Abstract
Background:
Chronic airway inflammation in asthma and/or chronic obstructive pulmonary disease (COPD) is presumed to be protumorigenic. The tumor inhibitory effect of inhaled corticosteroids (ICSs) used to reduce airway inflammation in patients with asthma and COPD remains unclear.
Objectives:
This study aimed to evaluate the impact of coexisting asthma and/or COPD on the survival of patients with lung adenocarcinoma. The effects of ICS treatment were also assessed.
Design:
This retrospective, real-world cohort study was conducted at a cancer center.
Methods:
The overall survival of a cohort of 1524 consecutive patients with lung adenocarcinoma who were enrolled between January 2011 and December 2019 and followed up until December 2022 was analyzed, followed by subgroup comparisons.
Results:
A total of 283 patients had coexisting asthma and/or COPD. Among them, 212 had used ICSs. ICS users were predominantly women, older, and had more advanced-stage disease; moreover, there were fewer tobacco smokers, fewer comorbidities, and relatively severe obstructive impairments than non-ICS users. When restricted to stage 0–II diseases, patients with coexisting asthma and/or COPD had a lower 5-year overall survival rate (77% vs 90%, p < 0.001), with a hazard ratio of 1.8, in contrast to no difference among patients with stage III–IV disease. ICS users had a lower 5-year overall survival rate in both subgroups, although the difference was not statistically significant.
Conclusion:
The impact of cancer on prognosis may overwhelm the effects of asthma and/or COPD in patients with advanced-stage lung adenocarcinoma. The evaluation of the effects of ICS treatment appears to be confounded by intent and compliance, which can introduce bias in the opposite direction. However, investigating the treatment effects on asthma and/or COPD control would be beneficial. A systematic prospective study is required to define the role of the ICS.
Keywords
Introduction
Lung cancer and chronic obstructive pulmonary disease (COPD) share common risk factors, particularly tobacco smoking.1,2 After multivariate analysis has excluded the effects of smoking, COPD is associated with an increased lung cancer risk and a poor survival rate.3,4 The underlying mechanism is not very well understood. Genomic, immune, and microenvironment dysregulation contributes to chronic inflammation have been proposed as major contributing factors. Similar trends were also noted for asthma and recently introduced asthma and COPD overlap, although they were less significant.5–7 However, as the results were inconsistent, more randomized controlled studies are required.
Inhaled corticosteroids (ICSs) are well tolerated in patients with asthma and/or COPD. 8 In addition to the beneficial control of airway diseases, there is epidemiological and circumstantial evidence of a protective effect of ICSs against lung cancer development in highly vulnerable patients with asthma and/or COPD. 9
Corticosteroids are effective in reducing the number of lung tumors in A/J mice and rats exposed to cigarette smoke. 10 The tumors that developed were all classified as adenocarcinomas and their precursor adenomas. The lack of efficacy of budesonide in regressing bronchial dysplasia may not reflect the lack of efficacy in the peripheral lung. 11 Whether ICSs decrease the risk of lung cancer in patients with COPD remains controversial.12,13 Results from randomized controlled trials have demonstrated that ICSs do not affect the risk, whereas observational studies have reported that ICSs reduce the risk, particularly at a higher dose. The interesting effects of budesonide on computed tomography-detected peripheral lung nodules suggest a biological effect that needs to be explored. 14
The release and action of the proinflammatory cytokines, such as interleukin (IL)-6 and tumor necrotic factor alpha (TNF-α), in the chronic inflammation microenvironment are believed to be protumorigenic. In addition to the reduced risk of lung cancer reported in the literature, ICSs may inhibit cancer progression by inhibiting proinflammatory and protumorigenic cytokines. In this retrospective cohort study, we aimed to evaluate the impact of asthma and/or COPD on the survival of patients with lung adenocarcinoma. We also hypothesized that patients with lung adenocarcinoma who used ICS would have longer survival than nonusers, regardless of asthma and/or COPD. This “proof of concept” study from a clinical practice may enhance the compliance for asthma and/or COPD control in patients with lung adenocarcinoma.
Methods
Study population
Consecutive patients with lung adenocarcinoma aged >18 years were prospectively enrolled in an electronic database, the Cancer Information System (CIS), at the Sun Yat-Sen Cancer Center, Taipei, Taiwan, between January 2011 and December 2019 and were followed up until December 2022. Lung adenocarcinoma was defined according to World Health Organization pathology codes (81402, 81403, 82503, 82523, 82533, 82543, 84803, and 85603). The patients were clinically and pathologically staged and received homogeneous treatment according to established guidelines. In particular, the CIS included data on smoking habits, which are decisive prognosticators for patients with lung adenocarcinoma. 15 The National Death Registry Database was linked for survival analysis. The index date was defined as the date of diagnosis of lung adenocarcinoma. The participants were censored at death and at the last follow-up. Patients with a history of cancer were excluded.
We identified patients with asthma and/or COPD among the cohort based on their conformance to meet the following criteria: (1) an International Classification of Disease, 9th or 10th revision-coded diagnosis consistent with asthma or COPD (ICD-9:496.x, 493.2, or ICD-10: J42, J 43.9, J44.9, J45.909); (2) detailed medical records review containing a clinical diagnosis through their history, which included self-report, physical findings, pulmonary function testing results, and prescriptions of bronchodilators.
Pulmonary function testing was performed using a SensorMedics Vmax System (Tucson, AZ, USA). The severity of obstructive ventilatory impairment was evaluated using the absolute forced expiratory volume in 1 s (FEV1), forced vital capacity (FVC) values, and the FEV1/FVC ratio. Based on the FEV1/FVC ratio, the patients were classified as mildly obstructive and scored 1, with a value between 60% and 69% predicted; moderately obstructive and scored 2, with a value between 45% and 59% predicted; and severely obstructive and scored 3, with a value <45% predicted. A positive response to a bronchodilator was defined as an increase of ⩾12% and ⩾200 mL as an absolute value compared with a baseline in either FEV1 or FVC.
ICS treatment
We identified patients with asthma and/or COPD who were prescribed ICSs as inpatients and/or outpatients. The ICSs included budesonide or fluticasone alone or in a combination inhaler with an inhaled β2 agonist. The indications for ICS treatment followed the guidelines of the Global Strategy for Asthma Management and Prevention (2022 update) and Global Initiative for Chronic Obstructive Lung Disease (2022 update).
For regular control, one canister of Symbicort® Turbuhaler (contains budesonide 160 μg and formoterol 4.5 μg/puff), Seretide® 50 Evohaler (contains fluticasone 125 μg and salmeterol 25 μg/puff), Duasma® MDI (contains budesonide 200 μg/puff), or Pulmicort® Turbuhaler (contains budesonide 200 μg/puff) was prescribed monthly for each patient. Equivalencies are based on the relative topical potency and dosages that experts deem comparable. On the assumption of good compliance, a daily dose of 640 μg budesonide in Symbicort Turbuhaler (two puffs twice daily), 800 μg budesonide in Duasma MDI (two puffs twice daily) or Pulmicort Turbuhaler (two puffs twice daily), and fluticasone 500 μg in Seretide Evohaler (two puffs twice daily) are considered equivalent. 16
Information regarding ICS prescriptions, including prescription dates, daily dose prescribed, and duration of prescription, was collected. This study was limited to ICS treatment-naïve patients prior to the index date. An ICS user was defined as a user with an ICS prescription for ⩾3 months after the index date. The date of the first ICS use was considered the initiation date.
Outcomes
Overall survival was analyzed. The clinical prognostic factors analyzed included stage, age, sex, smoking history, and the presence of coexisting asthma and/or COPD. Among patients with lung adenocarcinoma and coexisting asthma and/or COPD, survival was compared between ICS users and nonusers. The degree of obstructive ventilatory impairment was also assessed.
Covariates
We included other covariates that probably affected the prognosis, such as educational level, geographic distribution, alcohol consumption level, betel nut chewing, body mass index, and comorbidities, in the subgroup comparisons.
The medications for asthma and/or COPD other than ICSs, including theophylline, leukotriene modifiers, and other inhaled medications, such as short-acting or long-acting β2 agonists and short-acting or long-acting muscarinic antagonists, were not adjusted in this study. The use of oral corticosteroids was also not adjusted for, considering the various indications in patients with cancer.
Statistical analyses
The patient demographics and subgroups were compared. Descriptive statistics of frequency, percentage, mean, median, standard deviation, and 95% confidence interval (CI) were used to process the demographic data. Categorical data were compared using Fisher’s exact test or Pearson’s χ2 test, and continuous data were compared using the Wilcoxon rank-sum test.
Survival estimates were derived using Kaplan–Meier plots, whereas log-rank tests were used to assess differences in survival among subgroups. Multivariate analysis of the overall survival was performed according to the Cox proportional hazards model using clinical prognostic factors with significant differences in univariate analysis. The two-sided p < 0.05 was considered statistically significant. Analyses were performed using the SAS statistical software package version 9.4 (SAS Institute, Cary, NC, USA).
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology Statement. 17
Results
There were 1703 patients with lung adenocarcinoma diagnosed, staged, and treated at the hospital during the study period. The median follow-up period was 7.16 (95% CI, 6.84–7.40) years. A total of 114 patients who were diagnosed with lung cancer after a previous cancer or secondary lung cancer were excluded. One patient who was <18 years was excluded. Staging data were unavailable for two patients. Through a three-step data validation process, 1524 patients with lung adenocarcinoma were eligible for analysis. Among these, 283 (18.6%) patients had been diagnosed with asthma and/or COPD.
We allocated the patients into three groups: those without asthma or COPD (n = 1241), those with asthma and/or COPD who had used ICSs (n = 212), and those with asthma and/or COPD who had never used ICSs (n = 71). The patient characteristics are compared in Table 1. Patients with coexisting asthma and/or COPD were predominantly men, older, had an early stage disease, had increased tobacco smoking, alcohol consumption, or betel nut chewing; they also had a lower educational level and a higher number of comorbidities.
Patients’ characteristics.

AIDS, acquired immunodeficiency syndrome; COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroid; PVD, peripheral vascular disease; SD, standard deviation.
Among patients with coexisting asthma and/or COPD, ICS users were older and predominantly women, had a more advanced disease stage, had less tobacco smoking or betel nut chewing, and fewer comorbidities. ICS users had a more severe obstructive impairment, that is, a lower FVC (2.5 ± 0.8 vs 3.1 ± 0.9 L), a lower FEV1 (1.6 ± 0.6 vs 1.9 ± 0.7 L), and a higher severity score (p = 0.02) than ICS nonusers.
The 5-year overall survival rate of patients with lung adenocarcinoma was stratified according to disease stage. Younger patients, women, and those without a smoking history had better overall survival in the univariate analyses (Figure 1(a)–(d) and Table 2). No significant difference was observed between patients with lung adenocarcinoma and coexisting asthma and/or COPD and those without (38% vs 39%, p = 0.69, Figure 1(e)). Multivariate analyses showed the disease stage (hazard ratio (HR) = 18.8, 8.4, and 3.2 for stages IV, III, and II, respectively, p < 0.001), age (HR = 1.5 for age ⩾65 years, p < 0.001), and smoking history (HR = 1.5 and 1.2 for current smokers and former smokers (p < 0.001 and p = 0.04), respectively) remained the independent prognostic factors (Table 2). Among patients with lung adenocarcinoma with coexisting asthma and/or COPD, ICS users had a lower 5-year overall survival rate than ICS nonusers (34% vs 49%, p = 0.02, Figure 1(f)).
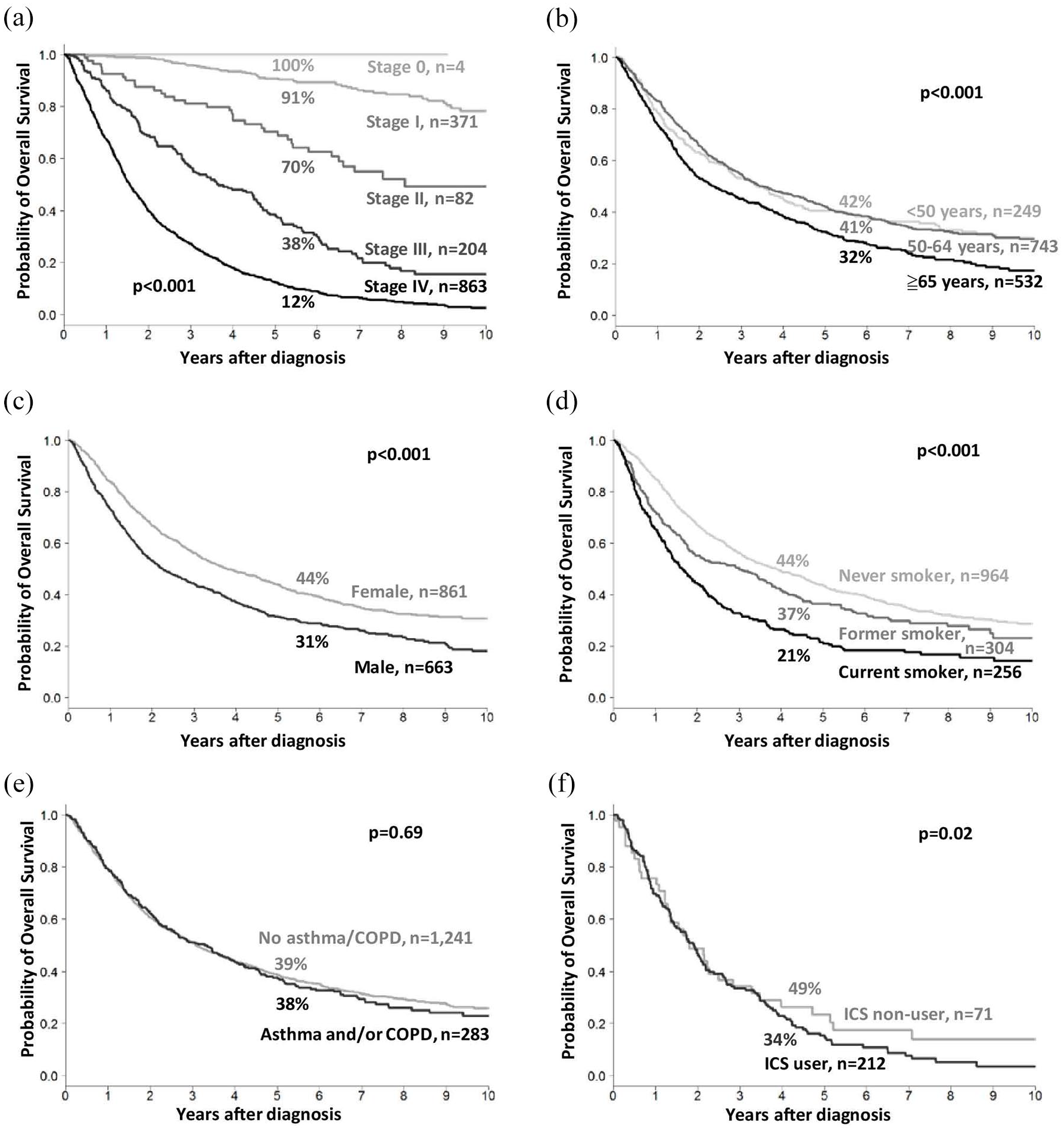
Kaplan–Meier overall survival curves of all patients with lung adenocarcinoma stratified based on (a) disease stage, (b) age, (c) sex, (d) smoking history, (e) coexisting asthma and/or COPD, and (f) inhaled corticosteroid use among patients with lung adenocarcinoma with coexisting asthma and/or COPD.
Cox proportional hazards model analysis of various factors affecting overall survival of patients with lung adenocarcinoma.

CI, confidence interval; COPD, chronic obstructive pulmonary disease; HR, hazard ratio.
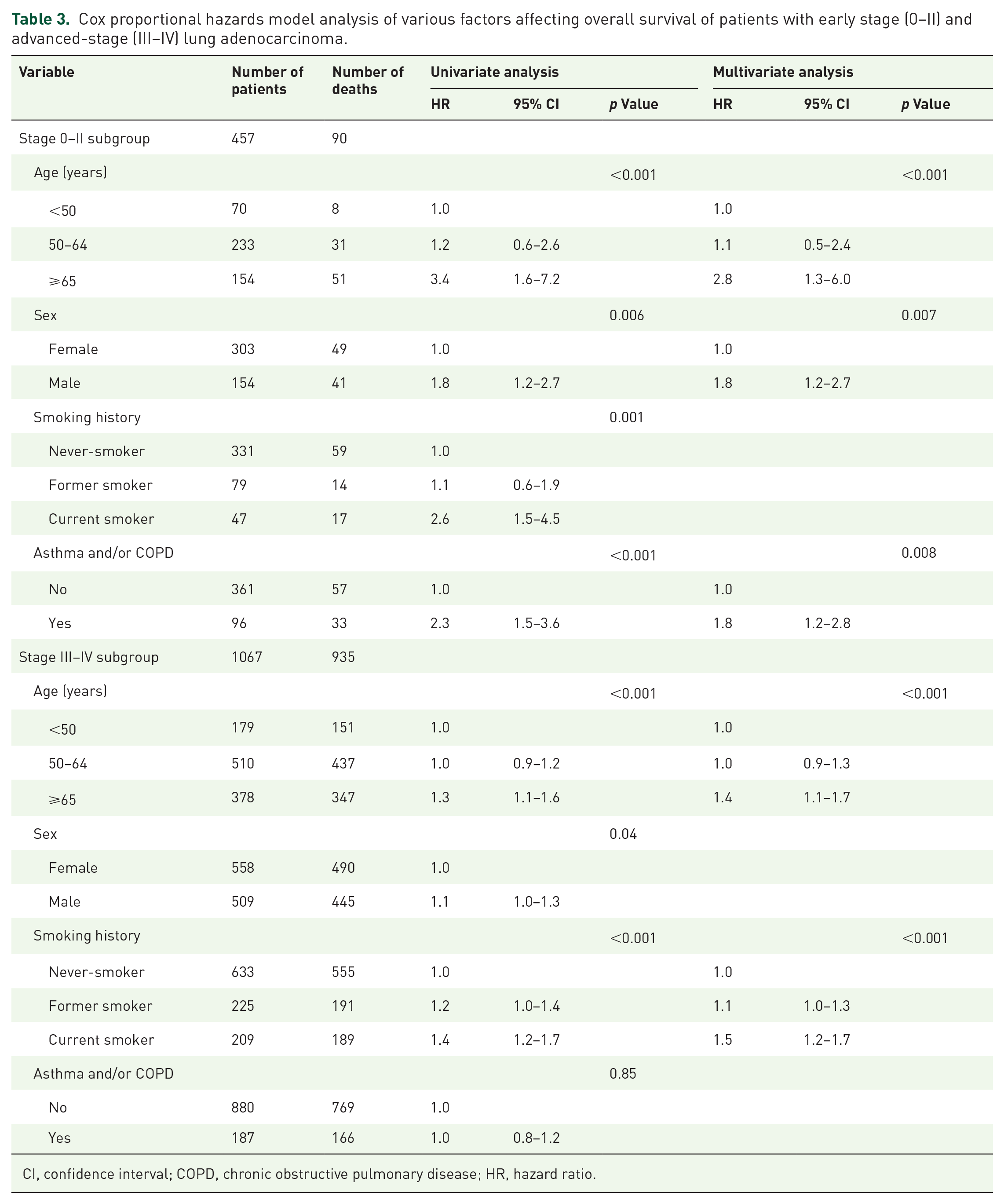
Analyses were subsequently completed for the early stage (stage 0–II, n = 457) and advanced-stage (stage III–IV, n = 1067) lung adenocarcinoma subgroups. There were 96 (21%) early stage and 187 (17.5%) advanced-stage patients with coexisting asthma and/or COPD. Among both subgroups, the patients with coexisting asthma and/or COPD were older, predominantly men, had higher tobacco smoking, alcohol consumption, or betel nut chewing, and had a lower educational level and a higher number of comorbidities.
Among the subgroups of patients with early stage lung adenocarcinoma, those with coexisting asthma and/or COPD had a lower 5-year overall survival rate than those without these conditions (77% vs 90%, p < 0.001, Figure 2(a)). Younger patients, women, those without a history of smoking, and those without asthma or COPD had better overall survival rates in the univariate analyses (Table 3). Subsequent multivariate analyses showed that age (HR = 2.8 for age ⩾65 years; 95% CI, 1.3–6.0; p < 0.001), sex (HR = 1.8 for male; 95% CI, 1.2–2.7; p = 0.007), and coexisting asthma and/or COPD (HR = 1.8; 95% CI, 1.2–2.8; p = 0.008) were the independent prognostic factors. ICS users (n = 67, 69.8%) had a lower 5-year overall survival rate than ICS nonusers (74% vs 86%); however, the difference was not statistically significant (p = 0.12; Figure 2(b)).

Kaplan–Meier overall survival curves of patients with early stage (0–II; a, b) and advanced-stage (III–IV; c, d) lung adenocarcinoma stratified based on coexisting asthma and/or COPD and inhaled corticosteroid use.
Cox proportional hazards model analysis of various factors affecting overall survival of patients with early stage (0–II) and advanced-stage (III–IV) lung adenocarcinoma.
CI, confidence interval; COPD, chronic obstructive pulmonary disease; HR, hazard ratio.
Among the advanced-stage lung adenocarcinoma subgroups, there was no significant difference in the 5-year overall survival rate between patients with coexisting asthma and/or COPD and those without (17% vs 17%, p = 0.85, Figure 2(c)). Younger patients, women, and those without a smoking history had better overall survival rates in the univariate analyses (Table 3). Subsequent multivariate analyses showed that age (HR = 1.4 for age ⩾65 years; 95% CI, 1.1–1.7; p < 0.001) and smoking history (HR = 1.5 for current smokers; 95% CI, 1.2–1.7; p < 0.001) were the independent prognostic factors. ICS users (n = 145, 77.5%) had a lower 5-year overall survival rate than ICS nonusers (15% vs 23%); however, the difference was not statistically significant (p = 0.35; Figure 2(d)).
Discussion
To the best of our knowledge, this is the first study to use a systematic approach to evaluate the impact of coexisting asthma and/or COPD on the survival of patients with lung adenocarcinoma and to explore the role of ICSs. To reduce the confounding effects of tobacco smoking, we restricted the phenotype to the most prevalent lung adenocarcinoma in Taiwan, with undetermined etiology and minimum relevance to smoking history.
A total of 283 patients with lung adenocarcinoma (18.6%) were diagnosed with asthma and/or COPD. Among these, 212 (74.9%) were treated with ICSs. This study demonstrated that coexisting asthma and/or COPD adversely affected the prognosis of patients with early stage lung adenocarcinoma (Figure 2(a) and Table 3). Coexisting asthma and/or COPD was not a significant prognostic factor in patients with advanced-stage lung adenocarcinoma (Figure 2(c) and Table 3). The impact of lung cancer on prognosis may overwhelm the effects of asthma and/or COPD.
The mechanisms by which asthma and/or COPD adversely affect the prognosis of patients with lung adenocarcinoma remain unclear. Severe lung inflammation, chronic systemic inflammation, DNA repair, apoptosis, and other associated comorbidities have also been reported.
Chronic inflammation was proposed as a factor of carcinogenesis and may have adversely affected the prognosis of lung adenocarcinoma.18–22 The effect of ICSs on carcinogenesis may be the result of an influence on chronic inflammation. 23 ICSs alone or in combination with β2-adrenoceptor agonists reduce systemic inflammation in patients with COPD, which is associated with major complications, such as weight loss, cachexia, osteoporosis, cardiovascular diseases, and cancer. ICSs increase the capacity of alveolar macrophages to express the anti-inflammatory cytokine, IL-10 mRNA, and release IL-10 in patients with asthma. 24 Chronic ICS therapy shifts the balance of pro- and anti-inflammatory cytokine expression and release from the alveolar macrophages in patients with asthma.
In addition, ICSs affect mouse lung carcinogenesis through the modulation of the Bcl-2- and caspase-2-regulated apoptosis pathways and the Mad2/3-regulated mitotic checkpoint. 25 Sohal et al. 26 demonstrated that ICSs caused a reduction in epidermal growth factor receptor (EGFR) expression in the airway epithelium. Dexamethasone induces estrogen sulfotransferase to decrease estradiol levels in tumor tissues and suppress A549 xenograft tumor growth. 27
Contrary to the trend toward a reduction in the lung cancer risk for ICS use in patients with asthma and/or COPD, there was no reduction in mortality from lung adenocarcinoma for ICS users in this study, contrary to our hypothesis. In both patients with early- and advanced-stage lung adenocarcinoma with coexisting asthma and/or COPD, ICS users had shorter survival than ICS nonusers, although the difference was not statistically significant (Figure 2(b) and (d)). These results appear to be confounded by the severity of lung diseases or the symptoms of lung cancer. ICS users were older and more likely to be women, had a more advanced stage, had less tobacco smoking or betel nut chewing, fewer comorbidities, and relatively severe obstructive impairments. The intent and compliance of the ICS treatment can introduce bias in the opposite direction. 12 According to Parimon et al.’s study, a strong dose–response relationship for ICSs and risk of lung cancer among patients with COPD was observed in the opposite direction of the anticipated results. However, ICSs reduced the risk when they restricted the eligibility criteria to never-smokers.
ICSs can decrease the local immunity of the lung and lead to an increased incidence of pneumonia or tuberculosis, resulting in cancer-specific survival being a better estimate. 28 ICSs simultaneously exert tumor-suppressive properties while diminishing the efficacy of immune-mediated therapies. We restricted the ICS users to the patients who used three or more canisters of ICSs, that is, chronic users, and analyzed them as categorical data. When cumulative doses are available and analyzed as continuous data, the dose–response relationship may be helpful in evaluating the effect of ICS use on mortality. Adjusting for significant covariate comorbidities using Deyo’s Charlson Comorbidity Index, a validated risk adjustment score that predicts mortality and resource utilization, may be more accurate. 29
This study has several strengths. First, we studied patients at a single cancer center, which minimized the variation in diagnostic or treatment patterns between physicians or institutes when compared with nationwide population-based cohort studies. Restricting the sample to patients with adenocarcinoma minimized the bias related to the cell type. Women comprised a significant proportion of the patients, which made the results generalizable. Smoking history was available in the registry, which is often lacking in nationwide databases, such as the National Health Insurance Research Database in Taiwan or the Surveillance, Epidemiology, and End Results program. We performed spirometry to confirm the diagnosis of asthma and/or COPD and estimate its severity. We controlled for important confounders, such as comorbidities.
However, this study has some limitations. The ambiguity in the diagnosis and classification made the differentiation between asthma and COPD difficult in some patients.30,31 Relied on ICD coding and clinical records may result in diagnostic overlap, such as asthma-COPD overlap, and missed diagnosis. Disease heterogeneity may have affected the results. Pulmonary function testing may not reflect the severity of asthma or COPD. The frequency of short-acting rescue inhaled bronchodilators was not determined. The effects of other drugs on asthma and/or COPD, such as oral corticosteroids, theophylline, and leukotriene modifiers, have not been evaluated. They may affect cancer progression through systemic immune suppression.32,33 This study was a retrospective cohort study. In addition to selection bias and residual confounding factors, it was difficult to completely avoid “reverse causality” bias. ICS users had a more severe obstructive impairment. They may receive treatment for more severe conditions, and their lower survival may be related to the severity of underlying diseases rather than the ICS itself. More severe symptoms in patients with advanced-stage lung adenocarcinoma led them to pursue medications, followed by better compliance, as mentioned above. Although some patients may be significantly sick to use ICSs.
Conclusion
The coexistence of asthma and/or COPD adversely affects the prognosis of patients with early stage lung adenocarcinoma. They need intensified asthma and/or COPD treatment to improve survival. In advanced stages of the disease, the impact of lung cancer on prognosis may overwhelm the effect of asthma and/or COPD. Asthma and/or COPD are still crucial in spite of many confounding factors. Determining the benefit of ICSs on survival was challenging owing to the data limitations and influence of various prognostic factors, treatment intentions, and adherence levels. To clearly establish the role of ICSs, a systematic prospective study with a larger patient cohort and more restrictive criteria, including disease severity, indication, and adherence, is required. The study results remained at the observation level. Detection of proinflammatory cytokines such as IL-6, TNF-α, cancer drivers such as EGFR, or immune checkpoint status such as PD-1/PD-L1 will allow delving into the biological mechanisms of “chronic inflammation promoting cancer.”34–37
