Abstract
Background:
Ovarian function suppression (OFS) and premature menopause lead to side effects that significantly worsen the quality of life for young women with hormone receptor-positive early breast cancer (HR+ eBC).
Objectives:
We retrospectively analyzed the most common side effects of OFS and endocrine therapy (ET) in young women with HR+ eBC.
Design:
This analysis was designed as a combined cohort study according to STROBE criteria.
Methods:
We retrospectively identified 118 patients (pts); after excluding pts with luteinizing hormone-releasing hormone analogs given for fertility preservation only and pts with permanent castration (surgical or by irradiation), 100 pts were included in the final analysis. We analyzed side effects that were represented by more than 10% of patients at any predefined time points. We looked at the frequency and dynamics of these side effects over time—baseline, 6th, 12th, 18th, 24th, 30th, and 36th month according to three categories—the entire population, according to age (younger and older than 40 years), and according to previous treatment (chemotherapy or not).
Results:
The most commonly reported side effects among the entire patient group were vasomotor symptoms, particularly hot flashes and night sweats. Sleep disturbances and anxiety were also reported, though less frequently. The most significant worsening of vasomotor symptoms occurred within the first 6 months. Women under 40 years of age reported night sweats and sleep disturbances more often than women aged 40 and older at all time points, with statistically significant differences observed at 30 months for both symptoms and at 12 months for night sweats alone. Among patients who did not receive CT, hot flashes were frequently reported; however, only night sweats showed a statistically significant difference at 6 months compared to the group that did receive CT. No significant differences were found in the frequency of sleep disturbances or anxiety between these groups.
Conclusion:
Young women with HR+ eBC undergoing OFS plus ET commonly report side effects in clinical practice. The most frequently reported symptoms are hot flashes, night sweats, sleep disturbances, and anxiety. All symptoms were more pronounced at the 30-month follow-up compared to baseline.
Keywords
Introduction
The initial connection between breast cancer and ovarian suppression was identified in 1882, when Nunn observed regression of breast cancer in a female patient after 6 months of menopause. This was followed by the first bilateral oophorectomy as an adjuvant treatment for breast cancer in 1897. 1
In their publication of collected study data, the Early Breast Cancer Trialists’ Collaborative Group confirmed the benefit of adjuvant ovarian function suppression (OFS) in early breast cancer (eBC). 2 Later, the prospective SOFT study showed a statistically significant difference in distant disease-free survival (DFS) for patients receiving OFS in addition to tamoxifen, compared to those receiving tamoxifen alone (OFS + TAM vs TAM: DFS 83.2% vs 78.9%; hazard ratio 0.76, 95% confidence interval 0.62–0.93, p = 0.0009). 3 The combined analysis of the SOFT and TEXT studies included a total of 4690 premenopausal women diagnosed with early hormone-receptor-positive (HR+) breast cancer. 4 The TEXT study examined the effectiveness of tamoxifen + OFS compared to exemestane + OFS over a 5-year therapy period, while the SOFT study evaluated the effectiveness of OFS + exemestane, OFS + tamoxifen, or tamoxifen alone over 5 years of treatment.4,5
Based on the results of the SOFT and TEXT studies, all major international guidelines (ASCO, European Society for Medical Oncology (ESMO), NCCN) recommend OFS combined with endocrine therapy (ET) in the adjuvant treatment of premenopausal women with HR+ breast cancer, starting at clinical stage II. The updated results of the SOFT and TEXT studies, with 12 years of follow-up, demonstrated an improved distant relapse-free interval, particularly in the high-risk patient group (which includes those with higher-risk clinical-pathologic features, such as prior neoadjuvant chemotherapy, age under 35 years, and grade 3 tumors) when OFS was added to tamoxifen or exemestane. 6
The SOFT and TEXT subgroup analyses examine the side effects of premature menopause and the impact of ET on quality of life. 7 Current data indicate that the most common menopausal side effects caused by luteinizing hormone-releasing hormone (LHRH) analogs are vasomotor symptoms, such as hot flashes and sweating. In addition, musculoskeletal pain, depression, sleep disturbances, and sexual problems, such as vaginal dryness, loss of libido, and dyspareunia, have also been reported by patients.3,4
The expression of adverse effects of OFS varies depending on age and the type of treatment used (aromatase inhibitor, tamoxifen, or prior chemotherapy).7 –9 According to the CANTO study, the side effects of ET differ between premenopausal and postmenopausal women. In younger patients, the primary complaints included more frequent hot flashes, night sweats, and leukorrhea, while postmenopausal women reported joint pain more often. 10
Adverse effects of therapy also reduce adherence; according to the literature, up to 20% of younger women (under 35 years old) discontinue therapy due to side effects. 7
Our analysis aimed to review the most frequently reported adverse events among patients with hormone receptor positive, HR+ (estrogen, ER, and/or progesterone, PR ⩾1%) eBC undergoing treatment with LHRH analogs during monthly visits at the Institute for Oncology and Radiology of Serbia (IORS).
We sought to indirectly compare the menopause-related side effects most frequently reported in our clinical practice with those described in published studies on the adverse effects of drug-induced early menopause in women with early-stage HR+ breast cancer.
We hypothesized that younger patients and those who had not previously received chemotherapy would report a higher frequency of adverse effects associated with early menopause.
Methods
The STROBE guidelines for reporting observational studies were followed. This retrospective analysis included consecutive premenopausal women with early HR+ breast cancer who began treatment with LHRH analogs at the outpatient department of the IORS from October 2019 to March 2021. Data were collected by reviewing the medical histories and hospital records of each patient included in the analysis. We included all women undergoing OFS with LHRH analogs, regardless of age. Patients could have previously received chemotherapy, either with LHRH analogs administered concomitantly or sequentially with chemotherapy and subsequent ET.
The analysis focused solely on women who received medical OFS with LHRH analogs. Women who began treatment with LHRH analogs but later underwent surgical or radiotherapy-induced permanent OFS were excluded. We also excluded patients with triple-negative or HER2-positive breast cancer who received LHRH analogs only concomitantly with chemotherapy for fertility preservation purposes. Medical OFS was administered using triptorelin or goserelin every 28 days for 3 years, which is standard practice in Serbia. ET with tamoxifen or an aromatase inhibitor was given for 5 or more years, depending on disease risk. Aromatase inhibitors were prescribed exclusively during LHRH analog therapy.
We collected data on the most common adverse effects (reported by more than 10% of patients at any time point) at specific time points: at the beginning of treatment and every 6 months thereafter (1, 6, 12, 18, 24, 30, and 36 months). We examined the frequency of adverse effects in relation to patient age (⩽40 and >40 years) and prior chemotherapy treatment (yes or no). We analyzed how the frequency of adverse effects changed at each prespecified time point. The severity of reported symptoms could not be graded, as this was a retrospective analysis of clinical charts, where data were recorded non-uniformly, making categorization difficult.
Premenopausal status was defined as regular menstrual cycles occurring monthly up until the initiation of adjuvant treatment for eBC. Women in perimenopause were also included in the analysis. Perimenopause was defined as the last menstrual cycle occurring within 1 year. All patients received LHRH agonists based on their premenopausal status prior to the initiation of oncologic therapy. The agonists were administered either concurrently with chemotherapy or sequentially afterward, without awaiting the return of menstrual cycles in cases of chemotherapy-induced amenorrhea.
The study was approved by the ethics committee (No. 01.1/2025/62). All patients provided written informed consent at inclusion and were aged ⩾18 years.
Statistical analysis
We employed graphs, including Normal Q-Q Plots and Histograms, along with the Kolmogorov–Smirnov and Shapiro–Wilk tests to assess the agreement of sampling distributions with the normal distribution.
We utilized descriptive statistics to describe key parameters based on their nature: frequencies, percentages, mean values (averages), medians, standard deviations (SD), and ranges. For statistical significance, a significance level of α = 0.05 was adopted.
To test differences between age groups and depending on the nature of the examined parameters, we applied the Pearson Chi-squared test, Fisher’s exact test, and the Wilcoxon rank-sum test. Spearman’s rank correlation coefficient was used to examine the linear association of significant parameters, while linear regression was employed to model their relationships.
Results
We identified 118 patients treated with LHRH analogs for early HR+ breast cancer at the outpatient clinic of the IORS. Eight patients received LHRH analogs only a few months before undergoing laparoscopic oophorectomy and were excluded from the analysis. Ten patients continued treatment at another medical center or were lost to follow-up. This analysis included 100 women who initiated therapy with LHRH analogs between October 2019 and March 2021. The median follow-up period was 35 months. The median age of the participants was 41 years (range: 30–50). Half of the patients (51%) had a T1 tumor size, the vast majority (94%) had a histological grade 2 tumor, and 70% had node-negative disease. All patients had HR+ breast cancer (Table 1).
Patients, disease, and treatment characteristics according to age – n (%).

HR, hormone receptor; LN, lymph node; NA, not applicable.
Chemotherapy was prescribed to 47% of patients, either in the neoadjuvant or adjuvant setting, and all patients underwent breast surgery. Oral ET was administered in the adjuvant setting following chemotherapy, with LHRH analogs initiated concurrently with chemotherapy (in 33% of patients) or afterward with ET. Most patients (95%) were treated with tamoxifen as adjuvant ET, while five patients with tamoxifen contraindications were treated with aromatase inhibitors (Table 1).
Relapse was diagnosed in four patients while on adjuvant ET, and one patient died due to relapse and progression of the disease.
The most frequently reported symptoms among patients were hot flashes, night sweats, sleep disturbances, and anxiety. Vasomotor symptoms, such as hot flashes and night sweats, were the most commonly reported at every time point during adjuvant treatment with LHRH analogs in all patients (Figure 1). Hot flashes (minimum 49% at baseline, maximum 73% at the 24–30 month time point) were reported more frequently than night sweats (minimum 30% at baseline, maximum 55% at the 12th month) at each time point. Vasomotor symptoms exhibited the most significant change in frequency during the first 6 months of therapy, compared to baseline values. After peaking in the first 6 months, the frequency of hot flashes remained consistent across all analyzed groups during the follow-up period.
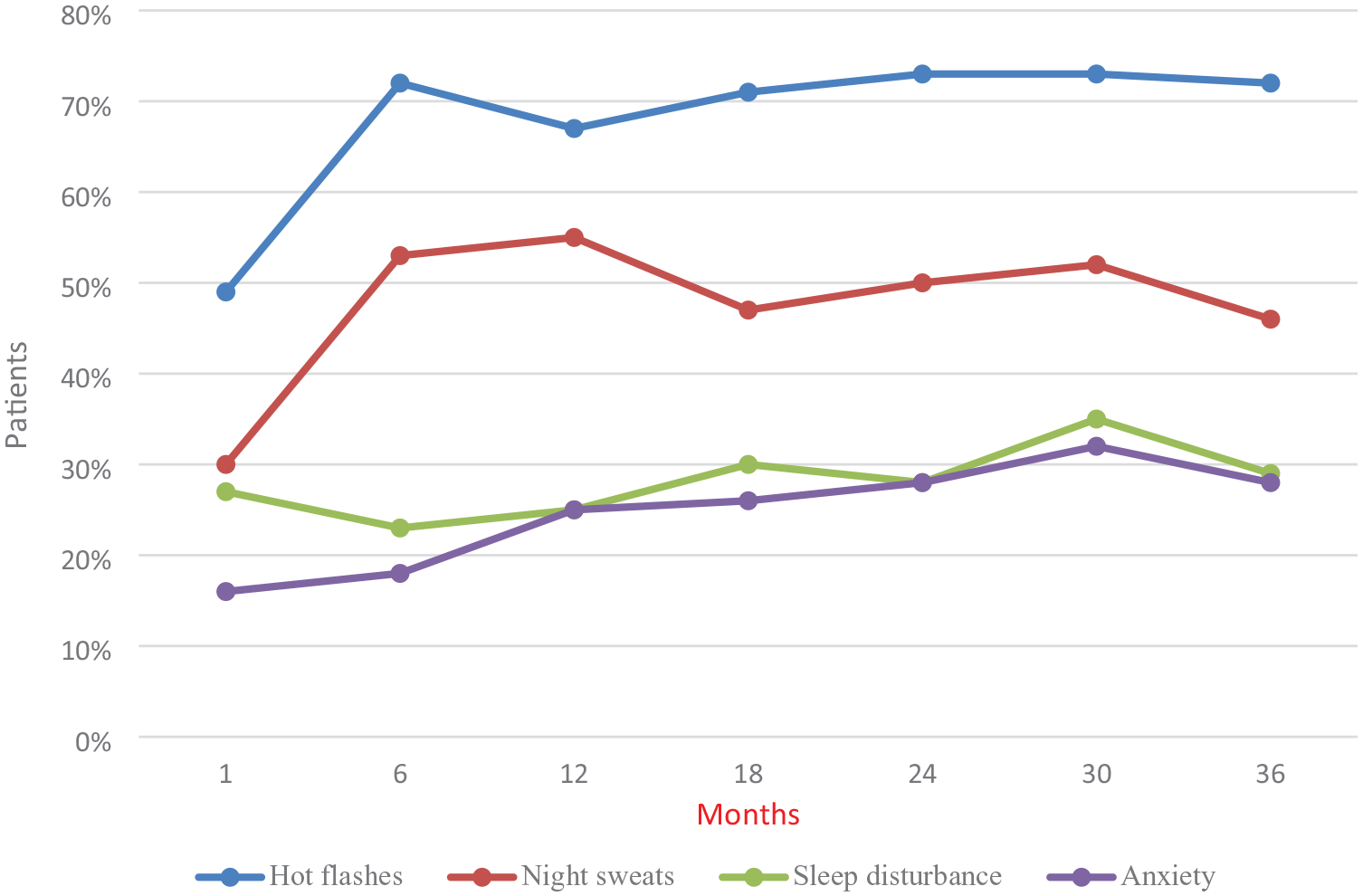
The frequency of complaints in the entire population. Vasomotor symptoms were more frequent than sleep disturbance and anxiety at any time point.
Sleep disturbances were reported in about one-third of the population, with a linear increase in frequency during treatment (minimum 23% at the 6th month, maximum 35% at the 30th month). Anxiety showed a slight increase in frequency during the first 12 months, then remained consistent (minimum 16% at baseline, maximum 32% at the 30th month).
Forty-six women were under 40 years of age (⩽40). Women in this group reported more frequent night sweats and sleep disturbances (Figure 2(a)). A significant difference in the frequency of night sweats (63.04% vs 42.59%, p < 0.04, Figure 2(b)) and sleep disturbances (45.65% vs 25.93%, p < 0.03) was observed at the 30th month of LHRH analog use, with younger women experiencing higher frequencies compared to women aged 40 years and older. In addition, at the 12th month of treatment, the frequency of night sweats in younger women was 65.22%, compared to 46.3% in older women (p < 0.05). Other symptoms showed similar frequencies between the groups, with no statistical significance.

(a) The frequency of complaints by age groups (⩽40 vs >40). The graph shows a statistically significant difference: at 12 months, night sweats 65.22% versus 46.32% (p < 0.05); at 30 months, night sweats 63.04% versus 42.59% (p < 0.04), and sleep disturbance 45.65% versus 25.95% (p < 0.03). All of the above complaints were more common in younger women (⩽40 group). (b) Statistically significant difference in night sweats’ frequency according to age groups (⩽40 vs >40), at month 30 of therapy.
Initially, anxiety was less frequently reported in the group of women under 40 years of age. During treatment, the frequency of anxiety worsened and increased linearly over time. Except at the baseline, anxiety was more frequently reported in younger women at subsequent time points (minimum 13.04% at baseline, maximum 36.96% at the 30th month).
The cohort with prior chemotherapy included 47% of the patients. Hot flashes were slightly more common in the group that did not receive chemotherapy (minimum 49.06% at baseline, maximum 81.13% at the 24th month) at each time point, though this difference was not statistically significant (Figure 3(a)). The frequency of night sweats reported by women treated with chemotherapy was generally lower, except at the 24th and 30th months, when the frequencies were nearly equal. A statistically significant difference in the frequency of night sweats was observed at the 6th month of treatment, where the frequency in patients not receiving chemotherapy was 64.15%, compared to 40.43% in the chemotherapy group (p < 0.01, Figure 3(b)). Furthermore, in the final month of follow-up, night sweats improved in the chemotherapy group compared to earlier months, although the frequency did not return to baseline levels.

(a) The frequency of complaints by previous ChT (without ChT vs with ChT). A statistically significant difference was found at month 6 of treatment for night sweats, 64.15% versus 40.43% (p < 0.01); night sweats were significantly more common in the group of women who did not receive chemotherapy. (b) Statistically significant difference in night sweats’ frequency according to previous ChT, at month 6 of therapy.
At all time points, sleep disturbances were less frequent in patients who had not previously undergone chemotherapy, except at the 12th month, when the frequency was nearly equal between the groups (26% vs 23%). After 36 months, there was no difference in the frequency of sleep disturbances between the groups (28% vs 29%). There was no statistically significant difference in the frequency of anxiety between the two groups. However, there was a numerical difference at some time points. In the first 6 months, anxiety was more frequent in patients who did not receive chemotherapy (20.75% vs 14.89%). No numerically significant differences were observed between the groups at other time points. Anxiety had the lowest frequency at each time point compared to other symptoms. After 36 months of treatment, there was no difference in the expression of anxiety between the groups (28% vs 27%).
All four analyzed symptoms showed a trend of declining by the end of therapy (36 months), but the percentage of patients reporting side effects remained higher compared to baseline values.
During a median follow-up period of 18 months, after completion of the LHRH analogs, 50% of patients continued to report symptoms. Among them, 72% indicated that their symptoms had diminished after stopping the LHRH analogs, with vasomotor complaints being the most prevalent issue.
The majority of patients did not utilize symptomatic therapy for their menopausal symptoms. Insomnia was managed using alternative therapies (in less than 4%) or benzodiazepines (16%). Gabapentin was utilized by 4% of patients to alleviate vasomotor symptoms; however, all patients discontinued it on their own due to poor tolerance. In total, 56% of the patients engaged in regular physical activity, and they did not report a noticeable reduction in vasomotor symptoms. In addition, 24% of the patients experienced weight gain, with an average increase of 9 kg during the treatment period.
Restoration of the menstrual cycle occurred in 36% of the patients, with an average time of 8 months following the conclusion of treatment with LHRH assays.
Discussion
The results of our analysis demonstrate that patients with early HR+ breast cancer, who are at moderate to high risk of recurrence and receiving adjuvant ET with OFS, frequently experience therapy-related symptoms. The most common symptoms include vasomotor disturbances, particularly hot flashes and night sweats, which primarily result from OFS-induced menopause. Hot flashes are common side effects of both tamoxifen and aromatase inhibitors, with patients receiving tamoxifen plus OFS experiencing them more severely. 8 These episodes can occur as frequently as once per hour, both day and night, and significantly impact quality of life. Furthermore, they are associated with negative health outcomes, such as an increased risk of cardiovascular issues and greater bone loss. In addition, hot flashes may contribute to lower treatment adherence.11,12 The side effects of OFS mirror the symptoms of estrogen deprivation and may include hot flashes, sweating, weight gain, reduced libido, muscle and joint pain, bone density loss, as well as sleep disturbances and new-onset depression. 3
Vasomotor symptoms, including hot flashes and night sweats, were the most reported side effects at each time point in our analysis. The highest frequency of hot flashes (72%) and night sweats (53%) occurred during the first 6 months of treatment, marking the most significant change compared to baseline data. Hot flashes were the most frequently reported symptoms throughout the entire analysis. Most patients experienced a stabilization of these symptoms after 12 and 24 months; however, a majority continued to experience some level of these symptoms throughout the entire treatment period. After the first 6 months and continuing until the end of treatment, about 70% of patients reported having hot flashes, and night sweats persisted in approximately half of the patients (ranging from 46% to 55%). These findings are consistent with other studies that report vasomotor symptoms as the most common issue among patients undergoing adjuvant ET with LHRH analogs, with hot flashes occurring in 80% of patients and sweating in 49%, with the hot flashes greatest worsening during the first 6 months of treatment.3,8 Women with breast cancer experience more frequent hot flashes than other women for several reasons. 13 Premenopausal women may suddenly become estrogen deficient due to chemotherapy, which causes early ovarian failure, or because of OFS with LHRH analogs or bilateral oophorectomy. In addition, they will receive tamoxifen or aromatase inhibitors, both of which are adjuvant ETs linked to hot flashes and night sweats in up to 80% of women. 13
We analyzed symptoms due to age difference in patients ⩽40 and >40 years. Hot flashes were less common at baseline in patients aged 40 and younger. Among these patients, 56% received chemotherapy, and 10% were under 35 years old, with 70% of this younger group undergoing chemotherapy. The greatest increase in the frequency of hot flashes was reported during the first 6 months of therapy. After this period, hot flashes were similarly distributed between the two groups (40 and younger vs above 40 years), maintaining relatively stable frequencies above baseline levels, up to 70% for both groups. Night sweats, like in the overall population of women, were less commonly reported compared to hot flashes in both groups (⩽40 and >40 years) at every time point. Night sweats were more frequent in patients older than 40 years at baseline, but after the 6th month, they became more common in the group of patients aged 40 and younger with the largest increase in night sweats within 12 months (from 26% in the first month to 65% in the 12th month). The difference in the night sweats was the most notable distinction between these two groups. While we currently do not have an explanation for this, some studies suggest a link between specific gene expressions and vasomotor symptoms. Literature also indicates an association between race, vasomotor symptoms (hot flashes, night sweats, and/or cold sweats), and certain polymorphisms in sex steroid-metabolizing enzymes and sex steroid receptors. 14 Neither hot flashes nor night sweats returned to baseline levels by the end of 36 months of LHRH therapy. While in the SOFT study, vasomotor symptoms were less severe in women under 35, it is important to note that 95% of these women received chemotherapy in comparison with 56% in our analysis. The CANTO study found that the toxicity of ET varied depending on menopausal status; premenopausal women receiving OFS experienced more pronounced hot flashes, night sweats, and mood disturbances. 10 Among patients included in our analysis, a significant difference between the two groups (⩽40 and >40 years) was found for night sweats and sleep disturbance at the 30-month point and the 12-month point for sleep disturbance, with higher frequency in patients ⩽40 years.
Symptoms reported by patients were analyzed based on prior chemotherapy treatment. We found that hot flashes and night sweats were more common in patients who had not undergone chemotherapy. It is important to note that during chemotherapy, patients are often not specifically asked about menopausal symptoms, which may result in underreporting of these issues, or symptoms can be masked by symptoms caused by chemotherapy. At each time point, but especially during the first 6 months, hot flashes were more frequently reported by those who had not received chemotherapy. In the SOFT study, patients who had previously undergone chemotherapy reported less severe vasomotor symptoms both at baseline and during follow-up. Symptoms were more pronounced in the group receiving tamoxifen plus OFS compared to those receiving an aromatase inhibitor plus OFS. 8 By month 60, there was little difference in the frequency of hot flashes and sweating between the tamoxifen plus OFS group and the tamoxifen-only group, and endocrine symptoms due to different treatments diminished after 2 years and became clinically insignificant after 5 years partially due to average age at that point was 48, which corresponds to the typical age of menopause. 9 Night sweats were also frequently reported in our analysis, with the most significant statistical difference within the first 6 months in women who had not received chemotherapy in comparison with those who had (64.15% vs 40.43%, p < 0.01). However, in the chemotherapy group, a slight increase in night sweats frequency was observed between 24 and 30 months. The median age in the chemotherapy cohort was 42 years, compared to 45 years in the non-chemotherapy group, which may explain this difference. By the end of the treatment (36th month), the frequency of night sweats was 52% in the non-chemotherapy group compared to 38% in the chemotherapy group, confirming previous observations. Since nearly 95% of patients in our study received tamoxifen along with an LHRH analog as adjuvant ET, we were unable to compare the differences in symptom frequency between tamoxifen and aromatase inhibitors. However, this made the group analysis more uniform. It is important to note that while our analysis focused on symptom frequency, the SOFT and TEXT subgroups specifically examined the intensity of symptoms.
Regarding non-vasomotor symptoms, the most reported in our analysis were sleep disturbance and anxiety, both of which tended to worsen over time. According to the SOFT-TEXT trial, more than 50% of patients receiving OFS and ET reported insomnia. 3 We observed that a higher percentage of women ⩽40 years reported insomnia and anxiety at each time point, with the peak occurring at 30 months after the start of therapy. At this point, the difference in insomnia between women ⩽40 versus >40 years became statistically significant. This was accompanied by a statistically significant increase in night sweats, which also peaked at 30 months and was the dominant symptom among women aged 40 and younger. The timing of these symptoms likely reflects the impact of night sweats on sleep quality, particularly global sleep dissatisfaction and difficulty maintaining sleep.15,16 In the general population, hot flashes are uncommon in premenopausal women, especially those under 40, and are typically followed by less sleep disturbance. However, peri- and postmenopausal women often experience more severe hot flashes, leading to worsened insomnia. 15 Anxiety, which is commonly present in peri- and menopausal women, is another contributing factor to insomnia.16,17 Despite treatment for vasomotor symptoms, sleep problems can persist due to anxiety and primary sleep disorders. 17 Although some differences were noted, there was no statistically significant variation in insomnia and anxiety between patients with and without a history of chemotherapy. However, women previously treated with chemotherapy did report a higher percentage of sleep disturbance and anxiety at the 30-month mark. In the SOFT and TEXT trials, patients who received prior chemotherapy experienced worsening vasomotor symptoms, insomnia, and anxiety from baseline to 6 months, but these symptoms improved over time, with anxiety returning to baseline levels. 7 This was different from our analysis, where both anxiety and sleep disturbance remained high at the 30-month point. This observation warrants further investigation and follow-up in clinical practice, as it may significantly impact quality of life and therapy adherence.
Our study provides valuable insights into the side effects and symptoms associated with adjuvant ET in young women with HR+ breast cancer, as it is based on real-world clinical data and patient-reported medical histories. We acknowledge that our study has some limitations. This is retrospective, not preplanned analysis, and might have biases depending on the attending oncologist who recorded patients’ symptoms, with the note that all oncologists at the institute have specialized training in supportive therapy. In addition, the Institute is an ESMO-designated center for supportive and palliative care. This analysis may be subject to bias due to the non-uniform collection of data on supportive measures used to treat or prevent well-known side effects of ET. Although most patients did not report using any symptomatic treatments, there is a need for greater consistency in delivering clear messages to patients about the value and importance of supportive care. Unfortunately, there are currently no validated questionnaires specifically designed to capture the adverse effects of endocrine suppression in premenopausal women within real-world clinical practice. The development of such tools—especially digital platforms accessible from home—would facilitate systematic monitoring of medical interventions and enable consistent data collection across clinical settings. Unlike the SOFT and TEXT trials, our analysis focused on the frequency of reported symptoms at defined time points, rather than assessing their severity, which would require additional time and resources in routine clinical practice.
This analysis does not include long-term follow-up data on symptom resolution or impact on quality of life, as patients transitioned to outpatient follow-ups, either every 6 months or annually, with their oncologist, within or outside the Institute, after completing LHRH treatment in the daily hospital. Nonetheless, we consider this study a valuable contribution to real-world data, providing oncologists with insights into the frequency and progression of side effects over 36 months of LHRH treatment. Our analysis also demonstrates that, even in routine clinical practice and with a large patient population, it is feasible to assess symptoms and adverse effects of therapy in greater detail. However, the findings also underscore the need for standardized monitoring practices to support more consistent conclusions and guide the implementation of targeted measures to alleviate these symptoms. Women undergoing long-term ET are generally considered healthy individuals, and we firmly believe that the quality of life in young women with breast cancer can be improved through the timely recognition and management of challenging side effects. Data from our analysis suggest that patients are not adapting to the effects of premature menopause. A significant worsening of vasomotor symptoms is observed not only within the first 6 months but also throughout the entire course of therapy. Recognizing this allows for the provision of effective supportive care and may help reduce treatment nonadherence. Notably, a high number of patients reported increased anxiety and sleep disturbances at the end of therapy, indicating the need for continued follow-up. One possible contributing factor, beyond the adverse effects of ET, may be the transition away from regular contact with their oncologist after completing LHRH therapy, which could lead to increased uncertainty regarding the further course of treatment.
Several important aspects of adjuvant ET in Serbian clinical practice require closer attention and consideration. In recent years, the use of aromatase inhibitors has evolved, particularly considering updated findings from the SOFT and TEXT studies.6,18 However, tamoxifen remains the standard treatment for many patients, with approximately 95% of patients receiving tamoxifen when they begin their adjuvant therapy. In addition, the current indication from medical authorities in Serbia specifies that aromatase inhibitors should be used in one of two ways: either sequentially after 3 years of tamoxifen therapy or as upfront therapy for patients who are not candidates for chemotherapy. In cases where chemotherapy is contraindicated, aromatase inhibitors are recommended as the initial treatment. Recently, adjuvant abemaciclib has been approved in Serbia for high-risk HR+ eBC patients, based on the criteria outlined in the MonarchE trial. However, none of the patients in our cohort received this treatment. Finally, a shorter duration of OFS, 3 years, is still considered the standard of care in Serbia. This contrasts with current international guidelines, which, based on the findings of the SOFT and TEXT trials, now recommend a 5-year duration of OFS. While one trial has explored a shorter, 2-year course of OFS with promising results, 19 no direct comparison has been made between 2 and 5 years of treatment. Current guidelines suggest that shorter durations of OFS should only be considered for patients who do not tolerate the standard treatment.
Finally, we would like to emphasize potential preventive measures that could help reduce the adverse effects of ET. One key finding in our analysis was that most women experienced weight gain, averaging up to 9 kg, while nearly half of them reported engaging in some form of physical activity. Unfortunately, our analysis did not include data on the type or frequency of physical activity, specific exercise programs, dietary habits, or any diets and supplements used by the patients. In recent years, leading oncology centers have increasingly established dedicated clinics for the treatment of young women with breast cancer to provide a comprehensive approach to their treatment and the supportive care they need. Further studies, utilizing dedicated and objective measurements, may help identify patients experiencing severe side effects of adjuvant ET for eBC treatment. In addition, these studies could explore how biological factors contribute to the inter-individual variability observed in treatment responses.
Conclusion
Adverse effects of adjuvant ET and OFS are common in real-world clinical practice. The most frequently reported side effects in our analysis were vasomotor symptoms, such as hot flashes and night sweats, with anxiety and sleep disturbances also commonly observed. Importantly, vasomotor symptoms worsened significantly during the first 6 months of follow-up in all patients and remained highly prevalent until the end of treatment. Anxiety and sleep disturbances showed a tendency to worsen, particularly toward the end of treatment, suggesting that supportive measures may not have been adequately implemented. These issues were especially pronounced in women under the age of 40. Given the complexity of breast cancer treatment in younger women, we emphasize the importance of understanding the dynamics, frequency, and severity of side effects through dedicated programs specifically tailored to their care in routine clinical practice.
