Abstract
Background:
Systemic therapy is a standard treatment option for pancreatic neuroendocrine neoplasms (Pan-NENs) with unresectable or metastatic disease. Streptozocin (STZ)-based chemotherapy is considered a standard treatment option for tumors with a high Ki-67 index or for cases refractory to molecular targeted agents. More recently, the combination of capecitabine and temozolomide (CAPTEM) therapy has emerged as a new treatment option.
Objectives:
This study aimed to compare the efficacy, safety, and clinical outcomes between the STZ-based regimen and CAPTEM therapy in patients with unresectable or metastatic Pan-NENs.
Design:
This was a single-center retrospective study of histologically confirmed Pan-neuroendocrine tumor (NET) patients treated with either STZ-based regimens or CAPTEM between November 2015 and June 2024.
Methods:
We compared efficacy, safety, and clinical outcomes between the two regimens. Tumor responses were assessed using Response Evaluation Criteria in Solid Tumors version 1.1, and adverse events were graded according to Common Terminology Criteria for Adverse Events version 5.0. The study was conducted in compliance with the STROBE guidelines.
Results:
Of the 371 patients diagnosed with Pan-NENs, 47 received STZ-based regimen and 21 received CAPTEM therapy. In the NET-G1/G2 patients, the STZ group showed a significantly higher tumor shrinkage rate compared to CAPTEM therapy. Although no significant differences were observed in progression-free survival (PFS) or overall survival between the two groups, subgroup analysis showed that the median PFS in the STZ group was significantly longer than that in the CAPTEM group in NET G1/G2 patients. Renal dysfunction was the main adverse event in the STZ regimen group, while gastrointestinal symptoms were common in the CAPTEM therapy group; however, both were manageable.
Conclusion:
Both STZ-based regimen and CAPTEM therapy are safe and effective treatment options for advanced Pan-NENs. STZ-based regimen was more beneficial in NET-G1/G2 patients, suggesting Ki-67 index and tumor grade may serve as indicators for treatment selection. Further prospective studies are warranted to validate these findings.
Plain language summary
Pancreatic neuroendocrine neoplasms (Pan-NENs) are rare tumors that start in hormone-producing cells of the pancreas. While surgery can cure early-stage tumors, many patients are diagnosed when the disease has already spread, making systemic treatment necessary. Two chemotherapy options used for these advanced cases are streptozocin (STZ)-based regimens and a combination of capecitabine and temozolomide (CAPTEM). STZ has long been used, especially in tumors that are growing quickly or do not respond to targeted drugs, but can have side effects like kidney damage. CAPTEM is a newer option that combines two oral drugs, offering a different approach to killing cancer cells. However, there have been few direct comparisons of these treatments in real-world settings. Our study analyzed patients treated at a single center in Japan between 2015 and 2024. We compared 47 patients treated with STZ-based regimen to 21 patients who received CAPTEM therapy. We looked at tumor shrinkage, progression-free survival, overall survival, and side effects. We found that in patients with lower-grade tumors, STZ-based regimen led to better tumor shrinkage and longer progression-free survival compared to CAPTEM. Both treatments had manageable side effects: kidney issues were more common with STZ, while gastrointestinal problems were more frequent with CAPTEM. These results suggest that the tumor grade and a measure called the Ki-67 index (which reflects how fast the cancer cells are dividing) could help guide which treatment to choose. STZ-based regimens may be more suitable for lower-grade, slower-growing tumors, while CAPTEM might work better for more aggressive types. Further research, especially larger prospective studies, is needed to confirm these findings and help doctors choose the best treatment for each patient.
Keywords
Introduction
Pancreatic neuroendocrine neoplasm (Pan-NEN) is a tumor arising from the endocrine cells of the pancreas. 1 Although its morbidity is relatively low,2,3 its incidence has been increasing in recent years.4,5 The prognosis of Pan-NEN largely depends on tumor grade, which is defined by the World Health Organization (WHO) as neuroendocrine tumor (NET)-G1, G2, G3, and neuroendocrine carcinoma (NEC), based on mitotic count or Ki-67 proliferation index. Ki-67 index of NET-G1 is <3%, G2 is 3%–20%, and G3 is >20%, and all of them are well-differentiated tumors. On the other hand, NEC is a poorly differentiated carcinoma, typically with Ki-67 >50%. 6 Genetically, NET and NEC are considered as different types of tumors 7 ; therefore, their treatment strategy is totally different. Many patients are diagnosed at an advanced stage with distant metastases, especially to the liver, 8 which significantly impacts the prognosis. 9 While surgical resection is the only curative treatment option, systemic therapy is essential for unresectable or metastatic cases.
Various systemic treatment options are available for Pan-NETs, and therapeutic strategies are selected based on tumor burden, grade, and proliferative activity.10,11 For well-differentiated tumors with low proliferative activity, somatostatin analogs (SSAs), such as octreotide or lanreotide, are often used as first-line therapy.9,12 For tumors that are progressive despite SSA treatment, molecular targeted agents such as sunitinib and everolimus are approved as standard therapies.13,14 On the other hand, for the tumors with a high Ki-67 index or those that are resistant to targeted therapies, cytotoxic chemotherapy, such as streptozocin (STZ)-based regimens, often combined with 5-fluorouracil (5-FU) or doxorubicin, or combination therapy with capecitabine and temozolomide (CAPTEM) therapy, remains a mainstay for Pan-NETs.
Among cytotoxic agents, STZ leads to cell death by inhibiting DNA synthesis, ultimately inducing apoptosis in cancer cells. 15 However, despite its efficacy, STZ is associated with serious side effects, including nephrotoxicity, which limits its long-term use.16,17 In recent years, CAPTEM therapy has emerged as an alternative treatment option.18–22 Temozolomide is an oral alkylating agent that induces apoptosis through DNA methylation. 23 Capecitabine is a prodrug of 5-FU that suppresses tumor proliferation by inhibiting thymidylate synthase. A recent randomized phase II trial has contributed to its establishment as a standard chemotherapy option for progressive Pan-NETs (E2211 study). 24 Although CAPTEM therapy is approved as a standard therapy in many countries, it is not currently approved for insurance coverage in Japan, which has limited its widespread usage in clinical practice.
Both the STZ-based regimen and CAPTEM include alkylating agents and have demonstrated efficacy against Pan-NETs; however, there was no study comparing their efficacy and toxicity directly. Therefore, this study aimed to compare the efficacy, safety, and clinical outcomes between the STZ-based regimen and CAPTEM therapy in patients with unresectable or metastatic Pan-NETs.
Materials and methods
Study design
This is a single-center retrospective study. We analyzed all Pan-NET patients who were histologically proven and received either STZ-based regimen or CAPTEM therapy between November 2015 and June 2024, as both regimens were first introduced at our institution in November 2015. This study was reported in accordance with the STROBE guidelines for observational studies. 25
Tumor grading was determined according to the 2022 WHO classification. All patients underwent contrast-enhanced computed tomography or magnetic resonance imaging every 2–3 months. Treatment efficacy was assessed based on the Response Evaluation Criteria in Solid Tumors, version 1.1. Objective response rate (ORR) was defined as the proportion of patients achieving complete response (CR) or partial response (PR) as the best overall response, and disease control rate (DCR) was defined as the sum of CR, PR, and stable disease. Best response rate was defined as the maximum percentage reduction in the sum of the target lesion diameters from baseline measurements. Adverse events were evaluated using the National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0. Efforts to reduce bias included using standardized criteria and including all eligible patients.
This study was approved by the Institutional Review Board of the Institute of Science Tokyo.
STZ-based regimen
We previously reported that STZ combined with oral administration of S-1 (a combination of tegafur, gimeracil, and oteracil) is effective with a tolerable adverse event regimen. 17 Therefore, we intensively used STZ with S-1, and as a result, all STZ-based regimens in this study were STZ with S-1. STZ 1000 mg/m2 was administered intravenously on day 1, and S-1 100 mg/body was orally administered twice a day on days 1, 3, 5, and 7 of a 7-day cycle (Figure S1). Although the initiation timing of S-1 was adjusted depending on the severity of adverse events following STZ administration, all patients in this regimen received S-1.
CAPTEM therapy
The CAPTEM therapy consisted of oral administration of capecitabine 1800 mg/body/day twice a day from days 1 to 14, and oral administration of temozolomide 200 mg/body/day once a day from days 10 to 14 of a 28-day cycle.
Statistical analysis
Progression-free survival (PFS) was defined as the time from treatment initiation to confirmed disease progression or death. Overall survival (OS) was defined as the time from treatment initiation to the last follow-up date or death. Survival analysis was performed using the Kaplan–Meier method with the log-rank test. A p-value of <0.05 was considered statistically significant. All statistical analyses were performed using SPSS version 24.0 (SPSS, Chicago, IL, USA).
Results
Baseline characteristics
There are 371 patients who were diagnosed with Pan-NENs in our hospital between November 2015 and June 2024. Among them, 47 and 21 patients who received the STZ-based regimen and CAPTEM therapy were included in the analysis. The median follow-up period was 36.5 months (IQR, 18.2–64.1). The baseline characteristics of the patients are shown in Table 1.
Baseline characteristics.
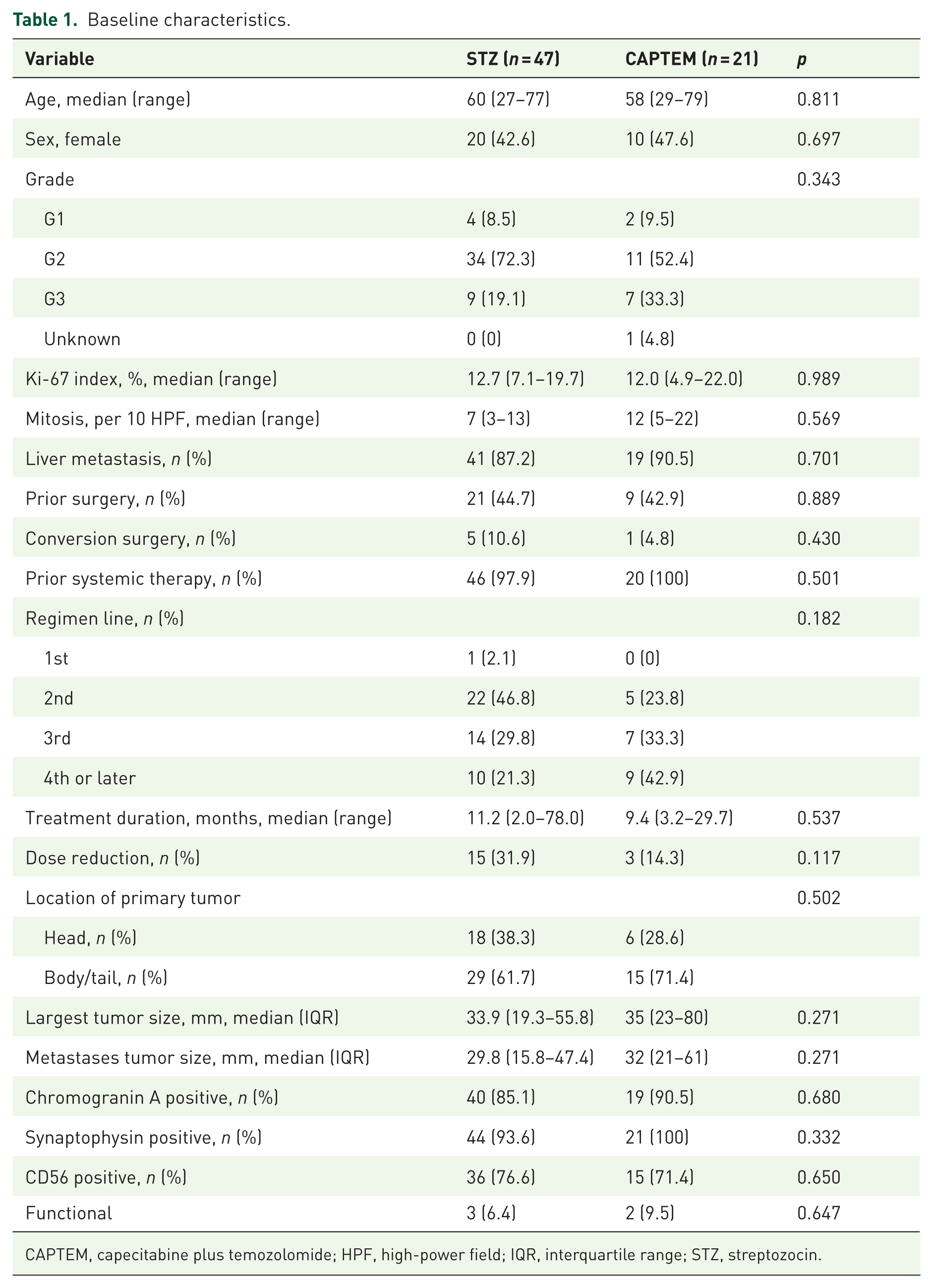
CAPTEM, capecitabine plus temozolomide; HPF, high-power field; IQR, interquartile range; STZ, streptozocin.
The median age in the STZ regimen and CAPTEM therapy groups was 60 and 58 years, respectively. The patient numbers of NET-G1, G2, and G3 in STZ regimen were 4 (8.5%), 34 (72.3%), and 9 (19.1%), and those in CAPTEM therapy were 2 (9.5%), 11 (52.4%), and 7 (33.3%), respectively. One patient in the CAPTEM therapy group had an unknown grade due to insufficient tissue for Ki-67 index evaluation. The median Ki-67 index of the STZ and CAPTEM groups was 12.7% and 12.0%, respectively.
Distant metastasis with liver involvement was observed in 41 patients (87.2%) in the STZ group and 19 patients (90.5%) in the CAPTEM group. The remainder in each group had a locally advanced disease. Among those with liver metastases, 21 (44.7%) and 9 patients (42.9%) in the STZ and CAPTEM group had metachronous recurrences following primary tumor resection.
In the STZ group, 46 patients (97.9%) had received prior systemic therapy, while all 21 patients in the CAPTEM group had also undergone prior systemic therapy. In both groups, most cases were treated in second-line or later therapy. In addition, six patients (12.7%) in the STZ group had previously received CAPTEM therapy, and three patients (14.3%) in the CAPTEM group had previously received the STZ regimen.
The median treatment duration was 11.2 and 9.4 months for the STZ and the CAPTEM group. Dose reductions due to toxicity were required in 15 patients (31.9%) and 3 patients (14.3%) in the respective groups. Five patients (10.6%) in the STZ group and 1 patient (4.8%) in the CAPTEM group achieved conversion surgery.
Higher tumor shrinkage rate of the STZ-based regimen in G1/G2 tumors
In the STZ group, the ORR was 38.3% (18 patients), with a DCR of 74.5% (35 patients; Figure 1(a)). In the CAPTEM group, the ORR was 23.8% (5 patients), with a DCR of 85.7% (18 patients; Figure 1(b)). The median best response rates in the two groups were −22.0% and −10.6%, respectively, with no significant difference (p = 0.116; Figure 1(c)).
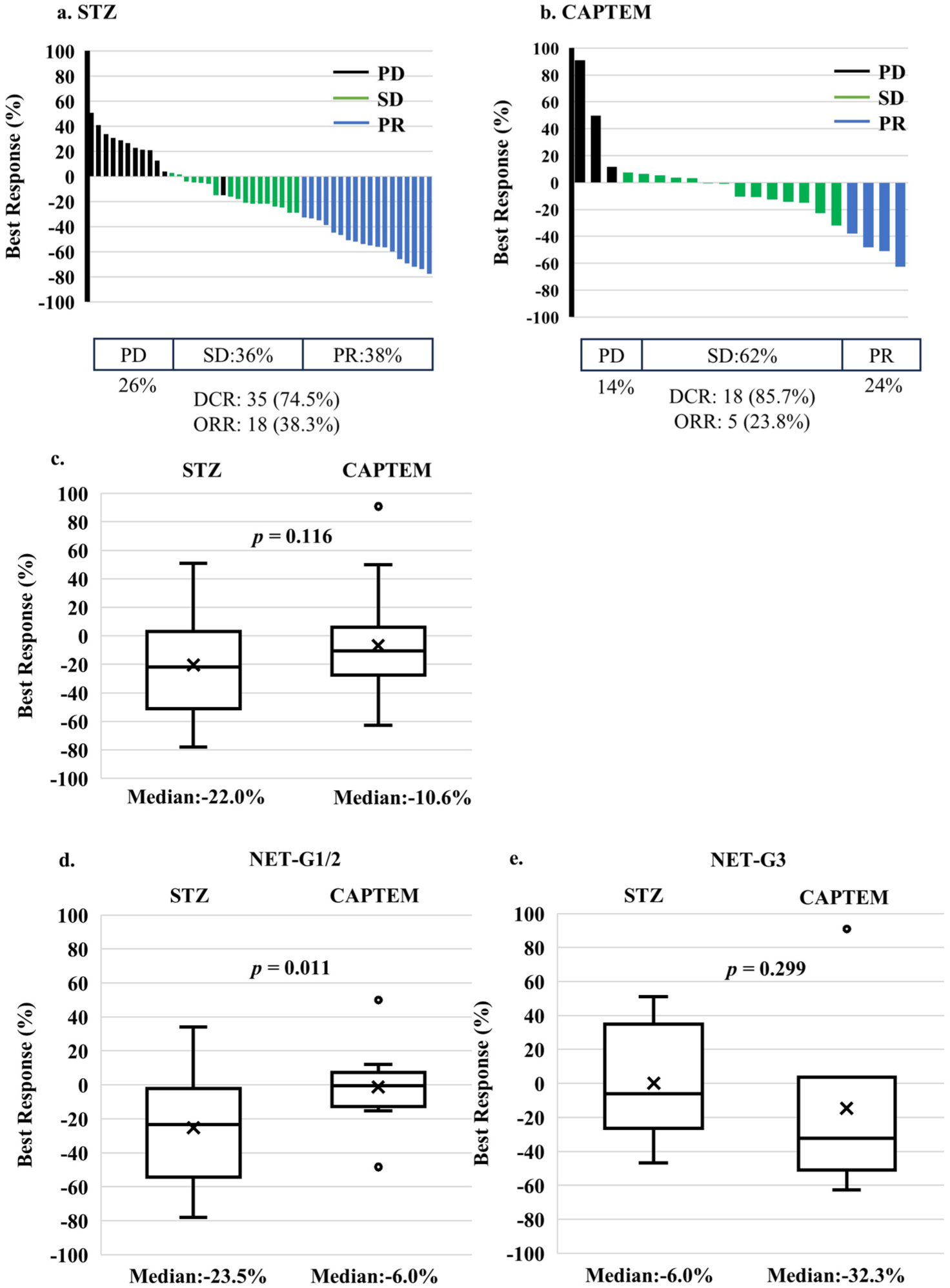
Best response rates of the target lesion after STZ-based regimen (a) and CAPTEM (b). Best response rate (%) = [(sum of tumor diameters with a maximum reduction/increase − baseline diameters)/baseline diameters] × 100 for each patient. The therapeutic response is evaluated by RECIST ver.1.1. Box-and-whisker plot comparing the overall best response between the STZ-based regimen and CAPTEM (c), NET-G1/G2 subgroup (d), and NET-G3 subgroup (e).
To evaluate treatment efficacy by grade, the best response rates were compared between the STZ and CAPTEM within the NET-G1/G2 and NET-G3 tumors. The results demonstrated that in the G1/G2 tumors, the best response rate in the STZ group was significantly greater than the CAPTEM group (−23.5% vs −6.0%, p = 0.011; Figure 1(d)). In the G3 tumors, the median best response in the STZ and CAPTEM group was −6.0% and −32.3%, however, it was not statistically different (p = 0.299; Figure 1(e)).
These findings suggest that the STZ-based regimen is more beneficial compared to CAPTEM therapy in the G1/G2 tumors. To further explore this, we investigated the relationship between the Ki-67 index and tumor shrinkage rate for each treatment. However, there was no correlation between Ki-67 index and tumor shrinkage rate, neither in the STZ nor the CATEM group (Figure S2). Of note, the regression lines crossed at a Ki-67 index of approximately 20%, suggesting the possibility that the STZ-based regimen may be more effective for tumors with a Ki-67 index ⩽20%, while CAPTEM therapy may be beneficial for tumors with a Ki-67 >20%.
Longer PFS of STZ treatment in G1/G2 tumors
Kaplan–Meier analysis showed that the median PFS was 23.2 months (95% CI: 5.5–41.0) in the STZ group and 11.3 months (95% CI: 4.0–19.1) in the CAPTEM group, with no significant difference (p = 0.408; Figure 2(a)). Median OS was 69.7 and 44.5 months, respectively (95% CI: not evaluable), also without a significant difference (p = 0.815; Figure 2(b)).
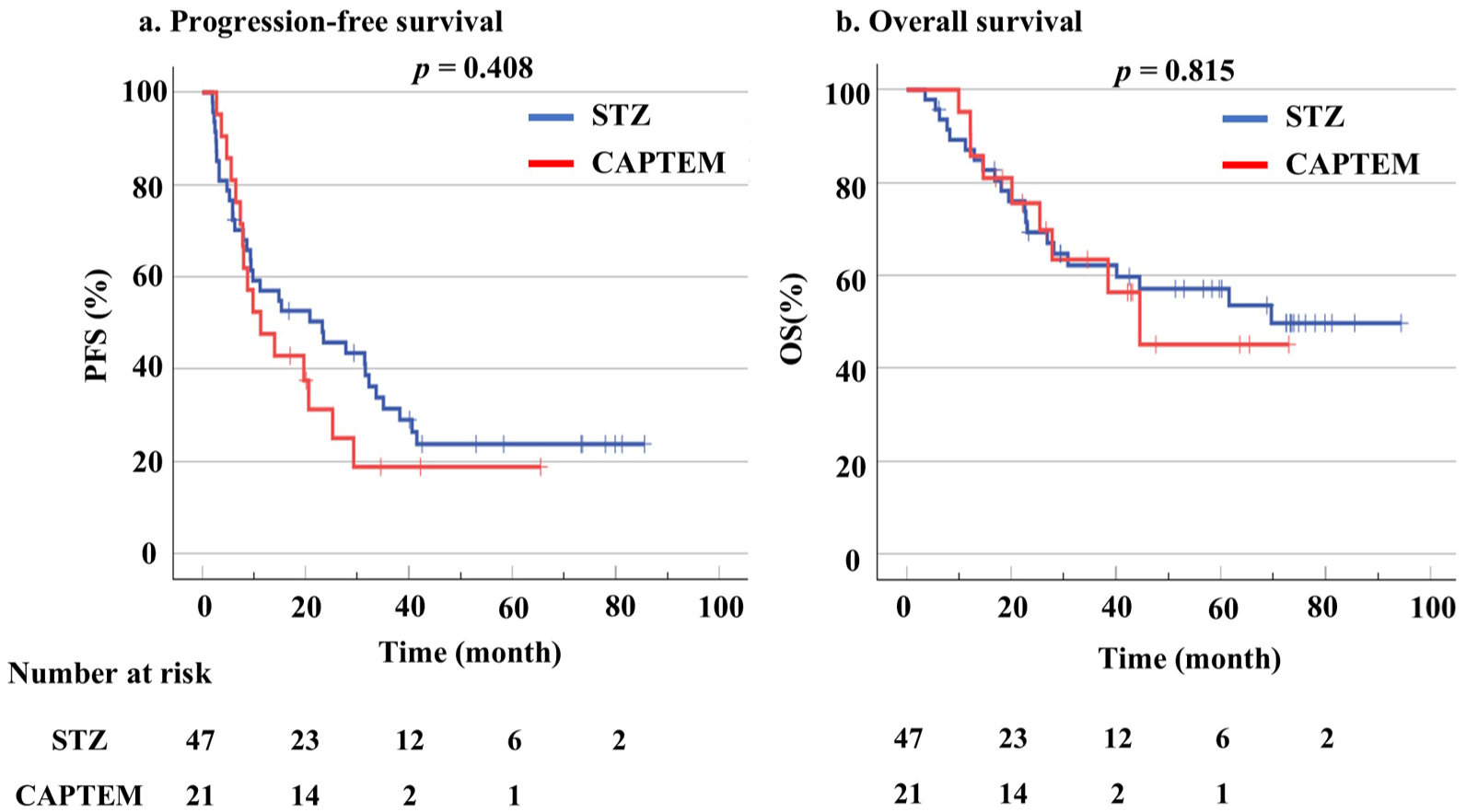
Kaplan–Meier curves of PFS (a) and OS (b) comparing STZ-based regimen and CAPTEM.
Since tumor shrinkage rate was different by grade, we proceeded with subgroup analysis. The result revealed that the median PFS was significantly longer in the STZ group (32.3 months; 95% CI: 8.0–48.1) compared to the CAPTEM therapy (7.8 months; 95% CI: 5.8–9.7; p = 0.030) in the NET G1/G2 group (Figure 3(a)). On the other hand, no significant difference was observed in OS between the two groups (p = 0.773; Figure 3(b)).
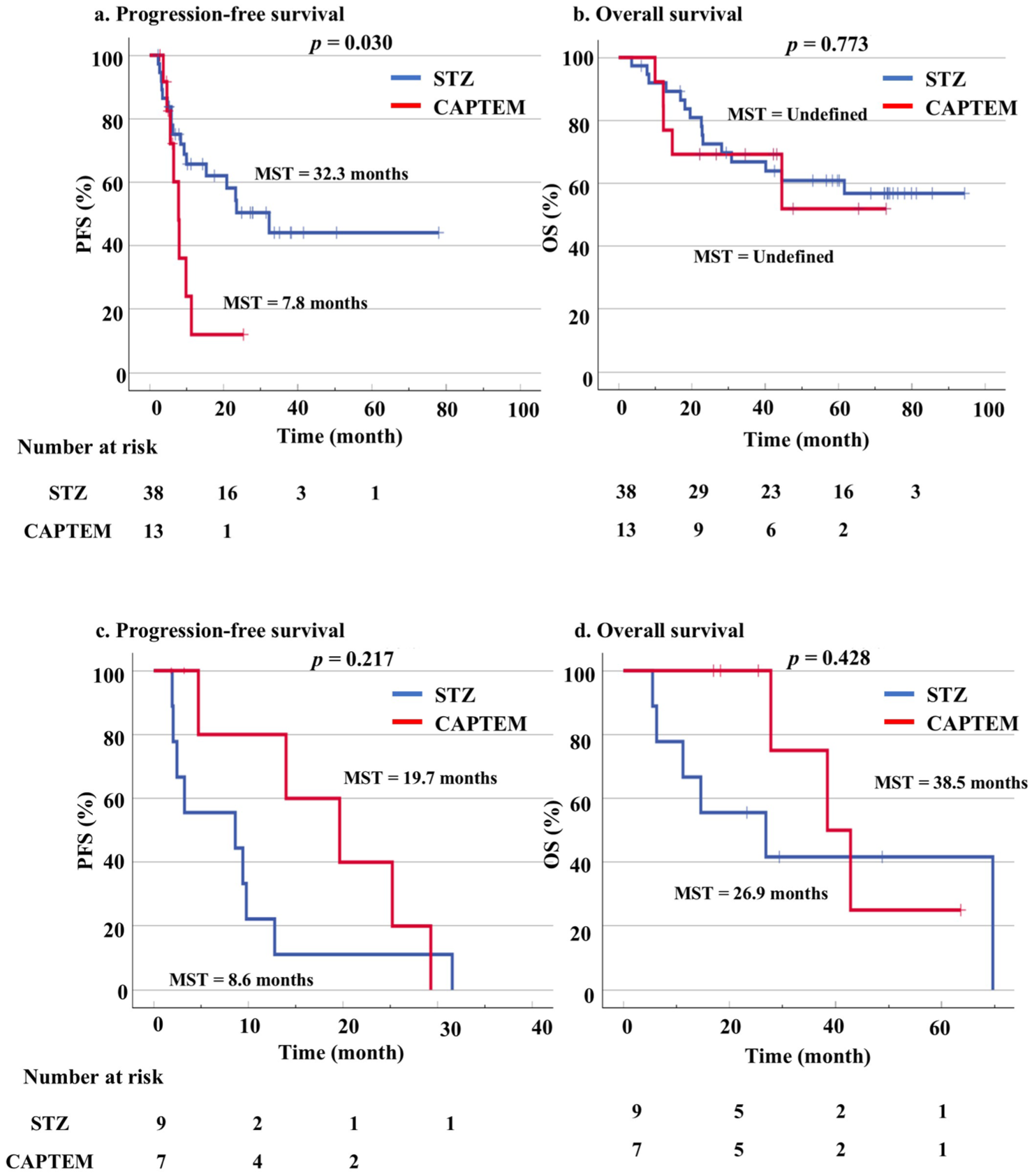
Kaplan–Meier curves of PFS (a) and OS (b) comparing STZ-based regimen and CAPTEM in NET-G1/G2, and PFS (c) and OS (d) in patients with NET-G3.
In the NET-G3 subgroup, the median PFS was 8.6 months (95% CI: 0.0–24.3) for the STZ group and 19.7 months (95% CI: 7.5–31.8) for the CAPTEM group, although it did not reach statistical significance (p = 0.217; Figure 3(c)). Similarly, no significant difference in OS was observed (p = 0.428; Figure 3(d)).
Adverse events
Adverse events were observed in 31 patients (66.0%) in the STZ-based regimen group and 10 patients (47.6%) in the CAPTEM group (Table 2). Grade 3 or higher adverse events were observed in three patients (6.4%) and two patients (9.5%), respectively. The most frequent adverse event in the STZ group was renal dysfunction, reported in 17 patients (36.2%), and one of them was Grade 3. In the CAPTEM group, the most common adverse events were anorexia, observed in seven patients (33%), including one of Grade 3. There was no statistically significant difference in the overall adverse events between the groups (p = 0.747).
Adverse event of each treatment.

CAPTEM, capecitabine and temozolomide; STZ, streptozocin.
Discussion
In this study, we directly compared the efficacy and safety of the STZ-based regimen and CAPTEM therapy in patients with Pan-NETs for the first time. Both regimens showed efficacy; however, the STZ-based regimen achieved a higher tumor shrinkage rate and was associated with longer PFS in patients with NET-G1/G2.
Previous reports on STZ-based regimen have suggested varying associations with the Ki-67 index; some showed improved ORR in patients with Ki-67 >5%, 26 while others reported longer PFS in those with Ki-67 <10%. 27 Fanciulli et al. 28 further proposed that a Ki-67 range of 6%–9% may represent the optimal window for predicting response in Pan-NETs. Meanwhile, the CAPTEM regimen is increasingly used in clinical practice, particularly for G2–G3 NETs, due to its mechanism of action and favorable toxicity profile.19,20 Although both regimens are considered effective for intermediate to high-grade Pan-NETs, prior studies have assessed each treatment independently, and no consensus exists regarding their optimal selection.
By contrast, we directly compared the efficacy of STZ-based and CAPTEM regimens, aiming to clarify their respective roles and support treatment selection. Our analysis demonstrated that the STZ-based regimen resulted in a higher tumor shrinkage rate than CAPTEM therapy among patients with NET-G1/G2. Although the statistical correlation was not shown, we observed a potential trend toward a crossover point at a Ki-67 index of approximately 20%, suggesting a shift in treatment efficacy according to tumor proliferative activity. In the overall cohort, no significant differences in PFS or OS were observed between the two treatment groups. However, subgroup analysis revealed that in patients with NET-G1/G2, the STZ-based regimen was associated with improved PFS compared to CAPTEM. In the NET-G3 subgroup, although no statistically significant difference was detected—likely due to the limited sample size—the Kaplan–Meier curves demonstrated a separation, suggesting a potential benefit of CAPTEM in this population.
One possible explanation for these findings may lie in the mechanistic differences between the two regimens and their interaction with tumor biology. STZ is selectively taken up by Pan-NET cells via GLUT2 transporters, leading to DNA alkylation and fragmentation independent of cellular proliferation rate. 15 This may explain its enhanced activity in well-differentiated tumors with lower Ki-67 indices. By contrast, the CAPTEM regimen exerts cytotoxic effects through both temozolomide-mediated O⁶-methylguanine formation and capecitabine-induced inhibition of thymidylate synthase, which impairs DNA synthesis.22,23,29 These mechanisms are most effective in rapidly proliferating cells, making CAPTEM more active in high-grade tumors. Another important consideration is the role of MGMT (O⁶-methylguanine-DNA methyltransferase), a DNA repair enzyme that counteracts the cytotoxic effects of temozolomide. MGMT expression is often downregulated in high-grade Pan-NETs, 24 rendering these tumors more susceptible to alkylating damage. Consequently, MGMT-deficient tumors may derive greater benefit from CAPTEM therapy, whereas MGMT-expressing, slow-growing tumors may be less responsive. These biological characteristics may underlie the observed divergence in treatment efficacy across tumor grades and Ki-67 levels.
Taken together, these mechanistic insights support a rational approach to regimen selection: STZ-based therapy may be more effective in low-grade tumors, while CAPTEM may be preferable for tumors with higher Ki-67 indices and potential MGMT deficiency.
Both regimens demonstrated manageable safety profiles, with no significant differences in the incidence of severe adverse events. Renal dysfunction was the most frequent adverse event associated with the STZ regimen, in line with previous reports on STZ-induced toxicity.26,27 By contrast, the CAPTEM therapy group experienced more gastrointestinal symptoms such as anorexia. Overall, both regimens were generally well tolerated.
In addition to chemotherapy, peptide receptor radionuclide therapy (PRRT) has recently emerged as an important treatment option for unresectable NENs.30,31 PRRT improved PFS in midgut NENs32,33 and showed promising first-line efficacy in Pan-NENs classified as NET-G2/G3. 34 While the role of PRRT in Pan-NENs continues to be explored, it remains a promising alternative or adjunct to chemotherapy. Future research should focus on optimizing treatment combinations and sequencing of PRRT and chemotherapy to further improve outcomes in patients with advanced Pan-NENs.
This study has several limitations. First, this is a single-center retrospective study with a relatively small sample size, and the imbalance in patient numbers between treatment arms reduced statistical power; therefore, subgroup analyses should be interpreted with caution. Second, while we actively employ the STZ with the S-1 regimen, this is not yet a globally established standard. The more widely used STZ-based regimen includes STZ with 5-FU; previous studies have reported ORR and DCR of 22%–34% and 70%–72%.26,27 In comparison, our study showed an ORR and DCR of 38.3% and 74.5%, respectively, for the STZ with S-1 regimen—suggesting comparable efficacy, although it remains non-standard. Third, CAPTEM therapy is not yet approved as a standard therapy in Japan, which made the strict uniform assessment of adverse events and imaging difficult. Fourth, baseline imbalances in prior treatment lines (with more late-line cases in the CAPTEM group) may have influenced efficacy outcomes, and the absence of propensity score matching or multivariate adjustment represents a potential source of bias. Finally, MGMT status, an important determinant of CAPTEM sensitivity, was not evaluated, and the observed Ki-67–PFS interaction was exploratory and not statistically validated. Taken together, these limitations highlight the need for cautious interpretation of our findings and underscore the importance of validating them in larger, prospective, multi-center studies.
Conclusion
In conclusion, both the STZ-based regimen and CAPTEM therapy are effective treatment options for advanced Pan-NETs. In the NET-G1/G2 subgroup, the STZ-based regimen achieved a higher tumor shrinkage rate and significantly prolonged PFS compared to CAPTEM therapy, although this did not translate into an OS benefit. In the overall cohort, no significant survival difference was observed between the regimens. Further studies of randomized controlled trials with large sample sizes will be essential to further validate our findings and refine treatment guidelines for Pan-NETs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251389742 – Supplemental material for Comparative analysis of streptozocin-based regimen and capecitabine–temozolomide therapy in advanced pancreatic neuroendocrine neoplasms: influence of tumor grade and Ki-67 index
Supplemental material, sj-docx-1-tam-10.1177_17588359251389742 for Comparative analysis of streptozocin-based regimen and capecitabine–temozolomide therapy in advanced pancreatic neuroendocrine neoplasms: influence of tumor grade and Ki-67 index by Yohei Tabe, Hiroaki Ono, Satoshi Matsui, Daisuke Asano, Yoshiya Ishikawa, Hiroki Ueda, Keiichi Akahoshi, Eriko Katsuta and Daisuke Ban in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251389742 – Supplemental material for Comparative analysis of streptozocin-based regimen and capecitabine–temozolomide therapy in advanced pancreatic neuroendocrine neoplasms: influence of tumor grade and Ki-67 index
Supplemental material, sj-docx-2-tam-10.1177_17588359251389742 for Comparative analysis of streptozocin-based regimen and capecitabine–temozolomide therapy in advanced pancreatic neuroendocrine neoplasms: influence of tumor grade and Ki-67 index by Yohei Tabe, Hiroaki Ono, Satoshi Matsui, Daisuke Asano, Yoshiya Ishikawa, Hiroki Ueda, Keiichi Akahoshi, Eriko Katsuta and Daisuke Ban in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
