Abstract
Background:
HER2-ultralow is an emerging subgroup of metastatic breast cancer (mBC). However, despite an increasing interest, limited data exist on its prevalence and outcomes, especially among patients receiving standard first-line chemotherapy.
Objectives:
This study assessed the prevalence and outcomes of HER2-ultralow mBC in a real-world cohort of hormone receptor-positive (HR+)/HER2-negative patients receiving first-line standard-of-care (SOC) chemotherapy.
Design:
A retrospective, single-center cohort study.
Methods:
We included HR+/HER2-negative mBC patients treated with SOC between January 2016 and February 2023. Patient data were reviewed from electronic health records. HER2-zero tumors (immunohistochemistry 0) were rescored by expert pathologists using the American Society of Clinical Oncology/College of American Pathologists guidelines to distinguish HER2-ultralow from HER2-null cases. Real-world progression-free survival (rwPFS) and real-world overall survival (rwOS) were estimated using Kaplan–Meier and multivariable Cox regression models.
Results:
Among 320 patients (median age, 62.4 years), 72.8% had visceral metastases, and 17.5% had bone-only disease. Previous CDK4/6 inhibitor treatment was reported in 43.4%. Rescoring identified 15.6% with HER2-ultralow, 61.9% with HER2-low, and 22.5% with HER2-null tumors. The median follow-up was 39.4 months (95% confidence interval (CI), 37.1–47.6). Median rwPFS was 7.9 months (95% CI, 4.6–19.0), 7.3 months (95% CI, 6.4–9.1), and 6.2 months (95% CI, 4.6–8.9) for HER2-ultralow, HER2-low, and HER2-null groups, respectively. Median rwOS was 19.5 months (95% CI, 10.7–33.4), 21.5 months (95% CI, 19.0–25.8), and 16.6 months (95% CI, 11.9–23.6). In patients previously treated with CDK4/6 inhibitors, median rwPFS was 4.6 months (95% CI, 2.7–21.4), 6.1 months (95% CI, 5.5–7.3), and 5.6 months (95% CI, 3.8–8.7).
Conclusion:
HER2-ultralow accounts for 15.6% of HR+/HER2-negative mBC cases and demonstrates outcomes comparable to HER2-low with SOC chemotherapy.
Introduction
Breast cancer classification has significantly evolved with the advent of novel antibody–drug conjugates (ADCs) targeting HER2. Initially, HER2-targeted therapies were limited to HER2-positive tumors characterized by HER2 protein overexpression or amplification (immunohistochemistry (IHC) 3+ or IHC 2+ with in situ hybridization (ISH) positivity). However, the recent DESTINY-Breast04 and DESTINY-Breast06 trials demonstrated that trastuzumab deruxtecan (T-DXd), a new HER2 ADC, outperformed standard-of-care (SOC) chemotherapy in patients with hormone receptor-positive (HR+) metastatic breast cancer (mBC) classified as HER2-low (IHC 1+ or IHC 2+ with ISH negativity), a subgroup previously considered HER2-negative.1,2 This breakthrough has defined HER2-low mBC as a distinct therapeutic category, reshaping treatment paradigms and validating the effectiveness of HER2-targeted ADCs in tumors with low HER2 expression. 3
HER2 expression exists along a continuum, and a new therapeutic subgroup is emerging within the HER2-zero category.4,5 HER2-null is defined by a complete absence of detectable HER2 staining and HER2-ultralow mBC is defined by incomplete and faint membrane staining in less than 10% of tumor cells.6,7 Interest in the HER2-ultralow subgroup has grown following the DESTINY-Breast06 trial, which demonstrated that T-DXd significantly improved progression-free survival (PFS) compared to the SOC in both HR+/HER2-low and HER2-ultralow mBC patients previously treated with endocrine therapy (ET). 8 These findings challenge the traditional dichotomous HER2 classifications and underscore the clinical relevance of identifying and addressing the HR+/HER2-ultralow population, which may soon become a distinct therapeutic subgroup. The prevalence of HER2-ultralow is not well understood, with estimates suggesting that it accounts for approximately 20% of mBC cases in clinical trials and retrospective studies.8 –10 Real-world data reflect the routine clinical practice from a more diverse and heterogeneous population. Investigating the true prevalence and clinical behavior of this population particularly in patients receiving first-line chemotherapy remains poorly defined. This study aimed to determine the prevalence of HER2-ultralow mBC and assess outcomes in HR+/HER2-negative mBC patients receiving first-line SOC chemotherapy in a real-world setting.
Materials and methods
Study population
This single-center cohort study included patients diagnosed with mBC between January 1, 2016, and February 28, 2023, who initiated first-line SOC chemotherapy for HR+/HER2-negative disease at the Institut de Cancérologie de l’Ouest (ICO). SOC chemotherapy at the time of study comprised taxanes, capecitabine, anthracyclines, or other agents, as a single agent or in combination according to the European Society For Medical Oncology (ESMO) guideline. 11 Eligibility required the availability of tumor slides for HER2 rescoring. Exclusion criteria included prior receipt of HER2-targeted therapies, participation in clinical trials, or a diagnosis of a second primary cancer within the preceding 5 years. The study protocol was approved by an independent ethics committee. As deidentified data were used, no formal informed consent was required; however, all participants provided written consent for the reuse of their electronically recorded data. The study adhered to the ethical principles outlined in the Declaration of Helsinki, Good Clinical Practice guidelines, applicable regulatory requirements, and the European General Data Protection Regulation. ICO ensures compliance with Reference Methodology 4, as required by the French Commission Nationale de l’Informatique et des Libertés, and this project is registered in the National Institutes of Health data’s public directory. Reporting adhered to the ESMO Guidance for Reporting Oncology real-World evidence and the strengthening of the Strengthening the Reporting of Observational Studies in Epidemiology guidelines.11,12
Available data
Electronic health records at the ICO were reviewed to gather baseline and demographic information, including age, Eastern Cooperative Oncology Group (ECOG) performance status, and metastasis sites. Data on medical history were collected, such as de novo metastatic status and prior treatments before first-line SOC, including ET with or without CDK4/6 inhibitors. Post-baseline information included treatment details, therapy response, dates of death, and last follow-up. Data collection continued until the study end date (February 28, 2023), death, or the last recorded contact for patients lost to follow-up.
Tumor subtype assessment and HER2 scoring
HER2 and hormone receptor statuses were assessed locally, using the most recent metastatic tissue sampling results when available, or the latest early-disease sampling if metastatic samples were unavailable. Breast cancer was classified as HR+ if estrogen receptor or progesterone receptor expression was ⩾10%, as determined by IHC and in line with European guidelines. 13 HER2 negativity was defined as IHC scores of 0, 1+, or 2+/ISH−, based on the American Society of Clinical Oncology (ASCO)/College of American Pathologists (CAP) guidelines. 14 HER2-negative cases were further categorized as HER2-low (IHC 1+ or 2+/ISH−) and HER2-zero. For patients with multiple tumor biopsies, a case was classified as HER2-low if at least one tumor specimen met the HER2-low criteria, following the ESMO expert consensus statement. 7 HER2 expression was evaluated on archived biopsy samples using the RxDx HER2 (4B5) VENTANA monoclonal antibody.
To determine the prevalence of HER2-ultralow cases, archived slides from patients originally classified as HER2-zero were rescored by the local breast cancer pathologist expert at each center. Each sample was reviewed at its center of origin to maintain consistency in interpretation. Rescoring adhered to ASCO/CAP guidelines, distinguishing HER2-null tumors (completely devoid of staining in infiltrating cancer cells) from HER2-ultralow tumors (IHC 0 with incomplete and faint staining in ⩽10% of tumor cells).
Outcomes and assessments
The primary endpoint was real-world PFS (rwPFS), as assessed by investigators. rwPFS was defined as the time from the initiation of the first SOC course to either disease progression or death from any cause, whichever occurred first. Secondary endpoints included real-world overall survival (rwOS), defined as the time from the first SOC course to death from any cause, and time to next treatment (TTNT), defined as the time from the first SOC course to initiation of subsequent systemic therapy or death from any cause, whichever occurred first. Patients without an event for these outcomes were censored at the earliest date of either loss to follow-up or the study’s end.
Statistical analysis
Baseline characteristics were described using frequencies and proportions for categorical variables and medians with ranges for continuous variables. Survival curves for rwPFS, rwOS, and TTNT with first-line SOC chemotherapy in the HER2-ultralow, HER2-low, HER2-null, and HER2-ultralow + HER2-low populations were generated using the Kaplan–Meier estimator and compared with the log-rank test.15,16 To assess the relationship between HER2 status and rwPFS, multivariable Cox models were used. 17 The proportional hazards (PH) assumption was graphically checked for each variable, and if violated, the model was stratified by the variable not meeting the PH assumption. PH assumption was also checked post-estimation using the Schoenfeld residuals on the final model. For continuous variables, the log-linearity assumption was tested in univariate analysis, and if the Bayesian information criterion decrease with natural spline transformation compared to using the variable in its natural scale, the variables were categorized. In such cases, cutoffs were selected based on graphical assessment and clinical relevance. In the multivariable models, the following baseline variables were adjusted for based on clinical relevance: patient age, ECOG performance status, presence of visceral metastasis, prior ET use, and previous CDK4/6 inhibitor use. CDK4/6 inhibitor use was treated as a stratification factor in the Cox models due to the violation of the PH assumption. Interaction terms were not considered. Patients with missing data for baseline covariates included in the multivariable models were excluded. In the multivariable model, categorical variables with more than two levels were tested globally using a Wald test, and individual contrasts were provided against the reference category. Data analysis was performed using R version 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Prevalence of the HER2-ultralow subgroup after HER2 rescoring by a pathologist
A total of 320 patients were included, with 198 (61.9%) classified as HER2-low and 122 (38.1%) as HER2-zero (Supplemental Figure 1). After HER2-zero tumors were reviewed by the breast cancer pathologists, the HER2-zero group was reclassified into HER2-ultralow (n = 50) and HER2-null (n = 72). In total, the HER2-ultralow, HER2-low, and HER2-null subtypes represented 15.6%, 61.9%, and 22.5% of the HR+/HER2-negative (IHC scores of 0, 1+, and 2+/ISH−) population, respectively (Figure 1). Overall, HER2 status was determined from metastatic tissue for 275 (85.9%) patients and from the most recent early-disease sample for 45 (14.1%) patients. The proportions of HER2 assessment from metastatic versus primary tissue were as follows: 90% versus 10% in the HER2-ultralow group, 84% versus 16% in the HER2-low group, and 87% versus 13% in the HER2-null group.

HER2 prevalence after rescoring. Each bar represents the proportion of patients based on the changes in HER2 scoring. HER2-negative patients were classified as HER2 0, 1+, or 2+ with ISH negative, HER2-zero as no staining or faint/incomplete membrane staining in 10% of the tumor, HER2-low as 1+ or 2+ ISH negative, HER2-ultralow as faint/incomplete membrane staining in less than 10% of the tumor, and HER2-null as no detectable membrane staining.
Patients’ baseline characteristics and treatments
The median age of the overall population was 62.4 years (range 27–91), with similar age distributions across the HER2-ultralow, HER2-low, and HER2-null groups (Table 1). The majority of patients had an ECOG performance status of 1, while 13 (26%), 43 (21.7%), and 19 (26.4%) patients had an ECOG score of ⩾2 in the HER2-ultralow, HER2-low, and HER2-null groups, respectively. The HER2-ultralow group had numerically a higher proportion of visceral metastases compared to the HER2-low and HER2-null groups. The median number of previous ETs for mBC was 0 (range 0–3) in the HER2-ultralow group, 1 (range 0–4) in the HER2-low group, and 1 (range 0–2) in the HER2-null group. Previous CDK4/6 inhibitor therapy was received by 20 (46%), 86 (43.4%), and 33 (45.8%) patients in the respective groups. Notably, 54%, 46.5%, and 45.8% of patients in the three groups received SOC as first-line therapy for mBC. First-line chemotherapy was primarily taxane monotherapy (64.0%) or anthracycline (23.6%) for HER2-ultralow patients. Further details of the SOC regimen are provided in Table 1.
Baseline characteristics of patients based on HER2 status.
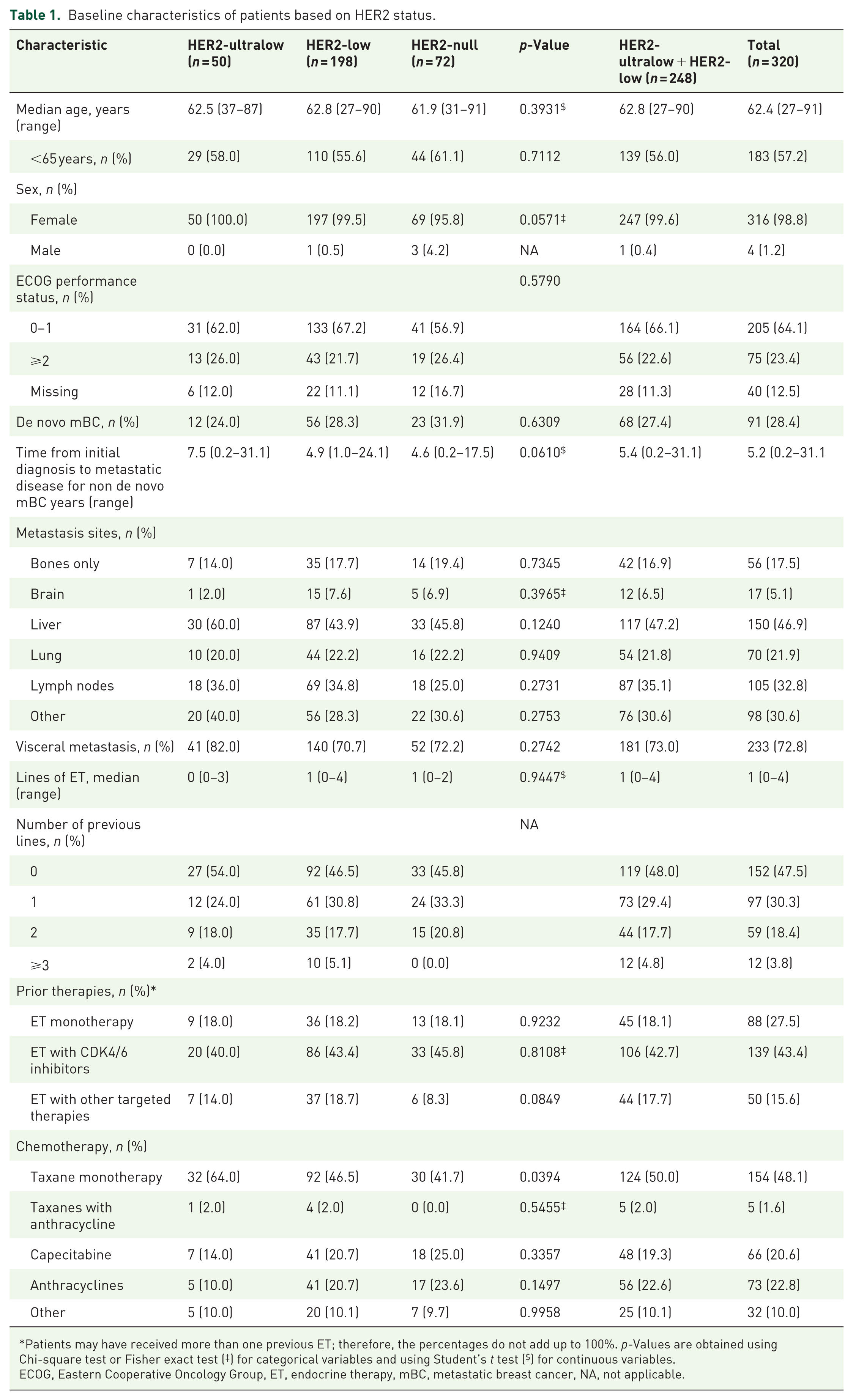
Patients may have received more than one previous ET; therefore, the percentages do not add up to 100%. p-Values are obtained using Chi-square test or Fisher exact test (‡) for categorical variables and using Student’s t test ($) for continuous variables.
ECOG, Eastern Cooperative Oncology Group, ET, endocrine therapy, mBC, metastatic breast cancer, NA, not applicable.
PFS, OS, and TTNT in the whole population and subgroups
The median time of follow-up for the 320 included patients was 39.4 months (95% confidence interval (CI), 37.1–47.6), 39.6 months for the HER2-ultralow group, 38.9 months for the HER2-low group, and 47.6 for the HER2-null group (Figure 2, Table 2). The median rwPFS with first-line chemotherapy was 7.9 months (95% CI, 4.6–19), 7.3 months (95% CI, 6.4–9.1), and 6.2 months (95% CI, 4.6–8.9) in the HER2-ultralow, HER2-low, and HER2-null groups, respectively (Figure 2(a)). The median rwOS was 19.5 months (95% CI, 10.7–33.4), 21.5 months (95% CI, 19.0–25.8), and 16.6 months (95% CI, 11.9–23.6) in the HER2-ultralow, HER2-low, and HER2-null groups, respectively (Figure 2(b)). The median TTNT was 8.7 months (95% CI, 4.8–19.5), 8.2 months (95% CI, 7.0–9.9), and 7.0 months (95% CI, 5.1–10.3) in the HER2-ultralow, HER2-low, and HER2-null groups, respectively (Figure 2(c)). For patients previously treated with CDK4/6 inhibitors, the median rwPFS was numerically lower, at 4.6 months (95% CI, 2.7–21.4), 6.1 months (95% CI, 5.5–7.3), and 5.6 months (95% CI, 3.8–8.7) in the HER2-ultralow, HER2-low, and HER2-null groups, respectively (Table 2).
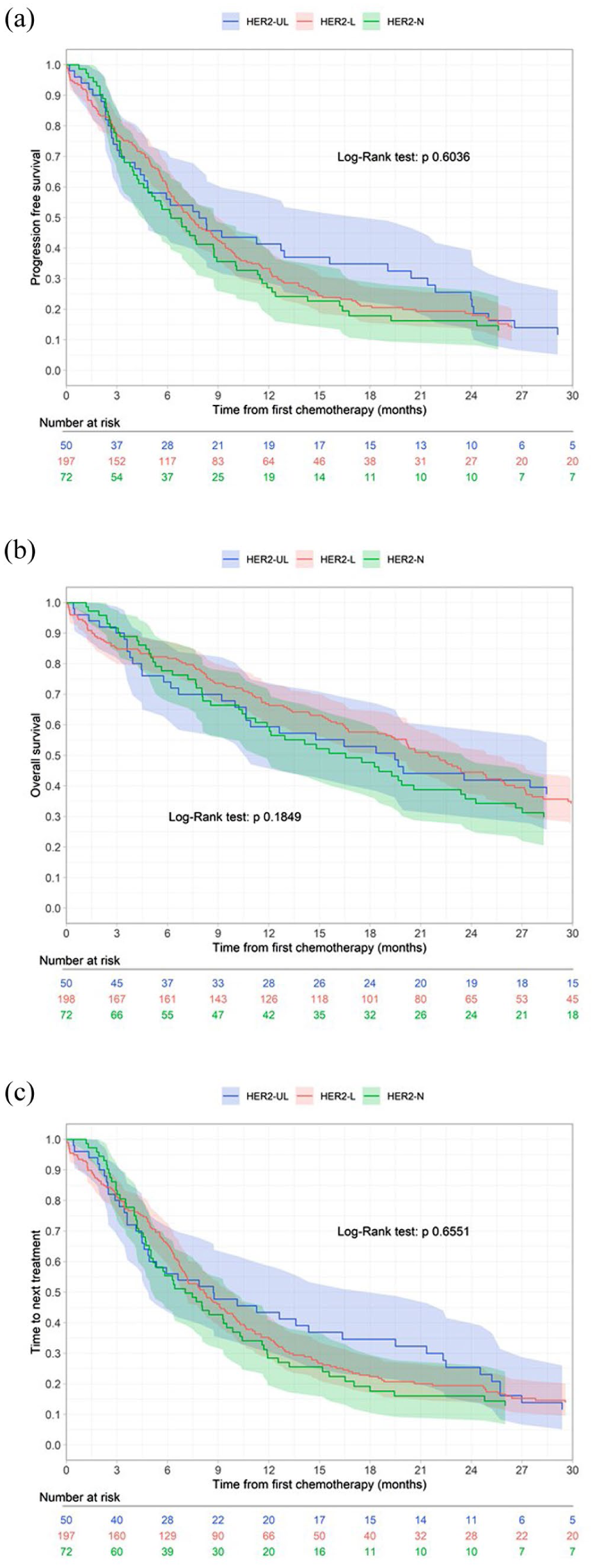
Survival curves of rwPFS (a), rwOS (b), and TTNT (c). The Kaplan–Meier estimator was used to compare survival curves according to HER2 status, with differences assessed by the log-rank test in the HER2-ultralow, HER2-low, and HER2-null groups.
Median progression-free survival with 95% CI by HER2 status across different subgroups.

CI, confidence interval; NA, not applicable.
Multivariable analyses of rwPFS and rwOS in the whole population
The multivariable Cox model assessing the risk of rwPFS and rwOS based on HER2 status in three categories was adjusted for patient age, ECOG performance status, presence of visceral metastasis, prior ET use, and previous CDK4/6 inhibitor use (Table 3). Of note, 40 patients were excluded due to the missing of ECOG performance status variable. HER2-ultralow status was not significantly associated with better rwPFS or rwOS. The hazard ratios (HR) for rwPFS, adjusted for confounders, were 0.97 (95% CI, 0.70–1.33) for HER2-low compared to HER2-null and 0.69 (95% CI, 0.45–1.06) for HER2-ultralow compared to HER2-null. Additional analyses of the HER2-ultralow + HER2-low group (similar to the DESTINY-Breast06 trial population) yielded similar results (Supplemental Tables 1 and 2).
Results of the multivariable analysis assessing the relationship between HER2 status (ultralow, low, null), PFS, and OS.

Forty patients were excluded due to missing data on ECOG performance status.
p-Value for global Wald test of the variable. The model was stratified based on the presence of prior CDK4/6 inhibitor treatment.
CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; ET, endocrine therapy; HR, hazard ratio; OS, overall survival; PFS, progression-free survival.
Discussion
This study offers important insights into the prevalence and outcomes of HER2-ultralow mBC after first-line SOC chemotherapy, aligning with the DESTINY-Breast06 trial setting. In a real-world population, our results show that using the ASCO/CAP guidelines to rescore HER2 status reclassifies 41.0% of HR+/HER2-zero mBC patients as HER2-ultralow. Overall, HER2-ultralow tumors represented 15.6% of HER2-negative patients.
HER2-ultralow is a newly identified subtype, with only a few studies reporting its prevalence. The 15.6% prevalence found in our study aligns with previous estimates. The largest prospective study in the metastatic setting, the DESTINY-Breast06 trial, found that 21.7% (402 out of 1856 HER2 assessments) were classified as HER2-ultralow. 18 In early-stage settings, two large cohorts, with 1363 and 1024 patients, reported slightly higher prevalence rates of HER2-ultralow subtypes, ranging from 24.9% to 29%.10,19 However, a Chinese retrospective study of 246 early-stage breast cancer patients found a much higher HER2-ultralow prevalence of 34.6%. 20 In the metastatic setting, a study conducted at three Mayo Clinic campuses reassessed HER2 status in 300 advanced breast cancer patients. 21 Among the clinically documented HER2-zero tumors, 60% were reclassified as HER2-ultralow by two pathologists. Notably, the interpathologist concordance rate was only 57%, with individual pathologists classifying HER2-ultralow tumors at rates ranging from 43% to 45%. Importantly, patients with HER2-ultralow tumors may become eligible for HER2-targeted therapies. The recent FDA approval of T-DXd for HER2-ultralow mBC in February 2025 highlights the evolving treatment landscape and supports the clinical relevance of precise HER2 scoring.
We found that the median rwPFS with SOC chemotherapy was 7.9 months (95% CI, 4.6–19) for the HR+/HER2-ultralow population and 7.3 months (95% CI, 6.4–9.1) for the HR+/HER2-low population. These results are similar to those reported in the DESTINY-Breast06 trial: 8.3 months (95% CI, 9.8–17.3) and 8.1 months (95% CI, 7.3–9.0) for the HR+/HER2-ultralow and HR+/HER2-low populations, respectively. 8 However, there are some differences between the two cohorts. In our study, 43.4% of patients received prior CDK4/6 inhibitors, compared to 90.4% in the DESTINY-Breast06 trial. Additionally, half of our patients received chemotherapy as first-line therapy for mBC, which is not in line with current standard practices. 11 This reflects the changing treatment landscape, as many patients in our cohort started chemotherapy before CDK4/6 inhibitors were widely approved and integrated into clinical practice.22 –25 These differences in treatment history and sequencing are important to consider, as they may influence clinical outcomes and response patterns. Therefore, direct comparisons with trial populations should be interpreted with caution. Patients who had prior CDK4/6 inhibitor treatment had lower rwPFS in our cohort. This finding is consistent with previous research showing that tumors resistant to CDK4/6 inhibitors often acquire additional genetic and molecular changes, which can reduce sensitivity to subsequent treatments, including chemotherapy. 26 These alterations may involve the activation of alternative growth pathways or changes in the tumor microenvironment, which can make the cancer more aggressive and less responsive to standard chemotherapies. 27 The association between prior CDK4/6 inhibitor use and shorter rwPFS may also reflect potential confounding factors, such as differences in disease burden at treatment initiation, tumor genetic alterations, or patient comorbidities. Another key difference is the inclusion of patients with bone-only disease, which accounted for 17.5% of the patients in our study but were not represented from the DESTINY-Breast06 trial. The optimal first-line SOC chemotherapy remains unclear, although capecitabine is often used due to its favorable efficacy-toxicity profile.
The efficacy of T-DXd in HER2-ultralow tumors supports the bystander effect as a key mechanism of action for this new ADC. This effect enables the cytotoxic payload of T-DXd to spread to adjacent tumor cells, even those without HER2 expression. 28 HER2-ultralow tumors, which exhibit faint or incomplete HER2 staining, may still express enough HER2 to facilitate ADC internalization, thereby enhancing the cytotoxic effect through the bystander effect. 29 Therefore, HER2 assessment is crucial for determining its therapeutic impact and must be carried out by a trained pathologist who understands the significance of accurate HER2 scoring, especially in the HER2-ultralow population. Notably, a real-world pathologist evaluation revealed high concordance for HER2-positive scoring but a high discordance rate for HER2-low and HER2-zero assessments. 30 In the DESTINY-Breast06 trial, the concordance rate for HER2-low status between local and central assessments was 77.8%, but 64% of tumors with a local HER2-zero IHC score were reclassified as HER2-low (24%) or HER2-ultralow (40%) in central assessments. 18 Our study highlights the importance of thorough and precise HER2 testing, including the reassessment of HER2 status in HER2-zero tumors. A single biopsy showing HER2-ultralow status could support the use of T-DXd in the future, potentially improving outcomes for this diverse patient population. In this context, the ASCO/CAP recommendations were updated in 2023 to reflect the emergence of HER2-targeted ADCs, highlighting the need for harmonization in HER2 scoring practice. 31 Advances in digital pathology, particularly artificial intelligence (AI)-based algorithms, may enhance the accuracy of HER2 scoring, reducing variability and improving patient selection for targeted therapies. 32 Two studies have shown that AI-assisted pathology significantly improved the accuracy and consistency in distinguishing HER2-zero from HER2-low cases.33,34 In both studies, pathologists initially assessed cases without AI assistance and then reviewed the cases with AI support. The use of AI increased concordance between pathologist assessments, particularly in cases with heterogeneity.
The outcomes based on HER2 expression levels were largely consistent in our population. The question of whether HER2-low and HER2-ultralow should be regarded as distinct clinical entities with unique molecular, clinicopathologic characteristics, and prognosis, or merely serve as biomarkers for HER2-targeted ADCs, remains unresolved. 35 To date, only two retrospective studies have evaluated outcomes in HER2-ultralow breast cancer patients. In a study of 1363 early breast cancer cases at Qingdao University, HER2-low and HER2-ultralow expression did not significantly affect disease-free survival (DFS), despite differences in clinicopathologic features. 10 Similarly, in a study of 1024 surgical patients, HER2-ultralow showed a nonsignificant trend toward better OS and DFS compared to HER2-low, with noted clinicopathologic differences. 19 The prognostic impact of low HER2 expression in breast cancer is still being studied, with current evidence showing mixed results. Some studies suggest that HER2-low expression does not significantly affect DFS or OS compared to HER2-negative cases under standard treatments, although differences in clinicopathologic features, such as HR status, are often noted between HER2-low and HER2-negative groups.36 –38 However, a large study from the National Cancer Database (24,636 patients) found significantly improved 3-year OS in HER2-low versus HER2-negative HR+ patients (60.9% vs 55.6%). 39 Similarly, a smaller study of 391 patients reported higher OS in HER2-low cases. 40
Although HER2-low and HER2-ultralow expression have not been definitively linked to a better or worse prognosis, they represent distinct subgroups with potential therapeutic implications. Current clinical trials are using these classifications as inclusion criteria. The
Our study complements the findings of a controlled clinical trial in an emerging population and highlights an unmet medical need. However, several limitations should be considered. First, the retrospective nature of our analysis may introduce bias, despite our careful methodological approach. This study was conducted at a single center, which may limit the generalizability of our findings to broader patient populations. Moreover, the relatively small size of the HER2-ultralow subgroup especially in stratified analyses reduces the statistical power to detect meaningful differences. Consequently, several estimates are associated with wide CIs, reflecting the inherent limitations in precision due to the smaller cohort size. Variability in HER2 testing and rescoring methods, even when adhering to established guidelines, could also lead to potential misclassification. The absence of a central pathology review may have contributed to inconsistencies in HER2 scoring across institutions. The lack of information on underlying genomic alterations (e.g., ESR1 mutations, PIK3CA alterations) prevents exploration of whether HER2-ultralow status is associated with distinct molecular profiles or resistance pathways. However, the breast cancer pathologists who reviewed the slides were aware of the possible therapeutic implications of HER2-ultralow status.
Conclusion
In conclusion, our findings support the view that HER2-ultralow breast cancer is a common subset within HR+/HER2-negative mBC. Given the demonstrated efficacy of HER2-targeted ADCs in this group, routine rescoring of HER2 status in HER2-negative patients may become increasingly important for guiding personalized treatment strategies.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251378863 – Supplemental material for Real-world analysis of HER2-ultralow in HR+/HER2– metastatic breast cancer: prevalence and first-line chemotherapy outcomes
Supplemental material, sj-docx-1-tam-10.1177_17588359251378863 for Real-world analysis of HER2-ultralow in HR+/HER2– metastatic breast cancer: prevalence and first-line chemotherapy outcomes by Laurent Mathiot, Olivier Kerdraon, Florent Le Borgne, Véronique Verriele, Anne Patsouris, Marie Robert, Jérôme Chetritt, Delphine Loussouarn, Mario Campone, François Bocquet and Jean-Sébastien Frenel in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
