Abstract
Background:
The evidence base for the management of oligoprogression in metastatic melanoma (MM) is limited. To our knowledge, this study presents the first analysis of the local benefit (LB) of radiotherapy combined with hyperthermia during systemic treatment in this diagnosis.
Methods:
Patients with oligoprogressive MM who were treated with radiotherapy (RTH) combined with hyperthermia (HT) at a melanoma center between 2019 and 2023 were evaluated. Oligoprogression was defined as up to five progressive metastases. Inclusion criteria was the availability of dimensions assessment of the lesion subjected to RTH before and after treatment, patients without follow-up imaging after radiotherapy were excluded. The benefit of RTH + HT was evaluated in terms of local control (LC) rates and LB rates. LC was defined as the percentage of patients who met the Response Evaluation Criteria In Solid Tumors (RECIST 1.1) criteria for stable disease (SD), partial response (PR), and complete response (CR). LB was defined as the proportion of patients who met the PR and CR criteria. In addition, overall survival (OS) rates were estimated. The association between BRAF status, age, concomitant systemic treatment, radiation total dose, and biologically effective dose and LC was estimated. Data regarding adverse effects associated with RTH + HT were compiled. Survival analyses were performed using the Kaplan–Meier estimator and log-rank tests and were used to compare between groups.
Results:
In total, 101 patients were included in the study, the median follow-up was 15.3 months (14–18 months). There were 56.4% BRAF(−) and 43.6% BRAF(+) patients. Most patients (71.3%) were irradiated during immunotherapy, 10.9% received concomitant BRAF and MEK inhibitors, and 3.9% had chemotherapy. LC and LB medians were not reached at the time of analysis. The 1- and 2-year LC rates were 93.5% (95% confidence interval (CI): 88.1%–99.3%) and 88.3% (95% CI: 79.9%–97.6%), respectively. The 1- and 2-year LB rates were 87.5% (95% CI: 80.5%–95.2%) and 78.1% (95% CI: 67.9%–89.9%), respectively. The mean reduction in irradiated tumor size across the entire cohort was 72%. The mOS from radiotherapy was not achieved at the time of our analysis, accordingly 1- and 2-year OS rates were 100% and 95% (95% CI: 90.4%–99.9%). None of the evaluated factors influenced LC among patients.
Conclusion:
Hyperthermia with radiotherapy is an effective treatment for patients with oligoprogressive melanoma. This approach has resulted in excellent LC.
Introduction
One of the first recommendations for radiotherapy (RTH) in melanoma incorporated adjuvant irradiation after lymphadenectomy (LND) in patients with risk factors for recurrence, such as a large metastasis diameter, multiple nodes involved, and extracapsular extension. 1 However, a clinical trial has demonstrated that adjuvant RTH after LND reduces the risk for relapse in the irradiation volume by approximately 50% but has no impact on recurrence-free survival (RFS) and overall survival (OS).2,3 The routine irradiation of the bed after LND was abandoned due to the introduction of effective systemic treatment and is now limited to selected patients. The indications for RTH in the current era of treatment are limited. The primary indications of RTH for melanoma are in the inoperable disease and palliative setting.4,5 The use of stereotactic radiotherapy with immunotherapy for melanoma brain metastases is of great importance, and this approach results in improved outcomes in terms of both progression-free survival (PFS) and OS.6,7 Also, RTH is employed in the treatment of metastases in patients with oligometastatic and oligoprogressive disease.7,8
Oligoprogression, as defined by the European Society for Radiotherapy and Oncology (ESTRO) and the European Organization for Research and Treatment of Cancer (EORTC), is the progression of a limited number of metastases under active systemic therapy.9,10 In several cancers, RTH represents a valuable treatment option in the scenario of oligoprogressive disease that could improve local control (LC), postpone systemic therapy commencement, elicit a full response, and prolong OS. In the case of progressive single metastatic lesions with a stable overall response to systemic treatment in other diagnoses, RTH can be successfully incorporated into the treatment. 11 The control of progressive metastases using local techniques has been demonstrated to significantly impact patient overall prognosis, for example, in breast, lung, or prostate cancers.12–14
The use of stereotactic RTH (SBRT) in the treatment of oligoprogression has become of increasing interest in recent years in the context of melanoma patients; however, data are still limited. Studies have demonstrated that the simultaneous administration of systemic therapy and SBRT is not only safe but also allows for a significant extension of the time to the next-line systemic treatment.15,16 Furthermore, the safety of the therapy has been demonstrated, with the majority of studies indicating no significant side effects of grade 3 or higher.15,17 The utilization of RTH in conjunction with immunotherapy (ITH) in solid tumors has been demonstrated to be a safe and efficacious approach, as evidenced by findings from a phase I study employing multi-site SBRT with a biologically effective dose (BED) exceeding 100 Gy in conjunction with pembrolizumab, nivolumab, or ipilimumab. The study demonstrated that only 27% of patients experienced grade ⩾3 AEs, with dual-agent immunotherapy being correlated with a higher incidence of toxicity than ITH monotherapy. 18 In a separate study of SBRT with pembrolizumab monotherapy, six patients experienced dose-limiting toxicities, and the objective response rate was 13.2%. The mOS was 9.6 months (95% CI, 6.5–NA months). 19 In the melanoma studies, the results in terms of LC and OS were also encouraging. In a phase I study investigating SBRT in combination with immunotherapy based on an anti-PD1 or anti-CTLA4 agent, the 2-year PFS rate was 21.9%, while the OS rate was 49.6%, only 4 out of 24 patients developed dose-limiting toxicities. 20 A retrospective analysis demonstrated that LC rates at 12 and 24 months were 96.6% and 82.8%, respectively, furthermore was a significant response in the irradiated lesions related to OS results (p = 0.019). 21 At present, research is also being conducted to improve outcomes with SBRT and ITH. It has been observed that elevated interleukin-8 (IL-8) levels are correlated with resistance to treatment. Consequently, a phase I clinical trial is being conducted with an anti-IL-8 antibody, nivolumab, and SBRT in melanoma. 22
Hyperthermia (HT) is an additional method of supraphysiological elevation of tissue temperature to enhance the efficacy of concomitant treatment in the form of RTH or systemic therapy. The biological evidence of its effectiveness is based on the following: increasing tumor perfusion and oxygenation, inducing cell death, inhibiting DNA repair mechanisms, and modulating the immune response within tumor tissue. 23 RTH has been demonstrated to exert a cytotoxic effect, primarily due to its oxygen-dependent production of reactive oxygen species, which results in DNA damage. 24 The addition of HT has a synergistic effect in this model, allowing for the dilation of blood vessels in the tumor microenvironment and the delivery of greater amounts of oxygen to the cells. Furthermore, cancer stem cells that are inaccessible to RTH alone can be activated by HT, thereby overcoming radioresistance. An additional beneficial effect on synergism is the immunostimulatory mechanism of HT, which increases the immunogenicity of tumor cells. 25 Thus, HT has been utilized with favorable outcomes in other diagnoses, including prostate cancer, colon cancer, soft tissue sarcoma, and breast cancer.26–28 The earliest and to this day to our knowledge, the only evidence of its use in metastatic or recurrent melanoma can be traced back to the 1990s, when a randomized trial demonstrated that LC without HT was 28% and with HT was 46% (p = 0.008), with good treatment tolerance. 29 Nevertheless, translating these results into current treatment guidelines is challenging due to the multitude of new systemic therapies available for patients with metastatic melanoma (MM).
Research on RTH in combination with modern systemic treatment is limited. A phase I clinical trial assessing the use of SBRT in combination with immunotherapy based on anti-PD1 and anti-CTLA4 antibodies demonstrated the effectiveness of a dose of 10 Gy in this indication, with only 16.7% of patients experiencing dose-limiting toxicities. 20 However, there are no available reports on the effectiveness of combining RTH with HT and immunotherapy or BRAF/MEK inhibitors. In our previous report, we assessed the outcomes of melanoma oligoprogressive metastases treated with radiotherapy combined with locoregional hyperthermia and demonstrated good LC and tolerability of this combination. The 1- and 2-year LC rates were 83% (95% CI: 74–94) and 54% (95% CI: 40–73), respectively. 30
Therefore, a retrospective analysis of a consecutive patient cohort treated with a multimodal therapeutic approach, comprising RTH combined with locoregional HT and systemic treatment, was conducted at our institution. The objective was to assess the local effectiveness of the combination of RTH and HT in oligoprogression in MM.
Materials and methods
Patients characteristics
Patients with histologically confirmed stage IV MM of the skin C43 per ICD-10 (the International Classification of Diseases, Tenth Revision) who were treated for oligoprogressive lesions with RTH combined with locoregional HT at the Maria Sklodowska-Curie National Research Institute of Oncology in Warsaw, Poland, between 2019 and 2023 were included in this study. All patients fulfilled the criteria of oligoprogression, as defined by the ESTRO and EORTC criteria, assuming limited progression of some lesions with stable disease (SD) responding to systemic treatment, with a maximum of five progressive metastatic lesions.9,10 Certain metastatic sites, including the brain or lungs, were excluded due to treatment guidelines. The clinical data were obtained from the medical records and included the age, BRAF status, gender, irradiated lesion site, details of the RTH, and information about concomitant systemic treatment administered during RTH or up to a month before/after (Table 1). Tumor size was determined through the analysis of radiographic findings, predominantly computed tomography (CT) scans, and defined following the dimensions of the lesion’s length and width. Follow-up data were obtained in accordance with current guidelines, with CT scans conducted approximately every 3 months.4,5 To unambiguously determine the local effect, it is necessary to perform a minimum of two CT scans after RTH with an appropriate time interval or to compare the image of the first diagnostic CT scan after RTH to the radiotherapy planning CT scan. Thus, patients without any follow-up imaging were excluded. The data on patient characteristics, such as age, gender, irradiated region, and concomitant treatment have been summarized using descriptive statistics.
Characteristics of patients included in the analysis.

Concomitant systemic treatment administered during RTH or up to a month before/after.
RTH, radiotherapy.
Radiotherapy and hyperthermia details
RTH planning and delineation were conducted by an experienced team comprising at least two radiation oncologists, with protocols in place for verification and validation (Figure 1). Fractionation regimens involved delivering 24–60 Gy in 3–20 fractions (Table 2). The calculated mean BED was calculated assuming an alpha/beta ratio of 2.5 Gy. 31 Immobilization strategies and bolus application were tailored on an individual basis. The gross tumor volume (GTV) was delineated on planning CT, in selected patients using image registration with contrast-enhanced magnetic resonance imaging or diagnostic contrast-enhanced CT, as required. The decision to define a clinical target volume (CTV) was contingent upon a thorough benefit–risk assessment. The planning target volume was established by expanding the GTV or CTV. Patients were treated using external beam RT using a linear accelerator. Most patients (96%) were irradiated with intensity-modulated RTH.
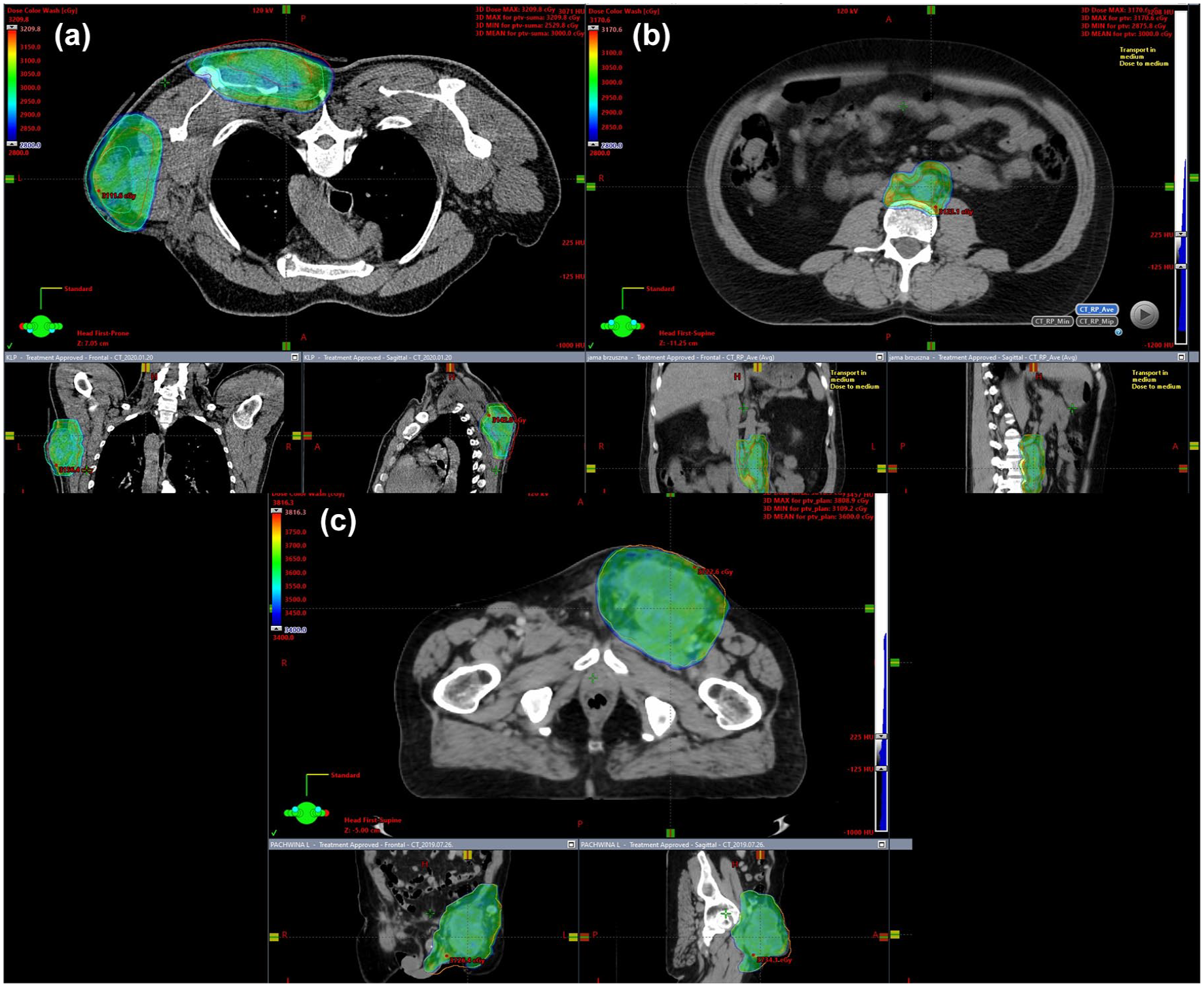
Radiotherapy planning of (a) two lesions of the chest wall, (b) a periaortic lesion, and (c) a lesion of the left groin area.
Characteristics of employed radiotherapy.
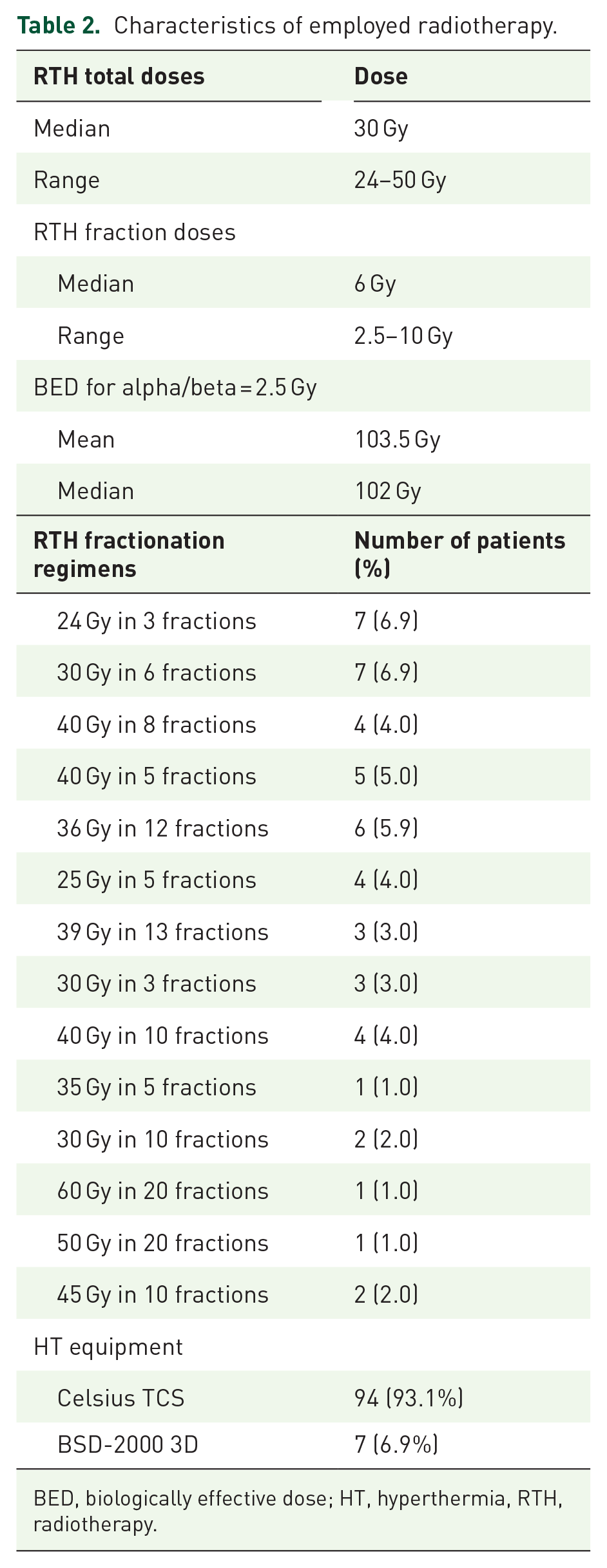
BED, biologically effective dose; HT, hyperthermia, RTH, radiotherapy.
HT was administered biweekly. For each patient, anthropometric measurements such as body weight and height, along with tumor-specific parameters, were assessed to select the appropriate thermal therapy equipment. The therapeutic objective was to elevate the temperature of the targeted tissue between 39°C and 42°C. After each HT session, an RTH fraction was delivered within 1 h. Two systems were employed for HT. First, the Celsius TCS system facilitates uniform temperature distribution within the treated area using a water bolus system and an adjustable dual-electrode configuration. This system employs 13.56 MHz electromagnetic waves to generate thermal energy, utilizing capacitive coupling for energy transmission. Second the BSD-2000 3D system, specifically indicated for deep-seated abdominal and pelvic tumors. This equipment encompasses the patient with 24 antennas, emitting electromagnetic waves ranging from 75 to 140 MHz, and employs specialized algorithms to focus the energy on the target volume. Contraindications for HT comprised implanted medical devices (e.g., pacemakers, stabilizers, or prostheses), significant tumor-related pain unmanageable with pharmacotherapy, severe cardiac or pulmonary conditions, uncontrolled hypertension, recent myocardial infarction or cerebrovascular incident within the past 6 months, or pregnancy.
Statistical analysis
LC was determined following the Response Evaluation Criteria In Solid Tumors (RECIST 1.1) criteria. These criteria correspond to the categorization of SD, partial response (PR), and complete response (CR). To gain deeper insight into the local impact of RTH with HT, we introduced the concept of local benefit (LB), which allows for the assessment of objective responses to treatment without the inclusion of SD. LC was defined as an event if any treated lesion exhibited growth of ⩾20%, and LB was defined as a reduction in tumor size of ⩾30%. The local results were estimated based on the observed changes in the irradiated lesion. 32 For this assessment, an elliptical tumor shape was assumed in 2D imaging, and the product of the dimensions was used, assuming that the tumor size was π/4 * A * B. The overall response to treatment was evaluated separately in accordance with RECIST 1.1. OS was calculated in two ways: from the date of diagnosis to the date of death and from the start of radiotherapy to death or last follow-up. For patients who were still alive at the time of the analysis, follow-up was censored at the time of the last observation. The Kaplan–Meier method was utilized to estimate the LC, LB, and OS rates. The median follow-up period was calculated using the reverse Kaplan–Meier estimator. A univariate analysis was conducted using a Cox proportional hazard regression model. The statistical analysis was conducted and presented using R version 4.3.2, with the “survival” package employed for this purpose. 33 The reporting of this study conforms to the STROBE statement (Supplemental File 1).
Results
Characteristics of the patients and radiotherapy details
A total of 101 patients with histologically confirmed stage IV MM who were treated for 118 oligoprogressive lesions with RTH combined with locoregional HT were included in this study. The data on patient characteristics, such as age, gender, irradiated region, and concomitant treatment have been summarized in Table 1.
The RTH technique and HT specifics are detailed in Table 2. The most commonly employed fractionation regimens involved delivering 30 Gy in five fractions. The calculated mean and median BED were 102 and 103.5 Gy, respectively. The majority of patients (93.1%) underwent a 60-min HT session utilizing the Celsius TCS system, the remaining patients received 90-min HT sessions with the BSD-2000 3D system.
Local tumor response
As was the case with mOS, both LC and LB medians were not reached at the time of analysis (Figure 2). Nevertheless, the 1- and 2-year LC rates were 93.5% (95% CI: 88.1%–99.3%) and 88.3% (95% CI: 79.9%–97.6%), respectively (Figure 2(a)). The mean reduction in irradiated tumor size across the entire cohort was 72%. It was observed that LC was primarily attributed to objective responses to treatment, rather than solely to the stabilization of progressive metastasis. The 1- and 2-year LB rates were 87.5% (95% CI: 80.5%–95.2%) and 78.1% (95% CI: 67.9%–89.9%), respectively (Figure 2(b)). A total of 11 patients (10.8% of the entire group) received a total RTH dose that was lower than 30 Gy. Eight patients received a total dose of 24 Gy in three fractions, while three others received 25 Gy in five fractions. It is noteworthy that, after incorporating the median dose of radiotherapy (6 Gy fraction dose, 30 Gy total dose) into the univariate Cox model, no significant difference was observed in the impact on LC between patients who received doses below the median and those who received doses equal to or above it (p = 0.3). To validate this result, we also included BED in the analysis, both with a mean cutoff of 103.5 Gy and a median of 102 Gy, we did not notice any effect on LC (p = 0.7). In analyzing the correlation between additional factors and response to RTH + HT treatment, no statistically significant correlations were identified. The analysis revealed that BRAF mutation status (p = 0.5), age (p = 0.4), and type of concomitant systemic therapy (p = 0.2) did not result in a more favorable local response among patients.

(a) Local control in patients with oligoprogressive disease treated with radiotherapy with hyperthermia (RTH + HT). (b) Local benefit in patients with oligoprogressive disease treated with RTH + HT.
The correlation between local treatment response and systemic response is illustrated in Figures 3 and 4. The results of the response to concomitant systemic treatment are also included. As illustrated in Figures 3, 4 and 5, a CR at the systemic level was observed exclusively in patients who also demonstrated a local response to RTH. In the cohort of patients whose progressive metastases did not demonstrate a decrease or increase following RTH, the final CT-based imaging assessment also revealed disease progression at the systemic level, predominantly associated with the emergence of new lesions or a notable increase in all existing metastases (Figure 3). Furthermore, as demonstrated in Figure 3(a) versus (b), there is tumor growth from the diagnostic CT, which was used to qualify the patient for RTH treatment, to the RTH planning CT, even among patients who were then undergoing active ITH/targeted therapy. This may indicate that the irradiated lesions have acquired resistance to systemic therapy.

Spider plot on the change in tumor relative to the initial measurement among patients who underwent radiotherapy with adjuvant hyperthermia. The degree of change corresponding to a partial response and progressive disease is indicated in the charts with a dashed line. (a) Change has been shown since the routine CT, the results of which determined patient eligibility for treatment. (b) Change demonstrated from CT scan performed for radiotherapy treatment planning.

Waterfall plot presenting a change in tumor size in correlation with systematic treatment outcomes among patients treated with radiotherapy with adjuvant hyperthermia. (a) Measurements from routine CT scans before treatment. (b) Measurements from CT for treatment planning. (c) Measurements from the first routine CT after treatment. (d) The correlation of overall survival rates for each evaluated patient.

The response to RTH with hyperthermia in CT. (a) 3.5 cm progressive melanoma metastasis in the adrenal gland as observed prior to treatment (02.02.2019). (b) Same patient, assessment of treatment response (04.2023). There was a complete response (CR) in the irradiated lesion. However, the patient exhibited PD at the systemic level. (c) 3.5 cm oligoprogressive metastasis to the axillary lymph nodes before RTH (09.10.2020). (d) The same patient exhibited CR in the irradiated lesion following treatment (07.04.2022). In addition, the patient achieved a systemic CR.
Survival outcomes
The average follow-up from diagnosis was 52.5 months (39–60 months), and the average follow-up for treatment with RTH with HT was 15.3 months (14–18 months). The mOS for the entire patient cohort from the time of diagnosis and the mOS from RTH were not achieved at the time of our analysis (Figure 6). The corresponding 1- and 2-year OS rates were 89.4% (95% CI: 83%–96.2%) and 73.6% (95% CI: 62.1%–87.3%) for the entire treatment cohort. The OS from RTH treatment rates were 100% and 95% (95% CI: 90.4%–99.9%).

OS among the patients treated with RTH with adjuvant hyperthermia: (a) OS for RTH treatment and (b) OS estimated from the initial diagnosis.
Discussion
This is the largest analysis to demonstrate the substantial efficacy of RTH and HT in the treatment of MM. Oligoprogression can be used to describe the initial stages of melanoma becoming resistant to systemic treatment.34,35 The optimal management strategy should aim to control progressive metastasis as early as possible, to improve the patient’s prognosis.36,37 Progressive lesions may be managed by local treatment, which enables the continuation of ongoing systemic therapy as a treatment beyond progression.38,39 The main local treatment options are surgical and RTH techniques.14,40 Also, extended early-line systemic therapy has been demonstrated to prolong OS.41–43 However, the local approach has been demonstrated to yield substantial benefits in comparison to prolonged systemic therapy. 44 RTH, particularly SBRT, is being increasingly employed in the case of oligoprogression and is included in the treatment guidelines.13,45 In melanoma, a phase II clinical trial evaluating the use of SBRT with interleukin 2 demonstrated an objective response rate of 54% in patients receiving the combination treatment, compared to 35% in patients treated with IL-2 alone. No significant difference was observed in OS. 46 Another study evaluating SBRT in combination with immunotherapy demonstrated an objective response rate of 45%. Furthermore, systemic CR was observed in patients who achieved CR in the irradiated lesion. Nevertheless, the incidence of adverse events was considerable, with 17 of the 20 patients experiencing complications related to the therapeutic regimen. 47 In addition, a phase I study evaluating the combination mentioned above demonstrated a 2-year PFS of 21.9% (95% CI, 7.1%–41.8%) and a 2-year OS of 49.6% (95% CI, 28.7%–67.6%). 20 A retrospective analysis of SBRT in melanoma oligoprogression also demonstrated its efficacy. In a retrospective analysis evaluating the use of SBRT among patients with oligoprogression of melanoma, the 1- and 2-year LC rates were 94.2% (87.0–98.1) and 90.3% (81.3–96.1), respectively. 17 In a similar clinical setting, another study demonstrated 1- and 2-year LC rates of 96.6% and 82.8%, respectively. 21
To the best of our knowledge, our study is the first to evaluate the use of RTH combined with HT in accordance with the most recent systemic treatment guidelines. The results of our study demonstrate the continued efficacy of RTH in the treatment of melanoma oligoprogression. In comparison to analyses conducted using only RTH, the 1- and 2-year LC rates were 93.5% and 88.3%, respectively. Notwithstanding the use of a combination of HT with active systemic treatment based on immunotherapy and other modalities, no severe treatment-related complications were observed. In consideration of the OS characteristics, the patients in our study cohort exhibited a 1-year OS rate of 89.4% and a 2-year OS rate of 73.6% for those who received RTH treatment. The 2-year OS rate of 73% was 1.5 times higher than those observed in clinical trials with only stereotactic radiation therapy in combination with ITH for MM. 20 Despite recent exemplary descriptions of the abscopal effect following immunotherapy with RTH, it remains unclear whether this phenomenon occurred among our patients. 48 Similarly, as in previous analyses, no relationship was observed between the total dose and fractionation regimens of RTH used and the local effects of treatment.20,21 It may therefore be surmised that both low and high doses are equally efficacious; however, this hypothesis and optimal doses require confirmation and evaluation through the conduct of larger prospective studies. 49 A comparison of our results with the only available study on the use of RTH + HT in melanoma reveals a significantly higher 2-year LC of 88% compared to the 37% reported in the aforementioned study. 29 The authors of the aforementioned analysis directly demonstrated the beneficial effect of HT on the results, with a p-value of 0.008. In contrast to our findings, the authors observed a positive correlation between higher RTH doses. However, the comparison was made between a total dose of 24 versus 27 Gy in three fractions, which in our study were rarely used. This discrepancy may explain the observed difference in results. Similarly, the authors did not observe any additional complications associated with the use of HT. 29
With regard to the RTH + HT studies in other diagnoses, there have been consistent findings. The incorporation of HT into treatment regimens for both breast and cervical cancer has been demonstrated to markedly enhance treatment outcomes. In a clinical trial for breast cancer, univariate analysis demonstrated that HT resulted in a superior local prognosis (68% for RTH alone vs 50% for RTH + HT, p = 0.04). 50 In cervical cancer, the 5-year OS rate in the HT group (81.9%) was superior to that observed in the non-HT group (72.3%; p = 0.04). 51 Furthermore, a study evaluating the efficacy of HT in the treatment of pelvic tumors demonstrated its effectiveness in a range of diagnoses, including bladder cancer, cervical cancer, and rectal cancer. The CR rates were 39% after RTH and 55% after RTH plus HT (p < 0.001). Also, the OS rates were markedly elevated in the HT group (27% vs 51%). 52 An increasing number of studies are examining the potential benefits of combining HT with RTH. Nevertheless, our previous analysis of the utilization of HT in soft tissue sarcomas demonstrated that the incorporation of HT into the therapeutic regimen resulted in 93% rates of 1-year local PFS. 53 It is noteworthy that, even in cases where the sole objective is the palliation of painful bone metastases, HT has been demonstrated to be superior in terms of efficacy. In the study, 78% of patients exhibited a complete or PR. Moreover, the utilization of analgesic medications declined from 74% to 48% post-treatment, and no significant adverse effects were documented. 54 These studies largely align with our analysis, indicating that this technique can enhance not only LC but also patient survival over the long term.
While our study represents a pioneering effort to assess the efficacy of RTH combined with HT in patients with MM, it is not without inherent potential for bias and limitations. As with any retrospective analysis, our study is subject to the limitations inherent to this methodology. In addition to its retrospective design, this study is a single-arm study, which precludes direct comparison of the effects of RTH + HT to RTH monotherapy alone. Furthermore, the HT employed excluded certain metastatic sites, including the brain or lungs, which introduced a potential selection bias. Also, the absence of precise temperature measurements in target is one of the Celsius TCS system drawbacks. Notwithstanding the favorable outcomes in terms of LC and OS, it is acknowledged that the observation period is insufficient to assess long-term outcomes. It can be hypothesized that the incorporation of hyperthermia may prove advantageous for patients, particularly those with substantial tumor sizes, accessible metastases, and those who exhibit rapid progression despite systemic therapy.
Conclusion
The combination of RTH and HT represents an efficacious treatment option for patients with oligoprogressive melanoma. This approach has resulted in optimal LC. Even when combined with various systemic therapies, this treatment is efficacious.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251316189 – Supplemental material for Radiotherapy combined with locoregional hyperthermia for oligoprogression in metastatic melanoma local control
Supplemental material, sj-docx-1-tam-10.1177_17588359251316189 for Radiotherapy combined with locoregional hyperthermia for oligoprogression in metastatic melanoma local control by Aneta Maria Borkowska, Paulina Chmiel, Piotr Rutkowski, Maria Telejko and Mateusz Jacek Spałek in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
