Abstract
Background:
Haematological toxicities (anaemia, neutropenia and thrombocytopenia), a known class effect of poly-ADP ribose polymerase inhibitors (PARPi) may limit exposure to PARPi and therefore impact efficacy.
Objective:
This study aimed to provide practical and detailed recommendations for the prevention and management of these toxicities in metastatic prostate cancer in the real world.
Design:
A national consensus study was performed using a modified Delphi methodology.
Methods:
A multidisciplinary steering committee of 9 French experts formulated and submitted 38 statements to the vote of 33 French healthcare professionals experienced in oncology and PARPi in prostate and/or breast/ovarian cancers.
Results:
All recommendations achieved a consensus. Before initiating PARPi, haematological disorders should be investigated and appropriate corrective measures implemented. The haemoglobin level should ideally be ⩾10 g/dl and a minimum delay of 4 weeks should be respected after chemotherapy. Monitoring should be frequent for the first 3 months of treatment, at least every 15 days, and even more frequent in patients at high-risk of toxicity. In the event of symptomatic grade 2 or 3 anaemia, grade 2 or 3 thrombocytopenia and grade 3 neutropenia, PARPi treatment should be discontinued until return to grade 1. Transfusion may be considered in symptomatic grade 2 or 3 anaemia. Myelodysplastic syndrome/acute myeloid leukaemia should be suspected in cases of cytopenia persisting beyond 4 weeks or changes in the blood count after maintenance of an optimal therapeutic dose over the long term.
Conclusion:
These proposals complement existing recommendations to guide healthcare professionals in real-world practice, and so optimise metastatic prostate cancer patient’s ability to maximally benefit from PARPi.
Introduction
In Europe, prostate cancer accounts for 20% of male cancers and is the third leading cause of cancer deaths in men. 1 While most prostate cancers are diagnosed at a very early stage and can be successfully managed by active surveillance, radical prostatectomy or radiotherapy, some will progress to metastatic, hormone-sensitive or castration-resistant prostate cancer (mCRPC), associated with an unfavourable prognosis. 2 The recent development of new therapies and precision medicine supports improved control of tumour progression and improved survival in advanced metastatic cancer patients. 3 These advances include poly-ADP ribose polymerase inhibitors (PARPi) – PARP being a family of proteins involved in the repair of DNA single-strand breaks by homologous recombination.3,4
Initially developed for breast/ovarian cancers, PARPi are now used in mCRPC as monotherapy or in combination with androgen receptor pathway inhibitors (ARPI), including abiraterone and enzalutamide.5,6 Indeed, two phase III studies have demonstrated a significant benefit in radiographic progression-free survival (rPFS) for rucaparib, and in overall survival (OS) for olaparib as monotherapy in patients with alterations in homologous recombination repair (HRR) genes. Moreover, in combination, two phase III studies have shown a benefit in rPFS (olaparib + abiraterone plus corticosteroids), and in OS (talazoparib + enzalutamide) in mCRPC patients in the general population, and one phase III study demonstrated a benefit in rPFS in HRR populations (niraparib + abiraterone plus corticosteroids).7 –12 These promising data led to approvals for olaparib, niraparib, rucaparib and talazoparib, either as monotherapy or in combination with ARPI, in patients with mCRPC with or without alterations in HRR genes, depending on the country.13 –16
Experience with PARPi in other cancers has demonstrated a relationship between exposure and efficacy, confirmed in mCRPC, underlining the importance of maintaining an optimal dosage.17 –19 However, the phase III studies have shown that PARPi induce haematological adverse effects, mainly anaemia (grade ⩾3: 16.3%–46.0%), neutropenia (grade ⩾3: 5.0%–18.0%) and thrombocytopenia (grade ⩾3: 0.3%–7.0%), which frequently lead to dose adjustment, interruption or discontinuation of treatment.7 –9,11,12 These observations have been supported by systematic reviews and meta-analyses confirming a class effect of PARPi.20 –22 It is therefore crucial to define appropriate monitoring and management to prevent unnecessary dose reductions, temporary or permanent discontinuations and unnecessary blood transfusions. However, in the Summaries of Product Characteristics (SmPCs) of the available PARPi and in published recommendations, the management of these haematological toxicities is not always detailed, particularly regarding prevention, nor is it specific to prostate cancer.13 –15,23 –25
In this context, a multidisciplinary group of French experts in prostate cancer and/or treatment with PARPi used a modified Delphi methodology to define more precise and standardised practical recommendations to help healthcare professionals (HCPs) better prevent and manage PARPi-associated haematological toxicities in patients with mCRPC in real-life practice to support treatment continuation.
Methodology
The Delphi method is a formalised consensus approach by an iterative multi-step process.26,27 Commonly used in healthcare, including oncology, it is based on four principles: anonymity, iteration, controlled feedback and statistical aggregation of responses.28,29 The key steps include the drafting of statements by a steering committee (SC), anonymous and iterative scoring by a panel of experts (voting group), feedback based on previous responses and finalisation of recommendations.27,28 Our modified Delphi-type national consensus, in line with French and international recommendations, took place from July to November 2024.
The SC, comprising nine experts (five medical oncologists, one onco-haematologist, one urologist, one pharmacist and one advanced practice nurse) with a strong commitment to, and experience in, prostate cancer and/or PARPi use, formulated statements on the prevention and management of haematological toxicities associated with PARPi in the treatment of mCRPC based on the literature and their practical experience.
These statements were submitted to the vote of experts selected according to several criteria: speciality (oncologists, onco-haematologists, urologists, haematologists), with strong expertise in oncology and experience in PARPi use in prostate or breast/ovarian cancers. HCPs from across France were invited to participate to ensure representativity.
In accordance with the HAS (French health authority) recommendations, the experts expressed their level of agreement with each statement via an online questionnaire, using a nine-point Likert scale, ranging from 1 (‘Strongly disagree’) to 9 (‘Strongly agree’). In the first round only, experts could explain their vote with a comment. A STRONG consensus was reached if more than 75% of the votes were ⩾7 and the median ⩾8. If only one of these criteria was met, the statement achieved a GOOD consensus. The process involved two voting rounds. After the first voting round, the SC reviewed the results and comments (Figure 1).
- Proposals that reached a consensus within the group were retained;
- those where divergences had been identified were revised based on the experts’ comments and submitted to a second voting round.
The participants remained anonymous throughout the process and had no interaction with the SC, whose members did not take part in the vote in accordance with the methodological recommendations in force. 28

Delphi study methodology.
All personal data collected as part of this programme were dissociated from the results and anonymised, in accordance with French data protection law (General Data Protection Regulation).
Results
Characteristics of respondents
A total of 33 HCPs took part in the consensus, including 25 medical oncologists, 5 urologists, 1 haematologist, 1 general practitioner working in a cancer centre and 1 radiation oncologist. Their profiles are detailed in Table 1.
Characteristics of voting experts.

aFour urologists reported treating between 2 and 15 metastatic prostate cancer patients per year with PARPi. One urologist was a member of a working group focused on oncology management involving PARPi, and participated as a speaker/trainer at scientific events dedicated to PARPi-related topics.
bOne general practitioner working in a cancer centre and 1 radiation oncologist treating 8 metastatic prostate cancer patient per year.
PARPi, poly-ADP ribose polymerase inhibitors.
Voting experts had a median age of 43 years (Q1 = 37, Q3 = 48) and had been practising for a median of 11 years (Q1 = 7, Q3 = 18), mostly in hospitals (91%). Nearly two-thirds (64%) had expertise in prostate cancer alone, 6% in breast/ovarian cancers alone, and 24% in both.
In addition to their clinical experience, 85% of voting experts also reported activities related to oncology management with PARPi, such as participating in clinical trials, research projects, working groups or scientific events as speakers or trainers, and authoring abstracts or publications.
Statements
Voting experts were invited to give their opinion on 38 statements formulated by the SC. Ten of these concerned the prevention of haematological toxicities associated with PARPi, with recommendations for initiation (pre-therapy assessment, what to do in the event of pre-existing haematological abnormalities or specific situations such as previous chemotherapy or moderate-to-severe renal failure); frequency and modalities of haematological monitoring, prophylactic measures and multidisciplinary coordination of care (Table 2). Twenty-seven statements concerned the management of haematological toxicities: biological assessment and action to be taken (Tables 3 and 4):
- grade 2 anaemia (haemoglobin 8–10 g/dl): 6 statements
- grade 3 anaemia (haemoglobin <8 g/dl): 7 statements
- grade 2 neutropenia (1000–1500 ANC/µl): 3 statements
- grade 3 neutropenia (<1000 ANC/µl): 5 statements
grade 2 thrombocytopenia (50,000–75,000 platelets/µl): 3 statements
- grade 3 thrombocytopenia (<50,000 platelets/µl): 3 statements.
The last statement addressed situations that should suggest a risk of myelodysplastic syndrome (MDS) or acute myeloid leukaemia (AML; Table 4).
Statements relating to the prevention of haematological toxicities with PARPi treatment in prostate cancer.
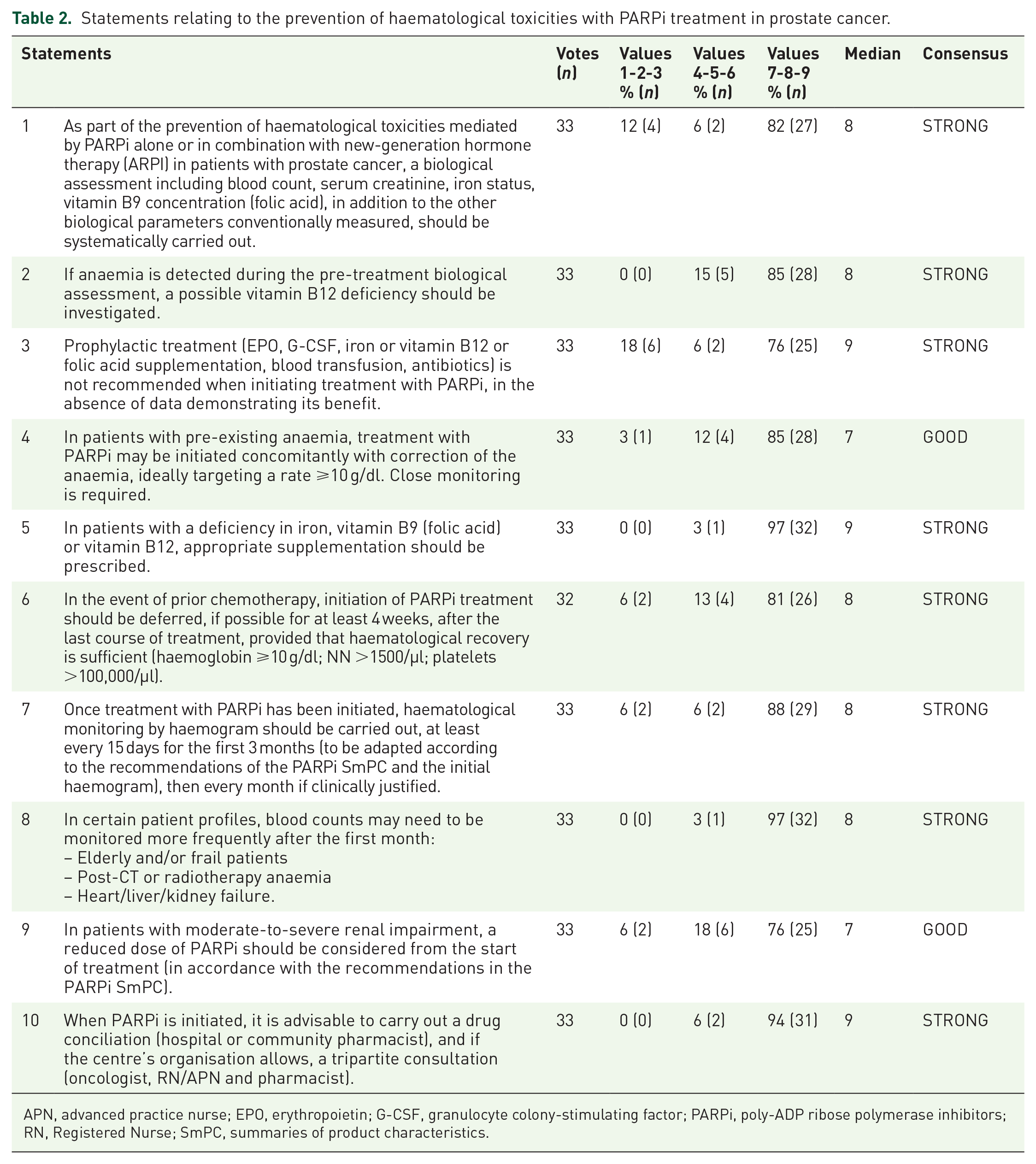
APN, advanced practice nurse; EPO, erythropoietin; G-CSF, granulocyte colony-stimulating factor; PARPi, poly-ADP ribose polymerase inhibitors; RN, Registered Nurse; SmPC, summaries of product characteristics.
Statements relating to the management of anaemia associated with PARPi treatment in prostate cancer.

EPO, erythropoietin; PARPi, poly-ADP ribose polymerase inhibitors.
Statements relating to the management of neutropenia and thrombocytopenia associated with PARPi treatment in prostate cancer and risk of MDS/AML.
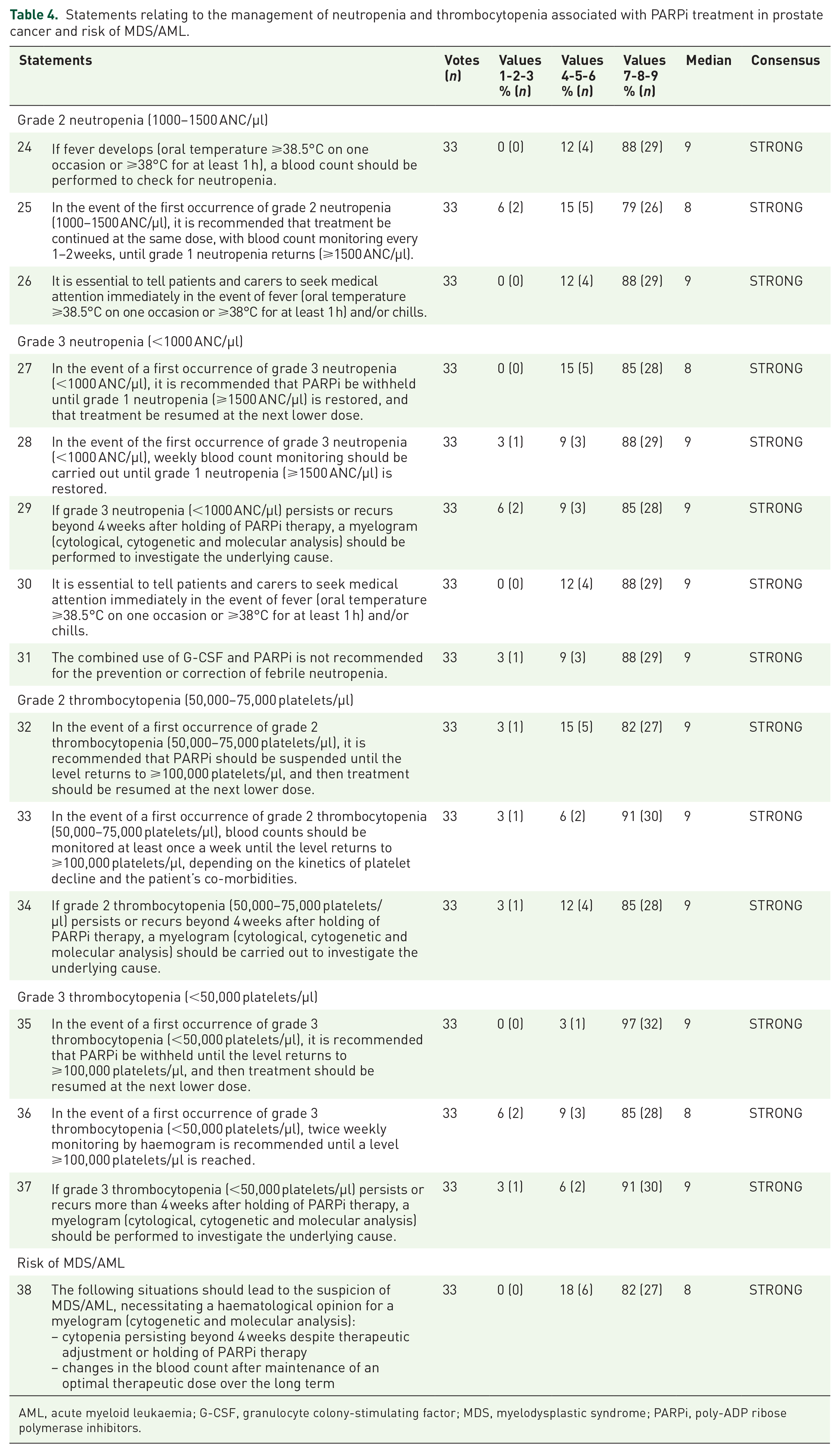
AML, acute myeloid leukaemia; G-CSF, granulocyte colony-stimulating factor; MDS, myelodysplastic syndrome; PARPi, poly-ADP ribose polymerase inhibitors.
From the first voting round, 30 statements achieved a STRONG consensus (⩾75% of votes ⩾7 and median ⩾8), 3 achieved a GOOD consensus (⩾75% of votes ⩾7 or median ⩾8) and 5 did not reach consensus. Based on the experts’ comments, seven statements – two with a GOOD consensus and five without consensus – were revised by the SC for a second voting round.
After this second round, 6/7 statements achieved a STRONG consensus, and 1/7 received a GOOD consensus.
In total, after two voting rounds, a consensus was reached for all the proposals. The distribution of votes, medians and results is shown in Tables 2–4.
Discussion
Prescription of PARPi requires particular attention to haematological toxicities, a known class effect which can lead to dose reductions or frequent treatment interruption and discontinuation. Using a modified Delphi methodology, this research has resulted in 38 practical and detailed recommendations (achieving 100% consensus among experts) to help HCPs prevent and manage anaemia, neutropenia and thrombocytopenia in the real world in patients with mCPRC.
Initiation of treatment with PARPi
The voting group strongly agreed with the recommendation to perform a full biological assessment before starting PARPi treatment, including blood count, serum creatinine, iron status and vitamin B9 (folate) concentration. Haematological disorders, in particular anaemia, are common in prostate cancer patients and may have different aetiologies, either linked to the patient’s general condition and cancer (iron deficiency, bone marrow invasion, etc.) or treatment (androgenic deprivation, systemic chemotherapy, etc.).30,31 A low haemoglobin level on PARPi initiation is associated with increased risk of grade ⩾3 anaemia, neutropenia or thrombocytopenia once on PARPi. 32 These haematological disorders should therefore be investigated before PARPi initiation so that appropriate corrective measures can be implemented.
Several statements offering guidance for specific patients were validated. The statement that pre-existing anaemia should not delay PARPi initiation, provided corrective measures and close monitoring are implemented, achieved a GOOD consensus, probably due to the proposed threshold of a haemoglobin level ⩾10 g/dl. Indeed, in some clinical studies, a level of 9 g/dl has been recommended.14,15 However, given real-life experience and general patient status (older, frailer and often anaemic), a threshold of ideally 10 g/dl was felt to be more clinically appropriate to initiate PARPi treatment under optimal conditions and limit subsequent dose interruptions or reductions.
Similarly, it was agreed that a delay must be observed before initiating PARPi after the last course of chemotherapy to allow sufficient time for haematological recovery. Selle et al. 23 have proposed a 6–8-week delay based on the results from studies in ovarian cancer patients treated with platinum-based chemotherapy. However, in the mCRPC clinical trials, a 3- or 4-week delay was allowed between the two treatments. Considering this and practical experience, the experts strongly agreed on the need to observe a minimum delay of 4 weeks.
Although the median age at mCRPC diagnosis is 72 years, 33 no statement specific to elderly patients was submitted to the experts. This fits with Selle et al. 23 who previously recommended not reducing the PARPi dose in elderly or frail patients without good pharmacological reason (significant haematological toxicity on previous chemotherapy or increased exposure to PARPi due to renal or hepatic impairment or drug interactions); advising patients to be vigilant for certain symptoms (malnutrition, gastrointestinal, sensory, balance, cognitive disorders, hyper- or hypotension); and organising a pre-therapeutic onco-geriatric consultation in the event of a geriatric syndrome, progressive co-morbidities or any doubt about the patient’s ability to take their treatment completely independently. In addition, pharmacokinetic studies have not found any impact of age on PARPi exposure, therefore PARPi SmPCs do not recommend any dose adjustment for the elderly population.13 –16,34
Haematological monitoring
As it is recognised that the onset of haematological toxicities induced by PARPi is more marked in the first few weeks, 35 there was STRONG consensus to monitor this frequently during the first few months of treatment. However, significant differences in onset have been reported between PARPi, with a median time to onset of 21, 23, 36 and 63 days, across all organs, for niraparib, rucaparib, talazoparib and olaparib, respectively. 35
The voters strongly agreed that this monitoring should be more frequent in profiles at high-risk of toxicity: patients with anaemia following chemotherapy or radiotherapy; with heart, liver or kidney failure and the elderly and/or frail.
Management of haematological toxicities
The 27 statements proposed for the management of grades 2 and 3 anaemia, neutropenia and thrombocytopenia all achieved STRONG consensus, providing precise guidance for assessment and implementation measures; this supplements existing recommendations and SmPCs which are not always detailed.23 –25 For example, SmPCs do not provide specific guidance for grade 2 anaemia, which is not surprising as, in MAGNITUDE and TALAPRO-2 trials, a haemoglobin level of 9 g/dl was permitted at initiation.13 –15
Voters agreed that symptomatic grade 2 anaemia – with symptoms including fatigue, weakness, pale skin, chest pain, fast heartbeat or shortness of breath, headache, dizziness, light-headedness, cold hands and feet and loss of appetite – should necessitate holding of PARPi therapy. 24 Furthermore, whether for grade 2 or 3 anaemia, neutropenia or thrombocytopenia, voters recommended waiting, ideally for a return to grade 1 before restarting treatment. Indeed, although less restrictive thresholds might be stipulated in trials, patients in the real world are often less fit, so a more conservative threshold may be more appropriate. However, clinicians should adapt the strategy according to the depth of toxicity, taking into account thresholds, the symptomatic nature of the disease and the general condition of patients.
Finally, the experts confirmed that patients with grade 3 anaemia (haemoglobin <8 g/dl) should receive blood transfusions, as well as those with grade 2 anaemia, depending on the intensity of symptoms, speed of onset, comorbidities, particularly cardiac, and any deficiencies needing correction. Although the ESMO guidelines on anaemia management in patients with cancer, and a recent study by Carson et al., suggest that a more restrictive transfusion threshold (7.0–8.0 g/dl) could be adopted, as patients with prostate cancer are often elderly and frail with cardiovascular toxicities, the group based its decision on the French recommendations, which are more in line with their clinical practice.25,36 –39
Risk of MDS and AML
There was STRONG consensus for confirming situations in which haematological expertise should be sought and a myelogram performed to rule out or confirm MDS/AML: persistent cytopenia beyond 4 weeks despite therapeutic adjustment or holding of PARPi therapy, or a change in the haemogram despite maintenance of an optimal therapeutic dose over the long term. However, the incidence of MDS/AML was ⩽2 cases per study in prostate cancer clinical trials, and estimated to be 0.73% (95% CI: 0.50–1.07) in a meta-analysis.7 –9,40 The risk of developing MDS/AML with PARPi therefore remains low but should be monitored, according to real-world data, particularly in prostate cancer. A number of risk factors have been identified, including previous treatments such as platinum-based chemotherapy (in ovarian cancers) or radiotherapy, and the presence of somatic or germline genetic abnormalities at tumour level.13 –15
Clinical implications
Recommendations have been published regarding PARPi management in other cancers, particularly gynaecological.23,41 While they provide relevant guidelines, they remain more general, addressing adverse effects in a broad sense. The management of haematological toxicities is not always detailed, particularly regarding prevention. In this work, we propose specific guidance on how to manage pre-existing anaemia and specific populations (elderly and/or frail patients, post-CT or radiotherapy anaemia, heart/liver/kidney failure), and prophylaxis. We also provide more detailed guidelines on the practical management of symptomatic low-grade anaemia and neutropenia, nutritional deficiencies and the place of supportive treatments such as EPO.
Limitations
These Delphi recommendations are based on French experience; therefore, the generalisability of the recommendations to other countries with different healthcare systems, patient populations and clinical practices must be considered.
Whilst the aim of this work was to propose detailed, standardised recommendations for PARPi, there are differences between the available PARPi, for example, in terms of monitoring, which is arbitrary and not necessarily based on clinical studies. In addition, in the absence of data, some of the proposals are based on the clinical experience and opinion of the SC and voters. Although the SC and voters are all experienced (median 11 years) in the management of prostate cancer and/or the follow-up of patients with mCRPC or breast/ovarian cancer on PARPi (5 and 10/year, respectively), and 89% of them have participated in research projects or clinical trials with PARPi over the last 5 years, the reliance on expert opinion, while highly valuable, inevitably introduces a degree of subjectivity.
Conclusion
PARPi represent a major advance in mCRPC management. However, haematological toxicities can affect patient exposure to treatment and so limit their effectiveness. Using a Delphi methodology, 38 practical recommendations on the prevention and management of grade 2 and 3 anaemia, neutropenia and thrombocytopenia associated with PARPi in mCRPC patients have been validated by a group of French experts. These proposals complement existing recommendations to guide HCPs in real-life clinical practice and so optimise treatment with PARPi.
