitumor activity against a wide range of tumor types. 8 Notably, the combination of enfortumab vedotin (EV) and pembrolizumab has become the first immune-combination ADC-based regimen to surpass traditional platinum-based chemotherapy. 9 This combination is now recommended as a standard first-line treatment by the National Comprehensive Cancer Network guidelines. 10 RC48, a HER2-targeted ADC, containing the novel humanized anti-HER2 antibody hertuzumab conjugated to monomethyl auristatin E via a cleavable linker. 8 HER2 plays a vital role in the progression of several cancers, including breast, gastric, and urothelial cancers. 11 HER2 expression is positive in up to 12.4%–36.1% of UC patients.12–14 This positive expression is strongly associated with tumor progression and poor clinical outcomes.12,13 Several recent phase II randomized controlled trials (RC48-C005, RC48-C009, and RC48-C011) have shown that monotherapy with RC48 achieves a promising objective response rate (ORR) and progression-free survival (PFS) in previously treated mUC, regardless of HER2 expression status.15,16 RC48-C014 study found that RC48 in combination with toripalimab was superior to RC48 monotherapy in terms of ORR and mPFS. 17 Furthermore, the RC48-G001 advanced the RC48 and pembrolizumab combination regimen as a first-line treatment for real-world mUC, with Cohort C reporting an impressive ORR of 61.1%. 18
However, patients were enrolled based on strict eligibility criteria in these clinical trials, which may exclude patients with specific conditions. In addition, research on RC48-ADC combined with programmed cell death protein 1 (PD-1) inhibitors as a first-line treatment for mUC is still limited. Results from previous phase I and phase II clinical trials have shown that the 3-weekly combination regimen is safe and feasible (ClinicalTrials.gov identifier: NCT05495724). 19 Therefore, this real-world study evaluated the efficacy and safety of the RC48 plus PD-1 inhibitor regimen (once every 3 weeks) as a first-line treatment therapy in a broader population of mUC patients, including those who may have been excluded from clinical trials.
Materials and methods
Study design
This was a multicenter, retrospective, real-world study conducted at three medical centers, including Fujian Medical University Union Hospital, Fuzhou University Affiliated Provincial Hospital, and The Second Affiliated Hospital of Fujian University of Traditional Chinese Medicine. Patients who received RC48 in combination with a PD-1 inhibitor as first-line treatment for mUC were enrolled in this study. The medical ethics committee of each study center approved the study protocol. The study complied with the Declaration of Helsinki. All patients or authorized family members were fully informed about the treatment, and provided written informed consent before receiving treatment.
Patients
Patients with mUC without prior treatment in three clinical institutions between January 2021 and February 2024 were included in this study. Inclusion criteria were: (1) UC confirmed by histological examination, with or without histologic variants; (2) refusal or intolerance to platinum-based chemotherapy, with RC48 combined with a PD-1 inhibitor administered as first-line treatment; (3) presence of metastatic diseases, including extra-regional lymph node, bone, or visceral involvement; (4) at least one available response assessment.
Platinum intolerance was defined as: Eastern Cooperative Oncology Group (ECOG) performance status ⩾3, or creatinine clearance <30 mL/min; or ECOG performance status = 2 with creatinine clearance <45 mL/min. 20
Treatment protocol
Patients received RC48 (2.0 mg/kg) combined with either toripalimab (3 mg/kg) or tislelizumab (200 mg) via intravenous infusion every 3 weeks. RC48 was administered on Day 1, followed by the PD-1 inhibitor on Day 2 or Day 3. Treatment continued until disease progression, intolerable toxicity, death, or discontinuation for other reasons. Meanwhile, for patients with metachronous oligometastases (⩽3 metastatic lesions) who meet the criteria after multidisciplinary team evaluation, stereotactic body radiation therapy can serve as an adjunct to systemic therapy.
Data collection and follow-up
The following data, in accordance with the STROBE guidelines, were collected: age, gender, ECOG performance status, primary lesion site, visceral metastasis status, metastatic lesion sites, imaging features, laboratory results, pathology findings, histological classification, HER2 expression, prior immunotherapy, treatment regimen, and treatment-related adverse events (TRAEs). 21
HER2 immunohistochemistry (IHC) was assessed using anti-HER2/neu (4B5) Rabbit Monoclonal Primary Antibody in accordance with the Chinese Expert Consensus on Clinical Pathology of HER-2 Detection in Urothelial Carcinoma; the results were interpreted by two experienced pathologists. 22
Programmed cell death ligand protein 1 (PD-L1) is a rabbit anti-human monoclonal antibody (SP263). According to the Consensus on the immunohistochemical tests of PD-L1 in solid tumors (2021 version). Tumor cell (TC) is used for evaluation. (① TC >25%, or ② ICP >1% and IC+>25%, or ③ ICP = 1% and IC+ = 100%). 23
Follow-up was conducted through chart reviews, outpatient visits, and telephone visits. Patients were followed up every 12 weeks after treatment completion (Data cutoff: June 30, 2024). Imaging assessments, including computed tomography scans of primary and metastatic lesions, bone scans, and magnetic resonance imaging, were performed before treatment and every 2–3 cycles. An objective response was defined as a response that lasted for at least two consecutive imaging evaluations (at least 4 weeks apart). Dose adjustments during treatment were documented for patients receiving standard-dose therapy.
Evaluation of treatment response and adverse events
Tumor evaluation included ORR, PFS, disease control rate (DCR), overall survival (OS), duration of response, and dose adjustments. ORR was defined as the sum of complete response (CR) and partial response (PR) rates. DCR included CR, PR, and stable disease (SD). PFS was measured from treatment initiation to tumor progression or death from any cause. OS was measured from treatment initiation to death from any cause. Antitumor efficacy was evaluated using the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. 24
TRAEs were recorded based on patient reports or laboratory results, graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0. Liver and kidney function, blood counts, biochemical parameters, thyroid function, adrenocortical function, coagulation parameters, and tumor markers were assessed before and during treatment. Specific tests performed were determined by the clinician based on the patient’s individual condition.
Statistical analysis
P Values between different subgroups were calculated using the Chi-square test and Fisher–Freeman–Halton, and the Wilson Score and Clopper-Pearson method was used to calculate the ORR and 95% confidence intervals (CIs). Time-to-event endpoints, including PFS and OS, were analyzed using the log-rank test. The survival package was used to perform proportional hazards testing and fit survival regression models. The survminer and ggplot2 packages were used to visualize the results.
Results
Baseline characteristics
Patient demographics and baseline characteristics are presented in Table 1. The median age of patients was 67 years (range, 50–92). Of these, 36.5% (23/63) were aged over 70 years, and the male-to-female ratio was 2.5:1, with 71.4% (45/63) being male. In terms of physical status, 39.7% (25/63) of patients had a score of 1, while 11.1% (7/63) had a score of ⩾2. Regarding renal function, 7.9% (5/63) of patients had poor renal function with a baseline creatinine clearance of ⩽30 mL/min. The primary lesion was located in the upper urinary tract in 46.0% of cases.
Patient demographics and clinical features in metastatic urothelial carcinoma treated.
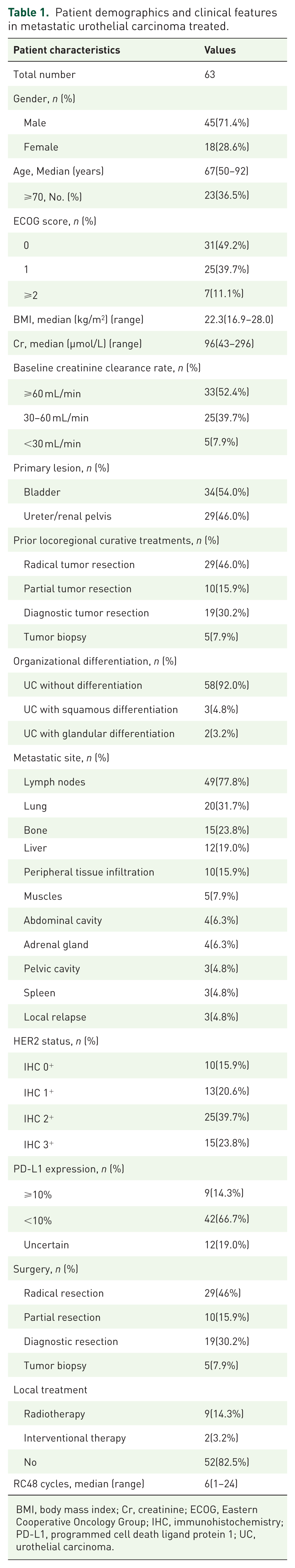
BMI, body mass index; Cr, creatinine; ECOG, Eastern Cooperative Oncology Group; IHC, immunohistochemistry; PD-L1, programmed cell death ligand protein 1; UC, urothelial carcinoma.
Prior surgeries included radical surgery (Radical Cystectomy and Radical Nephroureterectomy) in 46% (29/63) of patients, partial surgery (partial cystectomy) in 15.9% (10/63), diagnostic tumor resection in 30.2% (19/63), and tumor biopsy in 7.9% (5/63). Pathological analysis revealed that 92.0% of cases were UC alone, 4.8% were UC with squamous differentiation, and 3.2% were UC with glandular differentiation. All cases underwent immunohistochemical testing. HER2 expression levels by IHC were distributed as follows: IHC 0 in 15.9%, IHC 1+ in 20.6%, IHC 2+ in 39.7%, and IHC 3+ in 23.8% of cases. All 63 patients had distant organ metastases, with the most common sites being lymph nodes (77.8%), lungs (31.7%), bones (23.8%), and liver (19.0%).
In this study, all 63 patients received RC48-ADC combined with a PD-1 inhibitor as first-line therapy. Of these, 50 patients (79.4%) were treated with RC48 plus Toripalimab, and 13 patients (20.6%) received RC48 in combination with Tislelizumab. Nine patients (14.3%) received radiotherapy in addition to systemic therapy, and two patients (3.2%) received interventional therapy in addition to systemic therapy.
Efficacy
As of June 30, 2024, the median follow-up duration was 15.8 months (range: 4.0–41.9 months). All patients underwent at least one response assessment. Based on RECIST 1.1 criteria, treatment outcomes included CR in 12 patients (19.0%), PR in 33 patients (52.4%), and SD in 10 patients (15.9%). The ORR was 71.4%, and the DCR was 87.3% (Figure 1).
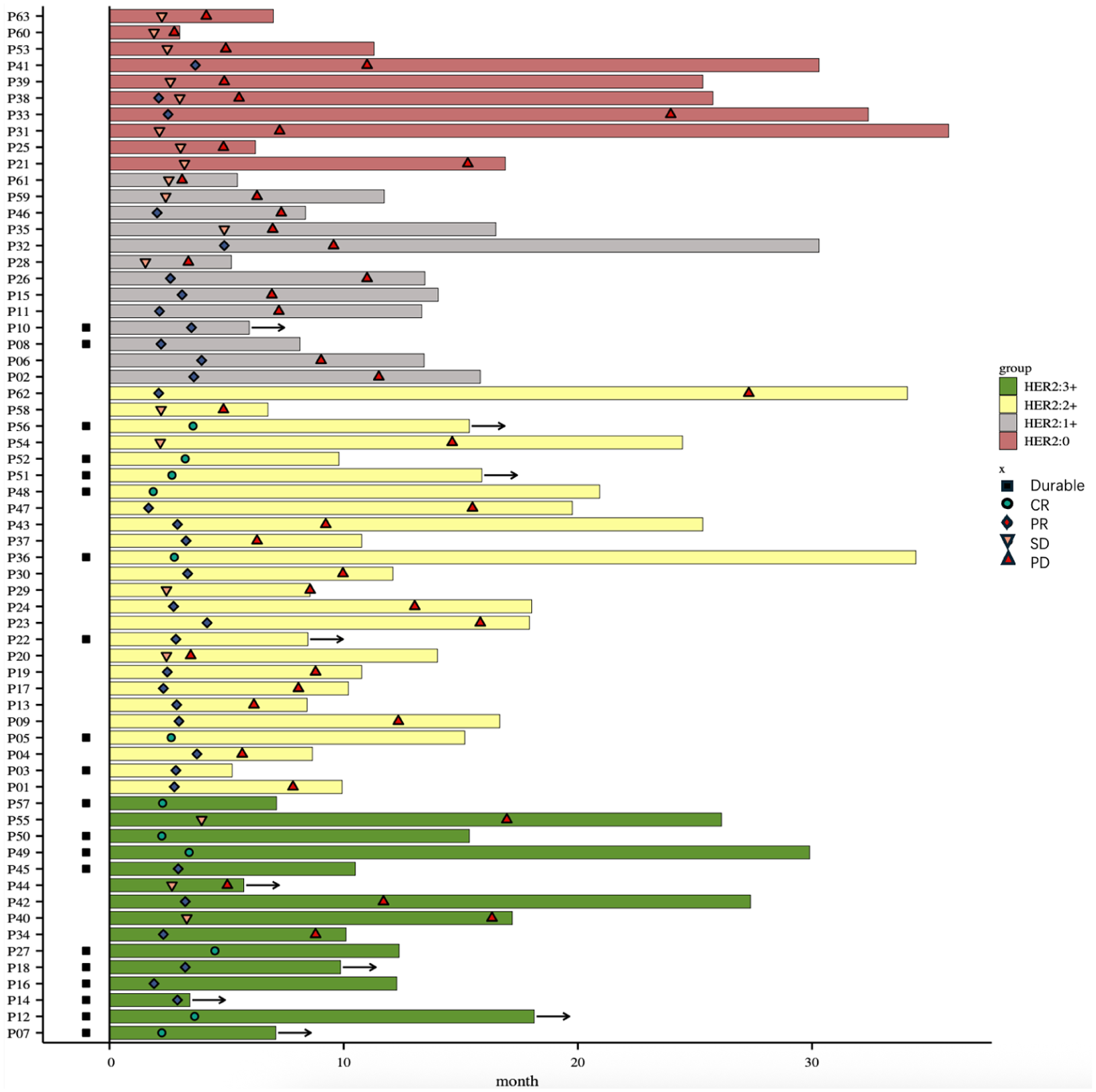
Swimmer plot of response assessment.
The ORR was 70.0% in patients aged <70 years and 74.0% in those aged ⩾70 years. The ORR was 81.8% (18/22) in patients with ECOG 0, 69.7% (23/33) in those with ECOG 1, and 50.0% (4/8) in those with ECOG ⩾2. The ORR was 78.8% (26/33) in patients with baseline creatinine clearance (Cr) ⩾60 mL/min, 64.0% (16/25) in those with baseline Cr 30–60 mL/min, and 60.0% (3/5) in those with baseline Cr <30 mL/min. The ORR was 60.0% in patients with lung metastases and 83.3% in those with liver metastases. The ORR for patients with HER2 3+, HER2 2+, HER2 1+, HER2 0-expressing patients was 80%, 84%, 69%, and 30% (p = 0.02). The ORR of patients with systemic therapy combined with radiotherapy, systemic therapy combined with interventional therapy, and systemic therapy alone was 78%, 100%, and 69% (Figure 2).

Optimal percent change from baseline in total target lesion diameter.
In all treated patients, 50 (79.4%) had a decrease in the total diameter of the tumor target lesion from baseline, 11 (17.5%) had an increase in the total diameter of the tumor target lesion from baseline, and 2 (3.1%) had no change in the total diameter of the tumor target lesion from baseline (Figure 3).

Forest plot of key subgroups of patients with ORR rows for multifactorial analysis.
By the last visit, disease progression occurred in 68.3% (43/63) of patients, and 30.2% (19/63) had died. The median OS was not reached, with an estimated median OS of 34.1 months and a 1-year OS rate of 72.4%. The mPFS was 10.5 months (95% CI: 8.8–14.6), with a 6-month PFS rate of 70.5% and a 12-month PFS rate of 22.7% (Figure 4).

Kaplan-Meier curves for (a) progression-free survival and (b) estimated overall survival calculated by statistical methods in patients receiving RC48 in combination with a PD-1 inhibitor.
The mPFS for HER2 (3+), HER2 (2+), HER2 (1+), and HER2 (0)-expressing patients was 16.3, 12.3, 7.3, and 5.3 months (p < 0.001). The mPFS for PD-L1 <10% and PD-L1 ⩾10% -expressing patients was 9.2 and 15.5 months (p = 0.12). The mPFS was 9.6 months for upper urinary tract lesions and 10.5 months for lower urinary tract lesions (p = 0.28). The mPFS was 12.3 months for patients with normal creatinine clearance and 8.1 months for those with impaired renal function (p = 0.08). The mPFS was 11.0 months for RC48 combined with toripalimab and 8.8 months for RC48 combined with tislelizumab (p = 0.90). Patients receiving systemic therapy with concurrent radiotherapy showed no significant improvement in median PFS compared to those receiving systemic therapy alone (ORR: 78% vs 69%, mPFS: 13.4 vs 9.2 months, p = 0.23; Figure 5).

Analysis of progression-free survival among patient subgroups treated with RC48-based therapy alongside PD-1 inhibitor. (a) HER2 expression. (b) Primary lesion location. (c) Age. (d) CCr. (e) PD-L1 expression. (f) ECOG. (g) Presence/absence of concomitant local therapy.
Safety
As of the data cutoff date, 8 patients remained on treatment, and all 63 patients were followed for safety and survival outcomes. TRAEs were reported in 82.5% of patients, primarily of low severity. Grade III TRAEs occurred in seven patients (11.1%), with five patients (7.9%) experiencing improvement after dose reduction or discontinuation. No grade IV or V TRAEs were observed.
The most common TRAEs (occurring in >20% of patients) were fatigue (36.5%), anemia (34.9%), pruritus (33.3%), peripheral sensory neuropathy (28.6%), nausea (28.6%), hypoalbuminemia (23.8%), and leukopenia (22.2%). In patients aged ⩾70 years, with ECOG PS ⩾2, CCr 30–60 mL/min, and CCr <30 mL/min, the incidence of grade II TRAEs was 34.8%, 75%, 44.0%, and 60%, respectively. The incidence of grade III TRAEs was 8.7%, 0%, 8.0%, and 40% in these subgroups. No grade IV–V events were observed.
Among patients who discontinued the RC48-ADC and PD-1 inhibitor combination, the main reasons were disease worsening (36.5%), adverse effects (11.1%), economic reasons (14.3%), the Novel Coronavirus Disease (9.5%), and unknown reasons (8.7%). Additionally, and 6 patients (9.5%) stopped treatment for CR assessment (9.5%). No deaths due to TRAEs occurred during treatment (Table 2).
Adverse events in all patients.

Discussion
RC48, a novel anti-HER2 ADC, has demonstrated promising efficacy in patients with locally advanced and metastatic urothelial carcinoma (la/mUC).16,25 The combination of ADC with PD-1 inhibitors has shown synergistic antitumor effects, which may be attributed to the tumor antigen exposure induced by ADC’s cytotoxic effects that potentially synergize with ICIs.26,27 Clinical trial data confirmed that the RC48 combination therapy demonstrated significant advantages over monotherapy RC48 alone or ICIs alone (RC48 combination with PD-1 inhibitors vs RC48 alone: ORR: 73.2% vs 50.5%; mPFS: 9.3 vs 5.9 months), which was also fully reflected in the study of Xu et al. and Sheng et al.19,25 In September 2020, RC48 was granted breakthrough therapy designation by the U.S. Food and Drug Administration for the indication of second-line therapy for HER2-positive la/mUC. On this basis, the Chinese Society of Clinical Oncology (CSCO) guidelines recommend DV combined with toripalimab as a first-line treatment option for mUC.
Based on phase I trials, RC48 Q3W has demonstrated acceptable efficacy and safety in advanced solid tumors. 20 A phase II study (NCT05495724) also adopted the Q3W cycle and initially validated its favorable efficacy in treating non-muscle-invasive bladder cancer (Clinical Trials.gov identifier: NCT05495724). The pharmacokinetic studies have affirmed the safety and efficacy of the Q3W regimen. 28 This study found that the efficacy endpoints of RC48 combined with PD-1 inhibitor Q3W showed good consistency with RC48-C014 trial data (ORR: 71.4% vs 73.2%; PFS: 10.5 vs 9.3 months). Remarkably, despite the higher proportion of HER2-low expression (HER2 1+: 39.7% vs 34.2%; HER2 0: 23.8% vs 7.32%) and poorer performance status (ECOG PS⩾2: 11.1% vs 0%) in this study population, the Q3W regimen demonstrated comparable antitumor activity to the C014 study. 29 In conclusion, the Q3W treatment program is safe and feasible. Besides, from the perspective of the Chinese healthcare system, the Q3W option may be more cost-effective and have a more favorable cost-benefit profile for patients with mUC, which requires further study.
In other real-world studies of RC48 monotherapy or combination therapy for mUC, the efficacy of the combination therapy was inferior to that in our study in terms of both ORR and mPFS (ORR: 38.8%–66.7% vs 71.4%; mPFS: 6.0–8.5 vs 10.5 months). This may be attributed to differences in treatment regimens. In other real-world studies, nearly half of the patients had received prior systemic therapy.28,30,31 In the present study, RC48 combined with a PD-1 inhibitor was investigated as a first-line therapeutic strategy. We aimed to compare this regimen with the current standard first-line treatment for mUC—gemcitabine plus cisplatin (GC) chemotherapy—to demonstrate RC48 combined with PD-1 inhibitors may represent a potentially advantageous therapeutic approach compared to the GC regimen (ORR: 71.4% vs 49%, mPFS: 10.5 vs 5.8–7.6 months).5–7
It is well known that TCs evade the immune system by overexpressing PD-L1. 32 This provides a theoretical basis for the clinical use of PD-1 inhibitors targeting the PD-1/PD-L1 axis. However, due to the complex and dynamic nature of the tumor immune microenvironment, the efficacy of ADC combined with PD-1 inhibitors remains difficult to predict.33,34 Our study also found no correlation between the efficacy of ADC combined with PD-1 inhibitor therapy and PD-L1 expression levels (p = 0.12), consistent with findings from the C014 study and previous research. It is relevant for clinicians to make reasonable clinical decisions based on the individual situation of patients, and they will choose different immunosuppressants based on the different expression levels of PD-L1 and the recommendations of the CSCO guidelines to seek the best therapeutic effect.
UC is more common in elderly patients. These patients often experience age-related health changes and usually have multiple underlying diseases and comorbidities. Many elderly patients can’t receive cisplatin-based chemotherapy due to poor general conditions and renal insufficiency. 35 Although PD-1 inhibitors such as pembrolizumab have been approved for first-line treatment, there remains room for improvement in the overall ORR among elderly patients.36,37 Current clinical data demonstrate that ADC exhibit promising therapeutic potential in elderly patients.16,17 Chen et al. 30 found no significant difference in ORR by age and no significant increase in TRAEs in elderly patients. Our study also found no significant difference in ORR and PFS in elderly patients, and no additional TRAEs were observed. Therefore, the combination of RC48 and PD-1 inhibitors is both effective and safe in elderly patients.
Renal function is a crucial factor that impacts treatment strategies for mUC patients. For mUC patients, a CCr rate below 60 mL/min is considered intolerable to cisplatin chemotherapy. 38 However, since the liver is the primary site of RC48 metabolism and PD-1 inhibitors gradually reduce their active components mainly through the regulation and clearance of the immune system, the RC48 and PD-1 inhibitor combination regimen will not exacerbate renal insufficiency in these patients with renal insufficiency.39,40 This is consistent with the previous UNITE study of RC48, in which 11.8% of patients had impaired renal function (eGFR <30 mL/min), and subgroup analyses showed no significant correlation between renal function and the efficacy of RC48. 41 The study data show that patients with normal renal function had a numerically higher ORR compared to those with renal impairment (CCr ⩾60 mL/min vs CCr 30–60 mL/min, CCr <30 mL/min: 78.8% vs 64.0%, 60.0%). Additionally, the mPFS was longer in patients with normal renal function than in those with renal impairment (12.3 vs 8.1 months), though the difference did not reach statistical significance (p = 0.08). In terms of safety, the incidence of TRAEs in patients with renal impairment showed an increasing trend as renal function worsened, but no grade 4–5 adverse events were observed. These findings are consistent with the results of other previous real-world studies.28,30 Thus, following clinical assessment, patients with renal insufficiency may be considered eligible for treatment with RC48 in combination with PD-1 inhibitors. Additionally, the administration of this regimen on a 3-weekly basis can mitigate drug toxicity, thereby enabling its safe application to a broader patient population.
For patients with advanced tumors, concurrent radiotherapy during systemic therapy following surgery is a well-established treatment strategy.42–44 Substantial clinical and preclinical evidence has demonstrated synergistic effects between these modalities, potentially mediated through the dissemination of tumor-specific antigens and stimulation of inflammatory microenvironments.45,46 Our study results showed that while outcomes of concurrent chemoradiotherapy were comparable to systemic treatment alone (ORR: 78% vs 69%, mPFS: 13.4 vs 9.2 months), the concurrent radiotherapy group demonstrated a favorable trend in hazard ratio (HR = 1.78, 95% CI: 0.70–4.54) suggesting better prognosis, though the difference did not reach statistical significance (p = 0.23). This may be attributed to insufficient sample size and short follow-up duration. We hope that subsequent phase III clinical trials or head-to-head comparisons will validate the efficacy of combining systemic therapy with radiotherapy.
In this study, most adverse reactions were Grade I–II. Fatigue (36.5%) and anemia (34.9%) were the most common. In addition, the incidence of grade III TRAEs was 11.1%, with no Grade IV or V TRAEs. All adverse events were effectively managed through symptomatic treatment. Compared with the data from the RC48-C014 trial report (all-grade: 82.5% vs 100%; grade ⩾3: 11.1% vs 36.6%), this study demonstrates significantly improved safety profiles.17,47 This favorable safety profile may be attributed to the reduced toxicity associated with the 3-week dosing interval.
This study has several limitations. First, as a retrospective analysis, it may be subject to patient recall bias. Second, the lack of a control group makes it impossible to directly compare the efficacy differences between RC48 combined immunotherapy and other treatment regimens. Thirdly, the relatively short follow-up period and the impact of COVID-19 during the treatment phase may have influenced the efficacy data. Fourth, the external validity of our findings may be limited by geographical constraints and inherent heterogeneity within the original study population. Therefore, when assessing the comparability and generalizability of these results, clinicians should incorporate individual variations specific to their local patient populations to enable more precise personalized treatment predictions.
Conclusion
In this study, we conducted a retrospective review and analysis of mUC patients from multiple centers. We included multifactorial analyses of age, ECOG performance status, and creatinine clearance. The results showed that the 3-weekly RC48 and PD-1 inhibitor regimen as first-line treatment had good efficacy and manageable safety in Chinese mUC patients. It also demonstrated excellent efficacy and survival in patients with poor general conditions (e.g., older age and impaired renal function), along with good tolerability.
