Abstract
Background:
There is a rapidly increasing incidence of early-onset colorectal cancer (EO-CRC) which threatens the survival of young people, while aging also represents a challenging clinical problem.
Objectives:
We aimed to investigate the differences in the clinical characteristics and prognosis in stage III rectal cancer (RC), to help optimize treatment strategies.
Design and methods:
This study included 757 patients with stage III RC, all of whom received neoadjuvant chemoradiotherapy and total mesorectal excision. The whole cohort was categorized as very early onset (VEO, ⩽30 years old), early onset (EO, >30 years old, ⩽50 years old), intermediate onset (IO, >50 years, ⩽70 years), or late onset (LO, >70 years old).
Results:
There were more female VEO patients than males, more mucinous adenocarcinoma, signet-ring cell carcinoma, pre-treatment cT4 stage, and higher pre-treatment serum carbohydrate antigen 19-9 compared with the other three groups. VEO patients had the worst survival with the highest RC-related mortality (34.5%), recurrence (13.8%), and metastasis (51.7%). LO patients had the highest non-RC-related mortality rate (16.6%). The Cox regression model showed VEO was a negative independent prognostic factor for disease-free survival [DFS, hazard ratio (HR): 2.830, 95% confidence interval (CI): 1.633–4.904, p < 0.001], distant metastasis-free survival (DMFS, HR: 2.969, 95% CI: 1.720–5.127, p < 0.001), overall survival (OS, HR: 2.164, 95% CI: 1.102–4.249, p = 0.025), and cancer-specific survival (CSS, HR: 2.321, 95% CI: 1.145–4.705, p = 0.020). LO was a negative independent factor on DFS (HR: 1.800, 95% CI: 1.113–2.911, p = 0.017), DMFS (HR: 1.903, 95% CI: 1.150–3.149, p = 0.012), OS (HR: 2.856, 95% CI: 1.745–4.583, p < 0.001), and CSS (HR: 2.248, 95% CI: 1.282–3.942, p = 0.005). VEO patients had better survival in the total neoadjuvant therapy-like (TNT-like) pattern on DFS (p = 0.039). IO patients receiving TNT-like patterns had better survival on DFS, OS, and CSS (p = 0.006, p = 0.018, p = 0.006, respectively).
Conclusion:
In stage III RC, VEO patients exhibited unique clinicopathological characteristics, with VEO a negative independent prognostic factor for DFS, DMFS, OS, and CSS. VEO and IO patients may benefit from a TNT-like treatment pattern.
Background
Data provided by the American Cancer Society and the North American Association of Central Cancer Registries show that for people up to the age of 50, the incidence of colorectal cancer (CRC) has increased by 22% between the years 2000 and 2013 and that CRC-related mortality has increased by 13% from the years 2000 to 2014. 1 The incidence is also rapidly increasing among people under 50 years old across Africa, Europe, and Asia more generally, specifically, countries such as Nigeria, Sweden, Denmark, and Japan.2–5 Among CRC patients under 50 years old, the incidence has increased sharply among those aged 20–34 in the United States. 6 The rising incidence of CRC among those 50 years old and younger is a troubling issue considering the declining overall incidence and mortality trend. 7 Meanwhile, an increasingly aging population is also a healthcare-related trend that is worthy of attention.8,9
Based on the results of a comprehensive demographic survey and early screening for CRC, previous studies defined those under 50 years old as having early-onset CRC (EO-CRC).10,11 Although some studies have found that EO-CRC exhibits different biological features, there is contentiousness regarding the age limit of EO-CRC. Khan et al. 12 found that those with CRC under 30 years old had more microsatellite instability. After an age-stratified analysis of those younger than 50 years old with CRC, a large sample-sized study found that those 30 years old and younger had more signet-ring cell carcinoma and poorly differentiated cancers compared with other age groups. 13 Elderly patients in particular have a poorer prognosis, although they are considered to have similar clinical features to middle-aged patients.14,15 Distinct from these above studies, a steadily increasing trend of early-onset (EO) incidence has been reported in China. 16 Therefore, our study aimed to document the clinicopathological characteristics and compare treatment outcomes among those with very early-onset (VEO), EO, intermediate-onset (IO), and late-onset (LO) stage III rectal cancer (RC).
The standard treatment strategy for locally advanced rectal cancer (LARC) is neoadjuvant chemoradiotherapy (NACRT) with total mesorectal excision (TME) with or without adjuvant chemotherapy (AC), which has been shown to improve local control and survival.17,18 Total neoadjuvant therapy (TNT) has also been widely recognized as a novel treatment pattern 19 but whether TNT is suitable for all aged patients remains unclear. At present, there is no study on the efficacy data among different onset age groups. This is important because comorbidities and treatment tolerance in elderly patients may mean they need to be more careful when choosing their treatment options.14,20 And more aggressive treatment strategies for younger patients are also controversial.21–23 Here, our study compares the survival outcomes of conventional chemoradiotherapy (CRT) patterns and TNT-like patterns based on the above age stratification, which may help clarify the value of TNT for these four onset age groups.
Materials and methods
Study population
A total included 799 in-patients with stage III (including cT1-4N1-2M0) rectal adenocarcinoma who were admitted to Sun Yat-sen University Cancer Center for the first time from August 2005 to September 2017, all of whom were treated with NACRT followed by TME with or without AC. Patients were staged according to the Union for International Cancer Control/American Joint Committee on Cancer, version 8. Excluding 42 patients, this study included 757 patients. The screening process is detailed in Figure 1.

The patient screening flow diagram.
Treatment
Radiotherapy techniques included Computed Tomography SIMulator (CT-Sim, 6 cases), three-dimensional conformal radiation therapy (131 cases), intensity-modulated radiation therapy (239 cases), volumetric-modulated arc therapy (379 cases), and tomotherapy (2 cases). The prescribed dose was 45–54 Gy/23–25 fractions to the planning target volume of the gross tumor volume (GTV) in the primary GTV and involved the lymphatic drainage regions. As for concurrent chemotherapy during radiotherapy, 200 cases received fluorouracil analog alone, and 553 cases received fluorouracil analog and oxaliplatin. Of the induction chemotherapy and consolidation chemotherapy in the NACRT phase, 5 patients received mFolfox6, 21 Capecitabine, and 401 Capeox. In all, 330 patients did not receive induction or consolidation chemotherapy.
To compare the differences between standard CRT and TNT patterns, we divided patients into CRT-like and TNT-like groups. A TNT-like pattern was defined as patients receiving induction chemotherapy before radiotherapy and/or consolidation chemotherapy besides concurrent CRT. A CRT-like pattern was defined as only concurrent CRT which was given without induction or consolidation chemotherapy.
Follow-up
The outcome events included disease-free survival (DFS), local recurrence-free survival (LRFS), distant metastasis-free survival (DMFS), overall survival (OS), and cancer-specific survival (CSS). DFS was defined as the time from diagnosis to the first relapse at any site, death, or date of the last follow-up. LRFS was defined as the time from diagnosis to the first local and regional recurrence at any site or date of the last follow-up. DMFS was defined as the time from diagnosis to the first distant metastasis at any site or date of the last follow-up. OS was calculated as the time from diagnosis to death from any cause. CSS was calculated as the time from diagnosis to the death from RC. Patients were assessed every 3 months during the first year after treatment, and every 6 months in years 1–3, and then every year thereafter. The evaluation included digital rectal examination, chest X-ray or computed tomography (CT), abdominal ultrasound or CT, pelvic CT, or magnetic resonance imaging, laboratory testing including serum carcinoembryonic (CEA) and serum carbohydrate antigen 19-9 (CA19-9), and other tests deemed applicable.
Statistics
To determine the study sample size, we assessed whether the number of cases in which DFS outcome events occurred met a 10-fold requirement of 16, which was the number of clinicopathological characteristic variables. 24 In our study, 195 patients had DFS events; thus, the whole cohort patient number 757 fulfills the sample size requirement. The reporting of this study conforms to the Enhancing the Quality and Transparency of Health Research reporting guidelines.
Based on early exploration (Supplemental Figure 1) and previous CRC onset-age studies, patients were further stratified into four groups: VEO (⩽30 years old), EO (>30 years old, ⩽50 years old), IO (>50 years old, ⩽70 years old), and LO (>70 years old) for analysis.
According to the rectal anatomical segments, the distance from the lower boundary of the tumor to the anal verge was defined as the lower segment (<5 cm), middle segment (⩾5 cm, <10 cm), and upper segment (⩾10 cm). The chi-square test, Fisher’s exact test, or T test was used for the correlation analysis and comparison rates. Analysis of variance was used to compare the continuous variables.
Kaplan–Meier survival analysis was used to calculate the survival rate, and the log-rank test was used to compare the differences. By reverse Kaplan–Meier analysis, the mean follow-up was obtained for the four subgroups. A Cox proportional hazards model was used to generate the hazard ratios (HRs) and confidence intervals (CIs) and to explore the significance of the clinicopathological characteristics on survival. All clinicopathological characteristics included four age-onset, gender, distance to the anal verge, histological type, cT stage, cN stage, family history, treatment pattern, induction chemotherapy regime, induction chemotherapy course, concurrent chemotherapy regimen, concurrent chemotherapy course, intermittent chemotherapy regimen, intermittent chemotherapy course, pre-treatment CA19-9, and pre-treatment CEA. A p value <0.05 (two-sided) was considered statistically significant. Statistical analyses were carried out using SPSS (version 26.0, IBM Corp., Armonk, NY, USA) and R (version 4.0.1, R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient characteristics
The mean follow-up was 83.4 months (95% CI: 71.4–95.4), 77.3 months (95% CI: 74.3–80.4), 78.6 months (95% CI: 76.3–80.9), and 85.8 months (95% CI: 76.9–94.8) in VEO, EO, IO, and LO, respectively.
In the VEO, EO, IO, and LO groups, the percentage of females followed a declining trend, with 48.3%, 39.0%, 36.2%, and 27.1% (p = 0.249). The proportion of signet-ring cell carcinoma also followed a declining trend, with 6.9%, 1.2%, 0.5%, and 0%. The percentage of mucinous adenocarcinoma was 3.4%, 2.4%, 1.6%, and 2.1% (p = 0.092). The proportion of cT4 stage in VEO was higher than in EO, IO, and LO (51.7%, 39.4%, 40.4%, 43.8%; respectively), although no statistically significant difference was found (p = 0.551). The proportion of the cN2 stage was 41.4%, 49.8%, 39.0%, and 39.6%, (p = 0.051). The median pre-treatment CEA was 5.1, 3.5, 5.1, and 5.17 ng/ml (p = 0.042). The median pre-treatment CA19-9 was 23.1, 13.4, 13.5, and 17.0 µ/ml (p = 0.002). The proportion of the four subgroups receiving induction, concurrent, and intermittent two-drug chemotherapy regimens showed a significant decreasing trend (p < 0.001, p = 0.002, p < 0.001). There was no significant difference in the ratio of the number of induction or concurrent chemotherapy courses received by any of the four patient groups (p = 0.597, p = 0.774). The proportion of intermittent-free chemotherapy presents a higher proportion at older ages (p = 0.05). The percentages of those receiving a TNT-like pattern were 72.4%, 61.0%, 53.6%, and 47.9% (p = 0.046) (Table 1).
Clinical, pathological, and treatment characteristics of VEO, EO, IO, and LO for stage III rectal cancer patients.

CA19-9, serum carbohydrate antigen 19-9; CEA, serum carcinoembryonic; CRT, chemoradiotherapy; EO, early onset; IO, intermediate onset; LO, late onset; TNT, total neoadjuvant treatment; VEO, very early onset.
Treatment outcomes and prognostic analysis
In the univariate analysis, the survival rates for the four onset-age groups had significantly different DFS [p < 0.001, Figure 2(a)], DMFS [p < 0.001, Figure 2(b)], LRFS [p = 0.041, Figure 2(c)], OS [p < 0.001, Figure 2(d)], and CSS [p = 0.001, Figure 2(e)], showing inferior survival results in the VEO and LO groups.

Survival analysis for the four-onset age groups with stage III rectal cancer on (a) DFS, (b) DMFS, (c) LRFS, (d) OS, and (e) CSS.
In the multivariable analysis, the IO group was set as a reference (Table 2). EO had similar DFS, DMFS, LRFS, OS, and CSS with the IO group.
Multivariable Cox regression analysis of prognostic factors for patients.

CA19-9, serum carbohydrate antigen 19-9; CEA, serum carcinoembryonic; CI, confidence interval; CRT, chemoradiotherapy; CSS, cancer-specific survival; DFS, disease-free survival; DMFS, distant metastasis-free survival; EO, early onset; HR, hazard ratio; IO, intermediate onset; LO, late onset; LRFS, local recurrence-free survival; OS, overall survival; TNT, total neoadjuvant treatment; VEO, very early onset.
VEO had a significantly lower 5-year DFS, 5-year DMFS, 5-year OS, and 5-year CSS of 57.7%, 57.7%, 67.3%, and 70.8%, respectively [Figure 2(a), (b), (d), and (e)]. The 5-year survivals in VEO were approximately 20% lower than EO and IO, which was a marked difference. As shown in Table 2, HRs on DFS, DMFS, OS, and CSS in VEO were 2.830 (95% CI: 1.633–4.904), 2.969 (95% CI: 1.720–5.127), 2.164 (95% CI: 1.102–4.249), and 2.321 (95% CI: 1.145–4.705), compared with IO.
LO had a significantly lower 5-year DFS, 5-year DMFS, 5-year OS, and 5-year CSS of 63.8%, 66.1%, 63.8%, and 71.2%, respectively [Figure 2(a), (b), (d), and (e)]. The 5-year survival rates of LO were reduced by about 10–20% compared with EO and IO. LO showed higher HRs than IO regarding DFS, DMFS, OS, and CSS, with HRs of 1.800 (95% CI: 1.113–2.911), 1.903 (95% CI: 1.150–3.149), 2.856 (95% CI: 1.745–4.583), 2.248 (95% CI: 1.282–3.942), exhibiting similar HRs on OS and CSS and lower HRs on DFS and DMFS compared with VEO (Table 2).
Excluding age, pre-treatment cT stage was an independent negative factor on DFS (HR: 1.385, 95% CI: 1.069–1.793), LRFS (HR: 2.056, 95% CI: 1.075–3.931), and OS (HR: 1.350, 95% CI: 1.011–1.803). Being female was an independent negative factor for DMFS (HR: 1.354, 95% CI: 1.010–1.815). Among the histological subtypes, signet-ring cell carcinoma was an independent negative factor for OS (HR: 5.121, 95% CI: 1.516–17.296) and CSS (HR: 6.213, 95% CI: 1.838–21.003); and mucinous adenocarcinoma was an independent negative factor for CSS (HR: 2.683, 95% CI: 1.177–6.114) (Table 2).
Impact of treatment strategies
As shown in Table 3, the mortality rates were 37.9% (VEO), 17.3% (EO), 17.2% (IO), and 47.9% (LO) (p = 0.030). Comparing the causes of death, the four subgroups showed statistical differences(p = 0.032). The RC-related mortality rates were 34.5% (VEO), 15.3% (EO), 15.3% (IO), and 31.3% (LO). LO had the highest non-RC-related death rate (16.6%), which was much higher than those of VEO, EO, and IO, with non-RC-related deaths dominated by chronic diseases such as diabetic complications, cardiovascular events, and cerebrovascular disease (75.0%).
Survival, recurrence, and metastasis characteristics of VEO, EO, IO, and LO for stage III rectal cancer patients.

EO, early onset; IO, intermediate onset; LO, late onset; VEO, very early onset.
The survival rates showed significance in the four subgroups(p = 0.003), whereas there was no statistical difference in alive with or without tumor recurrence (p = 0.174) (p = 0.113) (Table 3).
In the four subgroups, local–regional recurrence rates did not differ(p = 0.079), and metastasis rates were different (p = 0.017). The local–regional recurrence rates were 13.8% (VEO), 3.6% (EO), 3.9% (IO), and 6.3% (LO). The distant metastasis rates were 51.7% (VEO), 23.3% (EO), 21.8% (IO), and 39.6% (LO). No statistically significant difference was found in concurrent local–regional recurrence and the distant metastasis rates among the four subgroups (Table 3).
The incidence of metastasis in the lung (p = 0.001), liver (p = 0.008), bone (p = 0.015), and other (p = 0.005) of the four subgroups was statistically different. In terms of the overall population, distant metastases occurred mostly in the lungs (45.5%), followed by the distant lymph nodes (16.7%) and the liver (15.9%). VEO patients had relatively more distant lymph node metastases (22.7%), and LO patients had more bone metastases (16.0%), and brain metastases (8.0%) (Table 3).
As shown in Supplemental Table 1, patients receiving the two treatment patterns were relatively evenly distributed, except patients with signet-ring cell carcinoma who were more likely to receive TNT-like in the overall population. The distribution of the two treatment patterns was not always balanced across the onset age groups. EO receiving a TNT-like pattern had more high-risk factors, such as being female (59.8%), cT3 stage (56.1%), cT4 stage (67.3%), and cN2 stage (65.3%).
For the whole group, TNT-like improved DFS (HR: 0.710, 95% CI: 0.533–0.945), OS (HR: 0.667, 95% CI: 0.481–0.926), and CSS (HR: 0.574, 95% CI: 0.403–0.819) (Table 2).
VEO patients receiving a TNT-like pattern were superior to a CRT-like pattern in DFS [5-year, 65.5% versus 37.5%; 10-year, 49.2% versus 12.5%; p = 0.039; Figure 3(a)]. Those patients also had a higher OS [Figure 3(b)], and CSS [Figure 3(c)] but the differences were not statistically significant. The efficacy of the two treatment patterns for EO patients did not show any differences in survival [Figure 3(d)–(f)]. IO patients receiving a TNT-like pattern showed a significantly improved DFS, OS, and CSS than those receiving a CRT-like pattern [p = 0.006, p = 0.018, p = 0.006; respectively; Figure 3(g)–(i)]. For LO patients, there was no difference in DFS, OS, and CSS between the CRT-like and TNT-like patterns [Figure 3(j)–(l)]. Although similar to the 5-year OS and CSS, a TNT-like pattern seemed to have a slight advantage over a CRT-like pattern for LO [10-year OS 52.3% versus 33.3%, 10-year CSS 75.0% versus 48.5%; Figure 3(k) and (l)].
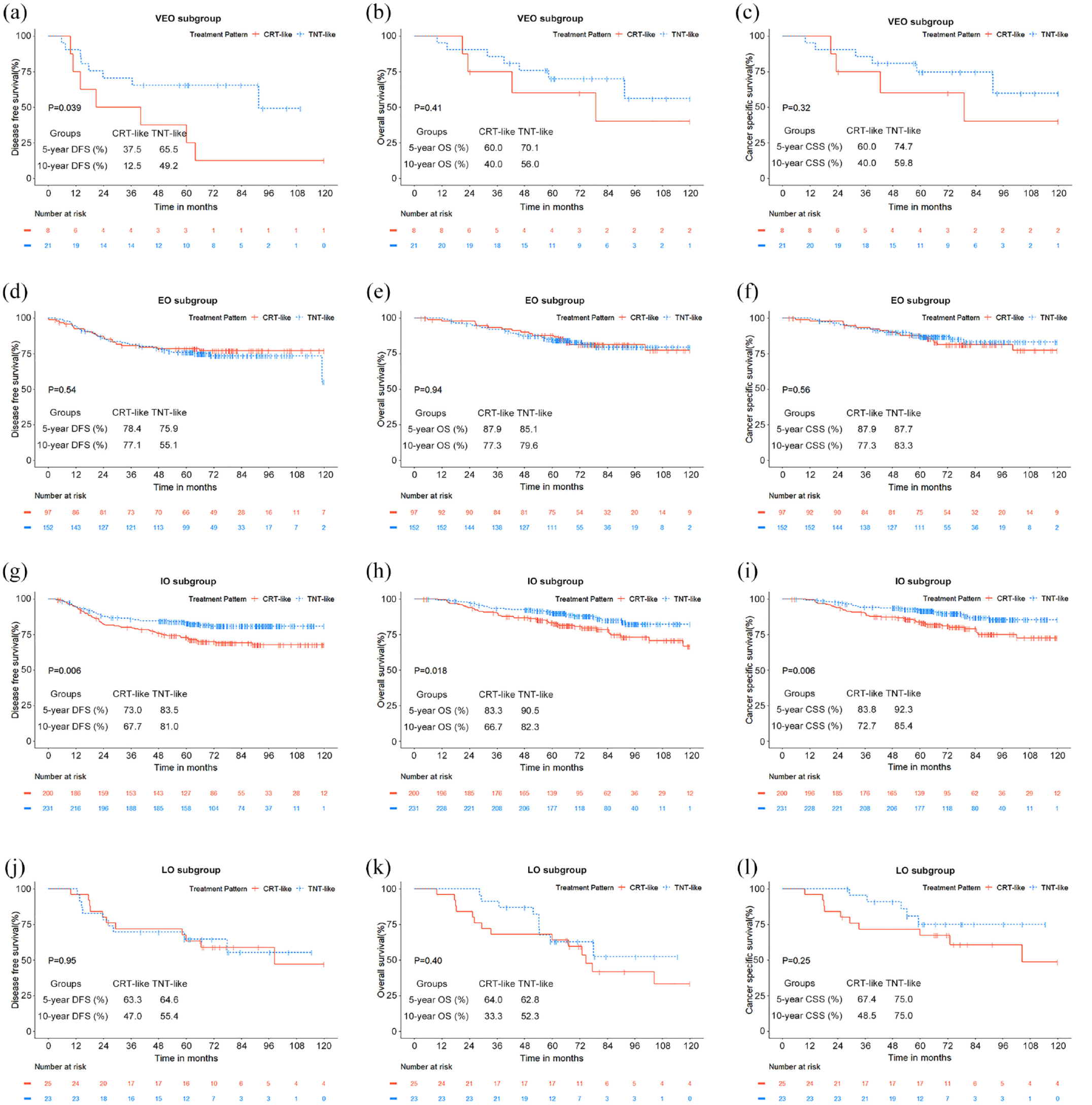
Survival analysis of stage III rectal cancer patients receiving different treatment patterns. Comparison of the CRT-like and TNT-like patterns of very early onset on (a) DFS, (b) OS, and (c) CSS. Comparison of the CRT-like and TNT-like patterns of early onset on (d) DFS, (e) OS, and (f) CSS. Comparison of the CRT-like and TNT-like patterns for intermediate onset on (g) DFS, (h) OS, and (i) CSS. Comparison of the CRT-like and TNT-like patterns for late onset on (j) DFS, (k) OS, and (l) CSS.
Discussion
The incidence of CRC is increasing worldwide among both the young and elderly. Our study found that VEO had higher RC-related mortality and that LO had higher mortality caused by non-oncologic chronic diseases compared with other onset ages.
Similar to previous studies,13,25 VEO exhibited distinctly more signet-ring cell carcinoma, mucinous adenocarcinoma, and a more advanced pretreatment cT stage. VEO had a higher median pre-treatment CA19-9 and comprised more female patients. VEO showed the highest rates of local recurrence, distant metastasis, and disease failure. Lung metastasis had the highest incidence across all onset ages, and distant lymph node metastasis was the second most common site among VEO patients.
The clinical and therapeutic characteristics of VEO patients may be related to their unique molecular characteristics. Published studies have observed higher rates of CMS1 in young CRC patients. 26 VEO exhibited the highest recurrence and metastasis rates in our study. In the era without programmed death 1/programmed cell death-ligand 1 antibody, studies have found that CMS1 CRC had worse survival than other types,27,28 which may be one of the molecular bases for poor prognosis in VEO. Moreover, some studies have found more SMAD4 mutations in VEO patients, 29 which have frequently been found in mucinous adenocarcinoma and are associated with an aggressive phenotype,30,31 as well as, poor prognosis in stages I–III. 32 In addition, SMAD4 mutations are thought to be associated with 5-fluorouracil resistance, 33 which may be the reason why VEO patients still exhibit lower survival rates than EO, IO, and LO patients even when receiving an aggressive TNT pattern. Another study has also found more poorly performing driver mutations in female patients, which may explain the strong immune selectivity, thereby making it difficult for females to benefit from immunotherapy. 34 Due to a lack of relevant molecular and genetic information in this study, further analysis was not possible.
In our study, younger patients were found to have worse DFS, which is similar to the finding of Foppa et al.35,36 In contrast to our study, Gao et al. 37 found no statistical difference in DFS and OS. Based on the results observed in our study, it is possible that the higher RC-related mortality in the VEO and LO subgroups led to a similar CSS in Foppa et al.’s 35 study when divided into two subgroups. Worse DFS may be associated with higher metastasis rates in younger patients, which is consistent with the findings of our study and Foppa et al.’s study. Unlike the consistent stage III RC patients in our study, the above studies were based on disease-characterizing observations of overall colon and/or RC. Therefore, the discrepancy between these studies and our findings is understandable. Our study had a more detailed age stratification, which allowed for a clearer observation of the clinical features and prognostic differences in stage III RC by age. This was also confirmed by the Foppa et al. 35 study, where matched post-comparison of DFS and CSS for stage III RC showed a trend toward poorer survival in younger patients in agreement with the results of our study.
LO had a lower rate of distant metastases, worse OS, and similar LRFS and CSS than VEO. LO has similar clinical characteristics to IO but worse survival. Comorbidity status and post-treatment frailty in LO patients may affect their prognosis.38,39 Therefore, we suggest constructing a scoring approach that matches the elderly’s overall health, thereby enabling personalized treatment suggestions. The 5-modified frailty index is a valid predictor of postoperative mortality and complications in CRC. 40 If the index is combined with clinical indicators such as Tumor Node Metastasis (TNM) staging it may have guiding significance for NACRT.
The different clinical features and prognostic presentation imply that it may be necessary to develop treatment strategies according to different onset ages. Treatment decisions may be preferential to a TNT-like pattern because of more signet-ring cell carcinoma, mucinous adenocarcinoma, and more advanced cT stage in VEO. Previous studies have not found marked survival differences among patients under 50 years old who received different CRT strategies.21,23 It may be because VEO patients are combined with patients aged 31–50 who show better survival, while the number of patients aged 31–50 years old is much higher than the number of VEO patients. In this case, the difference may have been weakened. Our results imply that an intensive pattern of chemotherapy given before and after concurrent CRT is necessary for VEO.
At present, several important studies have shown TNT reduces distant metastasis and improves DMFS and DFS relative to CRT in LARC. 41 In our study, we found that a TNT-like pattern has an advantage over a CRT-like pattern regarding DFS, OS, and CSS, for the overall population and the highest percentage of IO. However, in our study, among patients receiving both treatment patterns, IO was capable of benefiting from a TNT-like pattern, while EO was not. These inconsistent results may be due to the imbalance of clinical factors between the two treatment pattern groups. Patients with high-risk factors were more commonly treated with a TNT-like pattern with EO.
The 5-year OS was lower than the 5-year CSS in LO patients receiving a TNT-like pattern, indicating that comorbidities may be adverse factors affecting survival. Balancing CRT and comorbidities is paramount for elderly patients, with a TNT pattern perhaps preferable for elderly patients in good general condition.
Taken together, we consider that VEO patients have poor awareness of seeking timely medical care and the specific molecular features that worsen prognosis. The recommended starting age for early screening is 50 years old, which is currently considered to have the greatest net benefit for the population. 42 However, given the incidence and mortality of VEO CRC are increasing this may need to be reassessed; with appropriate screening strategies developed for VEO. Males and females have an equal chance of developing VEO CRC, with no gender selection tendency at screening. We did not find a typical family history of cancer among VEOs. Some studies have found that a family history of CRC may be instructive for early screening, 26 with rectal bleeding as the most common symptom at diagnosis.26,43 Even earlier screening may strengthen the follow-up of those with a family history. It may be good practice to refer to a risk scoring system, 44 to screen for high-risk groups and perform regular fecal tests for early detection. Screening strategies for the VEO population are still needed to collect and explore more extensive and comprehensive information to establish a suitable screening model.
There are some limitations of this study. First, the preoperative chemotherapy regimens were not completely consistent because of the retrospective nature of this study. Second, this study is a real-world demonstration of the different onset age states in stage III RC, while the VEO of age cases were potentially insufficient in number.
Conclusion
Among different onset age stage III RC populations, VEO patients comprised of more females had the most aggressive clinical features including a more advanced cT stage, more signet-ring cell carcinoma, mucinous adenocarcinoma, and higher pre-treatment CA19-9. Clinical characteristics were similar among EO, IO, and LO. Age was an independent factor for prognosis. DFS, DMFS, OS, and CSS were significantly lower among VEO and LO than IO. EO was similar to IO regarding DFS, DMFS, OS, and CSS. A TNT-like pattern may bring about more survival benefits for VEO and IO.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241229434 – Supplemental material for Distinct clinical characteristics in stage III rectal cancer among different age groups and treatment outcomes after neoadjuvant chemoradiotherapy
Supplemental material, sj-docx-1-tam-10.1177_17588359241229434 for Distinct clinical characteristics in stage III rectal cancer among different age groups and treatment outcomes after neoadjuvant chemoradiotherapy by Baoqiu Liu, Huilong Luo, Bin Li, Haina Yu, Rui Sun, Jibin Li, Yuanhong Gao, Peirong Ding, Xicheng Wang and Weiwei Xiao in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-2-tam-10.1177_17588359241229434 – Supplemental material for Distinct clinical characteristics in stage III rectal cancer among different age groups and treatment outcomes after neoadjuvant chemoradiotherapy
Supplemental material, sj-jpg-2-tam-10.1177_17588359241229434 for Distinct clinical characteristics in stage III rectal cancer among different age groups and treatment outcomes after neoadjuvant chemoradiotherapy by Baoqiu Liu, Huilong Luo, Bin Li, Haina Yu, Rui Sun, Jibin Li, Yuanhong Gao, Peirong Ding, Xicheng Wang and Weiwei Xiao in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
