Abstract
Background:
Body mass index (BMI) and weight change are commonly explored as prognostic factors in breast cancer. However, their impact on outcomes with sacituzumab govitecan (SG) in metastatic triple-negative breast cancer (mTNBC) remains poorly defined.
Objectives:
To evaluate the association between baseline BMI, weight change during treatment, and clinical outcomes—including survival and adverse events (AEs)—in a real-world Polish cohort of women with mTNBC receiving SG therapy.
Design:
A retrospective, multicenter cohort study.
Methods:
A total of 83 women with mTNBC treated with SG across four oncology centers in Poland between August 2021 and September 2024 were analyzed. BMI was categorized by World Health Organization (WHO) criteria: underweight (BMI < 18.5 kg/m2), normal weight (18.5–24.9 kg/m2), overweight (25.0–29.9 kg/m2), and obese (⩾30.0 kg/m2). Weight changes were recorded from baseline to last treatment cycle, indicating loss or gain. Median progression-free survival (mPFS) and median overall survival (mOS) were assessed using multivariate Cox regression models (significance level α = 0.05). AEs were classified, using the National Cancer Institute Common Terminology Criteria for AEs (NCI-CTCAE), version 5.0, and compared across BMI and weight stability categories.
Results:
The mPFS was 4.07 months (95% CI: 3.05–6.18), and the mOS was 8.01 months (95% CI: 6.05–9.75). The median follow-up time was 7.42 months (95% CI: 6.07–8.96). Neither baseline BMI nor weight change significantly influenced PFS (BMI p = 0.09, weight change: p = 0.68) or OS (BMI: p = 0.09, weight change: p = 0.21). No BMI categories predicted disease progression or mortality. In addition, there were no significant differences in the frequency or severity of AEs (neutropenia, diarrhea, nausea, or hepatic toxicity) based on BMI or weight stability (p > 0.05).
Conclusion:
In this real-world Polish cohort of women with mTNBC treated with SG, neither BMI nor weight changes were prognostic indicators for survival or AE profiles. These findings underscore the necessity for identifying more reliable prognostic biomarkers in this patient population.
Keywords
Introduction
The systemic treatment landscape for triple-negative breast cancer (TNBC) is rapidly changing. Combining chemotherapy with immunotherapy has improved outcomes in both early and advanced stages. 1 In metastatic TNBC (mTNBC), antibody–drug conjugates (ADCs)—especially sacituzumab govitecan (SG), either alone or in combination with immunotherapy—have marked a major therapeutic advancement.2,3 Identifying reliable prognostic factors is essential to optimize patient selection and balance therapeutic efficacy with toxicity.
Body mass index (BMI) and weight loss are frequently investigated for their potential influence on treatment effectiveness and survival outcomes in oncology. 4 BMI has been identified as a significant risk factor for developing breast cancer (BC) and has also been extensively studied as a prognostic marker in early breast cancer (EBC). 4 This has prompted growing interest in its role in advanced breast cancer (ABC), including mTNBC. 5
The relationship between BMI and BC outcomes is complex and depends on multiple factors, including disease stage, tumor biological subtype, and the type of treatment administered. The literature provides evidence indicating that a higher BMI may have no significant impact, may negatively influence prognosis (e.g., in hormone receptor-positive (HR+) human epidermal growth factor receptor 2-negative (HER2−) EBC), or may even confer a favorable prognosis (e.g., in HER2-positive (HER2+) ABC or mTNBC).5 –7 However, being underweight is consistently associated with poorer survival outcomes, particularly in mTNBC.4,7
Weight change after diagnosis—including both weight loss and weight gain—is another important factor. Even minor weight loss, especially when exceeding 5% or 10%, is associated with shorter overall survival (OS).4,8 Less attention has been given to weight gain; nevertheless, an increase in body weight after diagnosis, particularly beyond 10% of baseline weight, also negatively impacts prognosis by raising the risk of recurrence, metastases, and death from any cause. 9 Despite these observations, the impact of weight gain on treatment efficacy in advanced disease varies depending on cancer type, highlighting the need for further research to elucidate its prognostic significance in ABC. 4
Inflammation and metabolic dysregulation, commonly associated with both obesity and cachexia, are believed to play a key role in modulating treatment response and disease progression in advanced cancers.4,8,10 –12 Chronic systemic inflammation can influence tumor biology and the surrounding microenvironment, contributing to therapy resistance and worse clinical outcomes. In addition, excess adipose tissue influences drug pharmacokinetics—obesity has been linked to altered drug distribution, metabolism, and clearance, which may impact the efficacy of both chemotherapy and targeted therapies. 12 Despite this, obesity-related inflammatory and metabolic pathways remain underexplored in the context of SG and other ADCs more broadly.
SG has emerged as a game-changing therapy for relapsed or refractory mTNBC. 2 Results from the ASCENT trial demonstrated significantly improved survival outcomes with SG compared to standard chemotherapy, with consistent benefits observed across BMI subgroups. Patients treated with this ADC experience longer progression-free survival (PFS), OS, and higher objective response rates (ORR), regardless of their baseline BMI. 13
Despite this progress, BMI and weight change remain underexplored in their potential influence on the safety and efficacy of SG therapy, particularly in real-world settings and diverse populations.14 –19 A recent systematic review of BC clinical trials highlighted a lack of consistent reporting on BMI and weight changes, including both loss and gain, with limited subgroup analyses addressing these variables. 12
The primary objective of this study is to assess whether baseline BMI correlates with OS in a Polish cohort of women diagnosed with mTNBC treated with SG. Secondary objectives include evaluating the impact of BMI on PFS, assessing the relationship between weight change and survival outcomes, and examining the frequency of grade ⩾3 adverse events (AEs) in relation to BMI and weight change. This study aims to address current gaps in the literature and provide a more nuanced understanding of how these factors influence therapeutic outcomes in a real-world cohort.
Materials and methods
Patients’ population and treatment
Between August 2021 and September 2024, a total of 83 female patients initiated treatment with SG therapy across four oncology centers in Poland (listed in Table S1 in the Supplemental Appendix 1). Patient identification was performed through hospital registry systems. Eligibility criteria aligned with Polish drug reimbursement program, based on the ASCENT trial protocol,2,20 and included the following: histologically confirmed TNBC per American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guidelines, inoperable or advanced-stage disease, prior receipt of one or two prior lines of systemic therapy, Eastern Cooperative Oncology Group performance status (ECOG PS) of 0–1, measurable disease according to Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1, 21 adequate organ function, and no active central nervous system metastases. Treatment was discontinued upon disease progression, unacceptable toxicity, deterioration of PS or quality of life, or by physician or patient decision. Detailed inclusion and exclusion criteria according to the Polish drug reimbursement program are provided in Supplemental Appendix 1. Patients treated within early-access reimbursement programs (outside of clinical trials) were also included.
The standard starting dose of SG was 10.0 mg/kg, unless otherwise specified by the physicians. AEs of all grades were documented according to the National Cancer Institute Common Terminology Criteria for AE (NCI-CTCAE), version 5.0. 22 In the event of an AE, dose modifications were guided by the Summary of Product Characteristics for the European Union. 23 Treatment response was assessed every 8 weeks using computed tomography or magnetic resonance imaging according to RECIST 1.1 criteria. 21
Data collection
For this retrospective cohort study, data were collected from digital and paper-based medical records. The data were grouped into several categories to facilitate comprehensive analysis. Patient-specific data included demographic information such as age and sex. Pathological data comprised detailed histopathological findings, including estrogen receptor (ER), progesterone receptor (PgR), HER2 status, and the Ki-67 proliferation index from the initial or most recent specimens. Clinical disease-related data included the date of diagnosis, the initial treatment intent (palliative or radical), and metastatic lesion sites.
Treatment-related data specific to SG therapy included the initiation date, dates of subsequent administrations, and the date of the final dose. Further data included information on subsequent lines of therapy post-SG, the date of the last clinical follow-up, and, if applicable, the date of death.
BMI and weight data collection
Baseline weight and BMI, as well as body weight prior to each SG administration, were documented by clinical staff. Measurement procedures and equipment calibration protocols were defined according to the standards of each oncology center. BMI was calculated as weight in kilograms divided by height in meters squared (kg/m2). According to the World Health Organization’s (WHO) criteria, patients were divided into four groups: underweight (BMI < 18.5 kg/m2), normal weight (18.5–24.9 kg/m2), overweight (25.0–29.9 kg/m2), and obese (⩾30.0 kg/m2). Weight change was defined as the difference between the baseline weight at the start of SG treatment and the weight recorded at the last treatment cycle before disease progression, or the last cycle received prior to the cutoff date, with weight loss indicating a negative difference and weight gain indicating a positive difference.
Data were last updated on October 31, 2024.
Primary and secondary objectives
The primary objective of this study is to evaluate whether BMI correlates with OS in a real-world Polish cohort of women diagnosed with mTNBC undergoing treatment with SG. OS is defined as the duration from the initiation of SG treatment until death from any cause.
Secondary objectives include assessing the relationship between baseline BMI and PFS, defined as the time from the start of SG treatment until disease progression or death, whichever occurs first. In addition, the study aims to examine how changes in patient weight during treatment influence survival outcomes. Specifically, multivariate Cox regression analyses will evaluate whether initial weight, categorized BMI, and weight change during treatment are predictive of both PFS and OS in this cohort. Radiological response evaluations were conducted according to RECIST 1.1, categorizing patient responses into complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD). The objective tumor response rate (ORR) was calculated based on the proportion of patients achieving either CR or PR. Moreover, the study investigates the relationship between weight stability or weight gain and loss with the occurrence of complications across various categories, particularly examining the frequency of grade ⩾3 AEs in relation to BMI and weight changes. Collectively, these secondary analyses aim to fill existing gaps in the literature and provide a nuanced understanding of how BMI and weight dynamics impact therapeutic efficacy and tolerability in clinical practice.
Statistical analysis
Continuous variables are presented as median and first and third quartiles. Normality was verified using the Shapiro–Wilk test. Categorical variables are summarized as numbers and percentages. Comparisons between two groups were conducted using the Wilcoxon rank-sum test, as the normality assumption was violated, or the sample size was smaller than 10. Comparisons between more than two groups were performed using the Kruskal–Wallis test, under similar conditions.
A multivariate Cox regression model was applied to evaluate the relationship between explanatory variables and PFS or OS. The selection of variables for the Cox regression model was based on a clinical approach, expert knowledge, and previously published research findings. Accordingly, baseline body weight, BMI category, and weight change during treatment were included in the multivariate model. These variables have previously been identified in the literature as potentially prognostically significant for treatment outcomes and have been frequently analyzed in the context of systemic therapies for BC, including mTNBC. The proportional-hazard assumption was checked using Schoenfeld residuals. The presence of influential observations was evaluated using deviance residuals and dfbeta residuals. The linearity assumption was verified using Martingale residuals, and multicollinearity was assessed using the generalized variance inflation factor. Statistical analyses were performed using R version 4.4.0 and Python version 3.11.4.
All statistical inference was conducted at a significance level of α = 0.05.
Chi-square tests or Fisher’s exact tests were used to compare the frequency of AEs, depending on sample size requirements. Considering the number of events observed for each respective grade, statistical analyses were conducted specifically for diarrhea, grade 1 and 2 nausea, elevated liver enzyme levels, grade 3 and 4 anemia and thrombocytopenia, as well as neutropenia. In addition, a binary variable was created to represent the occurrence of any complication, coded as 1 for at least one complication and 0 for no complications, which was also analyzed using these methods.
The reporting of this study conforms to the STROBE statement for cohort studies (Supplemental Appendix 2). 24
Results
Baseline patient characteristics
Clinical data were collected from 83 women treated with SG. The median age at diagnosis was 51 (interquartile range (IQR): 41–62) years, while the median age at the start of treatment was slightly higher at 54 (IQR: 46–65; SD = 13.02). A detailed characterization of the patients and their disease is presented in Table 1. At the initiation of SG therapy, participants were categorized based on BMI as follows: underweight (n = 3, 3.6%), normal weight (n = 41, 49.4%), overweight (n = 19, 22.9%), and obese (n = 20, 24.1%). This classification provides a clear baseline distribution for further analysis.
Characteristics of patients and the disease.

BRCA, BReast CAncer gene; HER2, human epidermal growth factor receptor 2, IHC, immunohistochemistry.
Treatment exposure and outcomes
At the time of data collection, 59 patients (71%) had completed treatment. Among these, 55 (96.6%) discontinued due to disease progression, 2 (3.4%) due to death, and another 2 (3.4%) due to complications. Meanwhile, 24 (28.9%) patients were still receiving treatment. SG was administered at various lines of therapy, with a median line of 2 (IQR: 2–3). Patients completed a median of 5 SG treatment cycles (IQR: 3–9).
During treatment, 33 (39.76%) patients experienced no weight change, nearly the same proportion of patients, 32 (38.55%) experienced weight loss, while 16 (19.28%) patients gained weight. Data were unavailable for 2 patients. The median weight change was 0 kg (IQR: −2 to 0). Survival outcomes were as follows: the median PFS was 4.07 months (95% CI: 3.05–6.18), while the median OS was 8.01 months (95% CI: 6.05–9.75), and the median follow-up time was 7.42 months (95% CI: 6.07–8.96). A total of 48 (57.83%) patients received systemic treatment following SG therapy. The median number of subsequent treatment lines was 1 (IQR: 1–2). An ORR was achieved in 25 (30.12%) patients, and additional details describing the treatment response are provided in Table 2.
Best response to SG treatment according to RECIST criteria.

CR, complete response; ORR, overall response rate; PD, progressive disease, PR, partial response; RECIST, Response Evaluation Criteria in Solid Tumors; SD, stable disease; SG, sacituzumab govitecan.
Adverse events
The most common toxicity was neutropenia, occurring in 63.9% of cases, while the rarest was hypersensitivity, appearing in only 2.4% of cases. Symptoms such as anemia and alopecia were also relatively frequent, observed in 36.1% and 31.3% of cases, respectively. Details regarding all complications are provided in Table 3.
Adverse events during sacituzumab govitecan treatment.

G, grade; NR, not reported.
Results of analysis
Impact of BMI and weight change on survival
None of the evaluated variables—initial weight, BMI categories, or weight change—showed statistically significant associations with PFS (all p-values > 0.05 for all variables). In addition, tests for multicollinearity and proportional-hazards assumption were satisfied, indicating the validity of the PFS model. Similarly, no significant associations were found between the variables and OS (p > 0.05 for all variables). However, the proportional-hazards assumption was violated in the OS model (p = 0.02), indicating that the results for OS should be interpreted with caution. Key findings are summarized in Table 4.
Multivariate Cox regression results for progression-free survival and overall survival.
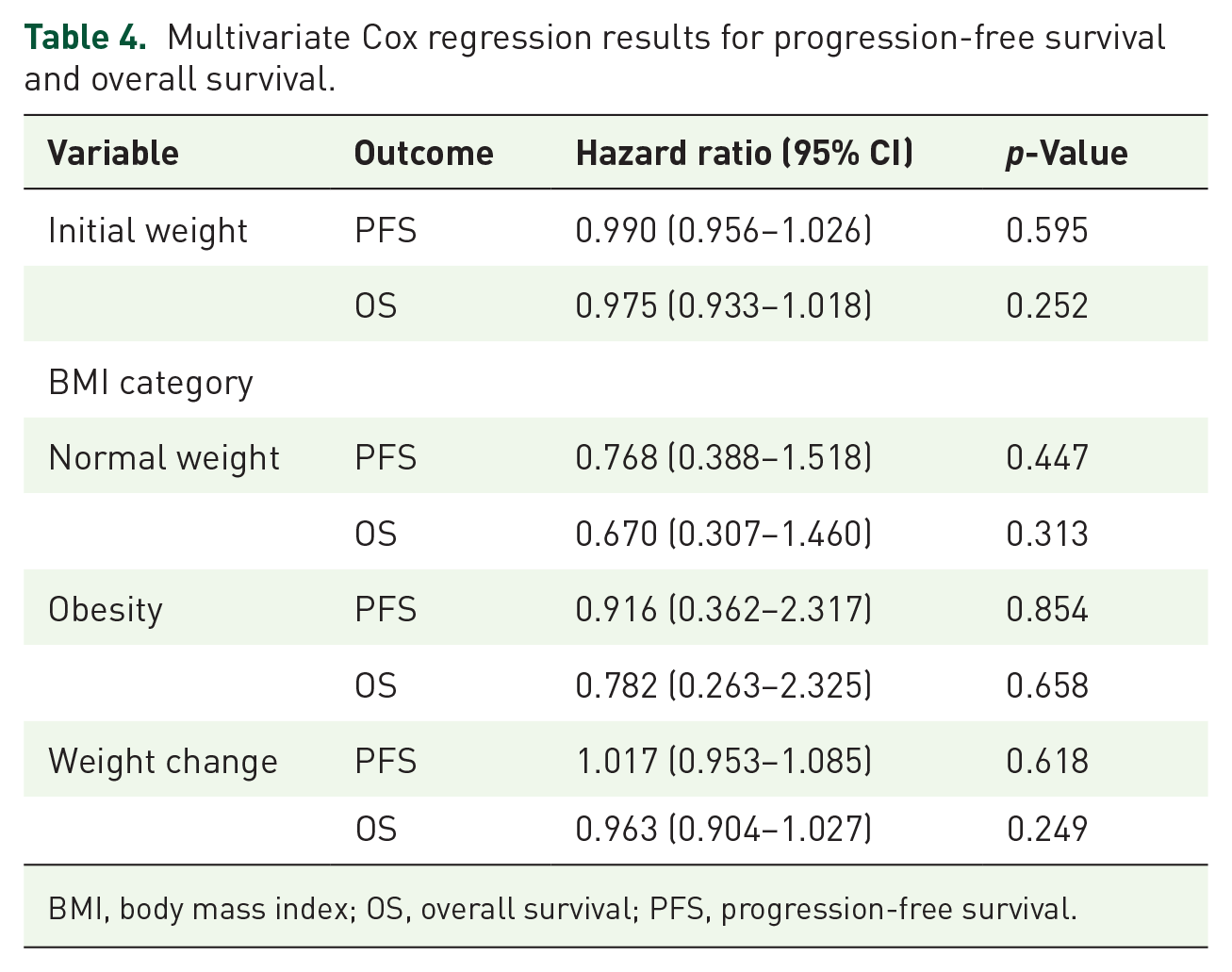
BMI, body mass index; OS, overall survival; PFS, progression-free survival.
Association of weight change and BMI with adverse events
Analysis of BMI categorized into WHO groups against the severity of specific AEs revealed no statistically significant associations (Figures 1–6 in the Supplemental Appendix 1), even after correcting for multiple testing. Using the binary variable to represent the occurrence of any complication, no statistically significant differences were found across BMI categories (p = 0.98 for non-categorized BMI and p = 0.76 for categorized BMI). Hepatic enzymes initially showed significance before multiple-testing corrections, but this significance was lost after applying Bonferroni adjustments.
Weight change was also not significantly associated with the occurrence, severity, or type of AEs, including diarrhea, nausea, anemia, neutropenia, and others (Figures 7–12 in the Supplemental Appendix 1). None of the tests for individual or combined AEs yielded statistically significant results (Wilcoxon and Kruskal–Wallis, p > 0.05).
Results of the analysis of the impact of weight stability/gain and loss on the occurrence of adverse events
None of the results demonstrated statistical significance (p > 0.05), both before and after applying corrections for multiple testing (Bonferroni adjustments). Specifically, no association was found for complications such as diarrhea (OR = 0.98; 95% CI: 0.21–5.14; p = 1.00), nausea (OR = 0.80; 95% CI: 0.16–4.38; p = 0.73), anemia (OR = 1.04; 95% CI: 0.49–2.19; p = 0.87), or alopecia (OR = 0.72; 95% CI: 0.31–1.66; p = 0.71).
In addition, an analysis of the impact of weight stability/gain and weight loss on the occurrence of any complication (logical OR) was conducted. Similarly, no significant differences were identified (OR = 1.02; 95% CI: 0.19–4.77; p = 1.00). These findings suggest no statistical evidence supporting a relationship between weight stability and the occurrence of the analyzed complications.
The analysis of the correlation between treatment discontinuation due to complications and BMI or weight loss was not conducted due to an insufficient number of patients (n = 2).
Weight change and subsequent therapy lines
No significant differences in weight change were observed between patients receiving subsequent therapy lines and those who did not (Wilcoxon, p = 0.58) (Figure 13 in Supplemental Appendix 1). Analysis of weight change across groups receiving varying numbers of subsequent therapy lines (0, 1, 2, or ⩾3) also showed no significant differences (Kruskal–Wallis, p = 0.84) (Figure 14 in Supplemental Appendix 1).
Discussion
In our real-world observation of Polish women with mTNBC treated with SG, we found a slightly shorter PFS compared to the ASCENT clinical trial. In addition, OS was significantly shorter, highlighting the complexity of translating clinical trial results into everyday clinical practice. Nevertheless, the observed AE profile remained consistent with previously reported clinical trial data. Furthermore, neither BMI nor weight change proved prognostic in this patient population nor did they influence the frequency or severity of AEs. These findings are particularly relevant given poor-prognosis BC such as mTNBC, which—as emphasized in recent literature—remains therapeutically challenging due to the lack of actionable molecular targets, despite the increasing availability of targeted therapies such as ADCs. 25
The relationship between BMI and outcomes in BC remains controversial. While some studies suggest that overweight and obesity do not significantly influence outcomes in BC, underweight status consistently emerges as a negative prognostic factor for survival, particularly in mTNBC.4,7 Conversely, evidence from other studies highlights an “obesity paradox,” where higher BMI appears to correlate with improved survival outcomes in ABC, including HER2+5,6 and TNBC subtypes. 7 These contradictory findings underline the complexity of the relationship between BMI, tumor subtype, and treatment modality. For instance, evidence indicates that higher BMI is associated with worse outcomes in HR+ EBC, while in HER2+ ABC, obesity paradoxically correlates with improved PFS and OS.6,13
Exploring the biological association between high BMI and weight changes with survival outcomes reveals that inflammation is a key underlying mechanism.8,10 Inflammation and metabolic dysregulation, frequently associated with both obesity and cachexia, are believed to modulate treatment response and disease progression in advanced cancers. Chronic systemic inflammation undeniably exacerbates cancer progression, influencing not only systemic physiology but also altering the tumor microenvironment. 11 Obese individuals often exhibit elevated levels of inflammatory markers such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α), which are produced by enlarged adipocytes that release cytokines and free fatty acids that activate inflammatory pathways (e.g., NF-κB). 10 Similarly, in cachexia, pro-inflammatory cytokines such as IL-6 and TNF-α promote muscle wasting and insulin resistance. 11 This shared mechanism disrupts metabolism, enhances oxidative stress, creates a tumor-promoting and treatment-resistant environment, and contributes to poor outcomes in cancer patients.4,8,10,11
The importance of BMI assessment and reporting in relation to drug efficacy in BC treatment was elucidated in the systematic review by Van Cauwenberge et al. 12 Despite the well-established impact of obesity on pharmacokinetics, pharmacodynamics, and cancer biology, BMI was documented in only 8% of the reviewed trials, with only two trials performing subgroup analyses based on BMI. Furthermore, fixed dosing regimens, which were used in 81% of the reviewed trials, often disregard weight-based variations that can influence both drug efficacy and toxicity. 12 The fixed-dosing regimen of SG (10 mg/kg on days 1 and 8 of a 21-day cycle) was supported by PK analyses, demonstrating no need for dose adjustments based on patient demographics or disease characteristics. 26 However, the observed correlation between body weight and PK parameters underscores the potential for BMI and adiposity metrics to influence treatment outcomes. As obesity is associated with altered drug distribution, metabolism, and clearance, tailored approaches for BMI-based dosing may further optimize efficacy and minimize toxicity, especially for drugs with narrower therapeutic windows. 12
To date, the only analysis addressing the correlation between BMI and treatment outcomes, as well as AEs associated with SG, was presented as a poster at the ESMO 2024 Congress. 13 It was an ad hoc subgroup analysis of the ASCENT trial, which demonstrated that, regardless of BMI, the efficacy of SG treatment (PFS and OS) was superior in the SG-treated group compared to those receiving chemotherapy per physician’s choice (TPC). Similarly to the ASCENT study, the proportion of overweight (n = 19, 22.89%) and obesity (n = 40, 48.19%) in the Polish cohort surpasses that of normal body weight, reflecting the ongoing epidemic of abnormal body weight, as highlighted by epidemiological data. 13 In contrast to the ASCENT trial—which suggested that overweight and obesity had higher rates of severe AEs, more frequent dose reductions, and treatment discontinuation, 13 we demonstrated that whether body weight remains stable or decreases during treatment, it does not correlate with the occurrence of AEs. Similarly, a case report of a 40-year-old patient with a BMI of 40 showed that high BMI did not negatively impact the efficacy of SG, even despite dose reductions due to AEs (e.g., neutropenia). Treatment with SG in this patient resulted in a sustained partial response and maintained a good quality of life. 27
Taken together, our findings suggest that the efficacy and safety of SG treatment are independent of body weight. This finding stems from evidence that differences in concentration and PK changes in SG caused by body weight are minimal. This confirms that the established therapeutic dose already accounts for interpatient variability. Moreover, not only body weight, but also other patient- and organism-related factors, such as age, sex, race, ECOG, albumin level, renal, and hepatic function, do not significantly impact SG PK. 26 Thus, the presence of the molecular target likely plays a more critical role in the effectiveness of this ADC compared to patient-related factors.
The available real-world data publications on SG therapy were reviewed.14 –19 None of these publications provided detailed data on BMI, weight change, or their correlation with treatment outcomes or AEs.
This study holds substantial potential for informing clinical practice by highlighting the need for more individualized treatment strategies, particularly in managing AEs and dosing. The identified knowledge gaps include a lack of prospective studies specifically addressing how BMI and body composition affect ADC therapies in BC. Researchers should prioritize large-scale, prospective trials with precise body composition metrics to better define these relationships. Future research must tackle these gaps by systematically collecting and analyzing comprehensive data on adiposity, muscle mass, inflammatory markers, and their interplay with pharmacokinetics and pharmacodynamics.
Looking ahead, we anticipate significant advances in personalized oncology. Integrative models combining biomarkers, molecular targets, pharmacokinetic parameters, and patient-specific physiological metrics, such as BMI and muscle mass, are expected to become more prevalent. This multidisciplinary approach will not only refine dosing strategies but also optimize the therapeutic index, especially for drugs like ADCs that exhibit narrow therapeutic windows. Over the next 5 years, we foresee a deeper integration of body composition analytics into clinical trial designs and real-world studies, further enhancing personalized medicine approaches. This evolution will likely contribute to optimized dosing strategies, improved therapeutic efficacy, and minimized toxicity, ultimately benefiting patient outcomes and quality of life.
Ultimately, a more personalized and holistic assessment of patient characteristics will enhance treatment efficacy, minimize toxicity, and significantly improve patient outcomes in challenging cancers such as mTNBC.
Therefore, to the best of our knowledge, the presented analysis is the first comprehensive, full-text study examining the association of BMI and weight change with treatment outcomes and AEs outside the context of clinical trial data. Our findings clearly indicate that BMI and weight fluctuations during SG therapy do not significantly influence efficacy or toxicity, contrasting with some subgroup analyses of clinical trials. Clinicians can confidently administer SG therapy in patients across a wide BMI spectrum without routine BMI-based dose modifications, although individual patient monitoring remains crucial due to inherent variability in pharmacokinetic responses.
Study limitations
The retrospective study design inherently limits the ability to establish causal relationships and introduces potential selection and reporting biases. The relatively small sample size and single-nationality cohort may reduce the statistical power and limit generalizability to other populations. In addition, the short median follow-up duration may not adequately capture long-term outcomes, and the absence of comprehensive data on important confounding factors, such as socioeconomic status, constrains the robustness of the analysis. While commonly utilized, BMI and weight change remain imperfect surrogates for body composition, as they do not effectively differentiate between adiposity and muscle mass. The heterogeneity of prior treatments and disease stages further introduces variability into outcomes. Despite adhering to standardized AE assessment criteria (CTCAE v.5) and response evaluation methods (RECIST), it should be noted that adverse events and treatment responses were assessed across multiple centers by different radiologists and laboratories, which may have slightly varying standards and practices. Collectively, these limitations underscore the need for prospective studies with larger, more diverse populations, longer follow-up durations, and comprehensive data on confounding variables and mechanistic pathways to validate and extend these findings.
Conclusion
Initial weight, BMI, and weight change did not serve as prognostic factors for survival outcomes or correlate with therapy-related AEs in patients with mTNBC treated with SG. These findings emphasize the need for alternative biomarkers to better stratify risk and tailor treatment in this patient population.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251363104 – Supplemental material for Weight change and BMI are not prognostic markers of survival outcomes in sacituzumab govitecan therapy for mTNBC in the Polish female cohort
Supplemental material, sj-docx-1-tam-10.1177_17588359251363104 for Weight change and BMI are not prognostic markers of survival outcomes in sacituzumab govitecan therapy for mTNBC in the Polish female cohort by Małgorzata Pienia̧żek, Anna Polakiewicz-Gilowska, Manuela Las-Jankowska, Jakub Wronowicz, Michał Jarza̧b, Aleksandra Łacko, Marek Ziobro and Mirosława Püsküllüoǧlu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251363104 – Supplemental material for Weight change and BMI are not prognostic markers of survival outcomes in sacituzumab govitecan therapy for mTNBC in the Polish female cohort
Supplemental material, sj-docx-2-tam-10.1177_17588359251363104 for Weight change and BMI are not prognostic markers of survival outcomes in sacituzumab govitecan therapy for mTNBC in the Polish female cohort by Małgorzała Pienia̧żek, Anna Polakiewicz-Gilowska, Manuela Las-Jankowska, Jakub Wronowicz, Michał Jarza̧b, Aleksandra Łacko, Marek Ziobro and Mirosława Püsküllüoǧlu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
