Abstract
Background:
Introduced by the International Association for the Study of Lung Cancer (IASLC) in 2005, uncertain resection (Run) categorizes a new subclass of residual tumor. Despite several studies, the prognostic significance of Run in operable non-small cell lung cancer (NSCLC) remains unclear.
Objectives:
This study aimed to investigate the prognostic influence of Run in operable NSCLC, focusing on the impact of the four elements that comprise R descriptors on patient survival.
Design:
A systematic review and meta-analysis were conducted to synthesize data from relevant clinical studies.
Methods:
We developed search strategies to identify relevant clinical studies across databases including PubMed, Embase, Cochrane Library, Web of Science, CNKI, and Wanfang up to June 2024. Quantitative analysis was performed with Stata 15 to investigate the prognostic influence of Run, the extent of mediastinal lymph node removal, and the highest mediastinal lymph node involvement (HMLI). We also summarized the main findings from studies on pleural lavage cytology (PLC) and carcinoma in situ in operable NSCLC.
Results:
Compared to complete resection, Run-associated patients exhibited inferior 5-year overall survival (OS) and disease-free survival (DFS; risk ratio (RR) = 1.31, 95% confidence interval (CI) 1.19–1.44; RR = 1.43, 95% CI 1.28–1.60). Limited lymphadenectomy (L-LA) in cI stage showed similar survival benefit (OS, RR = 0.97, 95% CI 0.90–1.06; DFS, RR = 1.06, 95% CI 0.97–1.15), in contrast with systematic lymph node dissection (SLND). For pN2-III patients, HMLI indicated poorer OS (hazard ratio (HR) = 1.22, 95% CI 1.14–1.31) and DFS (HR = 1.25, 95% CI 1.14–1.36).
Conclusion:
IASLC’s residual tumor classification correlated with significant survival differences. Compared with R0, Run was associated with inferior 5-year OS and DFS. L-LA seemed to provide equivalent survival benefits, in contrast to SLND. For patients with low invasiveness in stage cI, L-LA could be considered as a preferred option. HMLI predicts poorer survival in pN2-III patients, and positive PLC significantly worsened long-term survival in operable NSCLC, particularly at early stage.
Keywords
Introduction
Despite a decline in global smoking rates over several decades, lung cancer remains the most prevalent cancer in China as of 2022 and continues to be the leading cause of cancer-related deaths among both men and women. 1 Non-small cell lung cancer (NSCLC) accounts for 85% of all lung cancer cases, with approximately 25% diagnosed at an early stage.2,3 Surgical resection is the recommended treatment for patients with curable stage.
The definition of complete resection is based on the residual tumor classification proposed by the Union for International Cancer Control (UICC), which includes three categories: R0, R1, and R2. R0 indicates no evidence of residual tumor, while R1 and R2 denote microscopic and macroscopic residual tumors, respectively. 4 Due to limitations in the traditional R classification for predicting regional recurrence, the International Association for the Study of Lung Cancer (IASLC) proposed a modified R classification system, comprising complete resection, uncertain resection (Run), and incomplete resection. 5 When determining postoperative treatment strategies for patients, the TNM classification and R classification serve as important references. However, in guidelines such as NCCN, Chinese Society of Clinical Oncology (CSCO), and European Society for Medical Oncology (ESMO), the presence of R1/R2 in the R classification affects treatment decisions. For example, in stage IIB NSCLC, adjuvant chemotherapy is recommended for R0 resection, while reoperation or concurrent chemoradiotherapy is suggested for R1/R2 resection. In contrast, there are no specific treatment recommendations for patients with Run resection. For a lung resection to be deemed complete, all the following criteria must be met: (a) microscopically confirmed negative resection margins; (b) systematic lymph node dissection (SLND) or lobe-specific systematic lymph node dissection (L-SND), including at least six groups of lymph nodes based on the primary tumor’s location in the lung lobe, with three groups from intrapulmonary and/or hilar stations and three from mediastinal stations, one of which must be subcarinal; (c) separately resected nodules or nodules at the margins of the major lung specimen should not exhibit tumor extension beyond the capsule; (d) the highest mediastinal lymph nodes removed must be negative.
Incomplete resection includes R1 (microscopic residual tumor) and R2 resection (macroscopic residual tumor). Any resection involving the following is considered as incomplete: (a) tumor involvement at the resection margins; (b) tumor extension beyond the capsule in separately resected nodules or at the margins of the major lung specimen; (c) known positive but unremoved lymph nodes; (d) positive cytology in pleural or pericardial effusion.
Uncertain resection must have tumor-free margins but may include one or more of the following situations: (1) intraoperative lymph node assessment fails to meet the standards of SLND or L-SND; (2) the highest mediastinal lymph node is invaded; (3) carcinoma in situ (CIS) is confirmed at the bronchial margin; (4) pleural lavage cytology (PLC) is positive. 5 Professional guidelines have provided stratified pathways for R0 and R1/R2, but there is currently no consensus on the follow-up management of Run patients.
In this article, we first conducted a literature search and meta-analysis on the prognosis of applying the IASLC R staging system. We then performed a meta-analysis focusing on the elements included in Run, particularly different lymph node dissection methods and the involvement of the highest mediastinal lymph node. To our knowledge, this is the first article to conduct a meta-analysis of observational studies on residual tumor classification based on IASLC descriptors. Currently, SLND remains the recommended practice for all operable NSCLC patients, regardless of tumor stage or location. 6 Although the value of accurate histopathological staging of nodal status with SLND is widely recognized, it typically requires longer operation times and may increase perioperative complications. Consequently, many surgeons prefer limited lymphadenectomy (L-LA), such as mediastinal lymph node sampling (MLNS) and L-SND, especially in early-stage NSCLC. 7 This highlights the need to explore the prognosis for patients reclassified as having uncertain resection (Run). According to most professional guidelines, patients with stage II and III are recommended for adjuvant chemotherapy or targeted therapy, regardless of residual tumor status. Therefore, we summarized the main findings regarding different lymph node removal methods in clinical stage I NSCLC, aiming to provide evidence for developing follow-up schemes and treatment plans for patients classified as Run, particularly those in clinical stage IA disease. Highest mediastinal lymph node involvement (HMLI) is the second most common reason for migration from UICC R0 to IASLC Run. 8 Thus, a meta-analysis was conducted to clarify the clinical evidence and its impact on patient survival, such as overall survival (OS) and disease-free survival (DFS), with the goal of informing the development of follow-up protocols and treatment strategies for patients classified as pN2-III NSCLC. Numerous studies have established the prognostic significance of PLC in early-stage NSCLC.9–12 Consequently, this article includes a brief systematic review of PLC’s prognostic value, specifically focusing on implications for stage I patients.
Methods
Protocol and registration
This protocol was registered with Prospective Registry of Systematic Reviews (PROSPERO) under the number CRD42023443935 (available at: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42023443935). This systematic review and meta-analysis were conducted in alignment with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 13
Literature source and search strategy
This systematic review aggregated data from multiple databases, including PubMed, Cochrane Library databases, Web of Science, EMBASE, CNKI, and Wanfang databases, covering publications from their inception up to January 2024. The search utilized keywords and Medical Subject Headings terms, such as lung neoplasms, NSCLC, early-stage lung cancer, surgical procedures, operative, surgical resection, resection margins, uncertain resection, incomplete resection, suboptimal resection, lymph node excision, SLND, complete lymph node dissection, selective lymph node dissection, limited lymph node dissection, lymph node sampling, highest mediastinal lymph node, PLC, cytological analysis, prognosis, survival outcome, and mortality. The detailed search strategies of the databases are provided in Supplemental File 1.
In addition to electronic database searches, we conducted manual searches of references within systematic reviews, meta-analyses, and clinical trial registries, including the US Clinical Trials Registry, the World Health Organization International Clinical Trials Registry Platform, and the Chinese Clinical Trial Registry. We also reviewed conference proceedings from prominent organizations such as the American Society of Clinical Oncology, the ESMO, the American Association for Cancer Research, the World Conference on Lung Cancer, and the CSCO. The initial database searches were performed in January 2024 and updated in June 2024.
Study selection
Studies were included based on the following criteria: (1) patients diagnosed with primary NSCLC who underwent surgical treatment; (2) study designs including randomized controlled trials (RCTs) and observational studies; (3) study outcomes including OS or DFS; (4) studies with full accessibility in Chinese or English languages. Excluded were case reports, non-English/Chinese publications, and studies lacking full-text access. No study was excluded for flaws in study design or quality. For the purpose of this analysis, subgroup analyses that presented data by tumor stage were considered as separate studies. In cases of multiple publications or patients overlap, the most recent publication was chosen.
Quality assessment and data extraction
The methodological quality of each included RCT was assessed by two reviewers, J.Y. and X.L., using the Cochrane Collaboration’s Risk of Bias tool. 14 The evaluation criteria included random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, completeness of outcome data, selective outcome reporting, and potential other biases. Randomized clinical trials were initially viewed as high-quality evidence but may be downgraded due to potential bias, imprecision, inconsistency, indirectness, and publication bias. The Newcastle-Ottawa Scale (NOS) was used by M.H. and M.Z. to evaluate study quality for observational studies, categorizing them into low, moderate, and high quality based on selection, comparability, and outcomes. Studies scoring 0–3, 4–6, 7–9 were categorized as low, moderate, and high quality, respectively. 15 The ROBIS tool was used to evaluate the level of bias in the included meta-analysis, while the AMSTAR-2 scale was used to assess their methodological quality. In cases of disagreement, another investigator (T.Z.) was brought in to discuss in order to reach a consensus.
Data extraction included authors, publication year, patient demographics, tumor stage, study design, sample size, intervention details, and survival outcomes. Survival data were analyzed using risk ratio (RR) for dichotomous data and hazard ratio (HR) for time-to-event outcomes. When both univariate and multivariate results were reported by the authors, we opted to use the multivariate analysis findings. Survival rates were directly extracted or estimated from survival curves using Getdata Graph Digitizer. 16
Statistical analysis
Survival discrepancies based on different groups were analyzed using RRs or HRs with 95% confidence intervals (CIs). Study heterogeneity was assessed using the I2 statistic, with fixed-effect models applied for I2 < 50% and random-effect models otherwise. Subgroup analysis was performed according to study design (RCTs vs cohort studies), lymph node removal method (SLND vs nSLND), tumor stage, and methods for controlling confounding factors. Publication bias was evaluated using funnel plots and quantitatively assessed through Egger’s and Begg’s tests. If necessary, the trim-and-fill method was used, considering a p-value <0.05 as statistically significant. Analysis was performed using Stata 15.0 (Stata Corporation, College Station, TX, USA), and graphical data were presented using GraphPad Prism 9.0 (GraphPad Software, San Diego, CA, USA).
Results
The validation of uncertain resection
Characteristics of the included studies
A total of 123 articles were identified, of which 13 studies involving 37,149 patients met the inclusion criteria.8,17–28 The PRISMA flow diagram illustrates the search process (Figure 1). For this meta-analysis, we searched Web of Science (n = 36), PubMed (n = 40), Embase (n = 107), Cochrane Library (n = 15), CNKI (n = 2), and Wanfang (n = 1). After removing 78 duplicates, 123 records were screened. We excluded 81 records (77 irrelevant, 4 reviews). Full texts of 42 reports were assessed; 30 were excluded (20 not meeting criteria, 8 no full text, 2 duplicates). One study was added from references and three from a previous review. Ultimately, 13 studies were included in the analysis. Key characteristics of the included studies—including data sources, study periods, sample sizes, 5-year OS/DFS rates, HRs for OS/DFS, and NOS quality scores are summarized in Table 1. All studies were rated as high quality based on NOS criteria (Figure S1(A), Table S1). Three studies focused on operable stage III patients, six included surgical patients across stages I–III, one targeted IA cases, and one examined patients with clinical N0 but pathological pN2 disease. Five-year OS rates were extracted from all studies, with three derived from survival curves using GetData Graph Digitizer. Direct HRs for OS were reported in 11 studies.

Flow diagram showing the search process of articles included in section “The validation of uncertain resection.”
Main characteristics of 13 eligible studies enrolled in section “The validation of uncertain resection.”.

The survival rate was extracted from the survival curves by Getdata Graph Digitizer.
DFS, disease-free survival; HR, hazard ratio; NA, not available; NOS, Newcastle-Ottawa Scale; OS, overall survival; RFS, relapse-free survival.
OS assessment based on IASLC R classification
Significant heterogeneity was detected among the 13 pooled studies (I2 = 81.2%). A random-effects model revealed a pooled RR of 1.31 (95% CI 1.19–1.44) indicating superior 5-year OS rate in the R0 group compared to the Run group (Figure 2(a)). Eleven studies provided HR data comparing R-un and R0 resections, demonstrating worse OS for Run with no significant heterogeneity (HR = 1.34, 95% CI 1.27–1.42, p < 0.001, Figure S2(A)). Patients in the R1/2 group had a 1.98-fold higher mortality risk than the R0 group (RR = 1.98, 95% CI 1.59–2.47, Figure S2(C)).

Forest plot of survival difference between Run and R0. (a) 5-Year OS difference. (b) 5-Year DFS difference.
DFS assessment based on IASLC R classification
Eight studies reported 5-year DFS rates comparing Run and R0 resections. Meta-analysis showed significantly lower DFS in the R-un group (RR = 1.43, 95% CI 1.28–1.60, Figure 2(b)). Pooled HRs from eight studies further confirmed worse DFS outcomes for Run (HR = 1.33, 95% CI 1.20–1.47, Figure S2(B)).
Subgroup and sensitivity analysis
Strong heterogeneity was observed among the 13 studies that were included in the present 5-year OS rate synthesis (I2 = 81.2%). A subgroup analysis was conducted to explore the sources of heterogeneity in the OS analyses. Participants were divided into six predefined subgroups based on factors such as gender, age, data source, stage, percentage of patients with HMLI, and percentage of patients in N0. The results showed that there was significant heterogeneity in 5-year OS among the subgroups. Notably, studies from China showed more inferior OS in Run group compared to those from non-China (China: RR = 1.48, 95% CI 1.38–1.59; non-China: RR = 1.16, 95% CI 1.11–1.21), with no significant heterogeneity in either group (China, I2 = 0%; non-China, I2 = 26.7%). Despite the different data source, the trends in the results of the two groups were consistent (Figure S3(A)). Sensitivity analysis was conducted to assess whether the results were influenced by any single study. The studies from Edwards and Ren were identified as the main source of heterogeneity in overall studies18,22 (Figure S3(B)). In Edwards’ study, patients reclassified as Run group had the highest 5-year OS rate, showing the least difference compared to those in complete resection group. In this study, 87% of the cases were clinically staged as I or II, and 8% as IIIA. According to most current studies, for early-stage patients, such as those with stage IA or ground-glass nodules, complete lymph node dissection does not significantly impact OS. Therefore, in section “Extent of removal for mediastinal lymph node stations,” we will discuss in detail the impact of different lymph node dissection methods on patient prognosis. As mentioned in other articles, the long-term prognosis of patients with HMLI is inferior to those with incomplete lymph node dissection.8,19 In Ren’s study, the proportion of HMLI was 37.27%, the highest among the enrolled studies. Hence, in Ren’s study, the comparison between Run and R0 resection showed the greatest difference. After excluding these two studies, the pooled RR remained unchanged (RR = 1.43; 95% CI: 1.29–1.58). Thus, we retained the two studies in the final analysis and applied the Mantel-Haenszel method for the final synthesis. In the combined analysis of HR, no significant heterogeneity was observed (Figure S3(C)).
Publication bias
For the OS synthesis, the potential publication bias was evaluated using a funnel plot. Neither the visual inspection of the funnel plots nor Egger’s test revealed any significant publication bias, either in the 5-year OS synthesis or in the HR synthesis derived from OS (Figure S3(D) and (E); Egger’s test, p = 0.620; Begg’s test, p = 0.669).
Extent of removal for mediastinal lymph node stations
Characteristics of the included research
In this section, we reviewed the surgical procedures in each study, as the definition and execution of SLND varied across the included studies, as did the lymph node maps used. We established SLND standards based on the ESTS criteria, which require complete removal of all lymph nodes and surrounding fat tissue between the anatomical landmarks of the lung lobe where the tumor is located. This includes a minimum of six stations, with at least three stations must be from mediastinal lymph node zones, including station 7. We identified 1695 articles and ultimately selected 30 studies involving 11,766 patients for further analysis29–58 (Figure 3). General characteristics of these studies are summarized in Table 2. These studies included 4 RCTs and 26 observational studies, with 2 studies derived from the registry databases. The data sources were from China, Japan, Korea, France, and Italy. Of these eligible studies, 16 compared long-term survival between SLND and MLNS, while 10 compared SLND with L-SND. The general characteristics of these studies are summarized in Table 2.

Flow diagram showing the search process of articles included in section “Extent of removal for mediastinal lymph node stations.”
Main characteristics of 30 eligible studies enrolled in section “Extent of removal for mediastinal lymph node stations.”.

The survival rate was not directly in article but could be extracted from the survival curves by Getdata Graph Digitizer.
Propensity score analysis was used in study.
DFS, disease-free survival; HR, hazard ratio; NA, not available; NOS, Newcastle-Ottawa Scale; nSLND, nonsystematic lymph node dissection; Os, observational study; OS, overall survival; RCT, randomized controlled trial; SLND, systematic lymph node dissection.
Risk of bias and quality of studies
All observational studies were rated as moderate to good quality by NOS grading system (Figure S1(A), Table S1). We identified four RCTs involving 443 patients. Performance bias during lung resection was unavoidable. Two trials failed to perform an intention-to-treat analysis, and the methods for sequence generation and allocation concealment were additional sources of bias (Figure S1(B)).
Assessment of OS
Among the 30 enrolled studies, 29 studies provided 5-year OS rates, and 13 studies provided HRs. We extracted 5-year OS rates as primary endpoint directly from the full text of 26 studies and used Getdata software to convert survival curves into 5-year OS rates in 3 studies.32,55,57 The combined analysis (Figure 4(a)) showed no significant difference in OS between SLND and MLNS (RR = 0.97, 95% CI 0.90–1.06, I2 = 31.5%, Figure 4(a)). Given the completely different nature of these study designs, we separately report the meta-analysis results of observational studies and RCTs. The analysis results of observational studies show that SLND provides comparable survival benefits to MLNS (RR = 0.99, 95% CI 0.91–1.08, Figure 5(a)). The analysis of four RCT studies also shows similar results (RR = 0.77, 95% CI 0.50–1.18, Figure 5(b)). This result suggests that SLND is an alternative to MLNS in clinical stage I NSCLC.

Forest plot of survival difference between SLND and nSLND in stage cI NSCLC patients. (a) 5-Year OS difference. (b) 5-Year DFS difference.

Forest plot of 5-year OS difference between SLND and nSLND in subgroups of stage cI NSCLC patients. (a) 5-Year OS difference in observational studies. (b) 5-Year OS difference in RCT studies. (c) 5-Year OS difference in L-SND group. (d) 5-Year OS difference in MLNS group. (e) 5-Year OS difference in tumor ⩽2 cm. (f) 5-Year OS difference in studies with equilibrated baseline characteristics.
Subgroup analyses were conducted based on control arm (MLNS or L-SND) and tumor stage. Given the different definitions of stage I across TNM editions, we performed subgroup analyses based on tumor diameter. Eight studies reported 5-year OS rates for tumors ⩽2 cm, with no significant difference observed (RR = 1.20, 95% CI 0.88–1.64, Figure 5(c)). Interestingly, subgroup analyses based on surgical procedure showed that SLND was associated with higher 5-year OS compared to MLNS (RR = 0.71 95% CI 0.63–0.80, Figure 5(d)), while SLND appeared to offer worse survival outcomes than L-SND (RR = 1.13, 95% CI 1.01–1.26, Figure 5(e)).
We conducted a comprehensive assessment of baseline characteristics in the enrolled literature comparing SLND and nSLND. The key characteristics examined included age, gender, pathological type, tumor size, and surgical method. Of the studies reviewed, 11 exhibited baseline imbalances. Among these, propensity score matching was employed in seven observational studies to adjust for potential confounding factors.50,52–55,57 However, the remaining four studies did not employ propensity score matching.42,45,51,58 Given this discrepancy, we excluded these four studies from further analysis. The reanalysis yielded a pooled RR of 0.99 (95% CI 0.91–1.08, Figure 5(f)).
Assessment of DFS
Among the enrolled studies, 15 provided DFS data (15 studies with 5-year DFS rates and 4 with HR). We extracted the 5-year DFS rates as the primary outcome directly from the full text of 13 studies. For the remaining two studies, we used Getdata software to transform the survival curves into 5-year DFS rates.57,59 The pooled results of the 5-year DFS rates are shown in Figure 4(b). No significant heterogeneity was detected among these 15 clinical studies (I2 = 12.3%). A fixed-effect model was applied for the DFS analysis. No significant difference was observed between SLND and nSLND with a pooled RR of 1.06 (95% CI 0.97–1.15, Figure 4(b)).
Publication bias
Risk of publication bias was assessed using a funnel plot in OS synthesis. The plots were asymmetrical on visual inspection, suggesting potential risks of publication biases (Figure S4(A)). The p value of Begg’s test was 0.358, of Egger’s test was 0.002. By trim-and-fill analyses, seven hypothesized studies were imputed. Meta-analysis incorporating these seven studies did not significantly change the results (RR, 0.96, 95% CI: 0.85–1.07, Figure S4(B)).
Highest mediastinal lymph node involvement
Characteristics of the included research
A total of 13 studies were identified, involving 6943 patients with operable pN2-stage NSCLC. These studies compared survival data that were compared between patients with and without HMLI metastasis8,18,22,26,59–67 (Figure 6, Table 3). All of the enrolled studies were observational and conducted in China, Korea, Japan, the USA, and Italy. With the exception of Marziali’s study, which included patients with clinical N0 status between 2015 and 2020, the remaining studies focused on patients with pN2 NSCLC. Regarding survival data, all of the 13 studies reported HR based on OS data and 6 studies provided data on DFS data. The quality assessment of the enrolled studies is shown in Figure S1(A) and Table S1.

Flow diagram showing the search process of articles included in section “Highest mediastinal lymph node involvement.”
Main characteristics of 13 eligible studies enrolled in section “Highest mediastinal lymph node involvement.”.

NA, the survival rate was not directly in article but could be extracted from the survival curves by Getdata Graph Digitizer.
DFS, disease-free survival; HR, hazard ratio; NA, not available; NOS, Newcastle-Ottawa Scale; OS, overall survival.
Assessment of OS and DFS
The meta-analysis results of the combined HR and 95% CI are presented in Figure 6. No significant heterogeneity was detected among these clinical studies included in the analysis (I2 = 8.3%). Therefore, a fixed-effect model was applied. The results indicated that HMLI was an indicator of inferior prognosis, with a pooled HR of 1.22 (95% CI 1.14–1.31, Figure 7(a)). Additionally, five studies provided relevant HR data for the 5-year DFS rate of HMLI in pN2 NSCLC. The presence of HMLI was associated with shorter DFS (HR = 1.25, 95% CI 1.14–1.36, Figure 7(b)).

Forest plot of survival difference between HMLI and nHMLI. (a) 5-Year OS difference. (b) 5-Year DFS difference.
Publication bias
The risk of publication bias was assessed using a funnel plot (Figure S4(C)). No evidence of significant publication bias was detected, as indicated by visual inspection of the funnel plots and formal statistical tests (Egger’s test, p = 0.331; Begg’s test, p = 0.428).
Prognostic significance of PLC
Malignant cells identified in pleural lavage fluid in patients without pleural effusion indicate micrometastasis, with the rate in operable NSCLC ranging from 3% to 13%. To date, four meta-analyses have reported on the prognostic significance of PLC, consolidating the conclusion that PLC+ is a strong predictor of survival in patients with lung cancer9–12 (Table 4). We conducted a bias risk assessment using the ROBIS tool, with two articles rated as high risk and two articles rated as low risk. AMSTAR2 was used to evaluate the methodological quality, and all four articles were rated as moderate quality (Table S4). Despite the differences in bias risk and methodological quality among these four meta-analyses, the overall results consistently support the conclusion that positive PLC is a significant factor for poor prognosis, especially in stage I patients. Although there are limitations in bias risk and methodological quality, the consistency of the results from these four meta-analyses provides a certain degree of reliability to the conclusion.
Main result of four meta-analysis about PLC value in operable NSCLC.
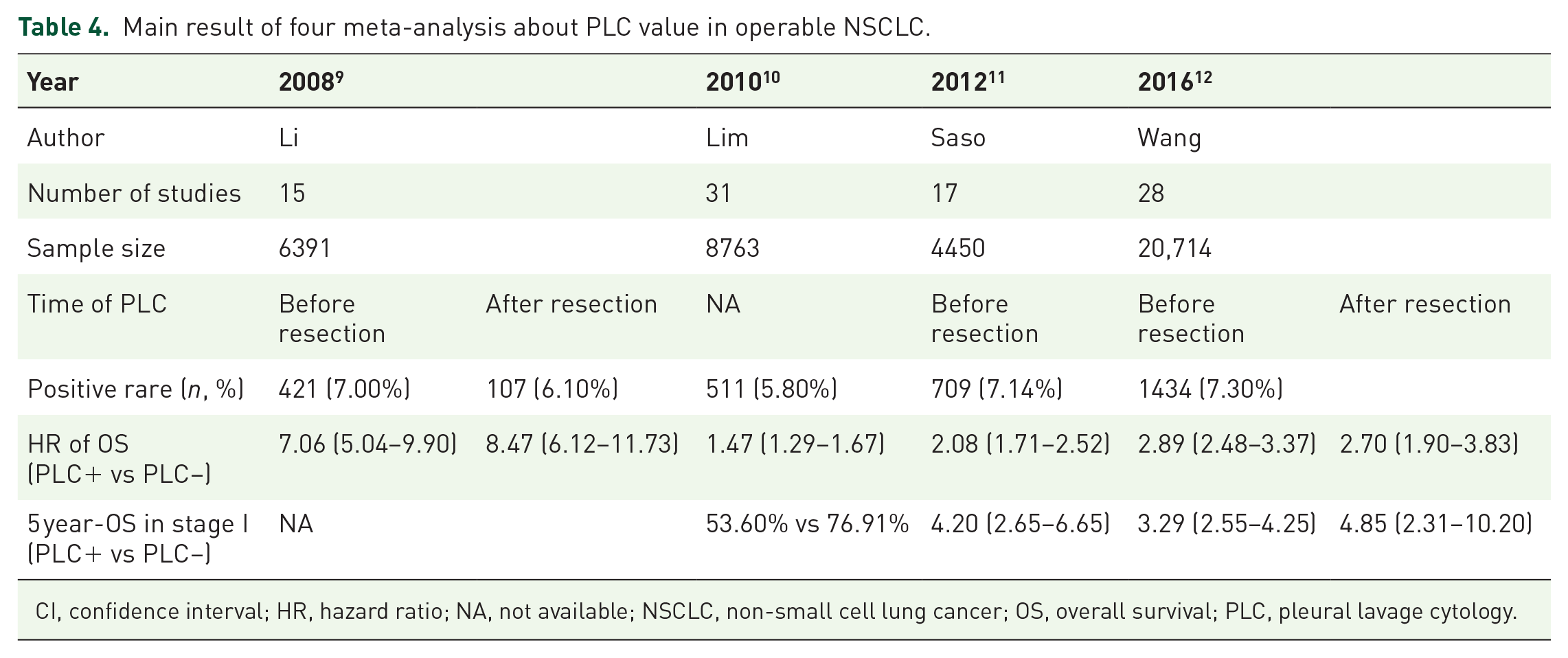
CI, confidence interval; HR, hazard ratio; NA, not available; NSCLC, non-small cell lung cancer; OS, overall survival; PLC, pleural lavage cytology.
Current studies can be categorized into two groups based on the timing of intraoperative pulmonary lavage: before and after lung resection. In 2008, Li et al. conducted a meta-analysis of 15 studies involving 6391 patients to assess the prognostic value of PLC on OS. Their results indicated that positive PLC, either before or after lung cancer resection, adversely impacted OS, with HR of 7.06 (95% CI, 5.04–9.90) and 8.47 (95% CI, 6.12–11.73), respectively. 9 In 2010, the International PLC Collaboration Group conducted a meta-analysis of 31 clinical studies from 22 medical centers, comprising individual data from 8763 patients, of whom 511 (5.8%) had positive PLC results. This analysis revealed that patients with positive PLC had lower OS rate, with the most significant difference observed in stage I patients. 10 A 2012 meta-analysis focusing on the prognostic value of preoperative PLC results, including 4450 patients, found that positive preoperative PLC was significantly associated with an increased risk of local and distant cancer recurrence. Among these, nine studies presented survival data for stage I patients, indicating a more pronounced effect in this group. 11 An updated pooled analysis in 2016 with a total of 20,714 patients. Subgroup analysis of stage I patients in Wang et al.’s 12 study also showed that pre-operation, post-operation, and combined PLC were associated with adverse survival rate, with HR of 3.29 (95% CI 2.55–4.25), 4.85 (95% CI 2.31–10.20), and 3.16 (95% CI 2.53–3.94), respectively. These consistent results suggest that PLC has stronger prognostic significance in stage I patients compared to the overall population.
Nevertheless, PLC status has not yet been incorporated into the TNM classification. Mizuno et al.’s 67 study found that the prognosis in patients with pI NSCLC with PLC+ was comparable to that in patients with p-IIIA disease. Several reports have recommended that NSCLC be staged as pT3 for all patients with a PLC+ status, while others have proposed increasing the pT stage by 1.10,68–70 Ogawa et al. conducted a study on the clinical benefits of adjuvant chemotherapy in 53 patients with positive PLC findings who underwent curative-intent surgery for lung adenocarcinoma. They found a significant difference in the 5-year DFS when comparing patients treated with surgery and adjuvant chemotherapy versus those treated with surgery alone. 71 PLC+ represents a negative prognostic factor that is likely to influence the long-term outcomes but not the treatment strategy of care in patients with Stage pII-III and selected cases of Stage pIB, as adjuvant therapy is already recommended for these patients. For patients with PLC+ in stage pI, it is important to evaluate the necessity of adjuvant therapy.
As discussed above, for early-stage patients, particularly those in stage I, positive PLC had greater prognostic value and is closely related to postoperative malignant pleurisy and distant recurrence. However, in clinical practice, methods such as the volume of lavage fluid used, type of solution, and timing of lavage still require standardization. Whether adjuvant therapy should be recommended for stage I patients with positive PLC needs validation through multicenter prospective clinical trials.
Discussion
More than a decade ago, the IASLC proposed definitions for complete, incomplete, and uncertain resections in lung cancer. This definition was based on the previous R classification of the UICC and considered various aspects related to surgical treatment, including intraoperative nodal assessment, assessment methods, criteria for pathological absence of nodal disease, and the prognostic significance of PLC and CIS. 5 Given the distinct prognosis, Run was introduced in the seventh edition of the TNM classification. 4 Distinguishing between complete resection and Run is clinically important and provides additional information beyond the R descriptor alone. In recent years, several independent groups have published articles on the prognostic value of the IASLC resection definitions, using institutional, international, and population-based databases. However, the results of these studies have been inconsistent. Therefore, we conducted the system review and meta-analysis to synthesize all available evidence. As far as I know, this is the first meta-analysis on this topic.
In this meta-analysis, we included 13 observational studies with sample sizes ranging from 68 to 14,712. The initial combined analysis showed a RR of 1.31 for 5-year OS (95% CI 1.19–1.44) and 1.43 for 5-year DFS (95% CI 1.28–1.60). These results indicate that Run is associated with inferior outcomes compared to complete resection. Thus, the IASLC R classification better identifies risk than the previous classification. Given the survival deficit observed in the Run group, additional treatment protocols after surgery should be considered. 22 Consistent with proposals for revising the classification of residual tumor, the OS of patients in the Run category consistently falls between that of the R0 and R1/2 groups. These differences in OS remained significant even after adjusting for potential confounders through multivariate analysis. 72 Due to the independence of the four elements that comprise the R descriptors, significant heterogeneity exists when analyzed together. Although evidence for specific descriptors is generally limited and partially consistent, we discuss each element individually for its impact on the prognosis of early-stage NSCLC.
Our research aimed to examine the differences in long-term survival between SLND and L-LA in patients with clinical stage I NSCLC. We found that patients with stage cI NSCLC had similar 5-year OS rates when comparing SLND to L-LA (RR = 0.97, 95% CI 0.90–1.06). We included 31 studies involving 12,231 patients from 1985 to 2023, based on TNM editions 5–8. To minimize the impact of varying tumor diameter definitions in stage I, we specifically analyzed the survival data from stage IA2 patients (tumor diameter ⩽2 cm). The results showed no heterogeneity among the studies and no difference in 5-year survival between SLND and L-LA. Stratified analyses based on study design and statistical methods yielded consistent results across subgroups, indicating that SLND provides equivalent OS benefits compared to L-LA. Interestingly, subgroup analysis based on the method of L-LA showed that SLND provided equal OS benefits, while L-SND seemed to offer a higher 5-year OS rate. The ACOSOG Z0030 trial is currently the largest prospective multicenter randomized controlled study comparing SLND and sampling. There were no statistically significant differences between the two groups in terms of postoperative median OS, 5-year DFS, or local and distant recurrence. Since survival and recurrence data of stage cI were not included in the published result, this RCT study was not included in section “Extent of removal for mediastinal lymph node stations.” Stratification based on T1 showed no difference in long-term survival between MLNS and SLND. Based on our current meta-analysis results, it is speculated that including this study would not affect the overall conclusion.
The pooled results in section “Extent of removal for mediastinal lymph node stations” should be interpreted with caution. First, surgical techniques for SLND are not yet standardized. The extend of SLND performed is not based on objective or reproducible criteria but primarily depends on the surgeon’s preference and the patient’s condition. There is a lack of details regarding partial or complete removal of lymph nodes and the number of lymph nodes removed from different stations in the studies reviewed. Secondly, although authors have used the term “systematic” to define lymph node dissection, their definitions of SLND are inconsistent. Organizations such as the IASLC, NCCN, ACOSOG, and ESTS have each published different standards for SLND. For example, in some Japanese studies involving bilateral upper lobe lung tumors, removal of stations 8 and 9 is not mandatory. Given the potential bias introduced by these inconsistent definitions, we established, the following criteria for SLND to minimize the confounding: complete removal of all mediastinal lymph nodes and surrounding fat tissue between the anatomical landmarks of the lung lobe where the tumor is located, and SLND involving the removal of at least six lymph node stations, with at least three groups coming from mediastinal lymph nodes, including station 7. The standard based on the number of lymph node dissections was not adopted in this section of the study.73,74 Even with a relatively consistent standard, some potential biases may still be inevitable. Lastly, certain high-risk factors for postoperative recurrence, such as spread through air spaces (STAS), have been identified. Several systematic reviews and meta-analyses have confirmed that tumor STAS is associated with shorter DFS and OS in stage I NSCLC.75,76 Some surgeons suggest that when STAS is found in sublobar resection specimens, subsequent lobectomy should be performed in T1N0M0 adenocarcinoma patients. Despite these limitations, the results of the meta-analysis in section “Extent of removal for mediastinal lymph node stations” indicate that less rigorous nodal assessment was not associated with worse prognosis, particularly, in early-stage NSCLC or in more favorable histologic subtypes. Considering the lower postoperative complications and the effectiveness of tumor-draining lymph nodes for immunotherapy, L-LA may be prioritized in less invasive stage cIA patients.
The presence of HMLI is an important category of Run. The results of this meta-analysis showed that both OS and DFS were inferior in the metastasis group compared to the nHMLI group. For this population, more aggressive postoperative follow-up and adjuvant therapy may be considered. Several retrospective studies have shown significant survival benefits with postoperative radiotherapy (PORT) in pN2-III patients.77,78 Nevertheless, recent RCTs, such as the Lung ART and PORT-C trials, showed that PORT did not improve long-term survival in pN2-III NSCLC.22,79 The Lung ART study included 501 patients, with 203 being Run patients. However, subgroup analysis of different surgical resection statuses, including patients with HMLI, has not been published.
Under the current circumstances, sub-classification of lymph node metastasis in patients with pN2-III NSCLC probably has more clinical implications for prognosis, including multiple station metastases of mediastinal lymph nodes, HMLI, and distribution of metastatic nodes (skip N2 or non-skip N2). 61 To my furthest knowledge, only two studies have reported survival data on PORT for HMLI in pN2 NSCLC. In Guo et al.’s retrospective study, patients with positive HMLI had worse prognosis in pN2 NSCLC. For these patients, PORT significantly improved both relapse-free survival (RFS; HR = 0.699, 0.59–0.83) and OS (HR = 0.79, 0.66–0.95). 65 Similar results were reported by Deng et al. 80 in a study of 140 patients in pN2-III. As suggested by this meta-analysis, HMLI is an independent factor for poorer OS and DFS, and further research is needed to clarify whether this population could benefit from PORT.
Bronchial margin involvement with CIS is a rare occurrence, with reported incidences ranging from 0.02%–2.4% in cohorts of 255–4493, totaling 138 cases (0.9%) out of 16,000 reported resections. 81 In several validation studies mentioned above, the incidence of CIS was 0.09% (13/14,293), 0 (0/3361), 0 (0/910), and 0.03% (2/5299) separately, with no reported survival data.8,18,20,22 Due to its low incidence, stratified analysis is not feasible in current large cohort or database studies. In a study of 1277 consecutive NSCLC patients, 1003 (78.5%) underwent complete resection, 185 (14.5%) had uncertain resection, and 89 (7.0%) had incomplete resection. Five patients showed in situ cancer at the bronchial margin, with a 5-year survival rate of 40.0%. The highest mediastinal lymph node positivity and less rigorous lymph node assessment had 5-year survival rate of 28.8% and 44.2%, respectively. 17 Based on the available literature data, the presence of CIS does not appear to influence the prognosis of patients after surgical resection.
According to the standards established by IASLC in 2005, lobectomy is mandatory for the definition of complete resection. Since the 1990s, the pathological types of early lung cancer have gradually changed due to the increasing detection of ground-glass opacities (GGOs). Preoperative thin-layer CT can predict the malignancy and lymph node metastasis of GGOs, allowing for sublobar resection in cases of low-grade malignant nodules identified through imaging. This raises questions about the necessity of lobectomy. After Japan Clinical Oncology Group0201 (JCOG0201) established radiologically noninvasive criteria for lung adenocarcinoma, JCOG and the West Japan Oncology Group conducted a series of prospective imaging-guided studies. These studies proposed more surgical options for some peripheral lesions, such as segmentectomy and wedge resection.82–86 Precise segmentectomy enables the simultaneous management of multiple lung nodules while maintaining the option for additional treatment for potential second primary cancer, respiratory diseases, or cerebrovascular diseases. This aspect was not addressed in the IASLC definitions of resection completeness. With evolving surgical techniques for lung cancer, a standardized approach to the extent and invasiveness of nodal resection may need revision, as suggested by Lee et al. 87
The presence of circulating tumor cells or tumor DNA (ctDNA) in the bloodstream indicates the existence of tiny residual lesions, and their persistence or recurrence implies a poor prognosis after resection. When the IASLC definition was published, signs of these minor residual diseases were not considered. A systematic review of 13 eligible studies demonstrated a poor prognosis in patients with postoperative molecular residual disease (MRD). Patients with positive MRD were most likely to benefit from adjuvant treatment in terms of RFS. 88 Similarly, the presence of circulating tumor cells (CTCs) at the baseline or postoperative period was associated with an increased risk of recurrence and death compared with the absence of CTC in lung cancer patients. 89 Cancer cells or their fragments in the blood could be considered as the cells in other body spaces or anatomic structures. They are in a fluid tissue, blood, in a well-defined anatomic system, the circulatory, but to date, they are not coded in the TNM classification. Rami-Porta 90 have suggested adding the letter B, for blood, to the TNM (TNMB) as a new component of the classification. That would certainly complement the TNM classification, but liquid biopsy is not universally available and perhaps it is too early to consider it in the TNM classification. It also requires more research to quantify the amount of cells or of circulating tumor DNA or RNA that make a difference in prognosis. However, the presence of tumor material in the blood after treatment should be considered as minimal residual disease, although more research is needed to see how to classify it and how to manage it.
Conclusion
IASLC has updated the definition of complete resection and reclassified the surgical resections into complete, uncertain, and incomplete resections. This updated classification has been validated in several studies, demonstrating its prognostic value. Our literature review and meta-analysis in section “Extent of removal for mediastinal lymph node stations” compared the prognosis of Run with R0, revealing that L-LA could be a viable option for some favorable early-stage NSCLC patients. Additionally, the presence of HMLI indicates poorer survival in pN2-III patients, while PLC is associated with a worse prognosis in operable NSCLC, particularly in stage I. To our knowledge, the proposed R-classification for the ninth edition of lung cancer stage classification maintains the same overall structure. 72 We believe this review will offer a foundation for further refining the R-classification, despite the limitations of existing data. Moreover, with advancements in imaging technology, more precise preoperative staging, and the accumulation of liquid biopsy data, the revision of IASLC definitions should be more comprehensive. This includes incorporating the appropriate extent of surgical resection, STAS, and the presence of CTCs/ctDNA. The definition of resection completeness will continue to evolve in line with emerging evidence, aiding in practical use and encouraging further research.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251344789 – Supplemental material for The IASLC uncertain resection, general overview, current evidence, and future prospects: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251344789 for The IASLC uncertain resection, general overview, current evidence, and future prospects: a systematic review and meta-analysis by Mingming Hu, Xiaomi Li, Maike Zheng, Jiaqi Yu, Baohua Lu, Ying Wang, Xiaoqing Cao, Chongyu Su, Yujie Dong, Xu Zhang and Tongmei Zhang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251344789 – Supplemental material for The IASLC uncertain resection, general overview, current evidence, and future prospects: a systematic review and meta-analysis
Supplemental material, sj-docx-3-tam-10.1177_17588359251344789 for The IASLC uncertain resection, general overview, current evidence, and future prospects: a systematic review and meta-analysis by Mingming Hu, Xiaomi Li, Maike Zheng, Jiaqi Yu, Baohua Lu, Ying Wang, Xiaoqing Cao, Chongyu Su, Yujie Dong, Xu Zhang and Tongmei Zhang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359251344789 – Supplemental material for The IASLC uncertain resection, general overview, current evidence, and future prospects: a systematic review and meta-analysis
Supplemental material, sj-pdf-2-tam-10.1177_17588359251344789 for The IASLC uncertain resection, general overview, current evidence, and future prospects: a systematic review and meta-analysis by Mingming Hu, Xiaomi Li, Maike Zheng, Jiaqi Yu, Baohua Lu, Ying Wang, Xiaoqing Cao, Chongyu Su, Yujie Dong, Xu Zhang and Tongmei Zhang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
