Abstract
Background:
Prostate-specific antigen (PSA) is widely used in the diagnosis and monitoring of prostate cancer. The prognostic relevance of very low PSA levels has not been clearly established in metastatic castration-sensitive prostate cancer (mCSPC). More sensitive PSA assays may provide more accurate estimates of clinical outcomes.
Objectives:
To evaluate the relationship between achieving very deep PSA levels (⩽0.02 ng/mL) within 6 months of treatment and radiologic progression-free survival (rPFS) in patients with mCSPC.
Design:
A retrospective, unicenter observational study conducted at a tertiary oncology center.
Methods:
A total of 203 patients with mCSPC who received first-line treatment with luteinizing hormone-releasing hormone analogs, androgen receptor pathway inhibitors, or docetaxel were included. Patients were stratified into four groups based on their PSA nadir levels: ⩽0.02, 0.02–0.2, 0.2–4, and >4 ng/mL. Kaplan–Meier and Cox regression analyses were used to assess the association between PSA nadir and rPFS.
Results:
Patients achieving PSA ⩽ 0.02 ng/mL had significantly longer rPFS (median: 59.2 months) compared to other PSA groups. Multivariable analysis confirmed PSA ⩽ 0.02 ng/mL as an independent predictor of rPFS (HR: 2.02, p < 0.001). The overall log-rank p-value for group comparison was < 0.001.
Conclusion:
A very deep PSA response (⩽0.02 ng/mL) is associated with longer rPFS and may serve as a prognostic marker in mCSPC. This threshold could be considered in future clinical trial design and treatment stratification.
Background
Prostate cancer is the second most common cancer type among men worldwide and ranks as the fourth most common cancer type overall. Although a small proportion of patients are diagnosed with metastatic prostate cancer, the large number of patients leads to a significant overall burden. Metastatic castration-sensitive prostate cancer (mCSPC) is a heterogeneous disease that can rapidly progress to castration-resistant prostate cancer (CRPC).1 –3
Prostate-specific antigen (PSA) was first identified in human prostate tissue extracts in 1970 and later detected in serum in 1980.4,5 In 1986, it received FDA approval for use in the monitoring of patients with prostate cancer. 6 PSA is a 33 kDa glycoprotein from the serine protease family, also known as human kallikrein 3 (hK3), encoded on the q arm of chromosome 19 (19q13.4). The structure of PSA consists of 237 amino acids, 5 disulfide bonds, and a carbohydrate component in the form of an N-linked oligosaccharide chain. 7 PSA measurement is utilized for screening, diagnosis, and monitoring treatment in prostate cancer. 8 Previous studies have reported that in patients with mCSPC, a rapid decline in PSA levels and the time to reach nadir PSA are associated with better outcomes when combined with androgen deprivation therapy (ADT) and treatments such as docetaxel, abiraterone, or enzalutamide.9,10
PSA decline has consistently been identified as a prognostic factor in patients with mCSPC receiving combination therapies, including ADT with docetaxel, abiraterone, enzalutamide, apalutamide, or even triplet regimens. However, the prognostic utility of extremely low PSA thresholds has not been extensively investigated across these treatment settings.
Previous prospective trials, including SWOG 9346 and CHAARTED, have consistently demonstrated that early PSA dynamics—such as rapid declines, time to nadir, and achievement of undetectable levels—are strongly associated with overall and progression-free survival. In particular, achieving a PSA nadir of <0.2 ng/mL within 7 months of initiating ADT has been identified as a robust, independent prognostic factor.9,10 Nonetheless, the prognostic value of more sensitive PSA thresholds, such as ⩽0.02 ng/mL, remains insufficiently validated, especially in the era of androgen receptor pathway inhibitors (ARPIs) and contemporary multimodal treatment strategies.
According to previous reports, the nadir PSA level for mCSPC has been defined as 0.2 ng/mL. However, the availability of more sensitive assays that can measure lower PSA levels may correlate with more accurate outcomes in patients with prostate cancer. In this study, we evaluated the relationship between very deep PSA levels (⩽0.02 ng/mL) and radiographic progression-free survival (rPFS) in patients treated for mCSPC.
Methods
This retrospective, single-center study was conducted at Bakırköy Dr. Sadi Konuk Training and Research Hospital. We included patients with newly diagnosed mCSPC who initiated systemic treatment between January 2019 and January 2025. A total of 245 patient records were reviewed. A total of 203 patients diagnosed with mCSPC and receiving first-line treatment (ADT, ADT plus enzalutamide or abiraterone, ADT plus docetaxel) were included in the study. Patients who received treatment for less than 3 months or had inadequate PSA and imaging follow-up were excluded. PSA measurements were conducted at a single institutional laboratory using a consistent assay methodology throughout the study period. PSA nadir was evaluated at the 6-month mark, in accordance with institutional follow-up protocols (at 1, 3, and 6 months), and supported by prior literature identifying this time point as prognostically significant. rPFS was assessed according to the Prostate Cancer Working Group 3 (PCWG3) criteria, based on imaging with either whole-body CT and bone scintigraphy or Prostate-Spesific Membrane Antigen Positron Emission Tomography (PSMA PET/CT), depending on clinical availability and physician discretion. Where appropriate, RECIP 1.0 criteria were also applied to evaluate radiographic progression. Demographic data, risk factors, pathological and molecular variables, and follow-up outcomes were analyzed descriptively across the entire cohort. Mean and standard deviation values were provided for continuous variables with a parametric distribution, while median and interquartile range (IQR = Q1 and Q3) values were given for non-parametric continuous variables. Categorical variables were expressed as percentages. The chi-square test was used for comparisons between groups. Overall survival (OS) and rPFS were calculated using the Kaplan–Meier method, and survival differences were assessed using the log-rank test. Cox proportional hazards analysis with a 95% confidence interval (CI) was used to estimate significance levels and relative risks. Median rPFS and OS were presented with two-sided 95% CIs. A p-value of < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS (Version 25, SPSS Inc., Chicago, IL, USA). Ethical approval for the study was obtained from the Ethics Committee of Bakırköy Dr. Sadi Konuk Training and Research Hospital with the decision number 2024-244. Verbal informed consent was obtained from the participants at the beginning of the treatment to access their medical records. The reporting of this study conforms to the STROBE statement, and the completed checklist is provided as a Supplemental File.
Results
Data from 245 patients were analyzed. A total of 30 patients were excluded due to insufficient data, and 12 patients were excluded due to inadequate follow-up, leaving 203 patients in the study. The median age was 71 years (38–91), and 50.2% of the patients had an Eastern Cooperative Oncology Group performance status (ECOG PS) score of 1 or higher. Of the patients, 51.2% had high-volume disease, and 70% presented with synchronous metastasis. In addition, 82.8% of the patients had bone metastases, 13.3% had lung metastases, and 4.4% had liver metastases. In the first-line treatment, 43.3% of the patients received ARPIs, 36.5% were treated with ADT alone, and 20.2% received docetaxel. When divided into four subgroups based on their lowest PSA levels, the distribution was as follows: PSA ⩽ 0.02 ng/mL, 31% (n = 63); PSA 0.02–0.2 ng/mL, 13.8% (n = 28); PSA 0.2–4 ng/mL, 33.5% (n = 68); PSA > 4 ng/mL, 21.7% (n = 44). Detailed characteristics of the patients are presented in Table 1.
Baseline characteristics.
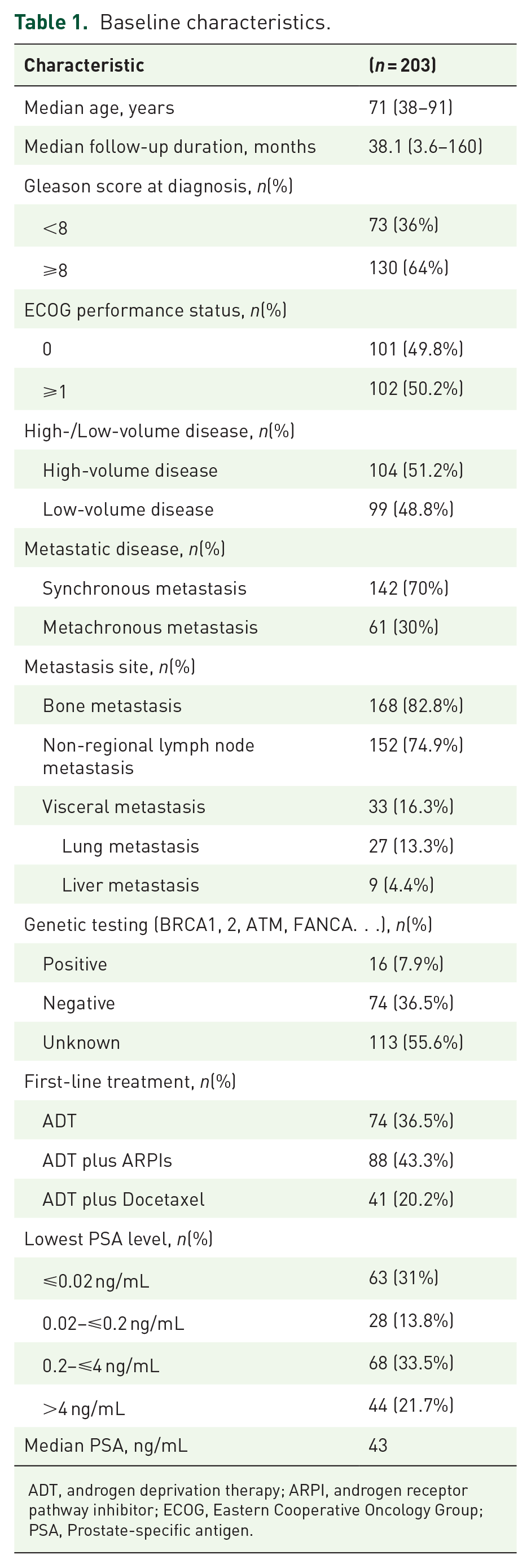
ADT, androgen deprivation therapy; ARPI, androgen receptor pathway inhibitor; ECOG, Eastern Cooperative Oncology Group; PSA, Prostate-specific antigen.
To further explore this finding, baseline patient characteristics were stratified according to the four PSA nadir subgroups and are presented in Supplemental Table 1. While overall distributions appeared generally balanced, patients with PSA ⩽ 0.02 ng/mL were more likely to have low-volume disease, whereas those with PSA > 4 ng/mL predominantly had high-volume disease. Modest differences were also observed in first-line treatment choices: ARPIs were more frequently used in patients achieving lower PSA nadirs, while docetaxel was more commonly administered in those with higher nadirs. Other characteristics, such as metastatic pattern and site, were relatively evenly distributed across the subgroups.
The median follow-up duration was 38.1 months (range: 3.6–160 months). The median rPFS for the entire cohort was 30.8 months (95% CI: 25.3–36.4 months). In the PSA ⩽ 0.02 ng/mL group, the median rPFS was 59.2 months (95% CI: 42.5–72.8 months); in the PSA 0.02–0.2 ng/mL group, it was 31.4 months (95% CI: 22.8–40 months); in the PSA 0.2–4 ng/mL group, it was 23.5 months (95% CI: 19.4–27.6 months); and in the PSA > 4 ng/mL group, it was 17 months (95% CI: 12.5–21.6 months). The relationship between very deep PSA levels and progression-free survival (PFS) was found to be statistically significant (p < 0.01). The 2-year rPFS rates were 81%, 60.7%, 39.7%, and 11.4% for the respective PSA groups (p < 0.01) (Figure 1).
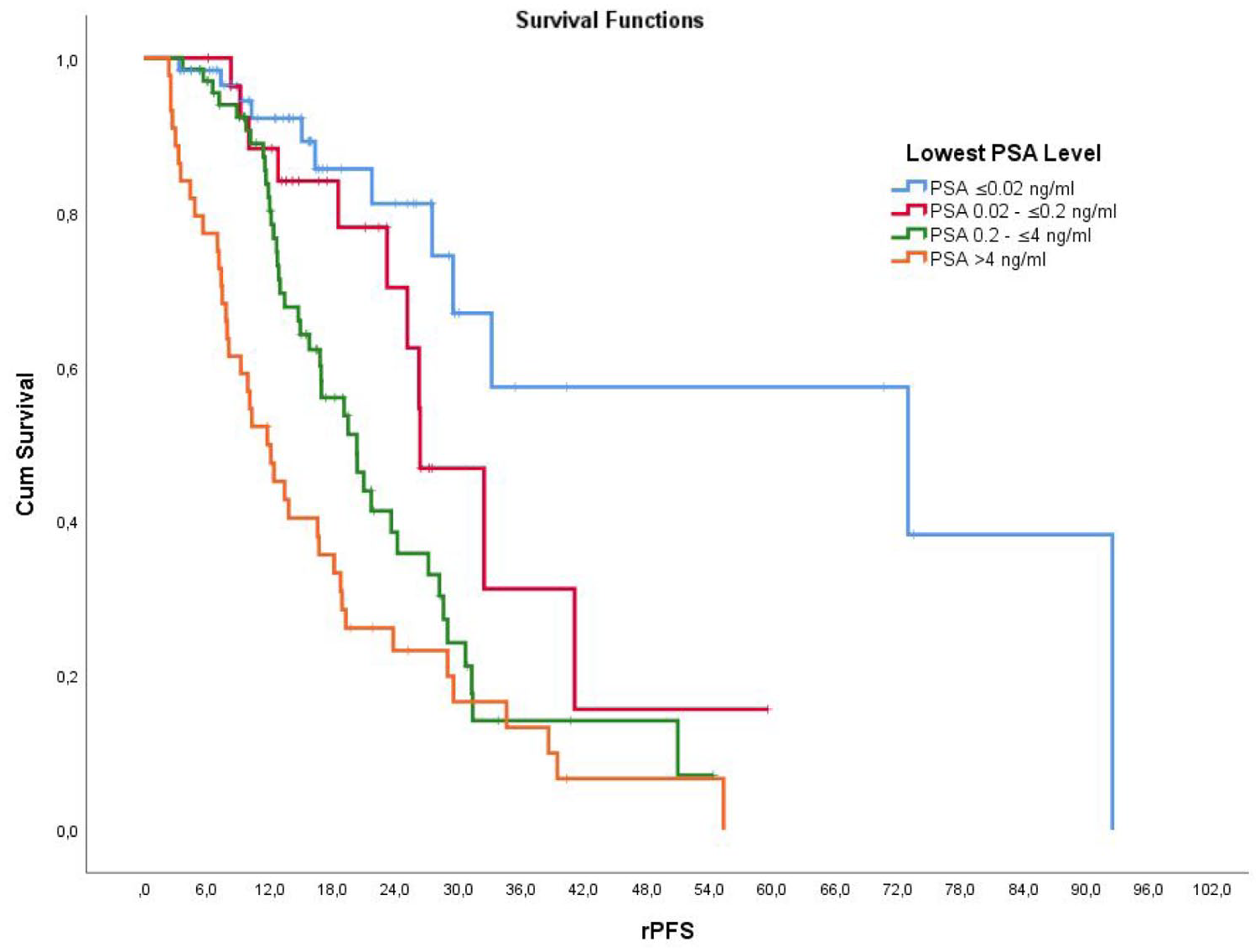
The relationship between the lowest PSA level achieved within 6 months after treatment and rPFS.
In the multivariable Cox regression analysis assessing rPFS, a very low PSA nadir (⩽0.02 ng/mL) remained an independent prognostic factor. Patients with PSA ⩽ 0.02 ng/mL had significantly longer rPFS compared to those with higher PSA nadirs (HR: 2.02; p < 0.001). Additional independent predictors of poorer rPFS included low hemoglobin (<12 g/dL; HR: 0.49, p = 0.004) and elevated lactate dehydrogenase (LDH) levels (HR: 1.85, p = 0.044). The prognostic significance of PSA nadir persisted after adjusting for other clinicopathological variables, including disease volume (high vs low), metastatic timing (synchronous vs metachronous), ALP levels, and type of systemic therapy (Table 2).
Multivariable Cox regression analysis evaluating rPFS.

ALP, alkaline phospatase; LDH, lactate dehydrogenase; ULN, upper limit of normal.
ADT alone was used as the reference group in the multivariable analysis.
Bold values indicate statistical significance (p<0.05).
ADT, androgen deprivation therapy; ARPI, androgen receptor pathway inhibitor; PSA, Prostate-specific antigen; rPFS, radiographic progression-free survival.
Furthermore, Kaplan–Meier survival analysis comparing the four PSA nadir groups (⩽0.02, 0.02–0.2, 0.2–4, and > 4 ng/mL) demonstrated a statistically significant difference in rPFS across the groups (log-rank p < 0.001). Notably, patients with PSA ⩽ 0.02 ng/mL had significantly longer rPFS compared to those in the 0.02–0.2 ng/mL group (log-rank p = 0.049) (Supplemental Figure 1).
Discussion
In our study, we aimed to evaluate the relationship between the lowest PSA level achieved within the first 6 months of treatment and rPFS in patients with metastatic hormone-sensitive prostate cancer (mHSPC). Unlike other studies comparing the relationship between nadir PSA levels and rPFS or OS, we specifically analyzed patients who reached a PSA level of ⩽0.02 ng/mL. We found that radiographic PFS in this patient group was longer than in the group with PSA levels between 0.02 and 0.2 ng/mL, as well as the other PSA subgroups. This difference was also statistically significant (p < 0.01).
In the CHAARTED study, a PSA level of <0.2 ng/mL at month 7 in patients receiving ADT was identified as a prognostic marker independent of the addition of docetaxel to ADT. 10 In the ARASENS study, an analysis published in March 2024 examining the relationship between nadir PSA levels and outcomes in patients with mHSPC receiving darolutamide (n = 651) found that achieving a PSA level of ⩽0.2 ng/mL within 24 weeks was associated with OS (HR 0.49 (95% CI 0.37–0.65)). 11 Similarly, in the SWOG 9346 study, patients were divided into three subgroups based on the lowest PSA levels achieved within 7 months of ADT treatment, and a significant difference in survival was observed between the groups. Median survival was 75 months in the PSA < 0.2 ng/mL group, 44 months in the PSA 0.2–4 ng/mL group, and 13 months in the PSA > 4 ng/mL group. 9 Another study involving mHSPC patients receiving apalutamide treatment also showed a significant association between a PSA level below 0.2 ng/mL and improved OS, better rPFS, and a longer time to progression to CRPC. 12 Similarly, in our study, we found that a PSA level below 0.2 ng/mL was associated with longer rPFS (p < 0.01).
As a result, achieving a PSA level of ⩽0.02 ng/mL within the first 6 months of treatment in mHSPC patients was associated with a longer rPFS compared to both the PSA 0.02–0.2 ng/mL group and the other PSA subgroups. The 2-year rPFS rate was calculated to be 81% in the group with PSA < 0.02 ng/mL, compared to 60.7% in the PSA 0.02–0.2 ng/mL group. This suggests that a PSA level of ⩽0.02 ng/mL may be a more favorable primary endpoint for achieving longer PFS.
Our findings demonstrate that a very low PSA nadir (⩽0.02 ng/mL) is not only prognostic in univariate analysis but also retains its independent prognostic value in multivariable Cox regression models, even after adjusting for established clinical factors such as disease volume, metastatic site and timing, hemoglobin, LDH, ALP levels, and type of treatment. This highlights the robustness of PSA nadir as a clinically meaningful biomarker. Although Kaplan–Meier curves for PSA ⩽0.02 and ⩽0.2 ng/mL may appear visually similar, only the very low threshold remained statistically significant in multivariable analysis. Notably, patients with PSA ⩽ 0.02 ng/mL had significantly longer rPFS compared to those in the 0.02–0.2 ng/mL group (log-rank p = 0.049). These findings suggest that achieving deeper PSA responses may provide more refined risk stratification and guide therapeutic decision-making in mCSPC.
PSA is a gene regulated by the androgen receptor (AR), and its expression directly reflects AR signaling activity in prostate cancer cells. Therefore, the extent of PSA decline following systemic therapy may reflect the tumor’s biological sensitivity to AR pathway inhibition. A very low PSA nadir may function as a biomarker of effective AR suppression, rather than merely serving as an indicator of tumor burden.13,14 This biological rationale supports the incorporation of PSA kinetics into early treatment assessment.
Although rPFS is commonly used in mCSPC clinical trials, it remains an incompletely validated surrogate for OS. Nonetheless, large-scale studies such as SWOG 9346, CHAARTED, and LATITUDE have consistently demonstrated that achieving a PSA nadir of <0.2 ng/mL within 6–7 months of treatment initiation is associated with improved OS.8,15,16 Our findings are consistent with these observations and further suggest that reaching a very low PSA nadir (⩽0.02 ng/mL) may provide additional prognostic value, although prospective validation is needed.
If validated in future studies, very deep PSA responses could inform adaptive treatment strategies in mCSPC. Patients who achieve early and profound PSA suppression may be suitable candidates for treatment de-escalation to reduce therapy-related toxicity, while those with suboptimal responses might benefit from earlier treatment intensification. Integrating PSA kinetics with clinical, radiographic, and genomic markers may support a more personalized approach to treatment selection. 17
The clinical benefit of local or metastasis-directed radiotherapy (RT) in patients with synchronous or oligometastatic mCSPC is increasingly recognized. Trials such as PEACE-1 have demonstrated improved outcomes with prostate RT in de novo mCSPC, 18 while STOMP and ORIOLE have confirmed the value of stereotactic ablative radiotherapy (SABR) in oligometastatic disease. 19 In addition, the RADIOSA, 20 EXTEND, 21 SATURN, 22 and SOLAR 23 trials have further supported the use of RT or metastasis-directed therapy (MDT) in selected patient subgroups. In our cohort, the impact of RT or MDT was not specifically assessed due to the retrospective nature of the study and limited sample size; however, future research exploring PSA kinetics in the context of local therapies may offer additional insights.
Our study has several limitations. First, its retrospective, single-center design and limited sample size may restrict the generalizability of the findings. Second, not all patients received current standard-of-care treatments, such as ARPIs or triplet regimens. Third, variability in imaging modalities (PSMA PET vs conventional scans) may have influenced the detection and timing of progression, potentially affecting rPFS estimates. Lastly, although rPFS is a widely accepted endpoint in mCSPC studies, it remains an unvalidated surrogate for OS. Therefore, our results should be interpreted as hypothesis-generating and require confirmation in prospective trials.
Conclusion
Very deep PSA level (PSA ⩽ 0.02 ng/mL) is associated with longer rPFS in patients with mCSPC. This very deep PSA level may serve as a more effective marker for predicting longer rPFS and could be considered as a new primary endpoint for future clinical trials.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251369037 – Supplemental material for Very deep prostate-specific antigen decline is associated with longer rPFS in patients with metastatic castration-sensitive prostate cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359251369037 for Very deep prostate-specific antigen decline is associated with longer rPFS in patients with metastatic castration-sensitive prostate cancer by Caner Kapar, Ilkay Gulturk, Volkan Tugcu, Selçuk Sahin, Haydar Guler, Saadettin Kilickap and Deniz Tural in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251369037 – Supplemental material for Very deep prostate-specific antigen decline is associated with longer rPFS in patients with metastatic castration-sensitive prostate cancer
Supplemental material, sj-docx-2-tam-10.1177_17588359251369037 for Very deep prostate-specific antigen decline is associated with longer rPFS in patients with metastatic castration-sensitive prostate cancer by Caner Kapar, Ilkay Gulturk, Volkan Tugcu, Selçuk Sahin, Haydar Guler, Saadettin Kilickap and Deniz Tural in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-3-tam-10.1177_17588359251369037 – Supplemental material for Very deep prostate-specific antigen decline is associated with longer rPFS in patients with metastatic castration-sensitive prostate cancer
Supplemental material, sj-jpg-3-tam-10.1177_17588359251369037 for Very deep prostate-specific antigen decline is associated with longer rPFS in patients with metastatic castration-sensitive prostate cancer by Caner Kapar, Ilkay Gulturk, Volkan Tugcu, Selçuk Sahin, Haydar Guler, Saadettin Kilickap and Deniz Tural in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
