Abstract
Patients with luminal breast cancer (BC) may develop central nervous system metastases in 20%–40% of cases. Radiation or surgical therapy represents the cornerstone of treating central nervous system metastases. Meanwhile, the best practice for metastatic luminal BC involves using cyclin-dependent kinase 4/6 inhibitors combined with endocrine therapy. To our knowledge, this is the first case to report a dramatic response of breast metastases to abemaciclib plus endocrine therapy without radiation therapy, particularly in a patient who presented with seizures and sudden coma. She received brain surgery to control a large bleeding metastasis. Abemaciclib was crushed and diluted in water for administration via the nasogastric tube, while an upfront fulvestrant was given since aromatase inhibitors cannot be diluted. Beyond the radiological response, the clinical improvement was notable, with complete symptom recovery to the point where she is again working. Our paper supports the activity of abemaciclib in brain metastases from luminal BC and includes a review of the medical literature. Further investigation is warranted in this clinical setting.
Introduction
Around 20%–40% of patients with breast cancer (BC) develop central nervous system (CNS) metastases in their lifespan, and metastatic breast cancers (MBC) seed the brain in 10%–24% of cases.1–3 Among BC metastatic to the CNS, hormone receptor-positive (HR+) and triple-negative carcinomas are the most common breast cancer subtypes. 4 Still, the incidence of CNS metastasis in HR+ BC is around 14%, ranking the lowest among all subtypes.4,5 The occurrence of CNS metastasis carries a poor prognosis. However, an extensive real-life study showed that molecular subtypes deeply influence the incidence, kinetics, and prognosis of CNS metastases in MBC patients.5–7 Overall survival (OS) was independently associated with molecular subtypes. Patients with HR+ human epidermal growth factor receptor-2 negative (HER2−) MBC had a median OS of 7.1 months; those with HR+/HER2+ or HR−/HER2+ carcinoma had a median OS of 18.9 and 13.1 months, respectively, and triple-negative ones had only 4.4 months, showing a large statistically significant difference (p < 0.0001). 6 The occurrence and prognosis of CNS metastases may also be related to the presence of BRCA1 or BRCA2 mutation. 8
The usual presentation of MBC involving the CNS may comprise headache, vomiting or nausea, various mental changes, seizures, and weakness or numbness on one side of the body. 7 To date, the best practice for HR+/HER2− MBC involves using cyclin-dependent kinase 4/6 inhibitors (CDK4/6i) combined with endocrine therapy (ET). 8 Among various CDK4/6i, abemaciclib (ABE) seems the most active against CNS metastasis, but the role of such agents in treating CNS metastasis is still unclear.8–10
In this paper, we report an unusual case of MBC to the brain presenting with syncope, coma, multiple CNS metastases, radiological total body imaging mimicking lung cancer, and successfully treated with ABE and fulvestrant hormonotherapy. The authors also present a review of medical literature. The reporting of this patient’s clinical case conforms to the CARE case report guideline.11,12
Case description
The patient is a 46-year-old Caucasian woman. Her medical history was positive for thyroidectomy for Basedow’s disease, and she was taking thyroid hormone replacement therapy. In September 2014, she underwent a super-central quadrantectomy at the left breast for a moderately differentiated ductal infiltrating carcinoma, with estrogen and progestin receptors at 100%, KI67 at 20%, and HER-20, followed by adjuvant chemotherapy and hormonotherapy with luteinizing-hormone releasing hormone (LHRH) and tamoxifen ended in 2019.
The timeline of cancer history is graphically displayed in Figure 1. On May 15, 2023, she suddenly experienced severe tonic-clonic seizures with syncope. Due to the very severe symptoms and lack of consciousness, she was immediately admitted to the resuscitation unit, and a nasogastric tube was positioned since the patient was entirely unable to swallow. Physical examination was non-significative.

Patient medical history timeline.
In the left frontal area at the vertex, the total body CT scan showed an intra-axial expansive cystic formation with a transverse diameter (TD) of approximately 33 mm and a solid eccentric parietal component along the posterolateral lesion profile with blood fluid level as per recent bleeding and severe perilesional edema (Figure 2). Another smaller solid mass of 2 cm in diameter was present in the left mesial occipital area close to the posterior surface of the trigone of the lateral ventricle, which appeared compressed (Figure 2). Other solid neoplastic foci were found in the white matter adjacent to the frontal horn of the left ventricle with minimal intralesional cystic quota (TD approximately 11 mm) and another likely and intrinsically idioventricular localized in the choroid plexus of the temporal horn of the right lateral ventricle (TD approximately 14 mm). Another solid lesion was present in the right cerebellar hemisphere (Figure 2). A mass of 4.1 × 2.1 cm was in the left upper hilar horn, invading the pleura and mediastinal fat with enlarged, metastatic lymph nodes in the left supraclavicular fossa (3 × 1.8 cm), para-esophageal area (1.8 × 1.7 cm), and the left hilum (2.3 × 1.4 cm) (Figure 3). Another subcutaneous deposit was present at the interscapular area (1.4 cm). Two oval formations were detected at the bilateral adrenal level, measuring 4 × 2.2 cm on the right and 2.5 × 1.8 on the left, compatible with metastatic deposits (Figure 4). Multiple enlarged lymph nodes in the para-aortic, inter-aortocaval, and the greater curvature of malignant significance. In the subcutaneous area at the level of the anterior abdominal wall (left paramedian) and left gluteal wall, two oval formations with nonhomogeneous enhancement are found, with a maximum dt of 2 cm, compatible with metastatic deposits (Figure 4).
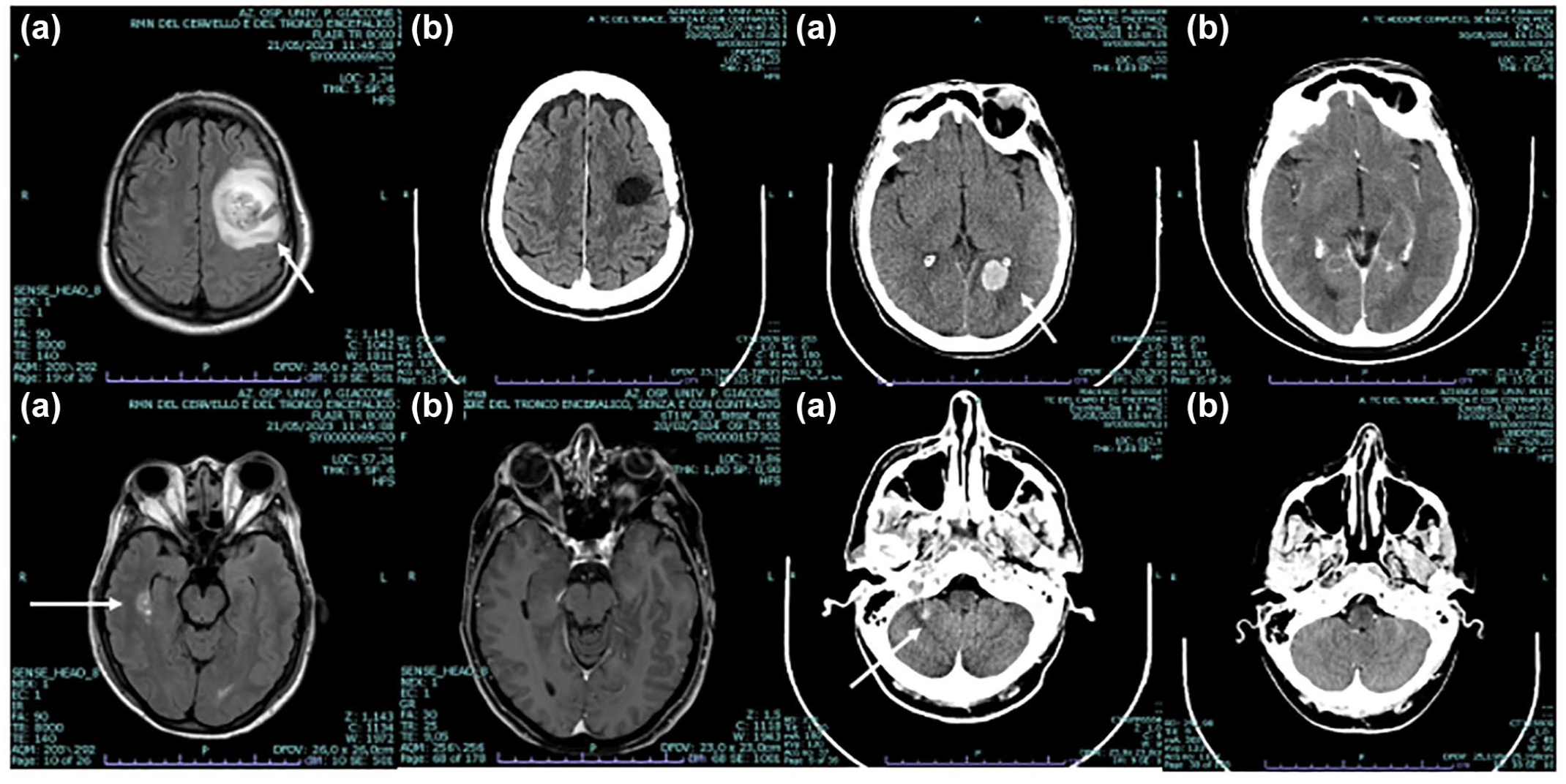
CT and Nuclear Magnetic Resonance scan of the brain before (a) and after (b) anticancer treatments. White arrows indicate tumor lesions at baseline. Brain scan shows cerebellar, temporal, parietal, and occipital metastases.

CT scan of the thorax before (a) and after (b) anticancer treatments. White arrows indicate tumor lesions at baseline. The CT scan shows lung and thoracic lymph node metastases.

CT scan of the abdomen before (a) and after (b) anticancer treatments. White arrows indicate tumor lesions at baseline. The CT scan shows renal, adrenal, abdominal wall, and gluteal muscle metastases.
The clinic-radiological picture mimicked a primary lung tumor, and therefore, oncologists decided to perform a new biopsy (Figure 1). On May 16, a biopsy of a subcutaneous mass in the peri-umbilical region was performed. Pathology showed fat tissue massively infiltrated by solid cord of malignant epithelial cells with Pan CK+, GATA3+, ER+, CK7-, CK20-, TTF1-negative, napsin-negative, CD56-, chromogranin, and synaptophysin negative, p40-, S100-, Pax8-, CD20-, CD3- immunophenotype compatible with a breast ductal infiltrating carcinoma. Further analysis showed positive estrogen receptors (ER) in more than > 95% of cells, progestin receptors (PR) in 25%, Her2 score 1+, and Ki-67: 40%. The pattern suggested a luminal BC. Despite medical attempts from May 19 until 21, the patient showed no signs of awakening. Serial brain CT scans carried out between 16 and 19 May did not reveal any modification of cancer, and brain NMR confirmed the CT scan picture.
On May 22, based on the clinical picture and the patient’s non-reawaken ability and considering the bleeding brain lesions, it was decided to perform surgical removal of the left frontoparietal lesion. Specimen obtained after craniotomy showed multiple fragments of brain parenchyma (GFAP+, Synaptophysin+) diffusely infiltrated by an atypical epithelial proliferation, with morphological and immunophenotypic characteristics (Pan CK+, GATA3+, ER+, E cadherin+, CK7-, p63-, and CK34βE12-) attributable to metastases from ductal carcinoma invasive with primary mammary origin. Immunophenotypic profile was ER+ >95%, PR+ 25%, Her2 negative (score 1+ according to ASCO/CAP 2018), Ki-67: 25%.
As shown in Figure 1, a medical oncology consultation suggested starting intramuscular fulvestrant at 250 mg bid every 2 weeks for the first month and then every 28 days plus a cyclin-dependent kinase inhibitor, considering the patient’s inability to swallow and the immunophenotypic pattern. Letrozole was excluded, considering the solubility characteristics that did not make it suitable for intake through the nasogastric tube. Oncologists preferred ABE because of its activity on CNS metastases. ABE was employed at 150 mg b.i.d. After pharmacological counseling, the pharmacist crushed the tablets and diluted them with water up to at least 25 mg/ml) to administer them through the nasogastric tube.
The patient experienced a slow but progressive recovery of both sensory and motor functions after surgery and medical anticancer therapy, although presented aphasia, apraxia, ataxia with faint right hemiparesis, and dysphagia. From June 22 until July 21, she underwent rehabilitation with a significant recovery of swallowing and speech, autonomous postural steps, reasonable trunk control, and autonomous verticalization at discharge (Figure 1).
A multigenic panel revealed a germline deletion NM_005732.4(RAD50): c.3476del (p.Asp1159fs), a hereditary cancer-predisposing syndrome. A liquid biopsy showed a sequence variant p.E545K (c.1633G>A) affecting exon 10 of the PIK3CA gene. Medical oncologists decided not to administer fulvestrant any longer, replacing it with letrozole, with a view to subsequent therapeutic lines. ABE was continued at the dose of 150 mg b.i.d. orally. The patient tolerated the treatment well, with only G1 diarrhea between the third and fourth cycles. The patient was treated with two tablets of loperamide after the first discharge, with a total resolution of the symptoms. Clinical and radiological monitoring showed no signs of interstitial lung disease. Therefore, the patient continued the full dosage of ABE.
On September 14, 2023, the disease was re-evaluated, and a brain CT scan showed a volumetric reduction of metastasis in the left perigonal area (approximately 1.3 cm). A subsequent NMR on October 23, 2023, indicated the disease was stable. In November 2023, a PET scan failed to show metabolic activity disease. In February 2024, brain NMR showed a partial response at all brain lesions. In May 2024, a total body CT scan reported a major partial response at all tumor sites (Figure 2–4). The poromalacic lesion size pre- and post-contrast graphic densitometric characteristics in the left frontal area of the ventricle were unchanged.
In contrast, the nodule in the left perigonal area (maximum TD 0.4 cm vs 1 cm) showed a dimensional reduction. The remaining finds remain unchanged. CT scan also revealed a volumetric reduction of the lymph node in the left supraclavicular area (maximum TD vs 2 cm approximately) and the subcutaneous nodularity in the left dorsal paramedian site (current maximum TD 1.3 cm). In the abdomen, a volumetric reduction of adrenal nodularities in the progress of 2.1 × 0.8 cm on the right (vs 3.3 × 1.5 cm) and 1.1 × 0.8 cm on the left (vs 1.9 × 2 cm) was observed. The lymph nodes in the lumbo-aortic retroperitoneum were further reduced in size (the largest at today’s control in the left para-aortic area of the trasverse diameter, DT, max of approximately 1 cm vs 1.9 cm). The subcutaneous nodularity on the left buttock was also significantly reduced in volume.
To date, the patient appears to be in good general clinical condition with no noteworthy disorders, without ongoing toxicity, and total recovery of motor and intellectual abilities. The resumption of daily life activities, including work, is complete. After cognitive recovery, the oncologists informed the patient about what had happened. The patient expressed her gratitude and excellent adherence to the proposed therapeutic program. She is still taking full-dose ABE pills orally plus letrozole and continues follow-up checks regularly.
Discussion
A systematic review of CNS metastasis treatment in patients with HR+/HER2− MBC reports that local radiotherapeutic or surgical therapy is still the cornerstone of the therapeutic strategy. 13 However, the therapeutic indications may vary according to the patient’s neurological symptoms, performance status, the number, appearance, and size of metastases, the burden of extra-CNS disease, and comorbid illnesses.13–17 Patients treated with abemaciclib must be carefully followed to prevent or early treat side effects, such as diarrhea, asthenia, cytopenia, and nausea/vomiting, and monitored for insidious events such as interstitial lung disease. Abemaciclib is linked to an increased risk of interstitial lung disease as soon as 1–2 months after the beginning of treatment regardless of patients’ age, suggesting monthly monitoring since no preventive measures exist.18,19 The mechanism of event onset is still unclear even if some data suggests a role of augmented inflammatory cell recruitment as a consequence of the cycline-dependent kinase inhibitor (CDKI)-induced cell cycle arrest with consequent cellular senescence. 19
In all tumors, the blood-brain barrier (BBB) prevents the diffusion of many drugs to the CNS, hampering their antineoplastic activity against brain metastases. 4 ABE can cross the BBB, reaching effective antitumor concentrations in the BM tissue as high as 96- and 19-fold above in vitro IC50 for cyclin-dependent kinase 4 and 6, respectively.14,15,20 A pharmacokinetic modeling study showed that ABE crossed BBB, reaching the brain tissue at cancer inhibitory concentrations, and persisted locally more widely than ribociclib and palbociclib.20,21 However, other CDK4/6i may cross the BBB. Bao et al. 21 showed that ribociclib can also cross the BBB, supporting further clinical development of this agent to treat CNS neoplasms. Preclinical data suggest that palbociclib can also prolong survival in murine brainstem glioma models and improve temozolomide activity in gliomas.22,23 On the other hand, preclinical studies showed that efflux transport in the BBB may limit the central nervous system (CNS) diffusion of palbociclib, and this has important implications in determining effective dosing regimens of palbociclib therapy in the treatment of brain tumors. 24
Medical literature on the activity of CDKI plus hormonal therapy in SNC metastases from HT+/HER2- MBC is scarce. Table 1 shows the studies published up to date. All reports but two are 1–3 case observations, and most report incomplete data.15,25–35 The sizable prospective phase III trials testing ribociclib (Monnaleesa-3) and palbociclib (Paloma 2 and 3) included only 6 and 25 patients with SNC metastases, respectively. Moreover, specific outcome data are unavailable for these patients due to a lack of data or concerns regarding patient de-identification. 10
Studies reporting the use of CDKI in CNS metastatic breast cancer.
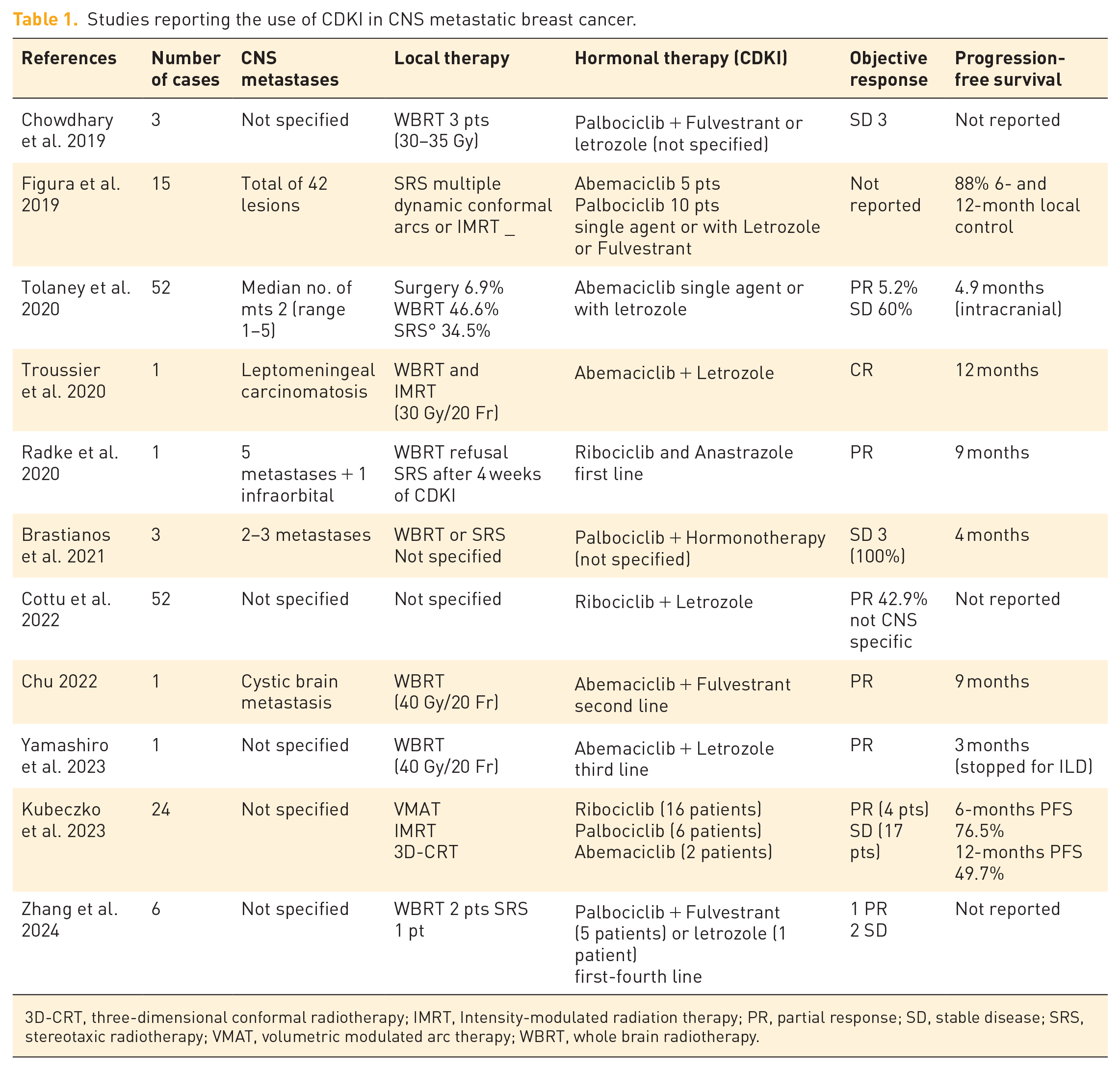
3D-CRT, three-dimensional conformal radiotherapy; IMRT, Intensity-modulated radiation therapy; PR, partial response; SD, stable disease; SRS, stereotaxic radiotherapy; VMAT, volumetric modulated arc therapy; WBRT, whole brain radiotherapy.
A phase II trial in patients with brain or leptomeningeal metastases secondary to HR+/HER2- MBC receiving ABE showed a 24% CNS response and good safety in line with prior studies. 15 The primary goal of this non-randomized, phase II study trial was to evaluate intracranial objective response rate in patients treated with ABE according to a two-stage design. The study included four cohorts of patients with brain metastases from HR+/HER− MBC, or HR+/HER+ MBC, or with leptomeningeal metastases, or with planned surgical resection of BM, secondary to HR+/HER+ and HER−/MBC. Secondary aims included extracranial response, ABE pharmacokinetics, brain metastases tissue exposure, and safety. Although the primary endpoint was not reached, however, response rate in patients with leptomeningeal metastases was higher than historical data, and pharmacokinetic data soundly proved the presence of ABE and its active metabolites in brain metastases and the cerebrospinal fluid (CSF) of high inhibitory drug concentrations.
In 2022, Chu reported the first case of ABE combined with fulvestrant in treating BM in a patient with HR+/HER2- MBC who responded at the brain. 33 The patient was previously treated with docetaxel plus capecitabine, followed by anastrozole maintenance therapy for 10 months, and palbociclib combined with exemestane for 22 months. The occurrence of SNC metastases and the discovery of a missense PIK3CA mutation (exon 10, c.1633G>A [p.Glu545Lys]) induced physicians to deliver WBRT for 40 Gy followed by ABE and FUL. After 3 months, SNC metastases demonstrated a partial response and extracranial bone metastases were stable. The patient enjoyed over 9 months of progression-free survival without apparent side effects.
Another issue is the tolerance and safety of CDK4/6i given simultaneously to radiation therapy. Generally, medical literature suggests that CDK4/6i may be safely administered with brain radiotherapy.13,25,26 Kubeczko et al. reported 24 patients treated with either ribociclib (n. 16), palbociclib (n. 4), or ABE (n. 2) before, during, or after radiation therapy without significant toxicity. 26 Six- and twelve-month progression-free survival (PFS) was 76.5% (95% CI: 60.3–96.9) and 49.7% (95% CI: 31.7–77.9), respectively, whereas six- and twelve-month local control was 80.2% (95% CI: 58.7–100) and 68.8% (95% CI: 44.5–100), respectively. 26 Figura et al. 28 reported a retrospective series of 42 BM treated with stereotactic radiotherapy in 15 patients within 6 months of CDK4/6i therapy with neurotoxicity as the primary endpoint. Ten patients received palbociclib and five ABE, and RT was delivered simultaneously, before, or after CDK4/6i inhibitors in 18 (43%), 9 (21%), and 15 (36%) lesions, respectively. Radiation-induced necrosis was recorded in two lesions pretreated with RT. These patients were treated with corticosteroids plus bevacizumab. Six- and 12-month local control of treated lesions was 88% and 88%, while 6- and 12-month distant brain control was 61% and 39%, respectively. Median OS was 36.7 months from the date of BM diagnosis.
In this paper, we report a unique case of HR+/HER2+ metastatic to SNC with a high burden of extracranial disease resembling lung cancer debuted with syncope. One of the brain metastases had a cystic radiological morphology and carried a particularly unfavorable prognosis. Cystic brain metastases in patients with BC are rare, less responsive to radiation therapy, and have a worse prognosis than solid brain metastases. The patient was bedridden and comatose. She was treated with intramuscular FUL, and a total dose of oral ABE was crushed and diluted in water after pharmacological consultation, achieving a rapid major objective response and a total functional recovery. The complete brain response induced treating oncologists and the patient herself to postpone radiation therapy. This case is, therefore, somewhat unique for both clinical presentations with severe neurological symptoms, radiological imaging, treatment decisions, and challenging drug administration and dramatic positive response.
The few case studies reported in the medical literature represent a limitation to definitive conclusions. Despite the above-reported limitations, the scientific and clinical interest in using ABE to treat BM remains high. The scientific papers reported in medical literature raise some interesting points about ABE activity and safety. In the adjuvant setting, ABE has improved invasive disease-free survival in high-risk patients with HT+/HER− BC, but the final data on OS are not mature yet. 36 Therefore, no data on BM occurrence is available yet due to the low rate of CNS metastases in this setting. Another issue is the activity of ABE in patients pretreated with other CDKI4/6i. Wander et al. showed that a subset of patients with progressive MBC after treatment with palbociclib may show further clinical benefit by the administration of ABE plus fulvestrant or aromatase inhibitor with a median PFS of 5.3 months and median OS of 17.2 months. 37
On the other hand, many patients experienced rapid progression on ABE, suggesting the existence of a resistance pathway that is common to other CDKI4/4i. Next-generation sequencing showed alterations in the RB1, ERBB2, and CCNE1 genes potentially linked to ABE resistance in these latter patients. Scientific data also suggest the efficacy of tyrosine kinase inhibitors in BM and support exploring the use of ABE with HER2 tyrosine kinase inhibitors and ET for patients with BM from HR+/HER2+ MBC.38,39 Although patients with untreated or progressive BM were not included in the monarch-HER trial, data suggest a significant role for ABE plus trastuzumab in treating HR+/HER2+ MBC, representing a potential alternative option to anti-HER agents plus systemic chemotherapy. 39 Many patients with BM are managed with various radiation therapy approaches. The issue of the safety of CDK4/6i administered concurrently with radiation therapy has been raised. ABE may be given concurrently with radiation therapy for BM with interesting results, although cautious toxicity monitoring is advised. 40
Conclusion
In conclusion, optimal management of CNS metastases in MBC still represents an unmet medical need. Published case reports suggest that all clinically available CDK4/6i have clinical activity in CNS metastases from HR+/HER2− MBC. However, no large controlled clinical trials demonstrate improved outcomes with these agents in this setting. Furthermore, scant information about the appearance of new CNS metastases precludes the possibility of concluding their ability to prevent the development of CNS metastases. Based on their potential clinical activity and acceptable toxicity profile, an expert opinion suggested using CDK4/6i for patients with HR+/HER2+ MBC to the CNS.
Some controlled trials are evaluating the activity of CDK4/6i in this clinical setting. The phase II Palina and the phase III Patina trials are currently assessing the efficacy and safety of palbociclib in MBC patients with CNS metastases.41,42 In the Phase I TEEL study, ribociclib is combined with tamoxifen with and without goserelin in HR+/Her2− MBC patients who may also have brain metastases (NCT02586675). Lastly, an expanded access study for HR+/HER2− metastatic BC will be treated by abemaciclib after disease progression on prior therapies. Immunotherapy may represent a possible future progress. Preclinical studies in immunocompetent mice have shown that adding ABE to immunotherapy reduces tumor burden and improves OS. This effect is probably due to increased recruitment and expansion of CD8+ effector T-cells, decreased CD4+ regulatory T (Treg) cells, and immunosuppressive cytokines in brain tumors. 43 The results of these trials are eagerly awaited. Therefore, the results of these clinical trials enrolling patients with CNS metastases are eagerly awaited.
