Abstract
Background:
Second-line options for biliary tract cancer (BTC) are limited. While nab-paclitaxel has demonstrated certain antitumor activity, evidence remains scarce. With gemcitabine–cisplatin plus anti-programmed death-ligand 1 (PD-(L)1) established as the first-line standard, the benefit of second-line immunotherapy—especially in patients without prior anti-PD-(L)1 exposure—remains unclear.
Objectives:
To evaluate the efficacy and safety of nab-paclitaxel-based second-line treatments for advanced BTC, and compare outcomes between regimens with and without anti-PD-(L)1.
Design:
This is a real-world retrospective study.
Methods:
This study reviewed BTC patients who received second-line nab-paclitaxel-based therapy at West China Hospital between August 2018 and August 2023. The primary endpoint was overall survival (OS) in the entire population. Secondary endpoints were progression-free survival (PFS), response rate, adverse events (AEs) in the entire population, and comparison of survival outcomes and response rate between the chemo-anti-PD-(L)1 and chemotherapy groups.
Results:
Among 84 patients (41 in the chemo-anti-PD-(L)1 group and 43 in the chemotherapy group), the median OS was 15.17 months (95% confidence interval (CI), 12.63–21.43), median PFS was 5.40 months (95% CI, 3.23–8.03), objective response rate (ORR) was 21.43% (95% CI, 13.22–31.74), and disease control rate (DCR) was 60.71% (95% CI, 49.45–71.20). Common grade 3–4 AEs were leukopenia (21.4%), neutropenia (17.9%), and anemia (13.1%). Hepatitis (14.6%) was the most frequent immune-related AE. Although a numerical trend favored the chemo-anti-PD-(L)1 group, no statistically significant differences were observed in OS (16.9 vs 14.6 months), PFS (7.3 vs 4.6 months), ORR (29.3% vs 13.9%), or DCR (68.3% vs 53.5%). In the entire cohort, radical surgery improved OS. In addition, patients with a baseline neutrophil-to-lymphocyte ratio ⩾3 and a ⩽30% reduction in carbohydrate antigen 19-9 from an initially elevated level during treatment had worse OS and PFS.
Conclusion:
Nab-paclitaxel-based regimens represent a promising second-line treatment option for BTC. Although the improvement was not statistically significant, adding anti-PD-(L)1 therapy showed a trend toward improved survival.
Trial registration:
ChiCTR2500096599.
Plain language summary
1. Patients who received nab-paclitaxel-based therapy survived for a median of 15.17 months.
2. Adding immunotherapy did not significantly improve survival, although a trend toward longer survival was observed.
3. Common side effects included bone marrow suppression. Overall, nab-paclitaxel-based treatment was generally safe and well tolerated.
4. Patients with an neutrophil-to-lymphocyte ratio (NLR) of 3 or higher at the start of treatment, and a CA19-9 decrease of 30 percent or less during treatment, had less benefit from nab-paclitaxel-based therapy.
Keywords
Introduction
Biliary tract cancer (BTC) accounts for less than 1% of all malignant tumors but is associated with a very poor prognosis. Approximately 50% of BTC patients are diagnosed at an advanced stage, with few candidates for surgery at initial presentation. The recurrence and metastasis rates within 1 year after surgery have been reported to be as high as 67%. The overall survival (OS) is typically less than 1 year, with a 5-year survival rate of only 5%–15%. 1
Second-line therapy options for BTC are still limited and provide only modest clinical benefits. Targeted therapies against actionable molecular alterations, such as RET or NTRK fusions, HER2 amplifications, FGFR2 fusions, IDH mutations, BRAF V600E, and KRAS G12C mutations, have shown favorable efficacy in BTC patients. However, these genetic alterations are relatively uncommon.2–6 For patients without targetable mutations, treatment choices are restricted. The FOLFOX regimen (folinic acid, fluorouracil, and oxaliplatin), recommended based on the ABC-06 study, extends OS by 0.9 months compared to active symptom control (6.2 vs 5.3 months). 7 The NIFTY trial demonstrated significant survival improvement with fluorouracil and leucovorin plus liposomal irinotecan 8 ; however, further validation in randomized phase III trials is needed. Therefore, there is a growing need to explore alternative second-line treatment strategies.
Nab-paclitaxel has shown promising antitumor activity in BTC.9–13 The combination of nab-paclitaxel and gemcitabine has been considered a viable first-line treatment option for cholangiocarcinoma. 13 Moreover, our prospective phase II clinical trial demonstrated that nab-paclitaxel combined with sintilimab (an anti-programmed death-1 (PD-1) antibody) exhibited promising efficacy and a manageable safety profile as a second-line treatment. 14 Even as a third-line therapy, weekly low-dose nab-paclitaxel monotherapy demonstrated encouraging efficacy in BTC (n = 6), achieving a disease control rate (DCR) of 83.3%. 15 While several studies suggest that nab-paclitaxel may be a potential option for second-line treatment of BTC, the sample size has ranged from 6 to 26, which is relatively small.
Immunotherapy has reshaped the treatment landscape of BTC. Both the TOPAZ-1 and KEYNOTE-966 trials confirm the favorable role of adding anti-PD-1/programmed death-ligand 1 (anti-PD-(L)1) immunotherapy to gemcitabine-cisplatin (GP) in the treatment of unresectable and metastatic BTC, making it a new standard of care.16,17 Based on the TOPAZ-1 trial, durvalumab was approved by the National Medical Products Administration in November 2023. Therefore, patients treated before this time generally did not receive anti-PD-(L)1 blockade as part of first-line therapy. In clinical practice, such patients are still encountered, yet evidence on the efficacy of combining chemotherapy with immunotherapy in the second-line setting for BTC remains limited.
In this study, we evaluated the efficacy and safety of nab-paclitaxel-based chemotherapy, with or without anti-PD-(L)1 blockade, as a second-line treatment for BTC in a real-world setting.
Methods
Patient selection
This was a single-center, real-world retrospective study approved by the Ethics Committee on Biomedical Research at West China Hospital, Sichuan University. We included all consecutive BTC patients who received second-line nab-paclitaxel-based therapy at West China Hospital between August 2018 and August 2023, with their data retrospectively collected. In this study, the term “real world” encompasses clinical data derived from standard clinical practice, along with investigator-initiated trials (IITs) conducted independently of industry sponsorship and protocol-driven randomized controlled trials. Of all the patients included in this study, 26 participated in an IIT conducted at West China Hospital (ChiCTR2100052118), all of whom were in the chemo-anti-PD-(L)1 group.
Eligibility criteria were as follows: patients aged 18 years or older with histologically confirmed locally advanced or metastatic BTC (including gallbladder cancer (GBC), intrahepatic cholangiocarcinoma (ICC), and extrahepatic cholangiocarcinoma (ECC)); patients who had experienced disease progression after first-line systemic therapy or adjuvant systemic therapy for metastasis or recurrence within ⩽6 months from the last dose; patients with measurable disease as defined by the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1; and patients who had received a second-line regimen containing nab-paclitaxel and had completed at least one radiographic response assessment at West China Hospital. Patients with a history of other malignancies and a disease-free survival of <5 years were excluded.
Treatment
In real-world settings, treatment decisions were made through multidisciplinary discussions and comprehensive communication with patients and their families, taking into consideration clinical factors (including Eastern Cooperative Oncology Group performance status (ECOG PS) and PD-L1 expression), patient preferences, and the family’s financial capacity.
Treatments were administered according to the recommended doses and schedules of the Guidelines of the Chinese Society of Clinical Oncology-Biliary Tract Cancer. A detailed description of the exact drug name and dose is in Supplemental Table 1. For adverse events (AEs) deemed attributable to the treatment agents and deemed intolerable, dose reductions were permitted for chemotherapy agents. However, dose modifications were generally not recommended for immune checkpoint inhibitors (ICIs). Temporary treatment interruption was allowed when clinically indicated.
Assessments
The primary endpoint was OS in the entire population. Secondary endpoints were progression-free survival (PFS), response rate, AEs in the entire population, and comparison of survival outcomes and response rate between the chemo-anti-PD-(L)1 and chemotherapy groups. An exploratory analysis was performed to assess factors influencing OS and PFS. These factors included prior radical surgery, baseline neutrophil-to-lymphocyte ratio (NLR), dynamic changes in carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9) levels during second-line treatment, original tumor locations, and receipt of third-line treatment.
AEs and abnormal laboratory findings were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0. Treatment-related adverse events were defined by the investigator as AEs for which a causal relationship with any study treatment was considered a reasonable possibility. Response was assessed according to RECIST, version 1.1. 18
Data collection
Data were extracted from the medical records and follow-up assessments of each eligible patient. The collected data encompassed patient demographics, cancer history, treatment details, and AEs, as well as laboratory, pathological, and radiological findings.
Statistical analysis
Continuous variables were summarized as medians and interquartile ranges (IQRs). Group medians were compared using the Kruskal–Wallis test. Categorical variables were presented as rates or proportions, with group differences assessed using Pearson’s χ2 test. Survival analysis was performed using Kaplan–Meier curves to estimate OS and PFS, with comparisons between groups conducted using the log-rank test. The Cox proportional hazards model was used to calculate hazard ratios (HRs) and their 95% confidence intervals (CIs). Missing data in biological variables, which constituted less than 20% of the total dataset, were imputed via multiple imputations (using the “mice” package in R statistical software). Specifically, CEA was missing in four cases (4.5%), CA19-9 was missing in nine cases (10.1%), and NLR was missing in eight cases (9.0%). Imputed data were included in subsequent analyses. Furthermore, sensitivity analyses were performed to assess the robustness of the results.
All statistical analyses were two-sided, with a p-value <0.05 deemed statistically significant. Analyses were conducted using R software (version 4.3.1; R Core Team, Vienna, Austria) and IBM SPSS Statistics (version 26.0; IBM Corp., Armonk, NY, USA). This cohort study was reported by the STROBE reporting checklist (Supplemental Table 2). 19
Results
Patients
A total of 84 patients were enrolled from August 2018 to August 2023, including 41 who received nab-paclitaxel-based chemotherapy combined with anti-PD-(L)1 blockade (chemo-anti-PD-(L)1 group) and 43 who received nab-paclitaxel-based chemotherapy alone (chemotherapy group). Detailed treatment regimens are provided in Supplemental Table 3.
Among all enrolled patients, the median age was 57.50 years (IQR: 51.75–64.25), with 49 (58.33%) being male. The primary tumor sites included ICC in 42 patients (50.00%), ECC in 23 patients (27.38%), and GBC in 19 patients (22.62%). The majority of patients (86.90%, n = 73) had an ECOG PS of 0–1. A total of 66 patients (78.57%) experienced recurrence or metastasis after undergoing intended curative resection. Immunotherapy was included in the first-line treatment regimens of only six patients (7.14%). Baseline characteristics were generally well balanced between the two groups (Table 1).
Baseline characteristics.
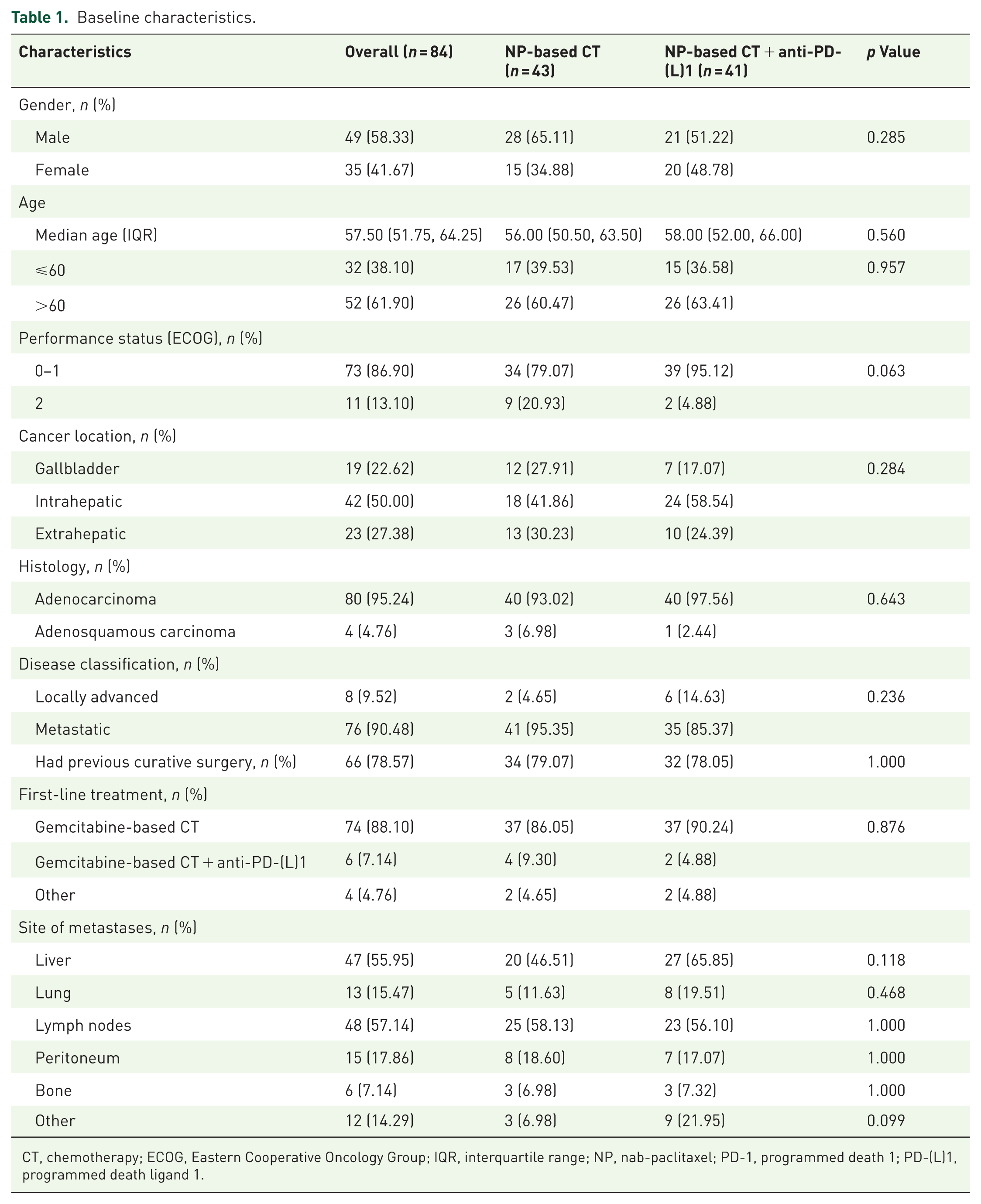
CT, chemotherapy; ECOG, Eastern Cooperative Oncology Group; IQR, interquartile range; NP, nab-paclitaxel; PD-1, programmed death 1; PD-(L)1, programmed death ligand 1.
Following treatment with a nab-paclitaxel-containing regimen, 55 patients (65.48%) proceeded to third-line therapy (Supplemental Table 4). In the chemo-anti-PD-(L)1 group, the most commonly used agents were S-1 (50.00%), oxaliplatin (40.00%), and sintilimab (35.00%). In the chemotherapy group, the most frequently administered agents were lenvatinib (26.47%), sintilimab (17.65%), and oxaliplatin (17.65%).
Efficacy
By the data cutoff date, 47 patients (55.95%) had died. The median follow-up duration for survival from the initiation of second-line therapy was 21.97 months (range: 3.37–45.57 months) in the overall cohort. The median OS for the entire cohort was 15.17 months (95% CI: 12.63–21.43; Figure 1(a)). No significant difference in OS was observed between the chemo-anti-PD-(L)1 group and the chemotherapy group (median OS: 16.90 vs 14.60 months; HR: 0.74; 95% CI: 0.41–1.32; p = 0.30; Figure 1(b)).
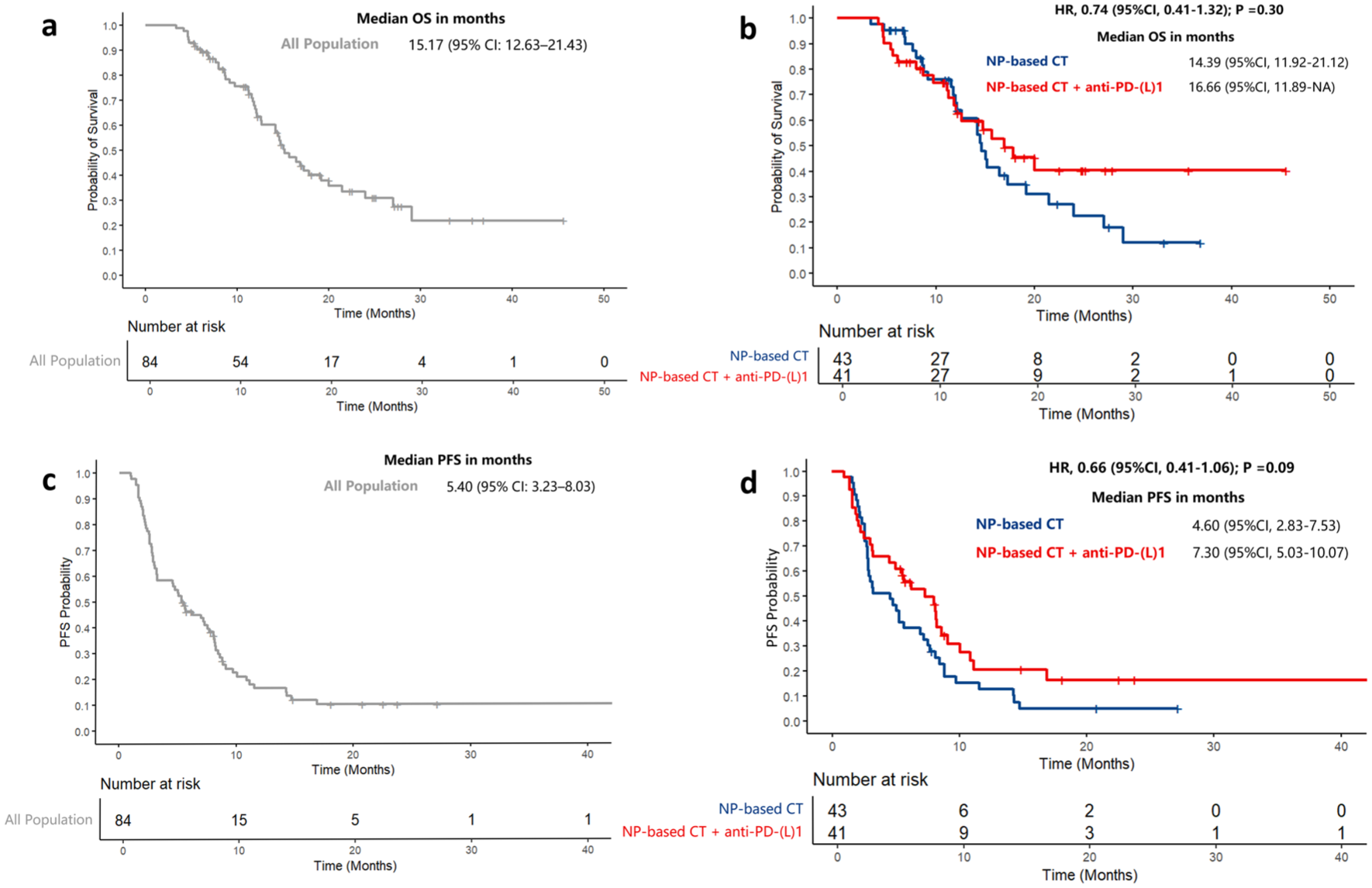
OS and PFS of NP-based therapy. (a) OS in all population. (b) OS of NP-based CT versus NP-based CT + anti-PD-(L)1 group. (c) PFS in all population. (d) PFS of NP-based CT versus NP-based CT + anti-PD-(L)1 group.
The median PFS for the overall cohort was 5.40 months (95% CI: 3.23–8.03; Figure 1(c)). Specifically, median PFS was 7.30 months (95% CI: 5.03–10.07) in the chemo-anti-PD-(L)1 group, compared to 4.60 months (95% CI: 2.83–7.53) in the chemotherapy group (HR: 0.66; 95% CI: 0.41–1.06; p = 0.09; Figure 1(d)). In all population, the objective response rate (ORR) was 21.43% (95% CI: 13.22–31.74), and DCR was 60.71% (95% CI: 49.45–71.20). The ORR was 29.27% (n = 12) in the chemo-anti-PD-(L)1 group and 13.95% (n = 6) in the chemotherapy group (odds ratio (OR): 2.55; 95% CI: 0.85–7.61; p = 0.09). The DCR was 68.29% (n = 28) in the chemo-anti-PD-(L)1 group, compared to 53.49% (n = 23) in the chemotherapy group (OR: 1.87; 95% CI: 0.77–4.56; p = 0.17). Although the PFS, ORR, and DCR appeared higher in the chemo-anti-PD-(L)1 group compared to chemotherapy alone, these differences did not reach statistical significance. Tumor response data are summarized in Table 2.
Tumor responses.
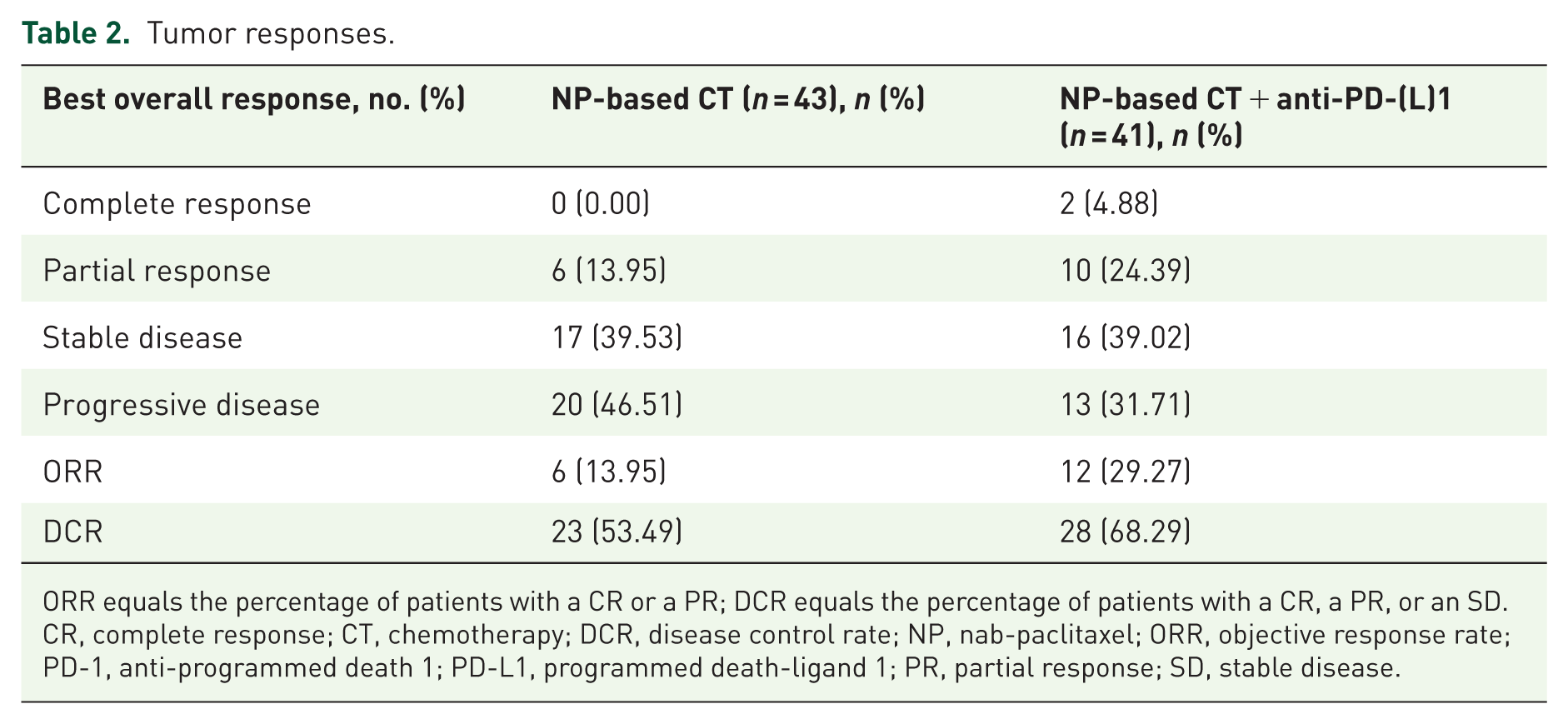
ORR equals the percentage of patients with a CR or a PR; DCR equals the percentage of patients with a CR, a PR, or an SD.
CR, complete response; CT, chemotherapy; DCR, disease control rate; NP, nab-paclitaxel; ORR, objective response rate; PD-1, anti-programmed death 1; PD-L1, programmed death-ligand 1; PR, partial response; SD, stable disease.
In the exploratory analysis, in all population, intended radical surgery significantly improved OS (p = 0.0034; Figure 2), but showed no significant impact on PFS. Besides, patients with a baseline NLR <3 had significantly better OS and PFS than those with NLR ⩾3 (OS: p = 0.0037; PFS: p = 0.0029; Figure 3). Regarding tumor markers, patients were categorized into three groups based on CA19-9 and CEA levels: (a) Tumor markers were initially abnormal but decreased by >30% during second-line treatment. (b) Tumor markers were normal at the baseline of second-line treatment. (c) Tumor markers were initially abnormal but decreased by ⩽30% during second-line treatment. Patients with initially elevated CA19-9 levels who exhibited a ⩽30% reduction during second-line treatment had significantly worse OS and PFS compared to those with a >30% reduction or normal CA19-9 levels at baseline (Figure 4). However, no significant difference in OS was observed between patients with a >30% reduction in CA19-9 and those with normal CA19-9 levels at baseline. None of the CEA change patterns (Supplemental Figure 1), original tumor locations (Supplemental Figure 2), or receipt of third-line treatment (Supplemental Figure 3) showed a statistically significant impact on OS in all population.
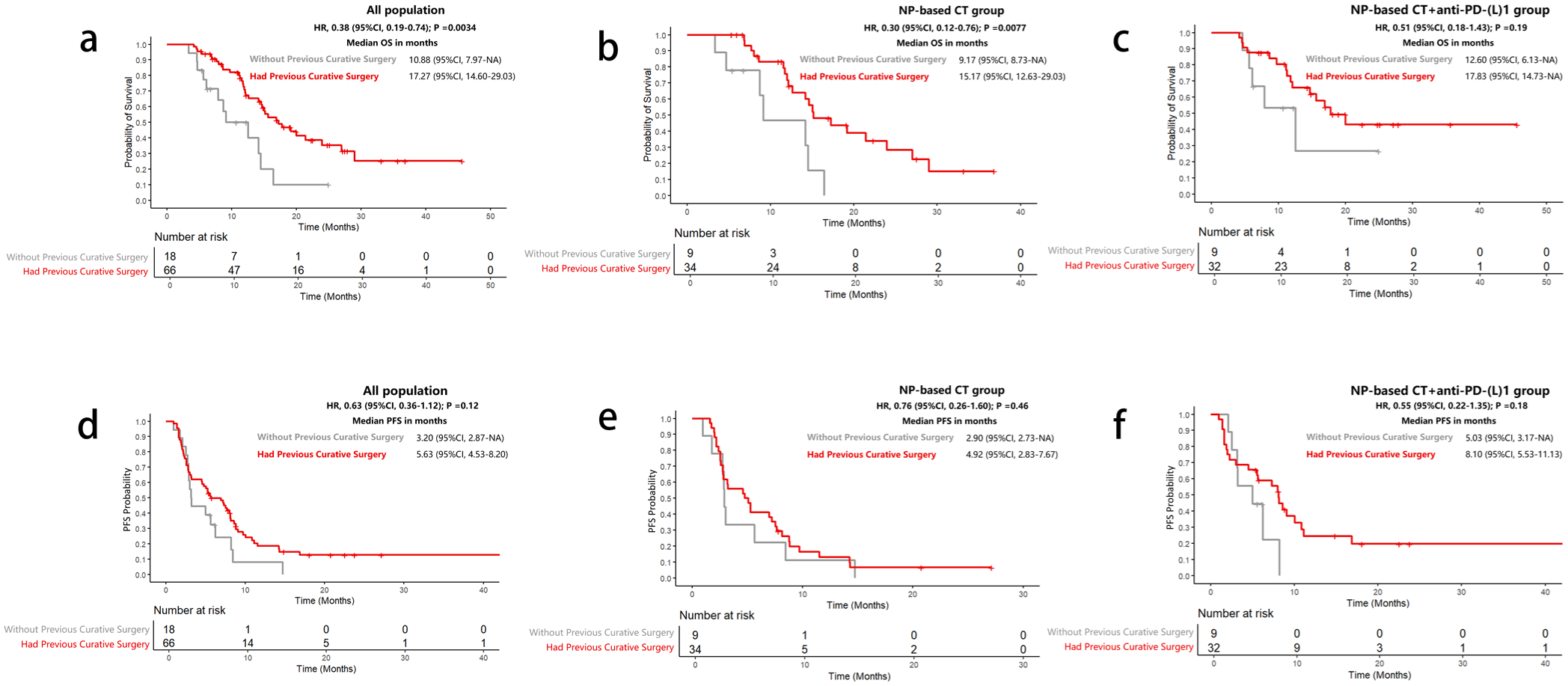
OS and PFS of patients with or without intended curative surgery. (a, d) In all population, (b, e) in the NP-based CT group, and (c, f) in the NP-based CT + anti-PD-(L)1 group.
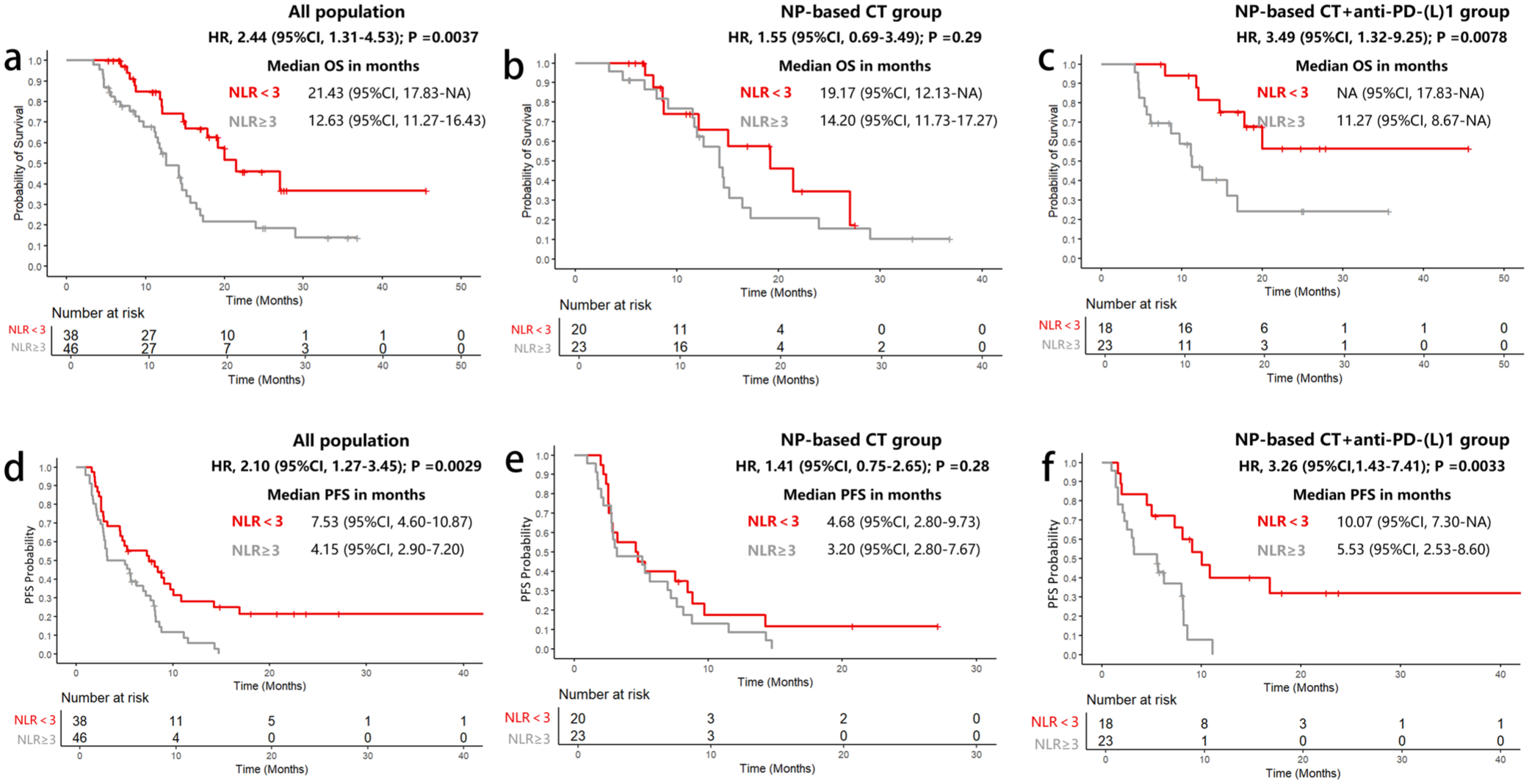
OS and PFS of different baseline NLR levels. (a, d) In all population, (b, e) in the NP-based CT group, and (c, f) in NP-based CT + anti-PD-(L)1 group.
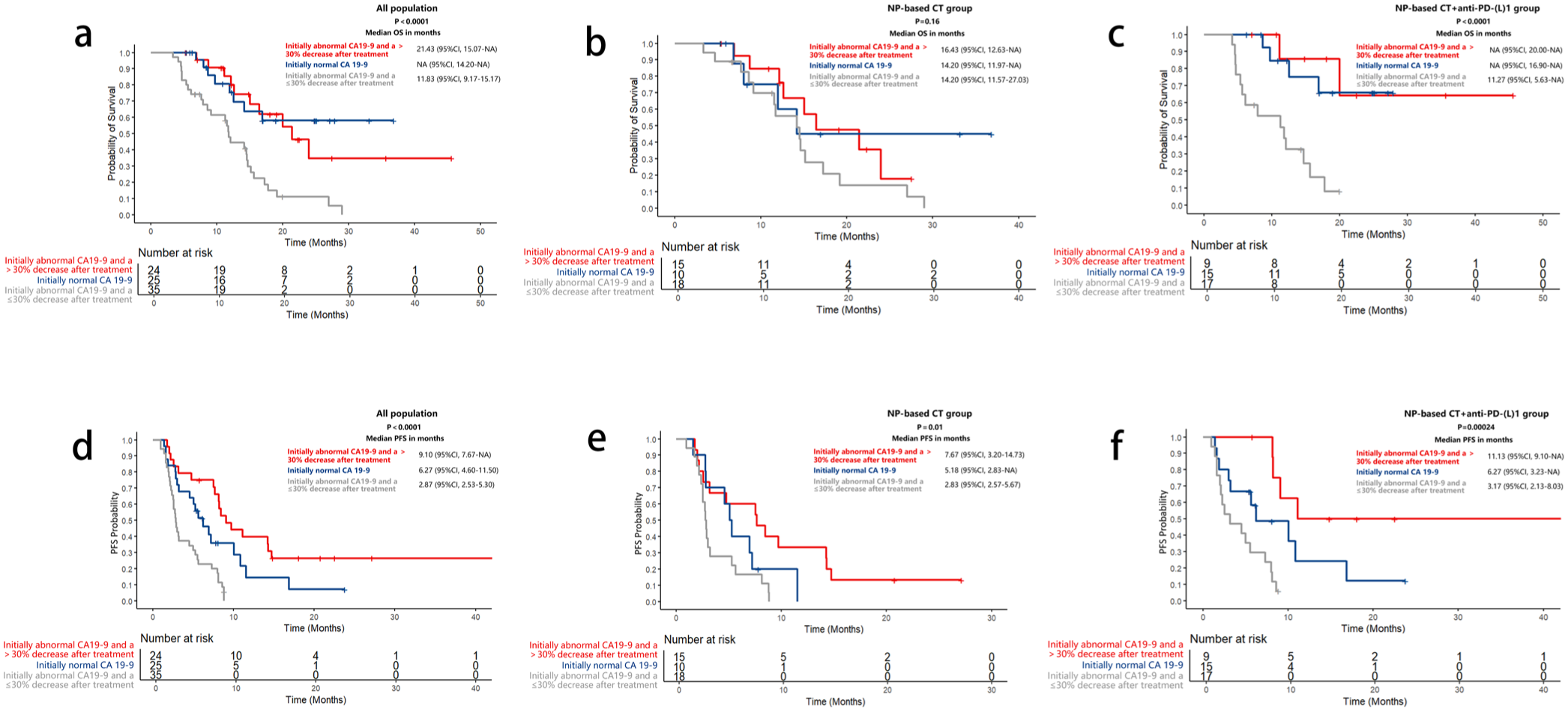
OS and PFS of different CA19-9 change. (a, d) In all population, (b, e) in the NP-based CT group, and (c, f) in NP-based CT + anti-PD-(L)1 group.
Multiple imputation was applied to three variables with missing data: CA19-9, CEA, and NLR. In all population, CEA was missing in four cases (4.76%), CA19-9 was missing in nine cases (10.71%), and NLR was missing in eight cases (9.52%). Sensitivity analysis was performed by excluding cases with missing values, and the results remained consistent, indicating the stability of our findings (Supplemental Figures 4–6).
Safety
Most patients experienced grade 1 or 2 AEs, and no grade 5 AEs were reported (Table 3). The most common grade 3–4 AEs in the overall population included leukopenia (21.43%), neutropenia (17.86%), and anemia (13.10%). Grade 3–4 immune-related AEs included hepatitis (14.63%), colitis (2.44%), and pneumonia (2.44%). Two (4.65%) patients in the chemotherapy group and one (2.44%) patient in the chemo-anti-PD-(L)1 group had dose reduction due to severe myelosuppression. Nine patients (20.93%) in the chemotherapy group and eight patients (19.51%) in the chemo-anti-PD-(L)1 group discontinued second-line treatment due to AEs. No treatment-related deaths were observed.
Adverse events.
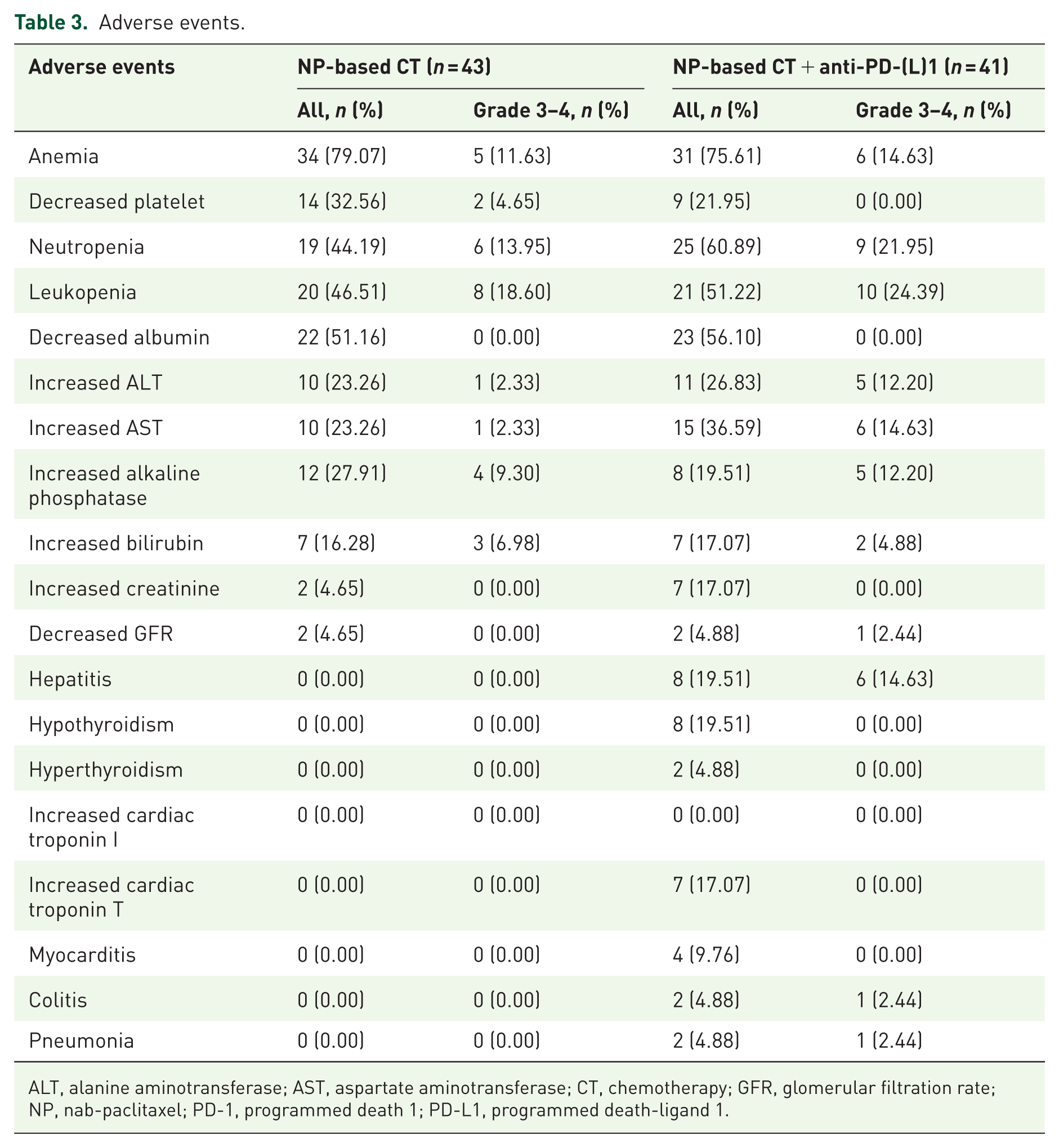
ALT, alanine aminotransferase; AST, aspartate aminotransferase; CT, chemotherapy; GFR, glomerular filtration rate; NP, nab-paclitaxel; PD-1, programmed death 1; PD-L1, programmed death-ligand 1.
Discussion
In our study, nab-paclitaxel-based therapy showed promising efficacy as a second-line treatment for advanced BTC. Although the addition of anti-PD-(L)1 therapy did not confer a statistically significant survival benefit, a trend toward clinical improvement was observed.
For patients with BTC without targetable mutations, the FOLFOX regimen has limited efficacy in the second-line setting. Recently, alternative therapeutic strategies, including multi-target tyrosine kinase inhibitors and various chemotherapy regimens, have been actively explored. Monotherapy with agents like surufatinib (median PFS: 3.7 months; median OS: 6.9 months) and regorafenib (median PFS: 3.0 months; median OS: 5.3 months) has demonstrated clinical activity.20,21 Second-line chemotherapy regimens have shown a PFS ranging from 1.7 to 4.2 months and an OS between 5 and 13.5 months.7,8,22–24
In this study, nab-paclitaxel-based therapy demonstrated promising clinical activity in patients with advanced BTC. Notably, 76 out of 84 patients (90.5%) had received chemotherapy alone as first-line treatment. These findings are consistent with previous small-sample studies investigating nab-paclitaxel as a second-line therapy following first-line chemotherapy.10,25,26 A study involving 10 patients revealed that treatment with nab-paclitaxel and capecitabine achieved a DCR of 80%, with median PFS and OS of 5.7 and 12.1 months, respectively. 10 With gemcitabine and cisplatin plus immunotherapy now established as the standard first-line treatment for advanced BTC, the efficacy of second-line nab-paclitaxel-based chemotherapy requires further evaluation.
In recent years, immunotherapy has been extensively investigated as a second-line treatment for BTC, with exploration of both ICI monotherapy27,28 and combination therapies.29–34 A multicenter phase II study evaluating nivolumab reported a median OS of 14.24 months. 27 In another study, lenvatinib plus immune checkpoint blockade in 14 patients with ICC achieved a DCR of 92.9%, and a median PFS of 5.9 months. 30 In addition, a phase Ib study assessing anlotinib combined with TQB2450 demonstrated a DCR of 72.73%, a median PFS of 6.24 months, and a median OS of 15.77 months. 31
Anti-PD-(L)1 blockade combined with nab-paclitaxel has demonstrated favorable clinical efficacy across several malignancies, including triple-negative breast cancer, non-small-cell lung cancer, and urothelial carcinoma.35–37 A recent study reports that nab-paclitaxel plus atezolizumab expands TCF7 + stem-like effector memory CD8 + T cells and CD4 + T follicular helper cells. Moreover, nab-paclitaxel boosts mast cells, potentially enhancing lymphocyte recruitment and improving the efficacy of PD-L1 blockade. 38 Our previous prospective phase II study of second-line nab-paclitaxel plus sintilimab in 26 patients with BTC demonstrated an ORR of 26.9%, a DCR of 61.5%, a median PFS of 5.6 months, and a median OS of 14.7 months. 14 Our study suggests a trend toward benefit from the addition of immunotherapy, although the difference was not statistically significant, possibly due to the limited sample size. Moreover, second-line immunotherapy does not benefit the entire patient population; it is possible that only a subset of patients derive benefit from immunotherapy. Therefore, further investigation is needed to identify predictive factors of efficacy and to select patients who are most likely to respond.
Exploratory analysis revealed that in the overall population and chemotherapy group, patients who had undergone intended radical surgery had significantly longer OS compared to those who had not. Similarly, the TOPAZ-1 study demonstrated that patients with postoperative recurrence had a higher proportion of long-term survivors. 39 In our cohort, 66 patients (78.57%) had previously undergone curative surgery, which may partially explain the relatively favorable OS observed.
NLR, an inflammatory biomarker reflecting the tumor immune microenvironment and systemic inflammation, has emerged as a promising, cost-effective biomarker for predicting responses to immunotherapy in gastrointestinal tumors. 40 An analysis of 864 BTC patients identified NLR ⩾3.0 as an independent prognostic factor, with a median OS of 12.0 versus 21.6 months for NLR ⩾3.0 and <3.0. 41 Our study corroborates these findings. Patients with NLR <3 had a better overall prognosis, particularly in those receiving anti-PD-(L)1 blockade.
CEA and CA19-9 have been used as prognostic biomarkers in BTC.42,43 Elevated CA19-9 levels have been associated with worse survival outcomes, 44 whereas patients with normal baseline CA19-9 levels exhibit higher ORR, longer PFS, and prolonged OS. 45 Besides, a decline of ⩾30% in elevated tumor marker levels after the first cycle of chemotherapy has been correlated with improved OS and overall response. 46 Our study found that a ⩽30% reduction in CA19-9 from an initially elevated level during treatment was associated with poor outcomes, whereas an initial normal level or a reduction of >30% correlated with similar survival benefits.
Our study has several limitations. First, as a retrospective study, some clinical data were missing or inconsistently recorded. Specifically, CA19-9 had the highest missing rate at 10.71% (nine cases), followed by NLR at 9.52% (eight cases), and CEA at 4.76% (four cases). To address this, we applied multiple imputation and conducted sensitivity analyses, which supported the robustness of our findings. Besides, biomolecular profiling and PD-L1 expression analysis were not performed in this retrospective study. Third, as a single-center study, the generalizability of our results is limited. Finally, to ensure accurate treatment response assessment, we excluded patients who did not undergo imaging follow-up at our hospital, which may have introduced selection bias and potentially skewed the results.
Conclusion
Nab-paclitaxel-based regimens may serve as a potentially safe and effective second-line treatment option for patients with advanced BTC. Large prospective studies are required for validation.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251389753 – Supplemental material for Nab-paclitaxel-based chemotherapy with or without anti-PD-(L)1 immunotherapy as a second-line treatment for advanced biliary tract cancer: a real-world retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359251389753 for Nab-paclitaxel-based chemotherapy with or without anti-PD-(L)1 immunotherapy as a second-line treatment for advanced biliary tract cancer: a real-world retrospective study by Nan Zhou, Xinyi Li, Mingyou Gao, Xiaofen Li, Sirui Tan, Qiancheng Hu, Zijian Lu, Xi Xiao, Tianhong Wang, Shunyu Zhang, Haiyan Luo, Cheng Yi, Yu Yang and Hongfeng Gou in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251389753 – Supplemental material for Nab-paclitaxel-based chemotherapy with or without anti-PD-(L)1 immunotherapy as a second-line treatment for advanced biliary tract cancer: a real-world retrospective study
Supplemental material, sj-docx-2-tam-10.1177_17588359251389753 for Nab-paclitaxel-based chemotherapy with or without anti-PD-(L)1 immunotherapy as a second-line treatment for advanced biliary tract cancer: a real-world retrospective study by Nan Zhou, Xinyi Li, Mingyou Gao, Xiaofen Li, Sirui Tan, Qiancheng Hu, Zijian Lu, Xi Xiao, Tianhong Wang, Shunyu Zhang, Haiyan Luo, Cheng Yi, Yu Yang and Hongfeng Gou in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The preliminary results of this study were selected for online publication on ![]() on May 22, 2025. Abstract Title: A real-world study of nab-paclitaxel-based chemotherapy with or without PD-1/PD-L1 blockade in second-line treatment of advanced biliary tract cancer. Abstract Number for Publication: e16242.
on May 22, 2025. Abstract Title: A real-world study of nab-paclitaxel-based chemotherapy with or without PD-1/PD-L1 blockade in second-line treatment of advanced biliary tract cancer. Abstract Number for Publication: e16242.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
