Abstract
Background:
Rectal cancer poses a significant global health burden. Conventional neoadjuvant chemoradiotherapy (nCRT) demonstrates limited efficacy in achieving disease-free survival (DFS) and organ preservation. Total neoadjuvant therapy (TNT), an emerging paradigm integrating systemic chemotherapy with radiotherapy, aims to address these limitations.
Objectives:
To evaluate the short- and long-term oncological outcomes and organ preservation feasibility of TNT in high-risk locally advanced rectal cancer (LARC) patients.
Designs:
A retrospective study was conducted to analyze the short-term and long-term results after total nCRT.
Methods:
This retrospective study involved 110 rectal cancer patients with high-risk features treated at the Fudan University Shanghai Cancer Center from 2008 to 2022. Patients underwent comprehensive staging and received nCRT followed by either total mesorectal excision (TME) or a watch and wait (W&W) strategy. The regimen included concurrent chemoradiotherapy with 50 Gy/25 fractions and at least six cycles of induction or consolidation chemotherapy. Both short-term and long-term outcomes were collected and analyzed.
Results:
Among the LARC patients, 73.6% were stage III, and 47.3% opted for the W&W strategy. The combined rate of clinical complete response or confirmed pathological complete response (pCR) reached 49.1%. During follow-up, 10% of patients experienced local regrowth. The 3-year DFS and overall survival (OS) rates were 75.7% and 92.4%, respectively. The W&W strategy could achieve superior outcomes than patients without pCR in DFS (p = 0.021) and OS (p = 0.006).
Conclusion:
TNT demonstrates durable survival outcomes and facilitates organ preservation in over 50% of high-risk LARC patients. Intensive surveillance is critical for the early detection of local regrowth.
Trial registration:
Our study is retrospective in nature, rather than a prospective clinical trial. Therefore, registration was not conducted, and the Clinical Trial Number is not applicable.
Plain language summary
Introduction
With the progress of treatment, the short-term efficacy of localized advanced rectal cancer (LARC) has been significantly improved. 1 By augmenting the number of chemotherapy cycles preceding and following radiotherapy, total neoadjuvant therapy (TNT) has emerged as the preferred strategy for high-risk LARC. 2 While TNT enhances pathological complete response (pCR) rates, it also expands opportunities for organ preservation via non-operative strategies like the watch-and-wait (W&W) approach in patients achieving clinical complete response (cCR).3–5 Consequently, a growing emphasis has been placed on optimizing organ preservation protocols while ensuring durable oncological outcomes.6,7 Nevertheless, more data are still required to elucidate the long-term efficacy of the TNT approach.
Total neoadjuvant chemoradiotherapy (nCRT) was considered because both chemotherapy and radiation therapy are given before surgery. Despite its clinical adoption, critical ambiguities persist in TNT implementation. The definition of TNT remains heterogeneous across trials, particularly regarding chemotherapy sequencing (induction vs consolidation) and regimen selection (fluoropyrimidine monotherapy vs FOLFOX/CAPEOX). In 2018, the landmark MSKCC retrospective study (2018) first demonstrated regimen-dependent efficacy variations, with FOLFOX-based TNT achieving superior pCR rates compared to capecitabine monotherapy. 8 Subsequently, in prospective phase III clinical trials such as CAO/ARO/AIO-12,9,10 OPRA, 11 PRODIGE 23, 12 STELLAR, 13 and RAPIDO, 14 which are primarily chemotherapy-based TNT clinical trials, there were variations in the scheme and number of chemotherapy cycles administered during the consolidation chemotherapy.
With the escalation of chemotherapy intensity, more pronounced tumor shrinkage consequently increased the rate of complete clinical remission and prolonged DFS.15,16 In response to clinical demands for organ preservation and enhanced quality of life, the strategy of W&W for patients, based on rigorous evaluation, has emerged as a pivotal focus in the current landscape of rectal cancer treatment.17,18
Investigations into TNT have revealed inconsistencies among diverse clinical trials, and the optimal application of TNT strategies remains a contentious issue, 19 thereby highlighting the significance of acquiring real-world data to fully comprehend the nuances of TNT. Summarizing and reporting on the status of the TNT research is crucial for selecting appropriate patients for this treatment regimen in the future.
Methods
Patient population
This study enrolled rectal cancer patients with high-risk factors who were treated at the Fudan University Shanghai Cancer Center (FUSCC) and underwent nCRT at the institution. Before treatment, all patients underwent thorough staging examinations, including rectal digital examination, colonoscopy with biopsy confirmation of rectal adenocarcinoma, pelvic magnetic resonance imaging (MRI) for disease staging, and chest or abdominal MRI/CT to rule out common distant organ metastases. Following extensive follow-up periods, the study reported the treatment status of patients utilizing the TNT approach at our center. All patients had complete follow-up data, and relevant clinical information was extracted from the electronic medical record system. The study was approved by the Institutional Review Board of FUSCC and adhered to the ethical standards outlined in the 1964 Helsinki Declaration and its subsequent amendments.
Neoadjuvant regimens
The patients, diagnosed with high-risk rectal cancer or LARC as per the eighth edition of the AJCC staging system (T3/T4/N+), underwent extensive preoperative assessment. The treatment protocol entailed long-course concurrent chemoradiotherapy, consisting of 50 Gy/25 fractions with either single-agent capecitabine or a capecitabine and irinotecan combination during radiotherapy. For patients receiving capecitabine alone, the dosage was adjusted based on their UGT1A1 genotype, with a dosage of 825 mg/m2 twice daily for those with the UGT1A1*1*1 genotype. Those on capecitabine and irinotecan received capecitabine at a dosage of 625 mg/m2 twice daily, alongside weekly irinotecan, which was administered at a dose of 80 mg/m2 among patients with the UGT1A1*1*1 genotype and 65 mg/m2 among those with the UGT1A1*1*28 genotype. The radiation techniques and treatment volumes were consistent with our prior Cinclare study 1 and Supplemental Material. Subsequently, patients underwent at least six cycles of consolidation chemotherapies before the surgery or the W&W strategy, with regimens tailored by physicians, including capecitabine-based, irinotecan, or oxaliplatin combined with fluoropyrimidine agents.
The variability in chemotherapy regimens and frequencies during induction and peri-radiation chemotherapy presents challenges for uniform comparison. The specific strategies and frequencies of consolidation chemotherapy are determined by clinical physicians after evaluation. To streamline subsequent research reporting, consolidation chemotherapy is categorized into four groups based on drug types: capecitabine monotherapy, irinotecan with fluoropyrimidine drugs (XELIRI/FOLFIRI), oxaliplatin with fluoropyrimidine drugs (XELOX/FOLFOX), and FOLFIRINOX. Detailed information on chemotherapy strategies exchange during TNT treatment is provided in Supplemental Table 1.
To ensure standardized comparison and analysis between oral (XELOX/XELIRI) and intravenous chemotherapy (FOLFOX/FOLFIRI/FOLFIRINOX), it is essential to equate drug efficacy and treatment duration differences. Given that two cycles of oral chemotherapy equate to three cycles of intravenous chemotherapy in time duration, the efficacy ratio is adjusted to 2:3. Consequently, all treatments are standardized as intravenous chemotherapy cycles for statistical analysis and comparison.
Treatment selection for surgery and W&W strategy
After undergoing long-course nCRT, subsequent interventions or follow-up strategies are chosen based on the patient’s disease remission status. For patients with residual disease or deemed at high risk of recurrence or metastasis, total mesorectal excision (TME) surgery is recommended. For patients with excellent disease regression and no residual tumor after rigorous assessment, they are advised to undergo curative TME surgery or adopt a W&W strategy with close surveillance. Patients declining TME surgery will undergo local excision following rigorous eligibility evaluation, with detailed selection criteria provided in the Supplemental Material. This stratified surgical approach ensures organ preservation while maintaining oncological safety for appropriately selected candidates. Given the early treatment timeline of these patients, we exclusively recommended the W&W strategy for those who met rigorously confirmed cCR criteria. The decision to recommend the W&W strategy is made when all of the following criteria are met: absence of observable residual tumor or presence of only residual fibrosis, the short axis of each target lymph node decreased to ⩽5 mm on MRI, absence of observable residual tumor or presence of only a scar on endoscopy, and absence of palpable tumor, nodules, or rectal wall stiffness on digital rectal examination. These criteria are established based on the Consensus of Chinese Watch & Wait Database Research Cooperation Group (CWWD), ESMO guidelines, and previous research.20–22 The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. 23
pCR and cCR
The pCR was defined as an absence of tumor cells in the surgical specimens from the primary tumor and the regional lymph nodes (ypT0N0) according to the described criteria before.24,25 Tumor regression grade was evaluated according to the criteria devised by Edge and Compton. 26 The cCR was defined as the absence of palpable tumor on digital examination, the absence of residual tumor on pelvic MRI and endoscopy, without metastasis and a sustained absence of residual tumor after the consolidation chemotherapy. Only rigorous evaluation confirmed for cCR patients was recommended for the W&W strategy. These patients received six cycles of consolidation chemotherapy and were monitored closely with digital examination and endoscopy every 1–2 months and chest and abdominal CT and pelvic MRI every 3 months.
Outcomes
Primary outcomes were disease-free survival (DFS) and overall survival (OS) of LARC patients. In this research, we also report local recurrence (LR) rate of the cohort. OS was defined as time from the patient was diagnosed with LARC. DFS was defined as time from the surgery for the patients received TME, for the patients with W&W strategy, the DFS date was from the patients finish the treatment of neoadjuvant consolidated chemotherapy.
Statistical analysis
The distribution of continuous numeric data was affirmed by the Kolmogorov–Smirnov test. Patient characteristics are presented as median and standard deviation for continuous variables and as numbers with percentages for categorical variables. The Kaplan–Meier method was used to determine recurrence, metastasis, and survival probabilities. The median follow-up time was estimated using reverse Kaplan–Meier analysis, all estimates of rates related to time are based on the Kaplan–Meier method in SPSS version 27 (IBM, NewYork, USA). And subgroup analysis within the survival analysis was conducted using the Log-rank test in SPSS version 27 (IBM, NewYork, USA). The standard error of the confidence interval is calculated based on the Kaplan–Meier method in SPSS version 27 (IBM).
Results
Patients
Between 2008 and 2022, 110 consecutive patients with histologically confirmed rectal adenocarcinoma were enrolled. The cohort primarily comprised AJCC stage I–III (cT3–4/N+) disease, with selective inclusion of cT2 cases exhibiting anatomical constraints for organ preservation (tumors ⩽5 cm from the anal verge), high-risk pathological features (lymphovascular/perineural invasion), and patient refusal of abdominoperineal resection. In total, 73.6% (81/110) of patients were diagnosed with stage III. The pretreatment demographic and clinical characteristics are shown in Table 1. The median follow-up time was 34.2 (95% CI: 30.7–38.1) months.
Characteristics for TNT patients.

The p-value for age was 0.20 from the Kolmogorov–Smirnov normality test, which is normally distributed.
cN, clinical nodal; cT, clinical tumor T stage; EMVI, extramural venous invasion; MRF, mesorectal fascia; W&W, Watch and Wait.
Treatment information
All patients with LARC underwent long-course radiotherapy (50 Gy/25 fractions) combined with either single-agent capecitabine or capecitabine-irinotecan chemoradiotherapy. The concurrent chemotherapy protocols during radiation therapy align with our prior Cinclare study methodology, while detailed consolidation chemotherapy regimens administered between radiotherapy and definitive treatment are specified in Supplemental Table 1. Chemotherapeutic agents were selected at physician discretion across ⩾6 treatment cycles, with regimen distribution as follows: induction chemotherapy in 10.9% (12/110), XELOX (capecitabine/oxaliplatin) in 4.5% (5/110), and XELIRI (capecitabine/irinotecan) in 6.4% (7/110). Treatment-related toxicity profiles during this phase are comprehensively documented in Supplemental Material. Definitive management—either TME or W&W strategy—was determined through multidisciplinary evaluation incorporating tumor response metrics, residual disease assessment, and patient preference after thorough informed consent. The detailed treatment information is systematically cataloged in Table 2.
Treatment information for TNT patients.

FOLFIRI, 5-fluorouracil plus irinotecan; FOLFOX, 5-fluorouracil plus oxaliplatin; W&W, Watch and Wait; XELIRI, capecitabine plus irinotecan; XELOX, capecitabine plus oxaliplatin.
The diagram of the clinical treatments and tumor response of TNT treatment patients was shown in Figure 1.
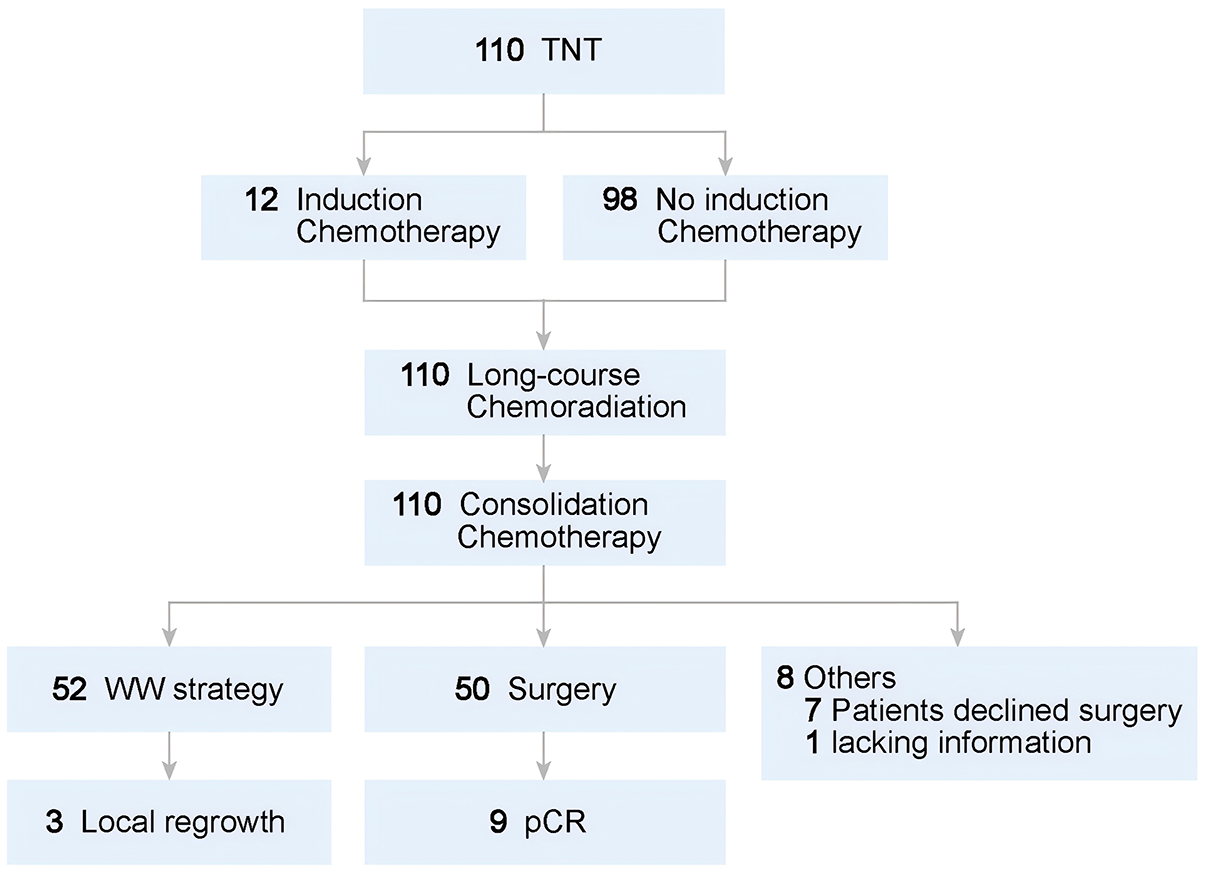
Diagram of the clinical tumor response of TNT treatment patients.
W&W, surgical information, regrowth, and organ preservation
A total of 47.3% (52/110) of patients underwent a W&W strategy, while 8.2% (9/110) achieved pCR post-surgery. Among those who pursued the W&W strategy, 5.77% (3/52) experienced LR with regrowth, and 9.6% (5/52) experienced metastasis during follow-up. Notably, all instances of regeneration were found within the intestinal wall. Following neoadjuvant therapy, the rate of cCR including patients with W&W or confirmed pCR reached 55.5% (61/110). Among these patients, 11.5% (7/61) experienced LR and metastasis.
Ultimately, among all the patients, 50 patients proceeded to surgery, with 7 individuals (7/110, 6.3%) forgoing surgical intervention, and 1 patient lacking surgical outcome data. Among the surgical cohort, 3 patients underwent local excision (3/50, 6.0%) and 25 patients (25/50, 50%) underwent the Miles procedure. Overall, the proportion of patients opting for W&W or organ-preserving surgical treatment was 70% (77/110) based on the entire study population. By the end of the follow-up period, a total of 10% (11/110) of the patients had experienced LR. The detailed pathological results are presented in Table 3.
Pathological results for TNT patients.

CRM, circumferential resection margin; dMMR, mismatch repair deficient; LVI, lymphovascular invasion; MMR, mismatch repair; pCR, pathological complete regression; pMMR, mismatch repair proficient; PNI, perineural invasion; W&W, Watch and Wait; ypN, pathological lymph node stage after neoadjuvant therapy; ypT, pathological tumor T stage after neoadjuvant therapy.
DFS and OS
At the time of statistical analysis, 23.6% (26/110) of patients experienced treatment failure. Within the entire study population, 10% (11/110) of patients encountered LR, and all of the LRs occurred within 18 months. The 2-year LR rate is 10.8% (95% CI: 4.72%–16.88%). Furthermore, 14.5% (16/110) of patients experienced metastasis, with 7.2% (8/110) developing liver metastases and 10% (11/110) experiencing lung metastases. Among all the patients with metastasis, 81.3% (13/16) were found in 2 years. The proportion of patients achieving a 3-year DFS and OS was 75.7% (95% CI: 67.1%–84.3%) and 92.4% (95% CI: 86.8%–97.8%), respectively. The patient follow-up data, including OS, DFS, metastasis-free survival (MFS), and cumulative incidence of LR, are illustrated in Figure 2.

The OS (a), DFS (b), MFS (c), and LR (d) for the whole cohort.
To further explore the relationship between treatment response and long-term outcomes, we stratified patients based on post-nCRT treatment strategies, excluding those with incomplete follow-up data. Nine patients achieved pCR through surgery but were excluded due to the small sample size. The analysis focused on “W&W” patients and those who had surgery without complete remission. The results indicated that treatment-sensitive patients undergoing the “W&W” strategy could also achieve superior outcomes in DFS (p = 0.021) and OS (p = 0.006), respectively. Corresponding results are shown in Figure 3.

The OS (a) and DFS (b) for different response of treatment patients. Analysis focused on “Watch and Wait” patients and those who had surgery without complete remission, patients achieved pCR through surgery were excluded due to the small sample size.
Discussion
Our retrospective analysis evaluated treatment patterns and clinical outcomes in 110 rectal cancer patients receiving TNT. The therapeutic concept of TNT remains inconsistently defined in existing literature, with most studies characterizing it as a strategic sequence involving specific chemoradiotherapy regimens, yet critical uncertainties persist regarding optimal treatment cycles and chemotherapy agent selection.
Significant studies on TNT include retrospective research such as research from MSKCC, and prospective studies like CAO/ARO/AIO-12, OPRA, PRODIGE 23, RAPIDO, and STELLAR research.8–14 The CAO/ARO/AIO-12 study has three cycles of chemotherapy before or after 50.4 Gy/28Fx chemoradiation,9,10 the STELLAR study has four cycles of chemotherapy after 25 Gy/5 Fx chemoradiation. 13 The other studies have 6–9 cycles with chemotherapy before or after chemoradiation. Consequently, differences were observed in the rates of pathologic complete response (pCR) and DFS times among the trials. Within the TNT treatment groups, the W&W or pCR patients accounted for 75%–78%, with cCR patients ranging from 51% to 58%. 11 In the 5-year follow-up of the OPRA study, it is indicated that patients on the OPRA regimen have a higher possibility of local regrowth. 27 We consider that this is due to the more lenient assessment of cCR in the OPRA study, as near-cCR patients are also included in the W&W cohort. Therefore, the lower regrowth rate in our retrospective study is also acceptable. Moreover, patients receiving consolidated TNT chemotherapy after radiotherapy have a lower local regrowth situation. This also suggests that consolidated chemotherapy can lead to greater tumor regression and thus may have better efficacy. Patients who have a better response to TNT will have more opportunities to enter the W&W approach, which brings a favorable prognosis for patients, like the corresponding population in our study. 2
Our center has previously reported on the long-term prognosis of patients treated with the W&W strategy as well as surgical pCR patients. Specific details can be found in the referenced paper. 28 In our previous study, rectal cancer patients treated with the W&W strategy after TNT showed similar local control and long-term efficacy as those undergoing surgery. The elevated W&W applicability in our cohort stems from its high proportion of low rectal tumors and patients prioritizing sphincter preservation—anatomically complex cases historically associated with higher permanent ostomy risks, which heightened motivation for organ preservation, combined with early-stage cases (10%) demonstrating better chemoradiation responses. While these factors improve W&W feasibility in selected subgroups, they preclude generalization to unselected populations. Comparatively, our TNT-treated W&W patients exhibited lower local tumor recurrence rates than the International Watch & Wait Database (IWWD; TNT vs IWWD: 9.9% vs 25.2%), similar DFS (TNT vs IWWD: 93.3% vs 94%), and a trend toward superior OS (TNT vs IWWD: 92.1% vs 85%).29–31 Despite inherent limitations stemming from population heterogeneity and therapeutic protocol disparities, the fundamental question persists regarding the necessity of TNT administration in all W&W-eligible candidates. Recent prospective phase III studies like Prodige23, OPRA, and RAPIDO, involving more than six chemotherapy cycles, reported around 76% 3-year DFS and approximately 90% 3-year OS.11,12,14,32 This aligns with our data. Preoperative chemotherapy regimens, such as the 3-cycle CAO/ARO/AIO-129,10 studies and the 4-cycle STELLAR study, 13 have shown slightly lower short-term and long-term treatment efficacy, highlighting the significant association between preoperative chemotherapy cycles and treatment efficacy. For detailed information about the studies summary, please refer to Supplemental Table 2. In addition, our findings regarding the implementation of TNT align with results from international multicenter studies, highlighting the strategy’s robustness in real-world practice.
Our study has several limitations that warrant consideration. First, the inclusion of postoperative chemotherapy in some cases—while clinically necessary for patients demonstrating inadequate tumor regression—introduces variability in treatment protocols. Second, heterogeneity in chemotherapy regimens and inconsistent cycle numbers (primarily 6–8 cycles) across the cohort complicate the evaluation of standardized therapeutic strategies in real-world settings. In addition, subgroup analyses were constrained by limited sample sizes, reducing their statistical reliability. Future studies with larger cohorts are required to systematically assess outcomes across diverse treatment response categories. These observations may inform subsequent investigations in this field. This study delineates the clinical implementation patterns and therapeutic outcomes of TNT for rectal cancer. Our findings provide foundational insights for refining TNT strategies. The observed outcomes underscore the potential clinical value of this approach while highlighting the necessity for standardized protocols and larger-scale investigations to optimize therapeutic decision-making in rectal cancer management.
Conclusion
TNT achieves durable survival outcomes and enables organ preservation in high-risk rectal cancer patients. Rigorous post-treatment surveillance remains imperative to promptly identify local tumor regrowth and metastasis.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251332466 – Supplemental material for Total neoadjuvant therapy in high-risk rectal cancer: organ preservation and survival outcomes in a single-center retrospective cohort
Supplemental material, sj-docx-1-tam-10.1177_17588359251332466 for Total neoadjuvant therapy in high-risk rectal cancer: organ preservation and survival outcomes in a single-center retrospective cohort by Zhiyuan Zhang, Ruiyan Wu, Zhenyu Ke, Fan Xia, Guichao Li, Juefeng Wan, Hui Zhang, Yujia Deng, Zhen Zhang, Yan Wang and Lijun Shen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251332466 – Supplemental material for Total neoadjuvant therapy in high-risk rectal cancer: organ preservation and survival outcomes in a single-center retrospective cohort
Supplemental material, sj-docx-2-tam-10.1177_17588359251332466 for Total neoadjuvant therapy in high-risk rectal cancer: organ preservation and survival outcomes in a single-center retrospective cohort by Zhiyuan Zhang, Ruiyan Wu, Zhenyu Ke, Fan Xia, Guichao Li, Juefeng Wan, Hui Zhang, Yujia Deng, Zhen Zhang, Yan Wang and Lijun Shen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251332466 – Supplemental material for Total neoadjuvant therapy in high-risk rectal cancer: organ preservation and survival outcomes in a single-center retrospective cohort
Supplemental material, sj-docx-3-tam-10.1177_17588359251332466 for Total neoadjuvant therapy in high-risk rectal cancer: organ preservation and survival outcomes in a single-center retrospective cohort by Zhiyuan Zhang, Ruiyan Wu, Zhenyu Ke, Fan Xia, Guichao Li, Juefeng Wan, Hui Zhang, Yujia Deng, Zhen Zhang, Yan Wang and Lijun Shen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-xls-4-tam-10.1177_17588359251332466 – Supplemental material for Total neoadjuvant therapy in high-risk rectal cancer: organ preservation and survival outcomes in a single-center retrospective cohort
Supplemental material, sj-xls-4-tam-10.1177_17588359251332466 for Total neoadjuvant therapy in high-risk rectal cancer: organ preservation and survival outcomes in a single-center retrospective cohort by Zhiyuan Zhang, Ruiyan Wu, Zhenyu Ke, Fan Xia, Guichao Li, Juefeng Wan, Hui Zhang, Yujia Deng, Zhen Zhang, Yan Wang and Lijun Shen in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors sincerely thank all participants and patients involved in this study for their invaluable cooperation, dedication, and trust throughout the research process. We deeply appreciate the time and effort they contributed, as well as their willingness to share personal experiences and data, which were essential to the success of this work. Their commitment not only advanced our scientific understanding but also holds the potential to improve future clinical care and outcomes for individuals facing similar challenges.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
