Abstract
Background:
Medulloblastoma (MB) relapse is typically resistant to available treatments. An emerging alternative strategy focuses on disrupting tumor angiogenesis at various stages, using a combined metronomic anti-angiogenic approach.
Objectives:
The study aims to assess the efficacy and safety of this modified treatment approach in managing recurrent MB in the pediatric population.
Designs:
This study is a retrospective observational analysis involving 14 pediatric patients diagnosed with first or multiple recurrences of MB.
Methods:
We analyzed clinical, molecular, radiological, and outcome data of our cohort treated using a modified Medulloblastoma European Multitarget Metronomic Anti-Angiogenic Trial (MEMMAT) strategy.
Results:
Median age of patients was 11.6 years (range: 6.4–26 years). All 14 patients presented with a metastatic relapse after conventional treatments. The median time from primary diagnosis/prior relapse to the start of “modified MEMMAT” was 22 months (range: 2–60 months). Fifty-seven percent received the “modified MEMMAT” schema as second-line treatment, while 43% received it as third-line or beyond after recurrence. At a median follow-up of 17.9 months, the median overall survival (OS) from the MEMMAT start date was 18.2 months, and the median progression-free survival (PFS) was 12.8 months. OS at 12 and 24 months was 78.6% and 28.6%, respectively. PFS at 6 and 12 months was 100% and 55.0%, respectively. Treatment was globally well tolerated.
Conclusion:
The modified MEMMAT strategy shows promise in treating recurrent MB, achieving a 12-month OS rate from date of starting treatment of 78.6%, with manageable toxicity. These findings suggest its potential as a viable option for heavily pre-treated pediatric patients, warranting further validation in larger prospective studies.
Keywords
Introduction
Medulloblastoma (MB) is the most common malignant pediatric brain tumor, representing 60% of all embryonal central nervous system (CNS) tumors of childhood.1 –3 Recent methylome and transcriptome analyses have shown that MB is a heterogeneous tumor consisting of distinct groups with unique developmental origins, transcriptional profiles, and diverse phenotypes, all of which influence clinical outcomes.4 –8 Four molecular subgroups, including Wingless-Related Integration Site (WNT), Sonic Hedgehog Signaling Molecule (SHH), Group 3, and Group 4, and further additional subtypes have been identified.9 –11
Surgical resection, craniospinal radiation therapy, and multi-agent chemotherapy continue to be the standard first-line treatment for the majority of patients. Over the past years, however, the 5-year survival rate remains between 65% and 80%.12,13 Recurrence of the disease occurs in about 30% of children14,15 and carries a poor prognosis, despite multimodal treatment.
Various therapeutic alternatives, encompassing high-dose chemotherapy (HDCT) as well as several conventional drug combinations for patients encountering a relapse, have regrettably yielded only marginal improvements in progression-free survival (PFS). Furthermore, these therapies did not succeed in decreasing the number of hospitalizations due to collateral effects, limiting severely the quality of life (QoL). 16
Metronomic chemotherapy (MC) involves administering chemotherapeutic agents at low doses over extended periods, thus reducing toxicity. 17 Recent studies have shown MC effectiveness in enhancing overall survival (OS) in pediatric patients with refractory and relapsed brain tumors.18,19
Low-dose, long-term administration of etoposide and cyclophosphamide can significantly inhibit tumor growth by targeting normal cells that support tumor proliferation, such as endothelial cells, even in tumors that have developed high resistance to these drugs.20 –22 In 2005, Kieran et al. 23 conducted a feasibility trial assessing a metronomic anti-angiogenic treatment in 20 children with various recurrent or progressive cancers. The initial regimen alternated 21-day cycles of low-dose oral cyclophosphamide and etoposide with continuous oral thalidomide and celecoxib, all administered at anti-angiogenic doses. Following studies demonstrated fenofibrate, a peroxisome proliferator-activated receptor (PPAR)-alpha agonist, had both anti-angiogenic and antitumor effects and that combining metronomic etoposide with PPAR modulation and COX-2 inhibition showed synergy. The phase II trial expanded to include fenofibrate, leading to a “5-drug” regimen.24,25 In 2022, Slavc et al. described a retrospective observational study involving 29 consecutive patients with first or multiple recurrences, who were prospectively treated according to a Medulloblastoma European Multitarget Metronomic Anti-Angiogenic Trial “(MEMMAT)-like” strategy (NCT01356290). The treatment regimen included daily oral thalidomide, fenofibrate, and celecoxib, along with alternating 21-day cycles of low-dose oral etoposide and cyclophosphamide. In addition, patients received intravenous bevacizumab and intraventricular therapy with alternating etoposide and liposomal cytarabine. 26 They reported a median OS after recurrence for the entire cohort of 29.5 months. The OS was 48.3% ± 9.3% at 3 years and 34.5% ± 8.8% at 5 years, while PFS was 42.0% ± 9.5% at 3 years and 29.4% ± 9% at 5 years. Treatment was primarily outpatient and generally well tolerated in all cases. 26
In our study, we describe a retrospective series of patients treated according to a “MEMMAT-like” regimen, with an assessment of both survival and toxicity.
Materials and methods
Patients and data collection
Eligible patients for this study ranged from 2 to 30 years of age and exhibited radiological or cytological evidence of recurrent/refractory MB. They underwent treatment with a “modified MEMMAT” regimen at Bambino Gesù Children’s Hospital in Rome over 5 years (2017–2022). The study imposed no restrictions on prior relapses, type of treatments received, or presence of ventriculoperitoneal (VP) shunts. Patients were required to have acceptable organ function and bone marrow recovery, with functional abilities assessed using the Karnofsky or the Lansky Performance Scale, with a minimum requirement of 50%.
Data collected for each patient included date of diagnosis, age, gender, tumor stage, histologic classification, molecular subgroup, treatment history, time and type of relapse, radiological data, outcome after treatment, toxicity, and follow-up.
Histological examination based on the morphological and immunophenotypic characteristics of the tumor biopsy was conducted for all cases.
Molecular subgroups were investigated with both immunohistochemistry (IHC) and methylation profiling. IHC was performed on formalin-fixed paraffin-embedded sections using an automated immunostainer (Dako Omnis) and included β-catenin, YAP1, GAB1, TP53, and sequencing of exon 3 of CTNNB1 to identify relevant mutations. The MB molecular subgroup was determined through DNA methylation profiling, which involved processing formalin-fixed and paraffin-embedded tissue (250 ng) following approved protocols. Written informed consent from the patient’s parents and the patient was obtained before conducting these procedures. The samples were analyzed using Illumina Infinium Human Methylation EPIC Bead Chip arrays on the Illumina iScan Platform, as per the manufacturer’s instructions. The MB subgroup classification was established through DNA methylation-based classification of CNS tumors using resources provided by the German Cancer Research Center in Heidelberg. 27
Adverse effects associated with this regimen were documented to provide insight into its side effects and tolerability. Clinical and radiological outcomes were analyzed to evaluate the effectiveness of this strategy within the study cohort.
Study treatment
Our five-drug “modified MEMMAT” regimen featured several key alterations compared to the MEMMAT regimen. We introduced topotecan (0.4 mg) for intrathecal therapy, administered through lumbar puncture (LP), to minimize multiple weekly anesthesia sessions and reduced LP to once every 4 weeks; oral regimen includes fenofibrate (90 mg/m2 daily), celecoxib (50–400 mg twice daily), thalidomide 3 mg/kg daily, and alternating 21-day cycles of low-dose oral etoposide (35–50 mg/m2/day) and cyclophosphamide (2.5 mg/kg/day, maximal 100 mg); intravenous bevacizumab (10 mg/kg/infusion) every 2 weeks remains the only anti-angiogenic agent, with the choice to exclude thalidomide due to its supposable anti-angiogenic mechanism to bevacizumab. 28
The original MEMMAT regimen is shown in Figure 1.

MEMMAT regimen versus “modified-MEMMAT” regimen.
Response evaluation
Brain and spine gadolinium-enhanced magnetic resonance imaging (MRI) was performed at baseline and every four cycles of the “modified-MEMMAT” treatment. Cerebrospinal fluid (CSF) cytologic examination was performed during each administration of intrathecal therapy. Response assessment followed the criteria outlined in the Response Assessment in Pediatric Neuro-Oncology Cancers (RAPNO). Since there are no specific criteria for adults with MB, we adapted the RAPNO criteria for patients over 18 years old as well.29,30
Complete response (CR) refers to the complete remission of disease on both MRI and CSF examination. Partial response (PR) denoted a tumor volume reduction of >50% compared to the previous MRI evaluation. Stable disease (SD) was characterized by a ⩽50% decrease and ⩽25% increase in the product of diameters. Progressive disease (PD) was defined as a ⩾25% increase in the product of diameters or the identification of new lesions or neoplastic cells in the CSF. Response evaluation was analyzed, separating patients into different groups depending on the treatment line: group A (second line) and group B (third and beyond line).
Toxicity and QoL evaluation
All patients underwent comprehensive assessments, including physical examinations, blood pressure measurements, and neurological evaluations, along with routine laboratory tests such as blood chemistry and urine analysis, at baseline and every 2 weeks. Side effects were retrospectively collected based on the Common Terminology Criteria for Adverse Events version 5.0. 31 Clinical benefits and patients’ perception of QoL were assessed using the Italian version of the PedsQL Cancer Scale. 32 This evaluation occurred before the treatment initiation (T0) and 6 months after the start of therapy (T1). The Pediatric Quality of Life Cancer Module Parent Proxy Report 3.0 (PEDSQL) is a specialized module within the PedsQL™ consisting of 23 items across 8 subscales. These subscales cover various domains, including pain, nausea, anxiety related to procedures and treatment, worry, cognitive issues, physical appearance, and communication. Scores range from 0 to 100, with higher scores indicating better QoL. Responses were assessed on a 5-point Likert scale, with zero indicating the absence of a problem (“never”) and four indicating frequent occurrence (“almost always”). Raw scores were then transformed to a 100-point scale, where higher scores represent better QoL.
In addition, we monitored the frequency of emergency department visits and hospitalizations.
Statistical analysis
OS was calculated both from the time of diagnosis and from the time of starting the “modified-MEMMAT” regimen to the date of death from any cause or last follow-up for alive patients. PFS was defined from starting the “modified-MEMMAT” regimen until relapse or progression of the disease. Kaplan–Meier survival estimates were used in the analysis of OS and PFS. Survival curves between patients treated as second line (group A) or later (group B) were compared with non-parametric log-rank tests. All comparison tests were two-sided and considered significant at the 5% level. GraphPad Prism version 10.4.0 was used for statistical analysis.
Results
Patient population
Fourteen patients met the eligibility criteria outlined above. Table 1 presents the baseline characteristics of these patients, including their age at the start of treatment (median age of 11.6 years, ranging from 6.4 to 26 years) and gender distribution (10 males, accounting for 71%, and 4 females, making up 29%). At diagnosis, six patients (43%) presented with a localized tumor pattern, while the remaining eight patients (57%) had metastatic disease. Histopathology at diagnosis revealed classic MB in 12 patients and anaplastic (LC/A) in two patients. Methylation profiling classification showed three subgroups 3, seven subgroups 4, three SHH, and one WNT subgroup. Initial chemotherapy treatments followed established standard protocols as first-line therapy according to national recommendations: the “Milan strategy” 33 in nine patients, PNET5 protocol, 34 in three patients, and PNET4 guidelines 35 in two patients. All patients received radiotherapy, with five undergoing proton therapy specifically.
Baseline population characteristics.

MEMMAT, Medulloblastoma European Multitarget Metronomic Anti-Angiogenic Trial; SHH, Sonic Hedgehog Signaling Molecule; WNT, Wingless-Related Integration Site.
All patients experienced metastatic relapse, with all cases detected through surveillance MRI scans. Eight patients experienced their first recurrence, while six of them had multiple recurrences at the start of “Modified-MEMMAT” therapy. Among the six patients with third and beyond relapse, previous treatments included oral temozolomide (TMZ) for two cases, a cyclin-dependent kinase inhibitor for the fourth relapse in two cases, and an enzyme poly ADP ribose polymerase inhibitor for the fifth recurrence in one case. Three patients had positive CSF cytology in addition to leptomeningeal metastases (M1–M3). None of the patients had undergone surgical resection before enrollment. The median time from diagnosis to the relapse that prompted the start of “modified MEMMAT” treatment was 41.4 months (range: 8–75 months). Fifty-seven percent (eight patients) of the cohort received the “modified MEMMAT” schema as second-line treatment (referred to as modified MEMMAT-A). By contrast, 43% (six patients) received it as a third-line or beyond treatment after recurrence (referred to as modified MEMMAT-B).
Response evaluation
The median duration of treatment was 13.2 months (range: 6–29) with a mean time to response of 90 days (±41.6 days).
Among the 14 patients, with a median follow-up of 20 months (7–42), the best responses included CR observed in one patient (7%), PR in five patients (36%), and SD in eight patients (57%). Among the three patients with positive CSF cytology, the results turned negative and remained so after the first cycle of intrathecal topotecan. Three patients remained alive after initiation of therapy, with one patient in PR, one patient in PD, and one patient in “modified MEMMT” ongoing treatment (Figure 2).

Timeline and outcome of patients included in the study.
In the whole population, OS from the onset of disease was 60.2 months (19.33–130). At the 2- and 5-year follow-up OS was 92.9% (95% confidence interval (CI): 59.1–99.0) and 57.1% (95% CI: 28.4–78), respectively (Figure 3).
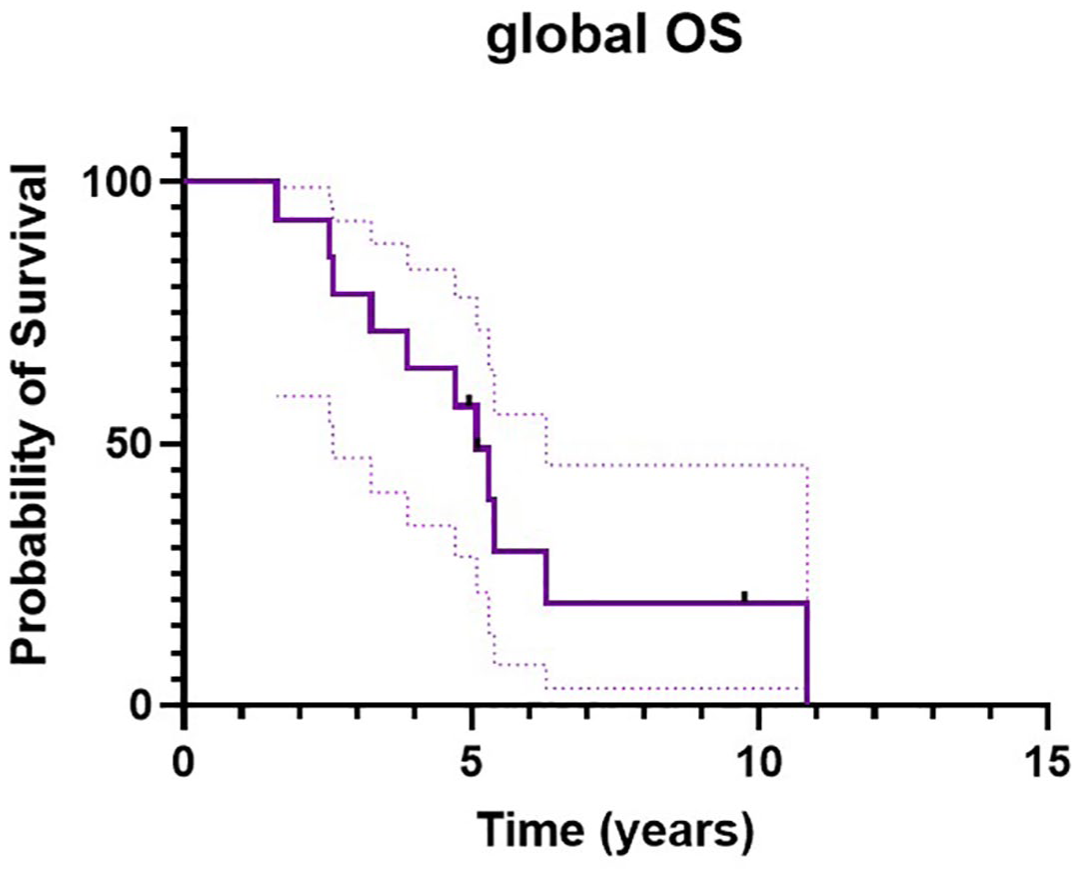
The overall survival from diagnosis for all patients.
The median OS from the date of start of the “modified-MEMMAT” regimen was 19.98 (8–42.4) months and the median PFS was 12.8 (6.2–32.3) months.
OS at 12 and 24 months was 78.6% (95% CI: 47.2–92.5) and 28.6% (95% CI: 8.8–52.4), respectively (Figure 4(a)). PFS at 6 and 12 months was 100% and 55.0% (95% CI: 25.8–76.8), respectively (Figure 4(b)).

(a) Overall survival from the date of start of the “modified-MEMMAT” regimen. (b) Progression-free survival from the date of start of the “modified-MEMMAT” regimen.
PD was observed in 71% of the cases; 11 patients died due to disease progression during the follow-up period.
Analysis of OS and PFS according to the number of lines of treatment (second line, A, or third and beyond, B) showed that OS from the time of start of “modified-MEMMAT” to death event or last follow-up had no difference between survival distributions of A and B (p = 0.22). At 1 year, the OS was 75.0% (95% CI: 31.5–93.1) for A and 83.3% (95% CI: 27.3–97.5) for B. At 2 years, the OS was 25.0% (95% CI: 3.7–55.8) for A and 16.7% (95% CI: 0.8–51.7) for B (Figure 5(a)).
There was no difference between the PFS distribution of A and B (p = 0.38). At 6 months, PFS was 100.0% for A and B. At 12 months, the PFS was 62.5% (95% CI: 22.9–86.1) for A and 41.7% (95% CI: 5.6–76.7) for B. At 2 years, the PFS was 25.0% (3.7%-55.8%) for A and 0.0% for B (Figure 5(b)).

(a) Overall survival according to number of treatment lines (A = “Modified-MEMMAT” as second line; B = “Modified-MEMMAT” as third and beyond line). (b) Progression-free survival according to the number of treatment lines (A = “Modified-MEMMAT” as a second line; B = “Modified-MEMMAT” as the third and beyond the line).
Toxicity and QoL
Overall, the “modified-MEMMAT” regimen was well tolerated, with no patients experiencing grade 4 toxicity. Grade 2 or 3 toxicities are detailed in Table 2. Hematological toxicity was the most common, observed in all patients, with 10 patients (55%) experiencing anemia. In addition, three patients exhibited grade 2 nephropathy with proteinuria, and two patients experienced hypertension of the same grade. One patient encountered grade 3 pancreatitis, necessitating the definitive discontinuation of treatment (Table 2).
Toxicity (CTCAE 5.0 version).

CTCAE, Common Terminology Criteria for Adverse Events; MEMMAT, Medulloblastoma European Multitarget Metronomic Anti-Angiogenic Trial.
The PEDSQL Cancer Module 3.0 was utilized to assess the QoL of the patient’s parents before the first dose of the drug (T0) and 6 months later (T1) indicating a significant improvement in the domain of nausea (p < 0.028), while the other investigated domains tended toward stability. This suggests that QoL did not deteriorate after the initiation of MC.
Discussion
MB is the most common malignant brain tumor in children, accounting for about 15%–20% of all pediatric brain tumors. 3
Despite progress in diagnostic techniques and treatment modalities, outcomes for high-risk patients have shown minimal improvement, with relapses in approximately 30% of MB patients and median survival following relapse of less than 1 year.36,37
Several salvage treatments, including various conventional drug combinations or HDCT, have been investigated for relapsed MB. These approaches have yielded discouraging outcomes. The OS rates after MB recurrence are reported, in the largest observational studies involving 581 children, as 38.2% at 1 year, 16.9% at 3 years, and 12.4% at 5 years. 38
Among the strategies employed for MB relapse, oral TMZ stands out as one of the most commonly utilized options. An Italian multi-center study showed a PFS rate for all patients at 6 and 12 months of 30% and 7.5%, respectively. Their median OS rate at 6 and 12 months was 42.5% and 17.5%, respectively. 39
Therefore, a definitive, impactful curative therapy for recurrent MB remains elusive, as evidenced by poor outcomes, short-lasting responses, and a high incidence of toxicity.3,40 –42
MC offers a novel approach in pediatric oncology, involving the repeated administration of low doses of chemotherapy to minimize toxicity. 43 These low, minimally toxic doses, without extended drug-free intervals, aim at disrupting vascular cells crucial for sustaining tumor cell proliferation, migration, and metastasis. 44 Besides its anti-angiogenic effects, MC seems to influence other cell populations within the tumor microenvironment, potentially triggering immunogenic pathways capable of activating immune responses. 45
Several studies have explored various MC combinations, initially for palliative treatment and later extended to high-risk solid tumors. 46
The MEMMAT protocol’s predecessor, the “5-drug” phase II trial 25 published by Robison, aimed to establish an outpatient-based approach for children with various relapsed solid tumors, adding fenofibrate, a PPAR-alpha agonist, to Celebrex, thalidomide, etoposide, and cyclophosphamide used in a prior “4-drug” pilot study. Fenofibrate was introduced based on its synergistic effects of anti-angiogenic activity with COX2 inhibition and cytotoxic metronomic therapy. 18
Based on this preliminary experience, a retrospective observational study using a modified “5-drug” regimen enhanced by IV bevacizumab every 2 weeks and alternating intraventricular therapy via an Ommaya reservoir consisting of etoposide and cytarabine, evaluated this alternative metronomic anti-angiogenic strategy in recurrent MB. The study conducted prior to the start of the formal MEMMAT trial, and termed “MEMMAT-like” included 29 patients with first or multiple MB recurrences and reported a median OS of 29.5 months. OS and PFS at 3 years were 48.3% ± 9.3% and 42.0% ± 9.5%, respectively, so higher than historically used regimens. 26
Even though the series are not numerous, several other studies corroborated the improvement of survival in pediatric patients treated with a preliminary MEMMAT/modified MEMMAT regimen in different types of brain tumors (ependymomas, high-grade gliomas, diffuse pontine glioma, refractory low-grade glioma, glial sarcoma). 47 The four-drug regimen originally reported by Kieran et al. 23 in a phase II trial showed a good safety profile as well as durable tumor control, particularly in the ependymoma setting; in fact the half of the patients maintained longer time to progression compared with what they had achieved on their most recent prior treatment regimen. The formulation was subsequently enhanced through the incorporation of intraventricular chemotherapy (etoposide and aracytin) alongside bevacizumab, culminating in the establishment of the regimen for metronomic multi-targeted anti-angiogenic treatment and used in several embryonal tumors (MBs, CNS PNET, embryonal tumor with abundant neuropil and true rosettes, pineoblastoma, and atypical teratoid rhabdoid tumor (ATRT)), showing both OS and EFS after 6 months of 75.0% ± 22% and equal to 0.0% at 12 months.44,48 The French Society for Children with Cancer highlighted a median OS of 26 months in children with various (MBs, ATRT, ependymomas) relapsed brain tumors. 47
Finally, Peyrl et al. 28 recently published the formal phase II results of the international transatlantic MEMMAT trial (ClinicalTrials.gov identifier: NCT01356290). The multicenter study evaluated the activity and toxicity profile of the combinatorial metronomic approach in pediatric patients who experienced first or consecutive MB recurrences. With a mean OS of 43.6% at 3 years and 22.6% at 5 years, the study supported the potential of MC in improving outcomes for pediatric patients with recurrent MB. The median OS of 25.5 months also favorably compares to the OS of 19 months in the better arm, including the addition of bevacizumab to TMZ and irinotecan, reported by the Children’s Oncology Group study. 41
Our analysis reports a real-life experience of 14 pediatric patients with relapsed MB treated with a modified MEMMAT regimen, to analyze the impact on survival and the treatment-associated morbidity. The combination demonstrated to be safe, prolonged median PFS and OS compared to historical controls (but not compared to original MEMMAT regimen), and did not impact QoL. The entire treatment for all patients was carried out on an outpatient basis.
Our modified MEMMAT regimen was characterized by some differences in using drugs; thalidomide was omitted due to its anti-angiogenic mechanism similar to bevacizumab. 49
Indeed, although the blood–brain barrier (BBB) remains a challenge in the treatment of brain tumors, the damage caused by previous treatments may enhance the therapeutic action of bevacizumab by improving its passage and contributing to the control of tumor growth in multi-treated MBs, where the BBB is often damaged by previous treatments. 50
In addition to bevacizumab, thalidomide and fenofibrate also have anti-angiogenic potential. Thalidomide is an immunomodulatory drug that has multiple effects, including anti-inflammatory, and anti-tumor properties. 51 However, its use is limited by side effects such as neuropathy, sedation, and teratogenicity, particularly when used in children. The primary evidence remains based on preclinical data and limited clinical trials, but its role in MB is still not fully established. 52
Bevacizumab, from our perspective, has represented the best choice for the mechanism of action (it directly targets vascular endothelial growth factor and effectively reduces blood flow to tumors), for the clinical evidence and safety and side effects (it is generally considered to have more manageable).53,54
Although we decided to omit thalidomide, we kept (because of the multiplicity of effects) the use of fenofibrate. Fenofibrate is a lipid-lowering agent that activates PPARs. These receptors play a role in regulating lipid metabolism, inflammation, and cell proliferation. Fenofibrate has shown potential in preclinical studies as an anticancer agent, as it may suppress tumor growth and enhance the effects of chemotherapy. Its effects on cell proliferation and apoptosis have sparked interest in its potential use as an adjunct therapy for cancers, including MB. It is thought to potentially enhance the anti-cancer effects of radiation and chemotherapy by reducing the proliferation of cancer cells and promoting cell death. 55
In addition, we chose to use intralumbar topotecan once every 4 weeks instead of intraventricular etoposide/cytarabine. The intrathecal administration of topotecan represents a targeted and effective therapeutic strategy for MB, particularly in relapsed or resistant cases, as it enables the drug to bypass the BBB and exert its action directly on the CNS.56,57
Compared with etoposide, topotecan has shown greater efficacy in controlling the disease, even in the presence of CNS metastases, allowing greater tumor focus and reducing systemic effects.58,59
Our modified MEMMAT regimen was characterized by some differences in using drugs; thalidomide was omitted due to its anti-angiogenic mechanism similar to bevacizumab. 49 In addition, we chose to use intralumbar topotecan once every 4 weeks instead of intraventricular etoposide/cytarabine. For the entire population, median OS from the date of start of the “modified MEMMAT” regimen was 19.98 (8–42.4) months and median PFS 12.8 (6.2–32.3) months. OS at 12 and 24 months was 78.6% (95% CI: 47.2–92.5) and 28.6% (95% CI: 8.8–52.4), respectively. PFS at 6 and 12 months was 100% and 55.0% (95% CI: 25.8–76.8), respectively. Analysis of OS and PFS according to the number of treatment lines (second line, A, or third and beyond, B) showed that OS and PFS had no difference between the survival distributions of A and B.
Results appear to be less encouraging than in the above-cited MEMMAT studies (Table 3). Major key differences between our series and all three other studies reside probably in both cohort characteristics and drugs used and may explain our comparatively shorter OS. Compared with the previous studies,26,27 a higher percentage of patients in our patient cohort received metronomic therapy from the third line onward, because of inconsistent drug distribution in cases with a VP-shunt in place. Furthermore, we used topotecan instead of etoposide/cytarabine for intrathecal therapy and applied it via LP rather than via an Ommaya reservoir. Additionally, to avoid repeated anesthesia, time intervals for intralumbar topotecan were extended to once every 4 weeks. In addition, the MEMMAT strategy allows for additional focal radiotherapy to persistent tumors after response evaluation, which was used in a proportion of the patients in all three above-mentioned studies.26,28,47
Comparison of MEMMAT and “modified-MEMMAT” studies.

ATRT, atypical teratoid rhabdoid tumor; MB, medulloblastoma; MEMMAT, Medulloblastoma European Multitarget Metronomic Anti-Angiogenic Trial; OS, overall survival; PFS, progression-free survival.
Like the study of Slavc et al. 26 and Peyrl et al., 28 our investigation focused exclusively on patients with recurrent MB. All three investigations aimed to assess the efficacy and toxicity of a MC regimen, with the phase II trial being the only one among them designed as a multicenter study. In addition to the differences in the therapeutic agents used, as previously discussed, it should be noted that our monocentric study included a sample size approximately half that of the study of Slavc et al. 26 and Peyrl et al. 28 Notably, PFS observed in our study was comparable to that reported in both the phase I and phase II trials. The difference in OS, however, may be attributed to the fact that, proportionally, our cohort included a higher number of patients treated beyond the second line of therapy.
Our data confirmed that the “modified-MEMMAT” regimen is well tolerated and safe, predominantly demonstrating grade 2–3 hematological toxicity, with only one patient experiencing grade 3 pancreatitis.
Any correlation between time of survival and molecular subgroup has been highlighted,26,28 though the pattern of relapse seemed to show association with the tumor’s biology in other series. 60 Our cohort, too heterogeneous and small to lend itself to analysis, comprised a WNT patient, usually the subgroup less involved in the relapse occurrence. This patient was the oldest of the cohort (26 years old), and it is commonly shared that, unlike those arising in children, adults with WNT tumors are postulated to have a worse prognosis.61,62 Moreover, the patient presented a germline pathogenetic gene APC mutation (well known for its negative regulator role of the WNT pathway). 63
QoL during metronomic therapy has rarely been reported but is even more important for patients with an overall poor prognosis. QoL, low at the baseline, due to prior therapy and associated sequelae did not worsen during modified-MEMMAT treatments.28,47
According to the literature, our study revealed that the “modified-MEMMAT” regimen did not significantly compromise patients’ QoL, highlighting a minor load in nausea, probably due to the daily use of ondansetron due to continuous oral chemotherapy. While documentation of the QoL during metronomic therapy has been limited, its significance is amplified for patients confronting a challenging overall prognosis. 64
Although our results in terms of PFS and OS appear comparatively inferior to those reported in the literature,26,28,47 this account emphasizes the treatment’s tolerability, with the overarching goal of extending survival through outpatient care.
Conclusion
In conclusion, there is a lack of standardization in treatment protocols for progressive and relapsed MB, presenting a challenge in determining the optimal approach for affected children. Our primary objective has been to assess the effectiveness and tolerance of a metronomic schema administered in children with recurrent MB. Our study confirms that the “modified-MEMMAT” regimen represents a promising approach for patients with relapsed/refractory MB. These findings provide valuable real-life data on a homogeneous group of patients. It is clear that an MC combination not only induces responses but also supports the continuity of PFS in patients with a history of multiple intensive prior treatments. In addition, it is crucial within this patient cohort, where curative options are lacking, to preserve QoL, preferably in an outpatient regimen.
