Abstract
Background:
The incidence of early-onset colorectal cancer (EO-CRC), defined as a diagnosis before 50 years of age, is increasing worldwide. However, its clinical characteristics and outcomes compared to average-onset colorectal cancer (AO-CRC) remain under debate, especially in the setting of locally advanced rectal cancer (LARC).
Objectives:
This study aimed to compare clinical characteristics, treatment responses, and survival outcomes between patients with early-onset and average-onset locally advanced rectal cancer.
Design:
A multicenter retrospective cohort study.
Methods:
We retrospectively analysed 305 patients with stage II–III rectal cancer treated between 2012 and 2022 across three Italian oncology centrers. Patients were categorised as EO-RC (⩽50 years) or AO-RC (>50 years). All patients underwent neoadjuvant chemoradiotherapy followed by total mesorectal excision. Pathological and radiological responses were evaluated, and survival outcomes were assessed through Kaplan-Meier methods.
Results:
Early-onset patients accounted for 10.5% of the cohort. Clinical and pathological characteristics were broadly similar between groups, although EO-RC patients had a higher prevalence of proficient mismatch repair status. Radiological and pathological response rates were comparable. After a median follow-up of 120 months, the 10-year overall survival (OS) was 73.3% in EO-RC and 91.9% in AO-RC (HR 7.60, 95% CI 2.22–26.06; p = 0.0012). Disease-free survival (DFS) at 10 years was 57.1% in EO-RC and 70.9% in AO-RC (HR 1.80, 95% CI 0.86–3.78; p = 0.1177).
Conclusion:
Early-onset rectal cancer patients exhibit similar response rates and DFS compared to older patients, but appear to have worse OS. Further studies are needed to explore biological factors and post-recurrence treatment strategies that may influence these outcomes.
Plain language summary
Colorectal cancer is a common and deadly disease, but the incidence in younger adults under 50 is increasing. It remains unclear whether younger patients with rectal cancer have different outcomes compared to older patients. In this study, we analysed 305 patients with locally advanced rectal cancer treated across three Italian hospitals. We compared the characteristics and treatment outcomes of younger patients (under 50 years old) with those of older patients. All patients received pre-surgery chemotherapy and radiotherapy followed by surgery. We found that younger patients had similar chances of cancer recurrence compared to older patients, but they were more likely to have a lower overall survival. This means that although the cancer may return at a similar rate, younger patients might still have a higher risk of dying earlier. This information suggests that rectal cancer in younger people could behave differently, possibly due to biological factors. More research is needed to understand why this happens and how treatments can be improved for younger patients.
Keywords
Introduction
Colorectal cancer is among the most common malignant tumours, ranking third in incidence and second in mortality worldwide. Specifically for rectal cancer, approximately 729,702 new cases are diagnosed globally each year, resulting in a mortality rate of about 343,761 deaths. 1 The data from Italy are consistent, indicating that there were 50,500 new diagnoses in 2023 and a mortality rate of 24,200 cases in 2022. The 5-year survival rate stands at 65%–66%. Thanks to the latest therapeutic innovations, survival rates have improved and are expected to continue rising in the coming years. 2
In recent decades, many high-incidence countries have experienced a decline in colorectal cancer cases. This positive trend is primarily attributed to changes in population lifestyles that encourage healthier habits and mitigate major risk factors. Enhanced diets and the implementation of screening programmes have significantly contributed to this decrease.3,4
In contrast to the overall trend of stabilisation or decline in cancer rates among the general population, recent reports indicate a rise in colorectal cancer cases among young adults (under 50 years old at diagnosis) in many Western, high-income countries, including Canada and the United States. The incidence of this type of cancer is increasing by 1% to 4% per year.5–11
The reasons behind the rising incidence in new generations remain unclear. However, it is suspected that certain risk factors during early childhood and young adulthood may have a significant impact. These factors include the increasing prevalence of obesity, dietary habits, physical inactivity, and the use of antibiotics, which may reduce the diversity of the intestinal microbiome. 12
We are collecting preliminary outcome data from retrospective studies that evaluate treatment responses in patients with early-onset disease. However, the literature remains inconsistent regarding the prognostic significance of early-onset colorectal cancer. Some studies suggest a worse prognosis, while others report similar or better outcomes than those with average-onset. A detailed review on this topic can be found in Gandini et al. 13 These mixed results highlight the need for further research. In cases of metastatic disease, data show that early-onset patients generally have lower overall survival (OS) and progression-free survival (PFS). Moreover, in stage II and III colon adenocarcinoma, these patients often present at a more advanced stage, which results in shorter disease-free survival (DFS) and OS.14,15
Due to the limited data on patients with early-onset rectal cancer (EO-RC), our study aimed to evaluate treatment responses and outcomes – specifically DFS and OS – in a cohort of early-onset patients diagnosed with locally advanced rectal adenocarcinoma. We compared these results with those from average-onset patients.
Methods
Patients and methods
We retrospectively collected data from 305 patients affected by LARC adenocarcinoma (cT3-4 +/− N1-2) treated at the Medical Oncology Unit of the University Hospital of Cagliari, Italy, the Medical Oncology Department, Fondazione IRCCS Istituto Nazionale dei Tumori, Milan, Italy, and at the Medical Oncology Unit, AOU Ospedali Riuniti, Ancona, Italy.
All patients were diagnosed and treated between January 2012 and December 2022. Those included in the study underwent neoadjuvant concurrent capecitabine (825 mg/m2/bid) and long-course radiotherapy (RT) (total dose of 50.4 Gy). CT and MRI were performed at baseline and before surgery to verify the radiological response according to RECIST v1.1 criteria. 16 Subsequently, all patients underwent total mesorectal excision at the local Colorectal Surgery Unit. Tumor Regression Grade (TRG) evaluation was conducted on the surgical sample according to Ryan’s score17–19 to describe the tissue response to chemoradiotherapy.
MMR expression was evaluated through immunohistochemistry. Immunohistochemical investigations were performed on the surgical samples to assess Mismatch Repair Proteins expression (MLH1, PMS2, MSH6, MSH2, and EPCAM). The study’s primary objective was to compare the OS of EO- and AO-RC patients. The secondary objectives included: PFS of EO- and AO-RC patients; overall response rate (ORR), differences in disease control rate (DCR) between EO- and AO-RC after treatment, and pathological responses measured through TRG. The reporting of this observational study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for cohort studies 20 (see Supplemental File 1).
Statistical analysis
Statistical analysis was performed with the MedCalc® Statistical Software version 23.0.2 (MedCalc Software Ltd, Ostend, Belgium; https://www.medcalc.org, 2024). The association between categorical variables was estimated by the Fisher exact test for categorical binomial variables or by the chi-square test in all other instances. Survival probability over time was estimated by the Kaplan-Meier method. Significant differences in the probability of survival between the strata were evaluated by the log-rank test. The independent role of variables that were statistically significant at univariate analysis was assessed with a logistic regression analysis.
OS was defined as the time interval between the date of the treatment start to death or the last follow-up visit for patients who were lost at follow-up. DFS was defined as the time from treatment start until the first cancer-related event, second cancer, or death from any cause. ORR is defined as the proportion of patients who have a partial or complete response to therapy. DCR was defined as the percentage of patients with stable disease or partial/complete response to treatment.
Results
Thirty-two (10.5%) patients were EO-RC and 273 (89.5%) were AO-RC. EO-RC median age was 45, while AO-RC median age was 68. Twenty-one out of 32 (65.6%) patients in EO-RC were male, and 186 out of 273 (68.1%) patients in AO-RC were male.
Overall population: Treatment response and survival outcomes
In EO-RC, the locations were distributed as follows: 10 (31.25%) lower rectum, 18 (56.25%) medium rectum, and 4 (12.50%) upper rectum. In AO-RC, the locations were: 76 (27.8%) lower rectum, 142 (52.0%) medium rectum, and 55 (20.2%) upper rectum. In EO-RC patients, 12 (37.5%) were diagnosed at stage II, while 20 (62.5%) were diagnosed at stage III. In AO-RC patients, 96 (35.2%) were diagnosed at stage II and 177 (64.8%) at stage III (Table 1). All patients achieved an R0 resection. Postoperative chemotherapy was administered in 78.7% of patients, with CAPOX used in 63.2% of cases and FOLFOX in 36.8%.
Baseline clinical and pathological characteristics of the study population.

Data are presented as number of patients with percentages in parentheses, unless otherwise specified. Italic values represent p-values from chi-square or Fisher’s exact test (as appropriate).
AO-RC, average-onset rectal cancer; cT/cN, clinical tumour/nodal stage; dMMR, deficient mismatch repair; EO-RC, early-onset rectal cancer; F, female; M, male; MMR, mismatch repair status; pMMR, proficient mismatch repair; ypT/ypN, pathological tumour/nodal stage after neoadjuvant treatment.
The median follow-up was 120 months (10 years; IQR 90–140 months). The median OS was not reached in either group. The 10-year OS rate was 73.3% in EO-RC and 91.9% in AO-RC. The hazard ratio (HR) for OS comparing EO-RC to AO-RC was 7.60 (95% CI: 2.22–26.06; p = 0.0012), indicating a significantly higher risk of death in the EO-RC group (Figure 1(a)).
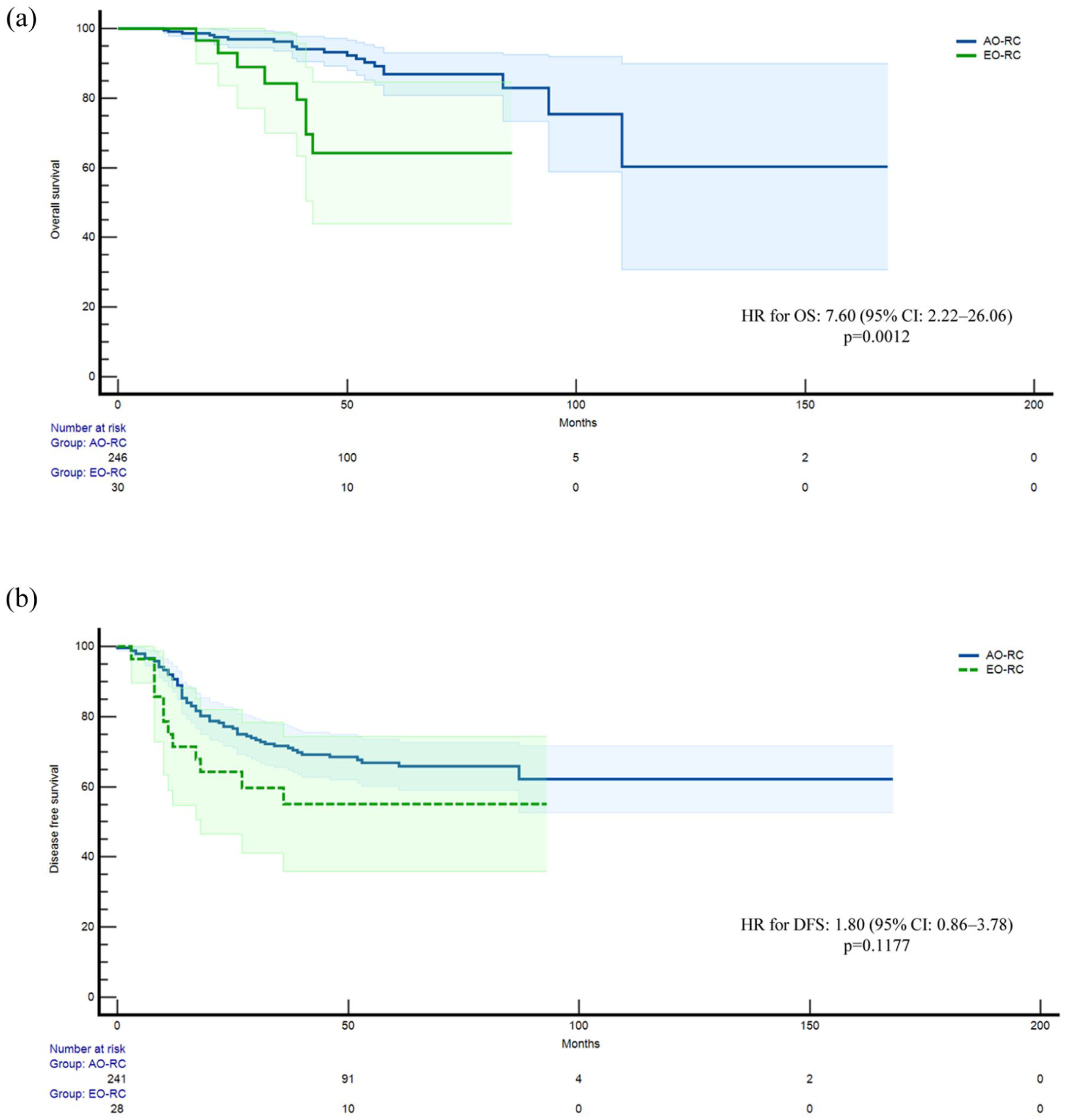
Kaplan-Meier survival curves for the overall population: (a) OS comparing EO-RC (green line) and AO-RC (blue line). (b) DFS comparing EO-RC and AO-RC. Shaded areas represent 95% confidence intervals. Number-at-risk tables are shown below each plot.
The median DFS was not reached in either group. EO-RC patients showed a 10-year DFS of 57.14% versus 70.95% in AO-RC. The HR for disease recurrence or death in EO-RC compared to AO-RC was 1.80 (95% CI: 0.86–3.78; p = 0.1177; Figure 1(b)).
In EO-RC, the radiological responses were as follows: 2 (6.3%) CR, 13 (40.6%) PR, 15 (46.8%) SD, and 2 (6.3%) PD. Radiological responses in AO-RC were as follows: 25 (9.1%) CR, 159 (58.2%) PR, 71 (26.0%) SD, and 18 (6.6%) PD (Table 2).
Radiological and pathological tumour response in early-onset and average-onset rectal cancer.

Italic values represent p-values from chi-square or Fisher’s exact test (as appropriate).
Pathologic responses in EO-RC were as follows: 3 (9.4%) TRG-0, 6 (18.7%) TRG-1, 10 (31.2%) TRG-2, and 13 (40.6%) TRG-3. Pathologic responses in AO-RC were as follows: 28 (10.2%) TRG-0, 66 (24.2%) TRG-1, 152 (55.7%) TRG-2, and 28 (10.2%) TRG-3 (Table 2).
pMMR subgroup: Treatment response and survival outcomes
Considering only proficient MMR patients, the OS rate was statistically significantly lower in EO-RC than in AO-RC (92.54% versus 70.83%). The HR for death was 11.97 (95% CI: 2.94–48.75; p = 0.0005), indicating a significantly worse OS in EO-RC patients (p = 0.0005; Figure 2(a)).

Kaplan-Meier survival curves for the pMMR subgroup. (a) OS comparing EO-RC (green line) and AO-RC (blue line). The 10-year OS rate was 70.83% in EO-RC versus 92.54% in AO-RC (HR 11.97, 95% CI: 2.94–48.75; p = 0.0005. (b) DFS comparing EO-RC and AO-RC. The 10-year DFS rate was 68.18% in EO-RC versus 72.65% in AO-RC (HR 1.19, 95% CI: 0.51–2.74; p = 0.6899).
EO-RC proficient mismatch repair (pMMR) patients showed a lower DFS rate than AO-RC (72.65% versus 68.18%). The HR for disease recurrence or death was 1.19 (95% CI: 0.51–2.74; p = 0.6899), indicating no significant difference between groups (Figure 2(b)).
dMMR subgroup: Treatment response and survival outcomes
Considering dMMR patients, even in this case DFS rate was lower in EO-RC compared to AO-RC (50.00% versus 14.29%). The HR for disease recurrence or death was 3.10 (95% CI: 0.79–12.13; p = 0.0508), indicating a trend toward worse DFS in EO-RC, although not statistically significant (Figure 3). OS rate data were immature.

Kaplan-Meier DFS curve for the dMMR subgroup. DFS comparing EO-RC (green line) and AO-RC (blue line). The 10-year DFS rate was 14.29% in EO-RC versus 50.00% in AO-RC. The hazard ratio for disease recurrence or death was 3.10 (95% CI: 0.79–12.13; p = 0.1045). Shaded areas represent 95% confidence intervals. Number-at-risk tables are shown below the plot.
In patients with pMMR EO-RC, the radiological responses were as follows: 2 patients (8.0%) achieved a complete response (CR), 13 patients (52.0%) had a partial response (PR), nine patients (40.0%) experienced stable disease (SD), and one patient (4.0%) had progressive disease (PD). For patients with pMMR AO-RC, the radiological responses were as follows: 25 patients (9.8%) achieved a CR, 154 patients (60.4%) had a PR, 60 patients (23.5%) experienced SD, and 16 patients (6.3%) had PD.
For dMMR patients, the radiological responses in the EO-RC group were as follows: 1 (14.3%) had SD, and 6 (85.7%) had PD. In the dMMR AO-RC group, the radiological responses were as follows: 5 (27.8%) had PR, 11 (61.1%) had SD, and 2 (11.1%) had PD (Table 3).
Radiological responses in pMMR and dMMR subgroups.

dMMR, deficient mismatch repair; ORR, overall response rate; pMMR, proficient mismatch repair.
For pMMR patients, pathologic responses in EO-RC were as follows: 3 (%) TRG-0, 6 (%) TRG-1, 10 (%) TRG-2, and 5 (%) TRG-3. Pathologic responses in AO-RC were as follows: 29 (10.6%) TRG-0, 61 (22.4%) TRG-1, 155 (56.8%) TRG-2, and 28 (10.2%) TRG-3.
In dMMR patients, pathologic responses in EO-RC were 7 (100%) TRG-3. Pathologic responses in LO-RC were as follows: 1 (%) TRG-1, 3 (%) TRG-2, and 14 (%) TRG-3 (Table 4).
Tumour regression grade according to Ryan’s scores in pMMR and dMMR subgroups.

Ryan tumour regression (TRG) score: TRG-0 no visible cancer cells (complete response); TRG-1 single cells or small group of cancer cells (near-complete response); TRG-2 residual cancer with evident tumour regression, but more than single cells or rare small group of cancer cells (partial response); TRG-3 extensive residual cancer with no evident tumour regression (poor or no response).
dMMR, deficient mismatch repair; pMMR, proficient mismatch repair.
Italic values represent p-values from chi-square or Fisher’s exact test (as appropriate).
Discussion
Our study revealed a correlation between young age at diagnosis and a worse prognosis in patients with rectal adenocarcinoma undergoing neoadjuvant therapies and surgical resection. Specifically, patients aged 50 or younger demonstrated a reduced response to treatments, assessed radiologically and pathologically. Research on this topic is currently limited and presents conflicting results. Lehtonen et al. 21 gathered data from 147 patients with early-onset rectal cancer, encompassing all disease stages from I to IV. They reported outcomes comparable to those of the general population.
Yin et al. 22 evaluated how the primary tumour size affects the prognosis of patients with early-onset colorectal cancer. Their study showed a correlation in the subgroup of rectal cancer patients but did not provide a comparison with those diagnosed later in life. Another aspect relates to the correlation between the deficit of MMR protein expression and prognosis and treatment responses. Specifically, within the same early-onset group, dMMR/MSI-H patients display a worse prognosis. Recent literature shows that MMR deficiency correlates with lower efficacy of treatments. 23 Emile et al. 24 evaluated the role of DMMR in this context, demonstrating a comparable prognosis for early-onset (EO) and late-onset (LO) patients. However, the study encompassed all disease stages; thus, the population was not stage-selected.
Our data suggest that EO-RC may exhibit distinct clinical behaviours compared to average-onset cases, especially regarding OS. However, these observations need to be interpreted with caution. In the pMMR subgroup, which makes up the majority of our cohort, DFS was similar between EO-RC and AO-RC, indicating that the prognosis for recurrence risk may not differ significantly based on age at onset.
Interestingly, the trend toward worse DFS in the overall population could be driven by the dMMR subgroup, where EO-RC patients appeared to have poorer outcomes, though this was based on a small sample and lacked statistical significance. This finding warrants further study with larger datasets.
A key finding of our analysis is the discrepancy between DFS and OS in EO-RC patients. While recurrence rates were similar among pMMR patients, OS was worse in EO-RC. This suggests that factors beyond recurrence – such as treatment-related complications, biological aggressiveness, or sociodemographic variables – might contribute to overall mortality. Still, the retrospective nature of our study limits the ability to explore these hypotheses fully.
In conclusion, our results do not definitively prove a worse prognosis for EO-RC compared to AO-RC, aligning with the varied evidence in the literature. Instead, our findings underscore the need to consider EO-RC as a distinct clinical entity, warranting specific prospective studies to better understand its biological features and prognostic implications.
In our study, neither median OS nor DFS was reached after a follow-up of 10 years. This is mainly due to the limited number of events, especially in the relatively small EO-RC subgroup, which diminishes the chance of the survival curves crossing the 50% threshold needed to determine median values. In such cases, long-term survival rates – like 10-year OS and DFS – serve as more reliable measures for comparing outcomes between EO-RC and AO-RC.
The discrepancy between DFS and OS in EO-RC raises several hypotheses. A more aggressive tumour phenotype in younger patients might be a key factor. Although the recurrence risk appears similar between EO-RC and AO-RC in the pMMR subgroup, the characteristics of recurrence – such as timing, anatomical sites, or response to salvage treatments – may differ. Younger patients could experience faster disease progression after recurrence or develop metastases in less treatable areas, ultimately affecting OS.
Furthermore, while our study lacks detailed data on post-recurrence therapies, it is plausible that EO-RC patients received more aggressive or experimental treatments, which may not have improved survival. Alternatively, cumulative toxicities from neoadjuvant, adjuvant, and subsequent treatments could have compromised their overall health, impacting long-term survival.
Another consideration is the potential influence of unmeasured biological factors, such as specific molecular alterations or tumour microenvironment traits, which might contribute to a more aggressive disease course in younger individuals. Although less likely, sociodemographic variables, disparities in access to specialised care, or delays in diagnosis could also play a role.
Without data on cause-specific mortality, we cannot exclude the contribution of non-cancer-related deaths, though this seems less probable in a younger population. Future prospective studies featuring comprehensive biological characterisation, detailed records of post-recurrence treatments, and analyses of progression patterns and causes of death are vital to better understand these dynamics and improve outcomes in EO-RC.
The study has limitations, including its retrospective design. These findings should be interpreted cautiously due to the relatively small sample size of the EO-RC subgroup, particularly when stratifying by MMR status, which may limit statistical power and the generalisability of the survival analyses. Additionally, the patients included were treated according to standard practices prior to the introduction of the total neoadjuvant therapy (TNT) strategy23–27 and the approval of dostarlimab26,28 for perioperative treatment in patients with dMMR rectal adenocarcinoma. Both TNT and perioperative dostarlimab use have since transformed the therapeutic approach. It will be interesting to assess these strategies in real-life settings and observe how EO-RC responds to these treatments.
Our study suggests that neoplasia developing at a young age may display biological aggressiveness. However, the literature on this topic remains limited and inconsistent, especially regarding hypotheses about pathogenesis and associated risk factors. The causes are thought to range from dietary and lifestyle factors to pollution from microplastics and other agents.27–31 Nonetheless, establishing a single definitive cause is currently impossible. An important factor is the often advanced stage of disease at diagnosis among younger patients, partly due to the absence of screening programmes and partly due to delays in diagnosis resulting from underestimating symptoms in this age group. Further studies are needed to better understand biological behaviour and clinical features. We also require precise information about risk factors and elements that may contribute to carcinogenesis in young individuals.
Conclusion
Our findings indicate that patients with EO locally advanced rectal cancer exhibit similar pathological and radiological responses, as well as comparable DFS, to those diagnosed at an older age. However, the observed worse OS in EO patients raises important questions about the biological behaviour of these tumours and the effectiveness of treatments after recurrence. These results emphasise the need for further prospective studies to better understand the underlying mechanisms and to optimise therapeutic strategies tailored to younger patients.
Supplemental Material
sj-doc-1-tam-10.1177_17588359251379744 – Supplemental material for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer
Supplemental material, sj-doc-1-tam-10.1177_17588359251379744 for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer by Andrea Pretta, Riccardo Giampieri, Pina Ziranu, Andrea Bottelli, Clelia Donisi, Elisa Tiberi, Erika Cimbro, Giovanni Randon, Matteo Fraschini, Dario Spanu, Luca Didaci, Veronica Dell’Utri, Gianluca Pretta, Stefano Mariani, Marco Puzzoni, Valeria Pusceddu, Rossana Berardi, Filippo Pietrantonio, Gavino Faa and Mario Scartozzi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-doc-3-tam-10.1177_17588359251379744 – Supplemental material for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer
Supplemental material, sj-doc-3-tam-10.1177_17588359251379744 for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer by Andrea Pretta, Riccardo Giampieri, Pina Ziranu, Andrea Bottelli, Clelia Donisi, Elisa Tiberi, Erika Cimbro, Giovanni Randon, Matteo Fraschini, Dario Spanu, Luca Didaci, Veronica Dell’Utri, Gianluca Pretta, Stefano Mariani, Marco Puzzoni, Valeria Pusceddu, Rossana Berardi, Filippo Pietrantonio, Gavino Faa and Mario Scartozzi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-doc-4-tam-10.1177_17588359251379744 – Supplemental material for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer
Supplemental material, sj-doc-4-tam-10.1177_17588359251379744 for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer by Andrea Pretta, Riccardo Giampieri, Pina Ziranu, Andrea Bottelli, Clelia Donisi, Elisa Tiberi, Erika Cimbro, Giovanni Randon, Matteo Fraschini, Dario Spanu, Luca Didaci, Veronica Dell’Utri, Gianluca Pretta, Stefano Mariani, Marco Puzzoni, Valeria Pusceddu, Rossana Berardi, Filippo Pietrantonio, Gavino Faa and Mario Scartozzi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251379744 – Supplemental material for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer
Supplemental material, sj-docx-2-tam-10.1177_17588359251379744 for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer by Andrea Pretta, Riccardo Giampieri, Pina Ziranu, Andrea Bottelli, Clelia Donisi, Elisa Tiberi, Erika Cimbro, Giovanni Randon, Matteo Fraschini, Dario Spanu, Luca Didaci, Veronica Dell’Utri, Gianluca Pretta, Stefano Mariani, Marco Puzzoni, Valeria Pusceddu, Rossana Berardi, Filippo Pietrantonio, Gavino Faa and Mario Scartozzi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-5-tam-10.1177_17588359251379744 – Supplemental material for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer
Supplemental material, sj-docx-5-tam-10.1177_17588359251379744 for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer by Andrea Pretta, Riccardo Giampieri, Pina Ziranu, Andrea Bottelli, Clelia Donisi, Elisa Tiberi, Erika Cimbro, Giovanni Randon, Matteo Fraschini, Dario Spanu, Luca Didaci, Veronica Dell’Utri, Gianluca Pretta, Stefano Mariani, Marco Puzzoni, Valeria Pusceddu, Rossana Berardi, Filippo Pietrantonio, Gavino Faa and Mario Scartozzi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-6-tam-10.1177_17588359251379744 – Supplemental material for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer
Supplemental material, sj-pdf-6-tam-10.1177_17588359251379744 for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer by Andrea Pretta, Riccardo Giampieri, Pina Ziranu, Andrea Bottelli, Clelia Donisi, Elisa Tiberi, Erika Cimbro, Giovanni Randon, Matteo Fraschini, Dario Spanu, Luca Didaci, Veronica Dell’Utri, Gianluca Pretta, Stefano Mariani, Marco Puzzoni, Valeria Pusceddu, Rossana Berardi, Filippo Pietrantonio, Gavino Faa and Mario Scartozzi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-7-tam-10.1177_17588359251379744 – Supplemental material for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer
Supplemental material, sj-pdf-7-tam-10.1177_17588359251379744 for Clinical characteristics and outcomes in patients with early-onset locally advanced rectal cancer by Andrea Pretta, Riccardo Giampieri, Pina Ziranu, Andrea Bottelli, Clelia Donisi, Elisa Tiberi, Erika Cimbro, Giovanni Randon, Matteo Fraschini, Dario Spanu, Luca Didaci, Veronica Dell’Utri, Gianluca Pretta, Stefano Mariani, Marco Puzzoni, Valeria Pusceddu, Rossana Berardi, Filippo Pietrantonio, Gavino Faa and Mario Scartozzi in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Preliminary results of this study were presented as an abstract at the 2024 ASCO Annual Meeting (![]() ). The views and opinions expressed are those of the authors only and do not necessarily reflect those of the European Union or the European Commission. Neither the European Union nor the European Commission can be held responsible for them.
). The views and opinions expressed are those of the authors only and do not necessarily reflect those of the European Union or the European Commission. Neither the European Union nor the European Commission can be held responsible for them.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
