Abstract

The concept of drug delivery has recently drawn the attention of the scientific community the claim that the efficacy of a new drug boils down to its ability to reach its target1,2 and ‘apparent’ resistance arises solely because of inefficient pharmacokinetics in the tumor: this could be called ‘pseudo-resistance’. Although it may seem an obvious concept, it receives scant attention during preclinical and clinical drug development. Clearly, achieving pharmacologically adequate drug delivery poses a challenge, particularly in oncology. 3 Successful treatment implies complete and homogeneous treatment of the tumor, meaning that the drug must reach all the tumor cells to the necessary extent, at the microscopic level.
New approaches in cancer research tend mainly to address omics and focus less on the intrinsic characteristics of each specific tumor. Often the omics are the reference markers to decide what type of drug, monoclonal antibody or Car-T-cell should be employed. This development is an integral part of the latest immunological discoveries that enable the body to make available all its immune resources to kill as many cancer cells as possible. This emerging new way to treat tumors or to make cancer a chronic disease is the basis of the old idea of personalized medicine, often referred to as ‘precision medicine’.
However, although this is a logical approach, attention focuses entirely on the tumor and the destiny of the drugs is neglected, when not altogether overlooked. 4 Scant attention is paid to drug concentrations in blood and even less to their distribution in the target, while we know that drugs are subject to absorption, distribution, metabolism, binding to proteins, and elimination, with broad variability among different subjects. Recent knowledge of the intestinal microbiome has now shown that drugs can be metabolized or stored by the different bacteria. For monoclonal antibodies, therapeutic proteins and enzymes, blood concentrations may change with time, because the antibodies that bind to the biological drugs can inactivate them.
Despite all our knowledge of pharmacokinetics, blood drug concentrations are not routinely measured, though it is the free drug available in the blood that enters the tumor and kills cancer cells. Moreover, even when systemic pharmacokinetic data are available they may not be indicative of the concentrations inside the tumor, adding even more complexity on account of their heterogeneous nature. The tumor is a complex tissue made up of different cell populations in the microenvironment, some of which are partially defective and contribute to extracellular matrix organization and angiogenesis. Therefore, the drug does not distribute homogeneously inside the tumor because the barriers made up of the tumor microenvironment prevent them from reaching all the cancer cells.
We can now study the fine distribution of a drug in a tumor with matrix-assisted laser desorption and ionization-mass spectrometry imaging (MALDI-MSI) which can quantitatively determine the concentrations of anticancer drugs in a slice of tumor, down to a few tens of micrometers, by laser scanning. 5 This technology visualizes the drug’s heterogeneous tumor distribution so we can see 6 how much of the tumor is adequately permeated (Figure 1). However, during treatment some of the cancer cells keep growing, not because they are intrinsically resistant, but because they are not reached by an adequate drug concentration. The concept of drug delivery here must necessarily be interpreted as drug penetration, and when the neoplastic cells do not receive a therapeutic dose, this contributes to the unsatisfactory treatment responses that are taken to imply drug resistance.
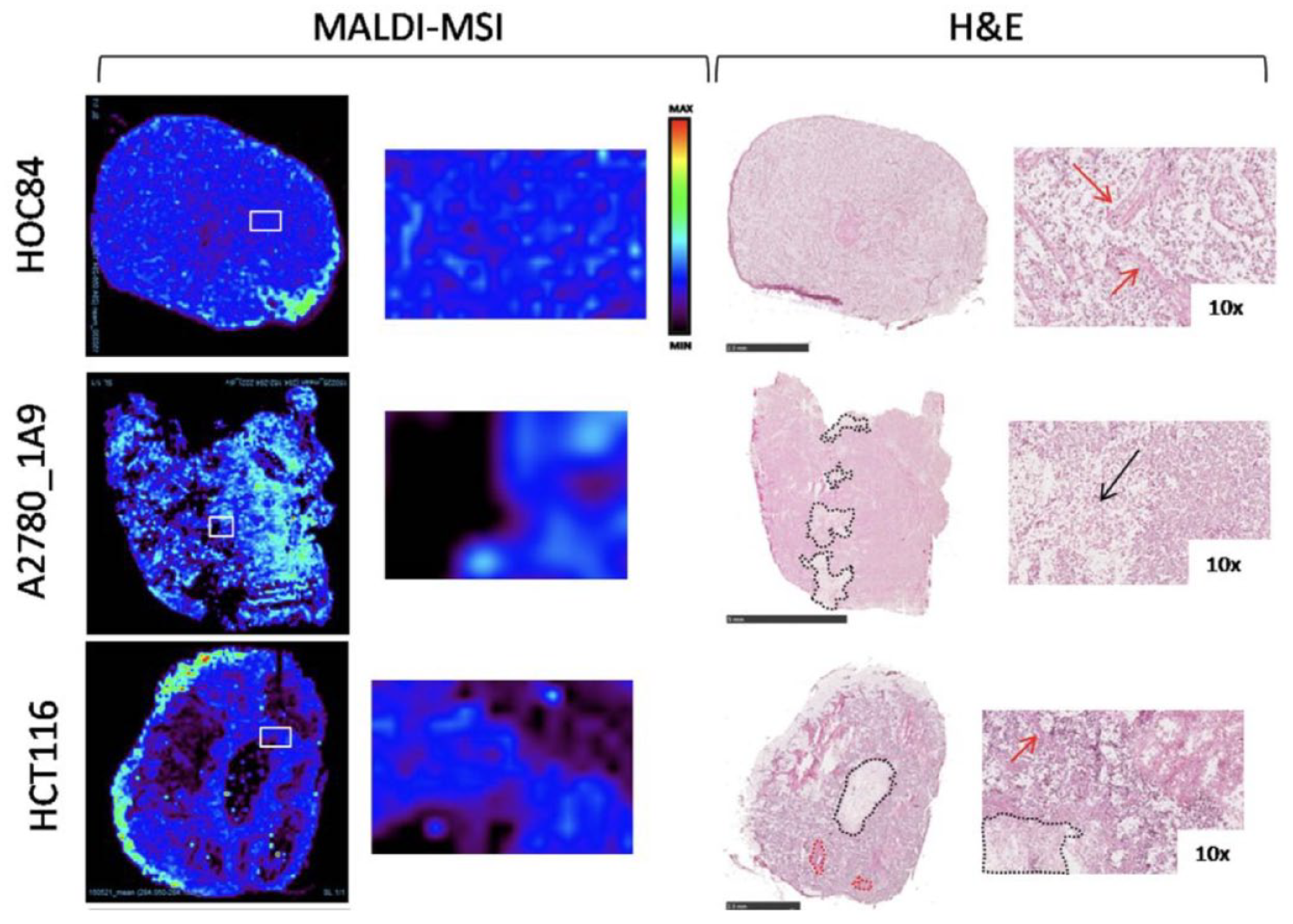
Spatial quantitative distribution of paclitaxel in human xenograft cancer models, two ovarian, HOC84, A2780_1A9 and one colon, HCT116. Drug distribution within tissues differs significantly in the three models. Black dotted lines and arrows in H&E images indicate necrotic regions. Red dotted lines and arrows indicate fibrotic region. H&E scale bar correspond to 2.5 mm except for A2780_1A9 (5 mm). Modified from Giordano et al. 6 .
How can we get round this ‘pseudo-resistance’? Better knowledge of the mechanism(s) that hamper homogeneous drug distribution in the tumor would have important therapeutic implications. For instance, we recently showed that if a tumor is rich in hyaluronic acid, drug distribution could be improved by pre-treatment with hyaluronidase allowing better entry of the drug and consequently a greater effect on the tumor growth. 7
