Abstract
Background:
Immune checkpoint inhibitors are used either as monotherapy or in combination with cytotoxic agents in metastatic gastric cancer (mGC). Studies show that circadian rhythms are essential for immune system functions, including anticancer immunity.
Objectives:
This study evaluated whether the time of day of nivolumab infusion altered the efficacy of mGC treatment.
Design:
This was a retrospective cohort study.
Methods:
We retrospectively analyzed the data of 296 consecutive patients with mGC who received nivolumab monotherapy between December 2014 and December 2022. Patients were divided into early (EA)- and late (LA)-nivolumab infusion groups: the EA and LA groups received ⩾70% and <70% of their infusions before 14:00, respectively. Treatment efficacy was compared between the groups.
Results:
A total of 248 patients were eligible: 140 and 108 in the EA and LA groups, respectively. Most baseline characteristics were similar between the groups, except for differences in the neutrophil-to-lymphocyte ratio. The progression-free survival (PFS) and overall survival (OS) were significantly better in the EA than in the LA group (median PFS, 2.3 vs 1.6 months; hazard ratio (HR), 0.65; p < 0.01; median OS, 7.6 vs 3.9 months; HR, 0.64; p < 0.01). In multivariate analyses, EA was an independent prognostic factor for PFS (adjusted HR, 0.70; p < 0.01) and OS (adjusted HR, 0.67; p < 0.01). Immune-related adverse events were more frequent in the EA than in the LA group (40.7% vs 29.6%, p = 0.07).
Conclusion:
Our data suggest that scheduling nivolumab infusions before mid-afternoon should be considered in daily practice for the treatment of mGC.
Introduction
Gastric cancer is the fifth most common cancer and the fourth leading cause of cancer-related mortality worldwide. 1 Systemic chemotherapy is the standard of care for improving survival and enhancing the quality of life of patients with unresectable locally advanced or metastatic gastric cancer (mGC).2,3 The prognosis of mGC remains poor, with a median overall survival (OS) of approximately 1 year. 4 However, with advances in drug development, there has been a gradual prolongation of the OS of patients with mGC in clinical practice. 5
In recent years, immune checkpoint inhibitors (ICIs), such as those targeting programmed cell death-1 (PD-1), have shown better clinical outcomes in patients with various solid tumors.6–8 Immune checkpoints downregulate the immune system, preventing its overactivation, and are used by cancer cells as an escape mechanism from the immune system. Nivolumab is a humanized monoclonal immunoglobulin G4 PD-1 antibody that has become standard of care as a third- or later-line treatment for mGC based on the ATTRACTION-2 trial, showing improved OS compared with placebo. 9
Chronotherapy is a treatment method that improves efficacy by administering drugs that align with a patient’s internal clock or circadian rhythm. The circadian rhythm uses cues from the environment, such as light and food intake, to establish an approximately 24-h rhythm for most physiological processes. The relationship between circadian rhythms and treatment efficacy has been reported not only for cytotoxic anticancer drugs but also for ICIs in various carcinomas, such as melanoma, non-small-cell lung cancer, renal cell carcinoma, and esophageal cancer.10–17 This phenomenon is thought to be influenced by the existence of a circadian rhythm in the cancer immune system, which affects the localization and proliferation of immune cells because of variations throughout the day.18,19
However, to our knowledge, there are no reports on whether the timing of nivolumab infusion affects the treatment efficacy and safety in mGC. This study aimed to evaluate the efficacy and safety depending on the time of day of nivolumab infusion as a later-line treatment in patients with mGC.
Materials and methods
Patients and treatment
This single-center retrospective study included patients with mGC who received nivolumab monotherapy as a third- or later-line treatment between December 2014 and December 2022 at Aichi Cancer Center Hospital. Patients received nivolumab at 3 mg/kg or 240 mg every 2 weeks or 480 mg every 4 weeks. The inclusion criteria were as follows: (1) histologically confirmed advanced unresectable or recurrent gastric/esophagogastric junction adenocarcinoma; (2) age ⩾20 years; (3) prior treatment with two or more lines of chemotherapy, including fluoropyrimidine and taxanes; (4) Eastern Cooperative Oncology Group (ECOG) performance status (PS) 0–2; (5) adequate bone marrow, hepatic, and renal function; and (6) received at least two injections of nivolumab. The study protocol was approved by the Ethics Committee and Institutional Review Board of the Aichi Cancer Center Hospital (approval number: IR051072). This study was conducted in accordance with the ethical principles of the Declaration of Helsinki. The requirement for written informed consent was waived by the Institutional Review Board of Aichi Cancer Center Hospital because of the retrospective, non-interventional design, and opt-out option available on the institution’s website.
Treatment group
Infusion times were retrieved from medical records and classified into two groups, with patients who received ⩾70% of infusions before 14:00 assigned to the early time infusion (EA), and patients who received <70% of infusions before 14:00 assigned to the late time infusion (LA). The cutoff value was set at 14:00 for the following reasons: (1) when the infusion times were divided into 30-min intervals, the interval with the highest number of infusions was between 13:30 and 14:00 (Supplemental Figure 1); and (2) based on a previous report. 10
Statistical analysis
The Chi-square test was used for categorical data, whereas the Mann–Whitney U test was used for continuous data. The objective response rate (ORR) was assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST) v1.1. 20 ORR was defined as the proportion of patients with complete or partial response. The disease control rate (DCR) refers to the proportion of patients with a complete response, partial response, or stable disease. Progression-free survival (PFS) was defined as the time from the initiation of nivolumab treatment until the date of disease progression or death due to any cause. Survivors without disease progression were censored at the last contact. OS was defined as the time from the initiation of nivolumab treatment until the date of death due to any cause or censored at the latest follow-up visit in surviving patients. PFS and OS were estimated using the Kaplan–Meier method and compared using the log-rank test. We conducted a Cox proportional hazards model analysis to calculate the hazard ratio (HR) of each factor in the different groups of patients. For the multivariate analysis, variables were selected based on factors with p values <0.10 in the univariable analysis. To investigate the impact of nivolumab monotherapy infusion time on survival, we conducted subgroup analyses with patients categorized into three groups: group 1 received at least 70% of their infusions before 14:00, group 2 received at least 30% of their infusions before 14:00 and 30% of their infusions after 14:00, and group 3 received at least 70% of their infusions after 14:00. 13 This three-group comparison was not included as a factor in the multivariate analysis. The modified Glasgow prognostic score (mGPS) was defined based on previous reports. 21 Pre-treatment blood test results were used in this study; the cutoff value for the neutrophil-to-lymphocyte ratio (NLR) was set at 2.4, which was the median value for the patients participating in this study. Adverse events related to treatment with ICIs were retrospectively assessed using medical records from the start of treatment to the last follow-up. A p value of <0.05 was considered statistically significant. All statistical analyses were performed using EZR version 1.64 (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R version 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria). This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Results
Patient characteristics
Of the 296 patients who received nivolumab between December 2014 and December 2022, 248 were eligible for this study, excluding 48 patients who received only one infusion (n = 44) or no prior treatment with fluoropyrimidines or taxanes (n = 4). A total of 140 patients were assigned to the EA group and 108 to the LA group (Figure 1).
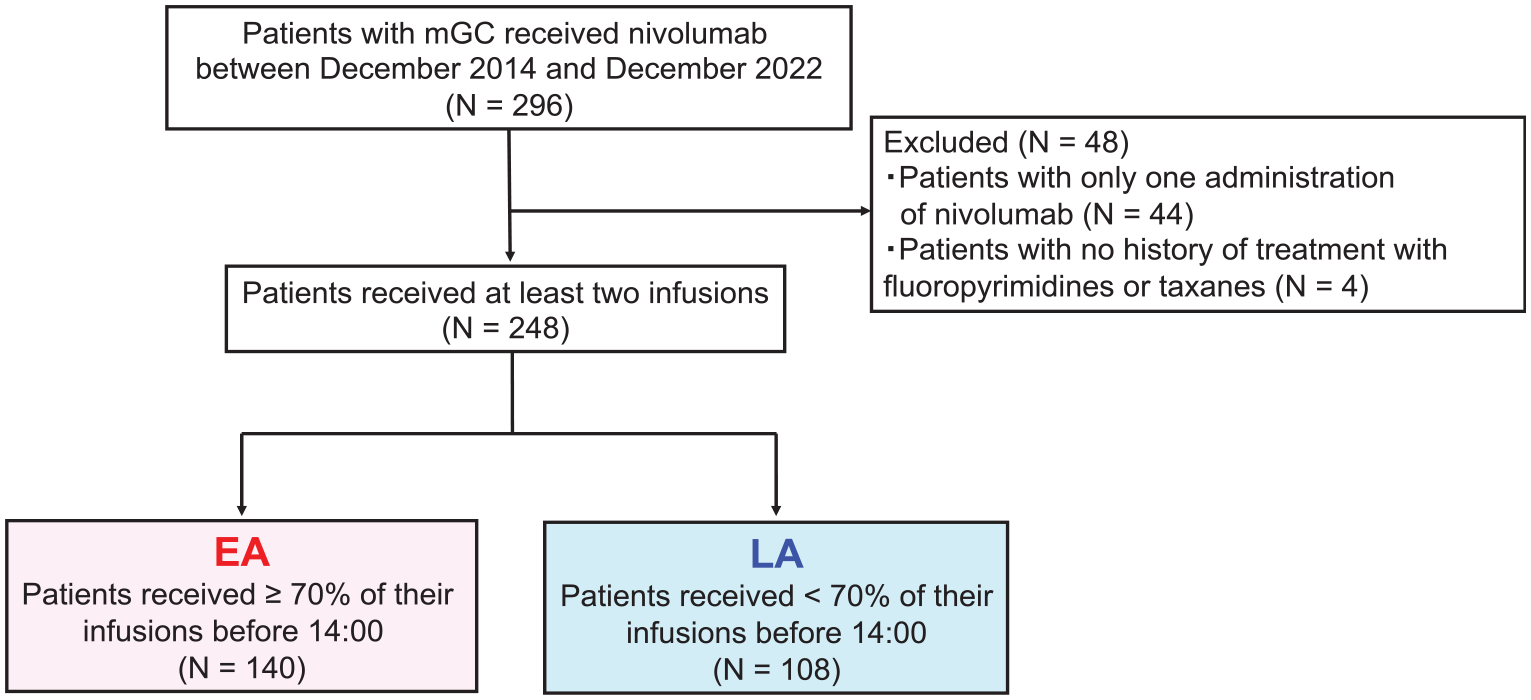
Flowchart of the selection process for the early or late infusion groups from cases receiving nivolumab monotherapy for mGC between December 2014 and December 2022.
Patient characteristics are summarized in Table 1. Most baseline characteristics were similar between the two groups. Significant differences were observed in the NLR (NLR < 2.4, 59% vs 38%, p < 0.01), median infusion start time (11:50 vs 14:22, p < 0.01), and total number of infusions (1039 vs 533, p < 0.01). In addition, the mGPS scores tended to be better in the EA group than in the LA group.
Patient characteristics.

EA, early group; ECOG PS, Eastern Cooperative Oncology Group Performance Status; HER2, human epidermal growth factor 2; LA, late group; mGPS, Modified Glasgow prognostic score; MSI, microsatellite instability; MSI-H, microsatellite instability high; MSS, microsatellite stable; NLR, neutrophil-to-lymphocyte ratio.
Clinical outcome
At the data cutoff date, the median follow-up period after initiating nivolumab therapy was 9.0 months (interquartile range, 4.5–15.3 months, EA: 11.7 months, LA: 6.2 months). Among the 149 patients with measurable lesions, ORR (17% vs 3%, p = 0.01) and DCR (47% vs 20%, p < 0.01) were significantly better in the EA than in the LA group (Figure 2(a) and (b), and Table 2).

Waterfall plot of the maximum percent change from baseline for the sum of measurable lesions (a) in the early group and (b) late group.
Tumor response.

ORR was defined as the proportion of patients who had CR or PR.
DCR was defined as the proportion of patients who had CR or PR, or SD.
CI, confidence interval; CR, complete response; DCR, disease control rate; EA, early group; LA, late group; NE, not evaluable; ORR, objective response rate; PD progressive disease; PR, partial response; SD, stable disease.
Patients in the EA group had significantly longer PFS than those in the LA group (median PFS, 2.3 vs 1.6 months; HR, 0.65; 95% confidence interval (CI), 0.50–0.86; p < 0.01; Figure 3(a)). The EA group also showed significantly prolonged OS compared with the LA group (median OS, 7.6 vs 3.9 months; HR, 0.64; 95% CI, 0.48–0.85; p < 0.01; Figure 3(b)). The multivariate analysis showed that the timing of nivolumab infusion was independently associated with longer PFS (adjusted HR, 0.70; p < 0.01) and OS (adjusted HR, 0.67; p < 0.01; Table 3).

Kaplan–Meier estimates of (a) PFS and (b) OS.
Univariate and multivariate analyses for (a) progression-free survival and (b) overall survival.

CI, confidence interval; EA, early group; ECOG PS, Eastern Cooperative Oncology Group Performance Status; LA, late group; mGPS, modified Glasgow prognostic score; NLR, neutrophil-to-lymphocyte ratio.
We conducted an analysis with varying cutoffs to examine whether there was a favorable trend in PFS and OS in the EA group, even with different proportions of infusion cutoffs. In the EA group, we observed a favorable trend in PFS and OS from early administration rates above 50% (Supplemental Figure 2(a)–(c)). In the subgroup analysis, there were 140 patients in group 1, 66 in group 2, and 42 in group 3. The efficacy results demonstrated consistent and statistically significant differences in both PFS and OS between the three groups (p < 0.01, respectively; Supplemental Figure 3(a) and (b)).
Safety
Adverse events associated with immunotherapy are shown in Table 4. Approximately one-third of patients had at least one immune-related adverse event (irAE). The most common irAEs were skin toxicity (36%), including rash and pruritus, thyroid dysfunction (14%), and transaminase elevation (9%). Overall, irAEs tended to be more common in the EA than in the LA group (40.7% vs 29.6%, p = 0.07), and skin toxicities of any grade were more frequent in the EA group (23.6% vs 12.0%, p < 0.01). Grade 3/4 irAEs were observed in 7% and 6% of the patients in the EA and LA groups, respectively, with no significant difference. No treatment-related deaths occurred.
Adverse events.

EA, early group; irAEs, immune-related adverse events; LA, late group.
Post-progression anticancer therapies
Four patients in the EA group and two in the LA group were still undergoing treatment at the time of the latest follow-up visit. Disease progression was the most common reason for discontinuing nivolumab in both groups (97.1% in the EA group and 98.1% in the LA group), and 0.7% and 4.6% of the patients in the EA and LA groups, respectively, discontinued nivolumab owing to adverse events. The number of patients who received subsequent chemotherapy was significantly higher in the EA than in the LA group (53.6% vs 32.4%, p < 0.01; Table 5).
Status of discontinuation/subsequent treatment.

EA, early group; LA, late group.
Discussion
To the best of our knowledge, this is the first report to demonstrate a correlation between the time-of-day of nivolumab infusion and clinical outcomes in patients with mGC who received it as a later-line treatment. Administering >70% of the nivolumab infusions before 14:00 was associated not only with better short-term treatment efficacy, including ORR, but also with long-term treatment efficacy, reflected in improved PFS and OS. Early nivolumab infusion was confirmed to be an independent prognostic factor for both PFS and OS in the multivariate analyses. Moreover, the incidence of irAEs of any grade was higher in the EA than in the LA group. Differences in subsequent treatment rates and patient backgrounds may have influenced OS; however, these findings suggest that optimal timing of ICI administration to stimulate the adaptive immune system can enhance the efficacy of ICI therapy.
The relationship between the timing of ICI administration and treatment efficacy has previously been reported in patients with metastatic melanoma. 12 The current study is in accordance with the MEMOIR study, which showed that patients with metastatic melanoma receiving <20% of ICI infusions after 16:30 had improved OS compared with those who had later infusions. Furthermore, our study is consistent with a meta-analysis of 13 clinical studies, demonstrating that patients with cancer who received most of their ICIs earlier rather than later in the day showed a statistically significant improvement in ORR, PFS, and OS. 17 One possible mechanism by which the timing of ICI administration influences treatment outcomes is as follows: immune cells, such as naïve T cells, memory T cells, and NK cells, as well as cytokine production, exhibit circadian rhythms. During the morning to early afternoon, naïve T cells tend to migrate to the lymph nodes. Therefore, when T cells are reactivated by ICIs during this time, they are more likely to be localized in the lymph nodes, making them more accessible to antigen presentation, thereby enhancing the therapeutic effect of ICIs. 22 These studies have examined anti-PD-1, programmed death-ligand 1 (PD-L1) inhibitors, anti-cytotoxic T lymphocyte-associated protein 4, and their combinations in various carcinomas; consequently, the patients’ baseline characteristics are not necessarily well balanced. Although not all potentially confounding factors could be assessed, a strength of our study is that the analysis included only nivolumab as a late-line treatment for mGC, and the baseline characteristics were well-matched.
In this study, the cutoff time was set at 14:00, although there was no clear basis for this specific time. The cutoff times in previous studies varied; two different cutoff times (16:00–16:30 or 12:00–14:00) have been adopted.10–16 These cutoff times were based on the results of vaccination studies that revealed reduced antibody formation in healthy individuals vaccinated after 16:00–16:30.12,23 By contrast, the 12:00–14:00 cutoff times correspond to the cutoff of the individual timing of ICI infusions in the study population. In our study, the cutoff time was defined as 14:00 based on the individual timing of the ICI infusions. Inconsistency in the timing of administration was caused by variations in patient schedules or the availability of treatment rooms, leading to difficulty in determining the cutoff values. However, even when the cutoff time for administration was changed to times other than 14:00, the early administration group consistently showed a favorable trend.
As the plasmatic half-life of nivolumab is approximately 3–5 weeks, 24 it is unclear why earlier administration results in higher efficacy. Immune cells undergo circadian rhythms many times within this plasmatic half-life period, implying that their efficacy and toxicity should be independent of the dosing time. However, the pharmacodynamics of nivolumab within the tumor microenvironment and in tumor-draining lymph nodes may significantly impact its efficacy. This suggests that when T-cell localization coincides with ICI distribution, there may be enhanced T-cell stimulation. 25 In preclinical models, pembrolizumab concentrations peaked in lymphoid organs 30 min after injection and were cleared approximately half within 12 h. 26 Therefore, irrespective of the plasmatic half-life of nivolumab, having its distribution into tissues and the localization of T cells coincide at the time of priming induction may be considered important for enhancing T-cell activation. In addition, the effects of corticosteroids on the circadian rhythm of the immune system further complicate these mechanisms. 27 The current investigation did not examine the levels of cortisol, white blood cells, such as lymphocytes, and immune-related cells, such as dendritic cells, or perform macrophage subset analysis using flow cytometry prior to and following nivolumab administration. Circulating cortisol levels correlate with the proportions of leukocyte subsets, such as naïve and memory T cells, and both fluctuate over a 24-h period; therefore, the timing of evaluation may influence the results and should be carefully considered. 19 However, further elucidation of the mechanism might help optimize the timing of ICI administration.
Previous studies have reported that the NLR, peritoneal metastasis, liver metastasis, ECOG PS, and GPS were prognostic factors for patients with mGC undergoing nivolumab therapy.28–31 From the real-world data of a large cohort (n = 487), peritoneal metastasis and ECOG PS were associated with poor prognosis for PFS. 29 In our study, multivariate analysis of previously reported prognostic factors revealed that early infusion time was the most significant factor. In addition, the microsatellite instability status and PD-L1 combined positive score have been reported as factors that predict susceptibility to treatment with ICIs. 30 However, in our study, because of the small number of patients in whom these biomarkers were measured, it was not possible to examine the relationship between the timing of administration and these biomarkers. Further studies are needed to clarify the significance of ICIs stratified by PD-L1 expression, microsatellite instability status, and timing of administration.
Previous studies have demonstrated the relationship between irAEs and the effectiveness of ICIs in various cancer types.32–34 The MEMOIR study demonstrated a higher number of patients who experienced toxicity leading to discontinuation of ICIs in those who had early rather than later infusions (22% vs 18%). 12 Moreover, an association between skin toxicity caused by nivolumab and treatment outcomes, such as PFS and OS, has been reported. 34 These findings support those of our study; the proportion of skin toxicity was significantly higher in the EA than in the LA group, correlating with a difference in survival.
Our study has some limitations. This was not a prospective randomized study. Furthermore, in the era of immunochemotherapy and approval for its use as a first-line therapy for mGC, this study only examined cases of nivolumab monotherapy as a later-line treatment. Further investigations are warranted to evaluate whether a consistent relationship exists between early administration and treatment response when treated with ICIs other than nivolumab, or in combination with chemotherapy.
Conclusion
Adjusting the timing of nivolumab administration may impact treatment efficacy in mGC without additional cost and complex examinations. The primary circadian-dependent clinical and biological mechanisms underlying nivolumab infusion timing and efficacy remain unclear, necessitating prospective randomized trials and translational research.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251339921 – Supplemental material for Impact of nivolumab monotherapy infusion time-of-day on short- and long-term outcomes in patients with metastatic gastric cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359251339921 for Impact of nivolumab monotherapy infusion time-of-day on short- and long-term outcomes in patients with metastatic gastric cancer by Yasunobu Ishizuka, Yukiya Narita, Tomoki Sakakida, Munehiro Wakabayashi, Hiroyuki Kodama, Kazunori Honda, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Masashi Ando, Masahiro Tajika and Kei Muro in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251339921 – Supplemental material for Impact of nivolumab monotherapy infusion time-of-day on short- and long-term outcomes in patients with metastatic gastric cancer
Supplemental material, sj-docx-2-tam-10.1177_17588359251339921 for Impact of nivolumab monotherapy infusion time-of-day on short- and long-term outcomes in patients with metastatic gastric cancer by Yasunobu Ishizuka, Yukiya Narita, Tomoki Sakakida, Munehiro Wakabayashi, Hiroyuki Kodama, Kazunori Honda, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Masashi Ando, Masahiro Tajika and Kei Muro in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
