Abstract
Background:
As metastasis drives the majority of breast cancer-related deaths, improving outcomes for metastatic breast cancer (MBC) remains a crucial challenge. Observational studies using target trial emulation are increasingly applied to estimate treatment effects, providing insights when randomized trials are not viable.
Objectives:
To estimate the effect of progression-free survival (PFS) of metronomic chemotherapy of weekly paclitaxel plus cisplatin (DP) compared with treatment of the physician’s choice (TPC) for MBC.
Design:
This analysis was designed as a retrospective cohort study according to STROBE criteria.
Methods:
This retrospective cohort study compared metronomic chemotherapy of weekly DP versus TPC in MBC. The DP regimen included paclitaxel (80 mg/m2) on days 1, 8, 15, 22 and cisplatin (25 mg/m2) on days 1, 8, 15 in a 28-day cycle. Women aged ⩾18 years with MBC treated with at least one prior therapy between April 2014 and January 2023 at Renji Hospital were included. Propensity score matching (1:3) adjusted for confounding. The matched cohort subsequently underwent emulation of a randomized target trial, including cloning, censoring and weighting. The primary endpoint was PFS.
Results:
Among 313 matched patients (83 on DP, 230 on TPC), DP showed a median PFS of 13.5 months (95% CI 10.3–16.6) versus 7.9 months (95% CI: 6.7–9.1) for TPC (hazard ratio (HR) 0.77, 95% CI 0.60–0.99). Target trial emulation further improved PFS in the DP arm to 14.1 months (95% CI: 13.3–16.6) versus 8.3 months (95% CI: 8.0–8.6) for TPC (HR 0.59, 95% CI: 0.54–0.64). Subgroup analysis and sensitivity analyses confirmed result robustness.
Conclusion:
This study demonstrates that weekly metronomic DP chemotherapy reduces disease progression in MBC patients, particularly in first-line settings. These findings support its potential as a treatment option and warrant further study for broader application.
Plain language summary
This study looked at a new way of giving chemotherapy to people with metastatic breast cancer (MBC), which is breast cancer that has spread to other parts of the body. MBC is hard to treat because it often becomes resistant to drugs and regular treatments don’t work very well. We wanted to see if giving low doses of two chemotherapy drugs, paclitaxel and cisplatin, every week (called metronomic chemotherapy) could help patients live longer without their cancer getting worse. We compared a method (called the DP group) to other treatments people usually get. We looked at medical records of 524 women who had MBC and had already received at least one treatment. We made sure the groups were similar in important ways like age, previous treatments, and cancer subtype. After matching, we found that the DP group had better results: their cancer took longer to progress compared to the other group. In the DP group, the average time before cancer got worse was about 13.5 months, while it was only 7.9 months in the other group. When we used special methods to make sure our results were accurate, the DP group still did better, with an average of 14.1 months compared to 8.3 months in the other group. For people getting this treatment as their first option, the results were even better, with an average of 19.2 months before cancer progressed. This study shows that giving low-dose chemotherapy with paclitaxel and cisplatin every week might be a good option for people with metastatic breast cancer, especially as their first treatment.
Keywords
Introduction
Breast cancer represents the most prevalent malignancy and stands as the leading cause of cancer-related mortality among women worldwide. 1 Despite significant advancements in early detection and treatment, the clinical outcomes for patients with metastatic breast cancer (MBC) remain unsatisfactory. Although the overall survival rate for breast cancer patients has improved in recent years, MBC continues to present a formidable challenge due to its complex biology, rapid progression, and high propensity for therapeutic resistance. 2 While targeted therapies and hormone treatments have shown great efficacy for human epidermal growth factor receptor 2 (HER2) positive and hormone receptor (HR) positive tumors, chemotherapy remains indispensable, particularly in cases of endocrine resistance or high-risk disease progression.3,4 For patients lacking HER2 and HR targets, chemotherapy still serves as a critical and non-substitutable modality in treatment.
The randomized controlled trials (RCTs), widely regarded as the highest-level evidence for evaluating therapeutic efficacy and safety, often entail substantial costs, prolonged timelines, and restrictive inclusion criteria. These constraints can limit the generalizability of findings to broader patient populations. 5 Such limitations are particularly pronounced in oncology, where rapid treatment advancements necessitate timely and broadly applicable evidence. In recent years, real-world data (RWD) has gained recognition as a valuable complement to RCTs, providing insights into treatment effectiveness across diverse patient populations and clinical settings. 5 Target trial emulation employs a methodological approach to emulate the structure and rigor of a hypothetical randomized trial using RWD, thereby enhancing causal inference and delivering timely evidence critical for clinical decision-making. 6
The combination chemotherapy of paclitaxel plus cisplatin (DP) has shown therapeutic efficacy across several malignancies, including ovarian, non-small-cell lung, and cervical cancers.7 –9 However, the significant toxicity related to platinum agents has limited their application in breast cancer, with the exception of certain subtypes, such as triple-negative breast cancer (TNBC), where platinum sensitivity is more pronounced. 10 Traditionally, platinum-based chemotherapy is administered on a 3-week cycle in breast cancer and other malignancies.7 –9 To explore alternative approaches, we previously developed a metronomic weekly DP regimen, which was evaluated in the neoadjuvant setting and demonstrated good efficacy with favorable tolerability.11,12 Building on these findings, the present study emulates a target trial using RWD to investigate the effectiveness of metronomic weekly DP in patients with MBC. By comparing the outcomes of patients treated with a metronomic weekly DP regimen to those receiving alternative treatment of physician’s choice (TPC), we aim to provide robust, real-world evidence regarding the effectiveness of this combination therapy.
Methods
Study population
The STROBE guidelines for reporting observational cohort studies were followed (Table S1). 13 We collected clinical data from the medical records of patients who participated in multi-disciplinary team (MDT) meetings in the Breast Cancer Center of Renji Hospital, School of Medicine, Shanghai Jiao Tong University, between April 1st, 2014, and January 31st, 2023. The MDT record encompasses comprehensive information regarding clinical presentations, diagnoses, prescriptions, laboratory tests, and imaging results for patients undergoing cancer treatment. All participants enrolled in this study were female. To reduce typing errors, checkboxes were used whenever possible to capture data instead of human entry. Eligibility criteria included: (I) patients aged 18 years or older; (II) a confirmed diagnosis of MBC; (III) receipt of at least one line of treatment following the onset of metastasis; and (IV) presence solely of unilateral primary breast cancer. Patients with brain metastases were also eligible and classified as having visceral metastases. During the study period, 610 patients were excluded due to de novo MBC, concurrent diagnosis of any other secondary primary malignancy, unknown HR status, or HER2 status, as well as incomplete progression-free survival (PFS) data. HR-positive status was determined based on immunohistochemistry (IHC) results, with HR positivity defined as an estrogen receptor or progesterone receptor expression of no less than 10%. HER2-positive was defined as HER2 IHC 3+ and/or fluorescence in situ hybridization amplification. The dataset was integrated using unique anonymized patient IDs. This retrospective study was approved by the Ethics Committee of Renji Hospital, School of Medicine, Shanghai Jiao Tong University (Approval ID: KY2024-159-B), with a waiver of informed consent due to the use of clinical data collected from routine care and the absence of foreseeable risk to participants. The study was registered with the Chinese Clinical Trial Registry (ChiCTR2400089679).
The enrolled patients were categorized into two groups: those who received at least one cycle of metronomic weekly DP regimen (DP arm), and those who had never undergone the DP regimen but instead received TPC (TPC arm). The DP regimen involved the metronomic weekly DP regimen consisted of paclitaxel administered intravenously at a dose of 80 mg/m2 on days 1, 8, 15, and 22 within each 28-day cycle and cisplatin administered intravenously at a dose of 25 mg/m2 on days 1, 8, and 15 within each cycle. 14 For HER2+ patients, targeted therapies (trastuzumab, pertuzumab) were added, with no patients receiving tyrosine kinase inhibitors (TKIs). In the TPC arm, treatment regimens were selected based on international guidelines and clinical considerations, including chemotherapy, endocrine therapies, and targeted therapies. Common regimens for TNBC included taxane (e.g., paclitaxel and docetaxel) or combination chemotherapy, while for HR+ MBC, endocrine therapies (tamoxifen, aromatase inhibitors) were used first-line, followed by chemotherapy upon endocrine resistance. HER2+ patients received standard targeted therapies with chemotherapy. Notably, 6 patients received abemaciclib (CDK4/6 inhibitor), and 4 patients received pembrolizumab (PD-1 inhibitor), while 14 patients were treated with TKIs (including lapatinib and pyrotinib). Specifically, taxane-based therapies were administered to 16.5% and platinum-based therapies to 6.5% of patients in the TPC arm. The overall exposure to these regimens was relatively low. To provide further context, Table S1 summarizes the most frequently used regimens and their usage frequencies across the entire cohort.
Sample size and power analysis
Based on prior research, 15 the median PFS for advanced first-line therapy in TNBC was reported as 9.7 months versus 5.6 months (hazard ratio (HR) 0.65, 95% CI 0.49–0.86). Assuming an HR of 0.65 for non-DP group patients compared to the DP group, with a two-sided α of 0.05 and 80% power, the required sample size was 230 patients. Accounting for a 10% dropout rate, the total enrollment target was 254 patients, with 64 in the DP group and 190 in the non-DP group.
Propensity score matching
To ensure comparability between DP arm and TPC arm while controlling for potential confounders, a propensity score matching analysis was conducted prior to evaluating therapeutic effects. We performed 1:3 matching between patients treated with metronomic weekly DP regimen and those receiving alternative TPC, based on age, HR status, HER2 status, chemotherapy sequence, radiotherapy history, endocrine therapy, disease-free interval (DFI), and metastatic sites. 16 Given the limited number of patients undergoing second-line treatment or beyond, we executed propensity score matching in stages, commencing with fourth-line patients. Patients successfully matched in the fourth-line cohort were excluded from subsequent matching processes. Following this step, we carried out propensity score matching for third-line treatment; those who were successfully matched were not included in the cohorts for second- and first-line treatment. Upon completion of batch-wise propensity score matching across first- to fourth-line treatment groups (ensuring no duplicates), these groups were aggregated to form the naïve population. The effectiveness of the propensity score matching was subsequently assessed by examining the distribution of propensity scores (excluding the treatment line variable) and calculating the standardized mean differences (SMD).
Target trial emulation
An overview of the methods is presented in Figure 1, with key components of this emulated target trial protocol detailed in Table S1. This study was not blinded due to the retrospective nature of the data collection. However, outcome assessors were blinded to treatment allocation during data analysis to minimize bias. The emulated target trial approach also addresses the limitations inherent in utilizing retrospective data by facilitating a priori implementation of appropriate measures to minimize selection bias, information bias, and confounding bias between the compared arms. 17 We modeled our hypotheses using a causal directed acyclic graph to visually elucidate causal relationships, identify potential confounders, and guide study design and analysis for proper confounding adjustment (Figure S1). Following propensity score matching, 18 we created two exact copies (clones) of all included patients and assigned each clone to either the DP arm or the TPC arm at time zero (index date). For patients receiving first-line treatment, we designated the date of confirmed metastasis as the index date; for those undergoing later-line therapy, we utilized the date of progression from the prior lines as their index date. During a 60-day grace period following the index date, clones were censored if they deviated from their assigned treatment strategies. Specifically, clones were censored on day 60 if no therapy had been prescribed within the preceding 60 days. Clones in the DP arm were censored upon initiation of alternative regimens, while clones in the TPC arm were censored when DP was initiated.6,18 Patients who remained alive and without disease progression at the last follow-up were censored. Inverse probability of censoring weights (IPCWs) were applied daily throughout this grace period. These weights, calculated using a Cox regression model, represented predicted probabilities that patients remained uncensored at observed times.6,18,19 The emulation process was tailored specifically for each arm to account for potential interactions between covariates and treatment, ensuring that these weights effectively mitigate any imbalances between treatment arms.6,20 Target trial emulation was similarly conducted across the matched HR−/HER2+ population and the matched HR+HER2− population.

Schematic diagram of target trial emulation. Cloning: Patients were virtually replicated and assigned to DP or TPC groups, mimicking randomization. Censoring: Immortal time bias was addressed by excluding pre-treatment periods. Weighting: Inverse probability weights adjusted for baseline imbalances and reduced selection bias.
Statistical analyses
Baseline characteristics were presented as means and standard deviations for continuous variables, and as frequencies and percentages for categorical variables. To assess the balance of baseline characteristics between treatment arms, SMDs were calculated for each covariate, with an SMD threshold of 0.1 indicating a significant imbalance. Missing data in baseline characteristics were addressed using the missMDA package in R. 21 Multiple imputation was conducted to generate complete datasets, ensuring that subsequent analyses remained unbiased by missing values. 22 The imputation model incorporated all baseline covariates to preserve the integrity of the data.
The primary endpoint was PFS in patients with MBC, based on the Response Evaluation Criteria in Solid Tumors (RECIST; version 1.1). 23 PFS was measured from the initiation of treatment until death or disease progression, which was confirmed through computed tomography or magnetic resonance imaging. The estimation of PFS distributions was done using Kaplan-Meier survival curves, and the log-rank test was employed to compare survival between different treatment arms. Cox proportional hazards models were utilized to estimate HRs and 95% confidence intervals (CIs) for evaluating the effects of treatment on PFS. Statistical significance was defined as an HR with a 95% CI excluding 1.
Sensitivity and subgroup analyses
To assess the robustness of our findings, sensitivity analyses were performed. First, we specifically matched two cohorts targeting distinct molecular subtypes of breast cancer. One cohort comprised the HR−/HER2+ population, which includes TNBC and HER2-positive regardless of HR status; the other consisted of the HR+HER2− population, encompassing HR-positive and HER2-negative breast cancer. Second, we applied univariable and multivariable Cox regression models to evaluate the association between treatment regimens and study outcomes in the naïve main population. Third, to investigate the impact of statistical methods employed to control for confounding bias in the emulated population, we utilized the following methods: IPCW Cox regression, multivariable adjusted Cox models, and propensity score-overlap weighting (PSOW).6,24,25
To compare the effects on PFS between the DP arm and the TPC arm across various patient groups, we conducted subgroup analyses stratified by age, HR status, HER2 status, HR and HER2 status, chemotherapy sequence, radiotherapy, endocrine therapy, DFI, metastatic sites, line of therapy, and Family history of breast and/or ovarian cancer. Chi-square tests were used to assess the distribution of patients in the DP and TPC arms across these predefined subgroups (Figure S2). The results demonstrated that there were no significant baseline differences between the simulated populations within these subgroups. All analyses were performed using R software version 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient characteristics
By leveraging the population-wide records maintained by the Breast Cancer Center at Renji Hospital, School of Medicine, Shanghai Jiao Tong University, we identified 524 patients with MBC who met the eligibility criteria. Among these individuals, 101 (constituting 19.3% of the total cohort) were treated with the metronomic weekly DP regimen across first- to fourth-line therapy (Figure 2). Following propensity score matching, a total of 313 MBC patients were successfully matched to form the total population (83 DP vs 230 TPC). The propensity scores for both groups were comparable, and their distribution exhibited significant overlap post-matching (Figure S3). In the matched population, the mean treatment duration in the DP arm was 169 days. In the TPC group, the overall mean treatment duration was 211 days, while the mean duration of other chemotherapy within this arm was 127 days. The naïve total population included individuals where 38.3% were over the age of 55 years, 68.1% were HR-positive, and 81.5% were HER2-negative at baseline. This cohort was utilized for subsequent target trial emulation. We employed cloning, censoring, and weighting methodologies to mitigate potential bias. Within the emulated population, 110 patients received a metronomic weekly DP regimen and 187 received TPC. The distribution of applied weights was deemed satisfactory as it indicated a well-balanced distribution of confounding factors between groups (SMDs ⩽0.1) (Figure S4). For instance, prior to applying the weights, the proportion of patients with visceral metastases in the DP arm was recorded at 62.7%, whereas in the TPC arm it stood at 53.9%; following weight application, these proportions adjusted to reflect values of approximately 55.8% and 56.3%, respectively (Table 1).
Patient characteristics by treatment arm in the naïve and emulated population.

BMI, body mass index; DFI, disease-free interval; DP, paclitaxel plus cisplatin; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; SD, standard deviation; TPC, treatment of physician’s choice; uSMD, unadjusted standardized mean differences.
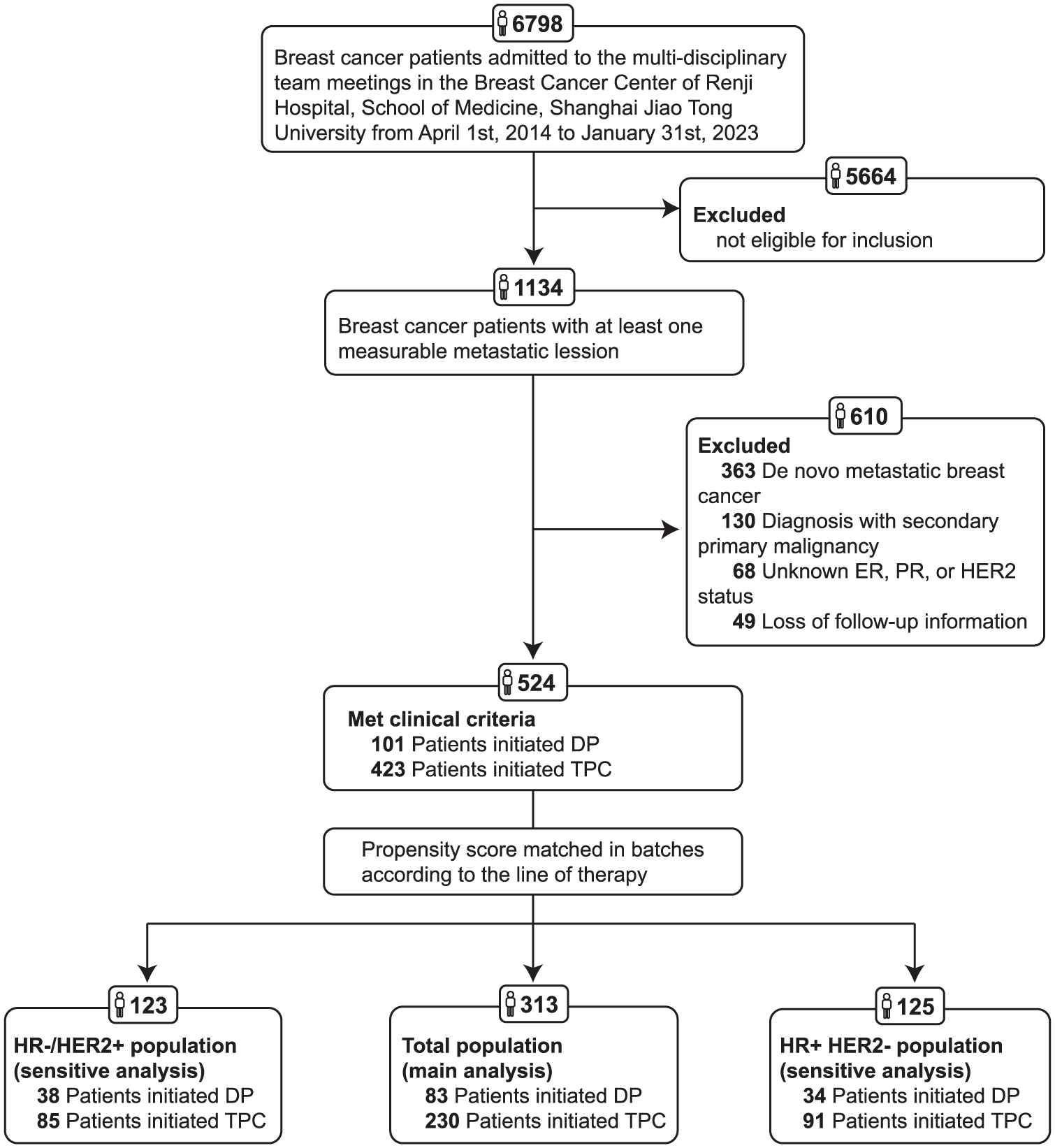
Study flowchart. Patient inclusion and exclusion in clinical and target trial cohorts based on eligibility criteria are presented.
Main outcome
Metronomic weekly DP regimen demonstrated a statistically significant improvement in PFS compared with TPC. In the naïve total population, patients receiving DP exhibited a median PFS that was significantly longer than TPC, with DP achieving a median PFS of 13.5 months (95% CI, 10.3–16.6) versus 7.9 months (95% CI, 6.7–9.1) for alternative treatments (HR = 0.77, 95% CI, 0.60–0.99). After emulation, DP median PFS remained superior at 14.1 months (95% CI, 13.3–16.6) versus 8.3 months (95% CI, 8.0–8.6) for TPC (HR = 0.59, 95% CI, 0.54–0.64) (Figure 3(a)). Furthermore, subgroup analysis highlighted enhanced PFS with DP in first-line treatment (19.2 months (95% CI, 17.8–19.5) vs 8.6 months (95% CI, 8.4–9.0); HR = 0.47, 95% CI, 0.43–0.52) (Figure 3(b)).
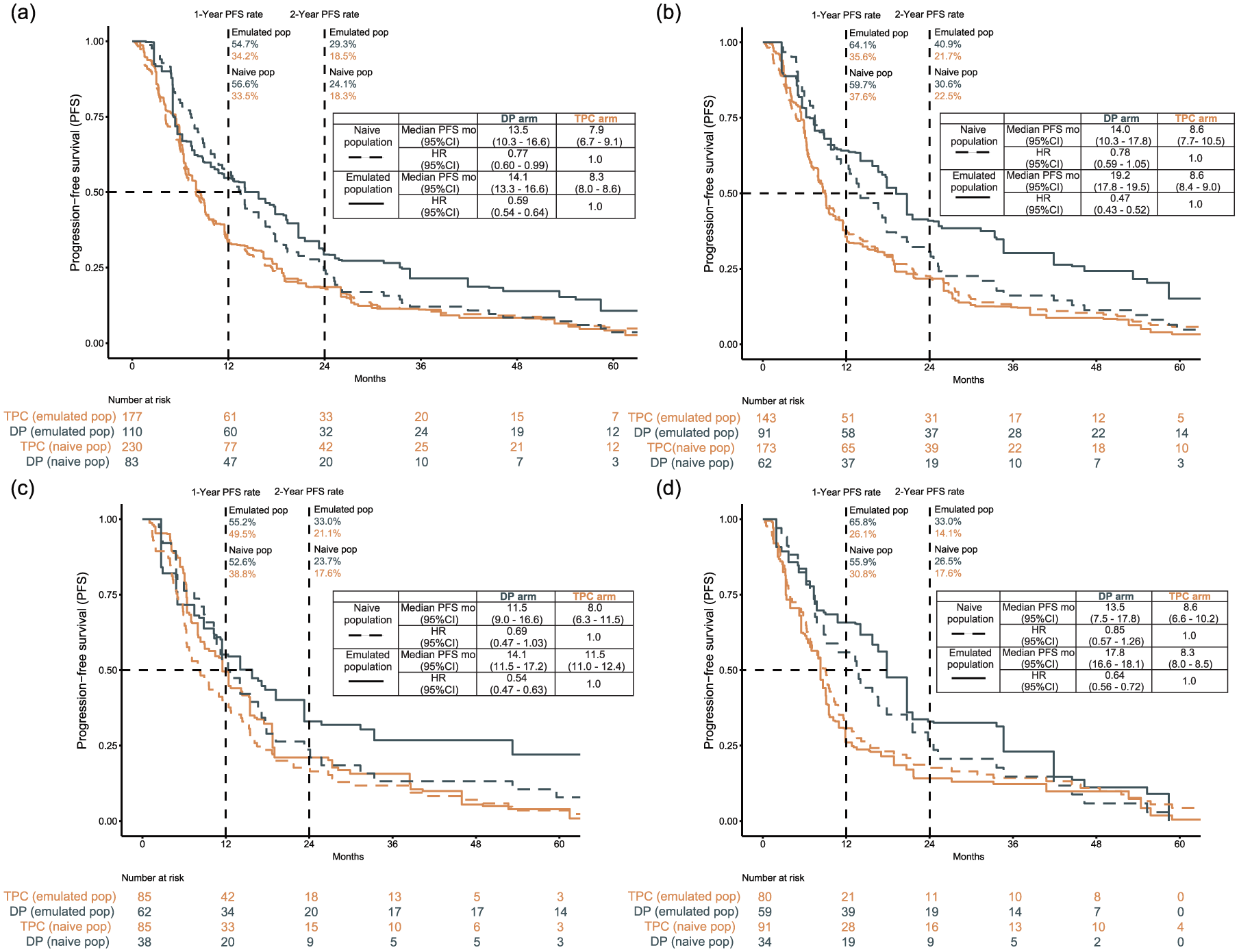
Progression-free survival in naïve and emulated population. (a) PFS in the overall breast cancer population, (b) PFS in patients receiving first-line treatment as a subgroup of the main analysis, (c) PFS in the HR-negative/HER2-positive population as a sensitivity analysis, and (d) PFS in the HR-positive/HER2-negative population as a sensitivity analysis. Dashed lines represent the naïve population and solid lines represent the emulated population. PFS was defined as the time from treatment initiation to disease progression or death, whichever occurred first. In the emulated population, hazard ratios were estimated using Cox proportional hazards models weighted by stabilized IPCW. The proportional hazards assumption was not fully satisfied in the naïve population; hazard ratios are therefore presented for exploratory interpretation only.
Sensitivity analyses
To validate the robustness of our findings, we conducted sensitivity analyses. Initially, we specifically matched two cohorts targeting distinct molecular subtypes of breast cancer. One cohort comprised the HR−/HER2+ population, which includes TNBC and HER2-positive, regardless of HR status; the other consisted of the HR+ HER2-population, encompassing HR-positive and HER2-negative breast cancer (Figure 2). In the HR−/HER2+ naïve population, patients treated with DP demonstrated a numerically longer median PFS compared to those treated with TPC; the median PFS was 11.5 months (95% CI, 9.0–16.6) for DP and 8.0 months (95% CI, 6.3–11.5) for TPC. However, the difference did not reach statistical significance (HR = 0.69; 95% CI, 0.47–1.03). After emulation, the median PFS was statistically longer for DP-treated patients than for TPC-treated patients (14.1 months (95% CI, 11.5–17.2) for DP versus 11.5 months (95% CI, 11.0–12.4) for TPC; HR = 0.54, 95% CI, 0.47–0.63) (Figure 3(c)). The DP regimen also showed favorable performance when compared with other specific chemotherapy regimens in an emulated HR−/HER2+ population (Figure S5(a)).
Similarly, in the HR+/HER2- naïve population, patients treated with DP demonstrated a numerically longer median PFS compared to those treated with TPC. The median PFS was 13.5 months (95% CI, 7.5–17.8) for DP versus 8.6 months (95% CI, 6.6–10.2) for TPC. However, the difference was not statistically significant (HR = 0.85; 95% CI, 0.57–1.26). After emulation, the median PFS was statistically longer for DP-treated patients than for TPC-treated patients (17.8 months (95% CI, 16.6–17.8) in DP vs 8.3 months (95% CI, 8.0–8.5) in TPC; HR = 0.64, 95% CI, 0.56–0.72) (Figure 3(d)). In the emulated HR+HER2-population, DP surpassed both other chemotherapies and endocrine therapies, while the efficacy between other chemotherapies and endocrine treatments was similar (Figure S5(b)).
Secondly, in the total population, the application of statistical methods aimed at controlling confounding bias was further scrutinized through five concurrent analyses: univariable Cox regression and multivariable Cox regression within the naïve population, IPCW, multivariable adjusted Cox model, and PSOW in the emulated population (Figure 4). The various methodologies indicated that patients receiving DP had a reduced risk of metastatic disease progression.

Sensitivity and subgroup analyses examining progression-free survival in DP-treated versus TPC-treated patients. Subgroup analyses were conducted in the emulated total population.
Subgroup analyses
The results from the subgroup analyses of the emulated population indicated that the PFS benefit associated with metronomic weekly DP compared to TPC was generally consistent across predefined subgroups, including patients with visceral metastases and those with nonvisceral metastases (Figure 4; Figures S6–S8). However, for patients receiving DP as second-line therapy or beyond, there was no significant reduction in risk when compared to those receiving TPC (HR = 1.04, 95% CI, 0.89–1.22). The subgroup analyses of the naïve population showed a statistically significant PFS benefit associated with the DP regimen compared to TPC in the subgroup of patients aged ⩾55 years. In other subgroups, including those with HR+/HER2− and HER2+ subtypes, the DP group demonstrated numerically longer median PFS than the TPC group. However, these differences were not statistically significant (Figures S6–S8).
Discussion
To the best of our knowledge, this study is the first to evaluate the real-world efficacy of metronomic weekly DP chemotherapy in MBC via target trial emulation. Our findings suggest that this regimen may offer a potential treatment option for select MBC patients, as it was associated with significantly improved PFS compared to TPC in both naïve and emulated populations. Although several subgroup analyses within the naïve population demonstrated only numerical advantages without statistical significance, corresponding analyses in the emulated population showed consistently significant PFS benefits. The consistency of metronomic DP’s efficacy across multiple sensitivity analyses supports its robustness, yet the retrospective nature of this study and potential unmeasured confounders must be considered when interpreting the results. The study also demonstrates the utility of target trial emulation in generating clinically meaningful evidence from RWD, especially when randomized trials are not feasible due to heterogeneity or recruitment limitations.
Our findings suggest that metronomic weekly DP represents a promising treatment strategy for MBC, offering both clinical efficacy and practical advantages. Metronomic chemotherapy, characterized by the frequent administration of lower-dose chemotherapeutic agents, minimizes toxicity while improving patient quality of life compared to conventional maximum tolerated dose (MTD) chemotherapy.26,27 Beyond its clinical advantages, metronomic DP also presents a cost-effective alternative to more expensive systemic therapies, making it particularly valuable in resource-limited settings. The affordability of paclitaxel and cisplatin, combined with their established efficacy, offers a high-value treatment option for patients in low- and middle-income countries and underserved regions. 28 By reducing the financial burden associated with cancer care while maintaining therapeutic efficacy, metronomic DP could improve treatment accessibility and equity, ultimately benefiting a broader patient population.
Metronomic chemotherapy exerts antitumor effects through anti-angiogenesis, immune modulation, and direct cytotoxicity. By sustaining endothelial suppression, it deprives tumors of essential nutrients and inhibits neovascularization, a process enhanced by paclitaxel’s disruption of endothelial function and cisplatin’s modulation of hypoxia-inducible factor 1-alpha (HIF-1α). Metronomic chemotherapy also reshapes the tumor immune microenvironment by reducing regulatory T cells and myeloid-derived suppressor cells, while enhancing cytotoxic T cell and natural killer cell activity, thereby alleviating immune suppression and promoting sustained antitumor immunity. 26 Beyond these effects, paclitaxel stabilizes microtubules, inducing mitotic arrest, whereas cisplatin cross-links DNA, disrupting cancer cell replication. 29 The efficacy of this regimen may also be attributed to the well-established benefits of platinum-based chemotherapy, which has demonstrated significant improvements in survival and quality of life for MBC patients, particularly in aggressive subtypes where the paclitaxel-cisplatin combination exhibits superior efficacy. 30 Unlike MTD chemotherapy, which relies on acute cytotoxicity, metronomic DP maintains prolonged tumor suppression with lower toxicity, making it particularly advantageous in aggressive MBC subtypes where sustained control is critical.26,27
For triple-negative MBC, where therapeutic options remain limited despite advances in immunotherapy, targeted therapy, and antibody-drug conjugates, our findings support DP chemotherapy as a promising regimen. Recent clinical trials have highlighted the benefits of incorporating platinum-based regimens in TNBC, demonstrating improved response rates and PFS, particularly in patients with breast cancer susceptibility gene (BRCA) mutations or homologous recombination deficiencies.31,32 For instance, the TNT trial showed that platinum significantly outperformed docetaxel in BRCA-mutated TNBC, with a higher objective response rate (68% vs 33%) and prolonged PFS (6.8 months vs 4.4 months). This combination has been shown to enhance pathological complete response rates in neoadjuvant settings. 32 Our study underscores the relevance of chemotherapy in the management of TNBC, not only as a first-line option but also as an effective treatment in subsequent lines of therapy when resistance develops to newer agents, aligning with emerging clinical evidence.
In HER2-positive MBC, our sensitivity analyses demonstrated that metronomic DP, when combined with concurrent HER2-targeted therapies, achieves significant efficacy. This aligns with clinical trial evidence indicating that the addition of platinum agents to paclitaxel and HER2 blockade improves patient outcomes. 33 Despite dual HER2 blockade being the standard of care, disease progression remains a challenge, underscoring the need for alternative strategies, particularly for patients who develop resistance to HER2-directed therapies. 34 Metronomic chemotherapy presents a promising approach by offering sustained cytotoxic effects with decreased adverse effects, potentially enhancing treatment tolerance and prolonging disease control.26,35 Preclinical studies suggest that platinum-based regimens may enhance antitumor immunity when combined with immune checkpoint inhibitors,14,36 supporting the potential integration of metronomic chemotherapy into immunotherapy-based combination strategies to improve treatment outcomes.
One of the key subtypes explicitly analyzed in this study is HR-positive and HER2-negative breast cancer, which comprises 65%—75% of all breast cancer cases.1,37 Currently, the preferred first-line therapy for HR-positive and HER2-negative MBC involves endocrine therapy (ET) combined with a CDK4/6 inhibitor, followed by additional endocrine therapies and targeted agents such as CDK4/6 inhibitors, protein kinase B (AKT) inhibitors, mechanistic target of rapamycin (mTOR) inhibitors, and poly (ADP-ribose) polymerase (PARP) inhibitors. 38 Chemotherapy and antibody-drug conjugates (ADCs) are typically reserved for cases where ET and targeted therapies have been exhausted.30,39 Our sensitivity analysis revealed that DP significantly reduced the risk of disease progression compared to first-line endocrine therapy, indicating its potential as a viable alternative, particularly for patients exhibiting resistance to endocrine therapy, targeted agents, or ADCs. Although chemotherapy is generally not the first-line treatment of choice for HR-positive and HER2-negative patients, some studies have shown that chemotherapy as a first-line treatment is not significantly inferior to ET, exhibiting comparable response rates and clinical benefit rates.30,39,40 In addition, initial chemotherapy can prevent rapid disease progression in endocrine-insensitive populations, maintaining patient stability at the beginning of salvage treatment. Future research should explore the optimal timing and sequencing of chemotherapy and ET, considering that most advanced HR-positive and HER2-negative patients will eventually develop endocrine resistance. This is crucial for improving long-term outcomes and quality of life for this substantial patient population.
Despite the strengths of this study, several limitations should be acknowledged, highlighting key areas for future research. First, as a retrospective observational analysis, residual confounding cannot be entirely excluded, even with the use of propensity score matching and target trial emulation. Future prospective studies or RCTs are needed to validate our findings and strengthen causal inference. Second, several subgroup analyses, particularly in the naïve population, did not achieve statistical significance, likely due to limited sample sizes in several subsets, such as HER2-positive pretreated patients. Expanding cohort sizes and incorporating molecular profiling could help identify patient subgroups that derive the most benefit from this regimen. Third, the extended study period spanning nearly a decade limited the inclusion of more recently developed treatment modalities, such as immune checkpoint inhibitors and advanced targeted therapies. A recent phase II trial demonstrated that metronomic chemotherapy combined with PD-1 blockade achieved high disease control rates and prolonged PFS in MBC, suggesting its potential as an effective treatment strategy. 35 Future research should explore the potential synergy between metronomic DP and novel immunotherapeutic or targeted approaches to optimize treatment strategies. Fourth, this study did not assess treatment-related toxicity, which limits our ability to evaluate the full therapeutic index of the metronomic DP regimen in the metastatic setting. Although prior studies in the neoadjuvant setting have demonstrated favorable tolerability of this regimen,11,12 future prospective analyses are needed to confirm its safety profile in real-world MBC populations. Another important limitation is the absence of key biological markers such as BRCA mutation status and the Ki-67 proliferation index. While BRCA status is a well-established determinant of platinum sensitivity due to its role in DNA repair mechanisms, Ki-67 serves as a useful prognostic and predictive marker.41 –43 Given their established prognostic and predictive value, future prospective studies with comprehensive biomarker profiling are needed to assess their impact on treatment outcomes. Finally, due to the retrospective nature of this study, long-term survival outcomes, including overall survival, as well as quality of life data, were not systematically collected. These important outcomes remain to be investigated in future prospective studies with structured follow-up. Further longitudinal studies with extended follow-up are essential to determine the durability of response and overall patient benefit.
Conclusion
This study demonstrates that metronomic weekly administration of paclitaxel combined with cisplatin may represent a viable therapeutic strategy for MBC, particularly in first-line treatment settings. By bridging the divide between RCTs and observational studies, our application of target trial emulation yields significant clinical insights derived from RWD.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251384228 – Supplemental material for Metronomic chemotherapy of paclitaxel plus cisplatin in patients with metastatic breast cancer: a target trial emulation study
Supplemental material, sj-docx-1-tam-10.1177_17588359251384228 for Metronomic chemotherapy of paclitaxel plus cisplatin in patients with metastatic breast cancer: a target trial emulation study by Yaorong Li, Liang Zhu, Jiayi Ma, Yan Wang, Yanping Lin, Yumei Ye, Liheng Zhou, Wenjin Yin, Jinsong Lu and Yaohui Wang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank the patients and their families participating in this study. We also thank Jie Zhang, Wenjuan Tang, and Mengya Fang for assistance in collecting data.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
