Abstract
Background:
Pulmonary lymphoepithelioma-like carcinoma (pLELC) is a rare subtype of non-small-cell lung cancer that predominantly affects younger, non-smoking individuals in southern and southeast Asia, where Epstein–Barr virus (EBV) prevalence is high. The efficacy and safety of immunotherapy in pLELC, especially in second-line settings, remain inadequately explored.
Objectives:
This study aimed to evaluate the efficacy of immunotherapy, either alone or in combination with chemotherapy, in improving progression-free survival (PFS) and overall survival (OS) in patients with advanced pLELC.
Design:
This was a multicenter retrospective study.
Methods:
A retrospective analysis was conducted on 252 patients with stage IIIB–IV pLELC treated across six centers. Patients received chemotherapy, immunotherapy, or a combination of both (chemoimmunotherapy). The primary outcomes measured were PFS and OS across different treatment regimens.
Results:
Chemoimmunotherapy significantly improved both PFS and OS compared to chemotherapy alone, in both first- and second-line settings. In first-line treatment, chemoimmunotherapy resulted in a median PFS of 17.6 months and OS of 26.1 months, compared to chemotherapy alone (PFS 8.7 months, OS 19.2 months). In the second-line setting, chemoimmunotherapy achieved a median PFS of 5.1 months and OS of 13.5 months, surpassing the outcomes with chemotherapy alone (PFS 3.3 months, OS 8.9 months). High baseline EBV-DNA levels (>2000 copies/mL) and low programmed death ligand 1 (PD-L1) expression (<50%) were associated with poorer outcomes. In addition, patients with high baseline serum tumor markers (STMs) and a dynamic reduction of ⩽20% in STMs exhibited significantly worse PFS and OS.
Conclusion:
The study suggests that immunotherapy, particularly when combined with chemotherapy, offers significant survival benefits for patients with advanced pLELC. Baseline EBV-DNA levels, PD-L1 expression, and both baseline and dynamic STM changes serve as important predictors of treatment response, highlighting the need for personalized therapeutic approaches in this unique subtype of lung cancer.
Plain language summary
This study focused on a rare type of lung cancer called pulmonary lymphoepithelioma-like carcinoma (pLELC), which mostly affects younger, non-smoking individuals in parts of Asia where the Epstein-Barr virus (EBV) is common. pLELC is a subtype of non-small cell lung cancer (NSCLC), and while immunotherapy has been successful in treating other types of NSCLC, its effectiveness in pLELC is not well understood, especially for patients who have already undergone other treatments. We reviewed medical records of 252 patients with advanced pLELC who were treated at six different hospitals. These patients received either chemotherapy, immunotherapy, or a combination of both. Our goal was to see which treatment option provided the best outcomes in terms of delaying the progression of the disease and extending patients’ lives. The results showed that combining immunotherapy with chemotherapy was more effective than using chemotherapy alone. For patients receiving this combination treatment as their first option, the average time before the cancer worsened was 17.6 months, and the average survival time was 26.1 months. In contrast, those who only received chemotherapy had an average of 8.7 months before the disease progressed and a survival time of 19.2 months. Similar benefits were seen for patients who received combination treatment after their cancer had already progressed on chemotherapy. We also found that patients with high levels of EBV in their blood and low levels of a protein called PD-L1 had worse outcomes. Additionally, those with high levels of certain tumor markers in their blood who did not show significant reductions in these markers during treatment also had poorer survival. These findings suggest that using immunotherapy alongside chemotherapy can offer significant benefits for patients with advanced pLELC. The study also highlights the importance of personalized treatment strategies based on specific biological markers.
Introduction
Numerous clinical studies have firmly established the efficacy and safety of immunotherapy in the treatment of advanced non-small-cell lung cancer (NSCLC).1–6 Pulmonary lymphoepithelioma-like carcinoma (pLELC), a rare subtype of NSCLC, predominantly affects younger, non-smoking individuals in southern and southeast Asia, where the prevalence of Epstein–Barr virus (EBV) is notably high.7–10
pLELC exhibits unique clinicopathological characteristics.11,12 Its genomic landscape reveals distinct alterations that impact cell cycle regulation, the JAK/STAT pathway, and the NF-κB pathway, potentially contributing to its tumorigenesis. 11 Multidimensional genomic analyses have shown that pLELC shares a closer genetic resemblance to nasopharyngeal carcinoma (NPC) than to other lung cancers. 11 Advanced EBV-positive NPC frequently presents with dense lymphocytic infiltration and positive programmed death ligand 1 (PD-L1) expression in tumor cells. 13 Several studies suggest that immunotherapy offers a promising treatment option for patients with advanced NPC.14,15 Furthermore, previous research has confirmed that PD-L1 expression is significantly higher in pLELC patients compared to those with lung squamous cell carcinoma or lung adenocarcinoma.12,16,17
In addition, a recent study highlighted the clinical significance of PD-L1 expression and its association with immunotherapy outcomes in pLELC, further supporting the potential benefits of immunotherapy in this subtype. 18 Our previous study demonstrated a significant improvement in 2-year progression-free survival (PFS) in the chemoimmunotherapy group compared to the chemotherapy group among pLELC patients (hazard ratio (HR), 0.38; 95% confidence interval (CI): 0.18–0.78; p = 0.007). 19 In addition, our retrospective analysis showed that patients with advanced pLELC and advanced lung squamous cell carcinoma who received first-line immunotherapy experienced improved PFS and overall survival (OS) outcomes. 20 However, due to small sample sizes, current research has primarily been limited to retrospective analyses of the efficacy and safety of first-line immunotherapy. 21 The study by Pang et al. 22 included some pLELC patients who failed first-line immunotherapy but did not conduct survival analyses. Consequently, there remains a lack of robust evidence regarding the effectiveness of second-line immunotherapy in advanced pLELC.
Building on previous research, our study includes a larger patient cohort to strengthen the existing evidence on first-line immunotherapy for advanced pLELC. In addition, we evaluated the efficacy and safety of second-line immunotherapy in these patients. Furthermore, we explored the relationship between baseline and dynamic serum tumor markers (STMs) and the efficacy of immunotherapy in both first-line and second-line treatments of pLELC, to deepen our understanding of therapeutic strategies for this rare NSCLC subtype.
Methods
The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement, as recommended by the EQUATOR Network. 23
This retrospective study included patients diagnosed with pLELC (stages IIIB–IV) from January 2008 to June 2022 at the following institutions: Guizhou Provincial People’s Hospital, the Third Affiliated Hospital of Sun Yat-sen University, the Third Xiangya Hospital of Central South University, the University of Hong Kong-Shenzhen Hospital, General Hospital of Southern Theater Command, and Jiangxi Cancer Hospital (Figure 1).
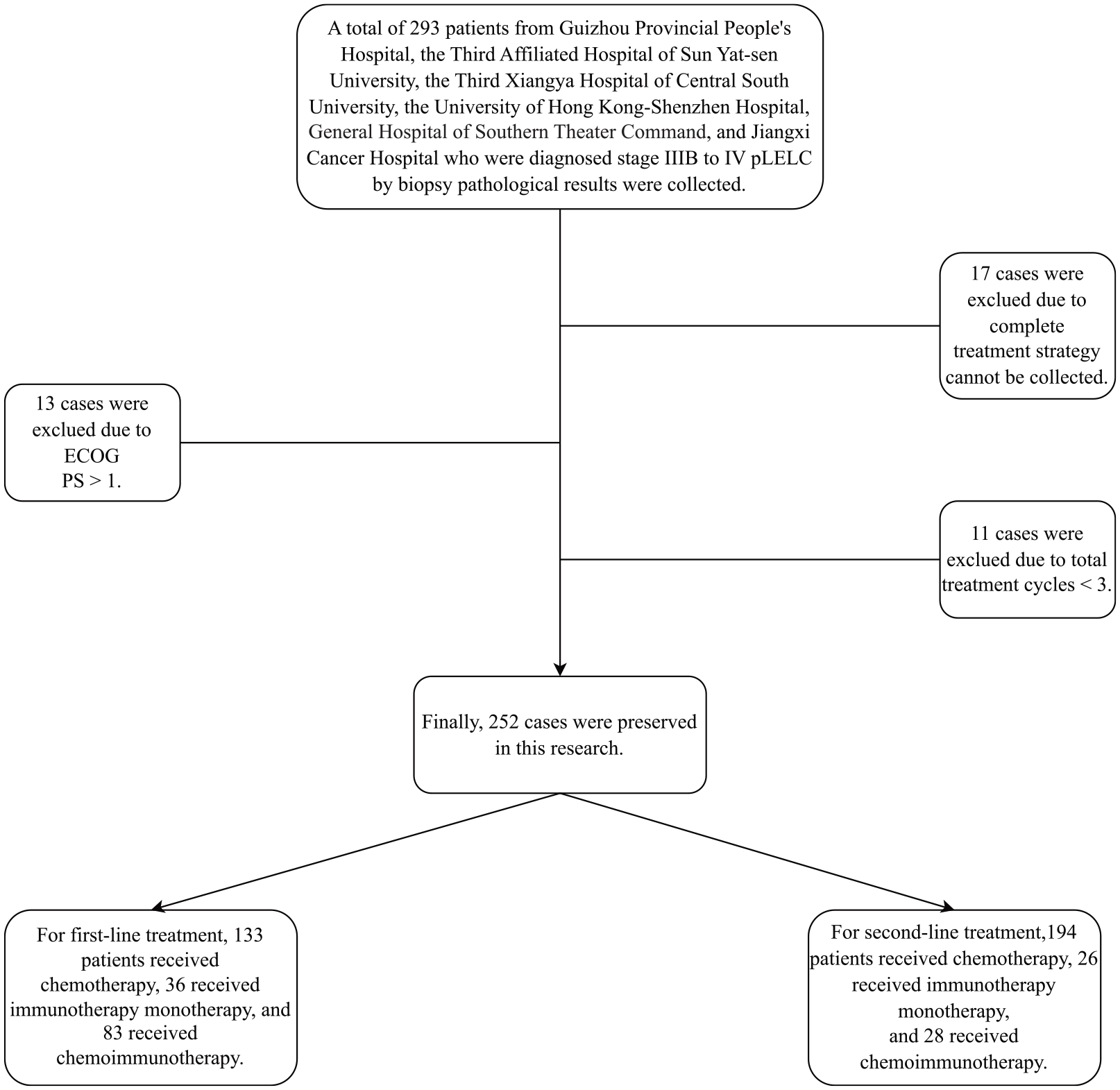
Flow chart of this study.
Inclusion and exclusion criteria
Patients were included based on the following criteria: a confirmed diagnosis of pLELC via imaging and biopsy, staging between IIIB and IV (eighth edition), and no history of surgical or radiotherapy treatment for pLELC. Exclusion criteria included incomplete treatment history, Eastern Cooperative Oncology Group performance status (ECOG PS) greater than one, fewer than three total treatment cycles, or fewer than three cycles of immunotherapy. We excluded patients who received fewer than three total treatment cycles or fewer than three cycles of immunotherapy to ensure that the treatment was administered for a sufficient duration to observe its efficacy and minimize the impact of early treatment discontinuation due to reasons unrelated to treatment efficacy. This exclusion criterion helps to reduce bias in outcome assessment and ensures more consistent and reliable comparisons across treatment groups. A total of 17 patients were excluded because their complete treatment history could not be collected due to transferring between multiple hospitals and the lack of comprehensive medical records. A total of 252 patients met the inclusion criteria and were included in the study.
Data collection
Collected patient data included gender, age, ECOG PS, smoking history, PD-L1 expression, clinical TNM stage (eighth edition), 24 tumor location, Epstein–Barr encoding region (EBER) status, EBV-DNA levels, treatment regimen, OS, and PFS. To ensure consistency across the study, all patients’ TNM staging was analyzed using the 8th edition of the lung cancer TNM staging system, regardless of the time of diagnosis. This allowed for a standardized approach to staging, despite the long time span of the study (January 2008 to June 2022).
PD-L1 expression
PD-L1 expression was assessed using the 22C3 antibody. Two pathologists independently evaluated all tissue sections for PD-L1 expression. Tumor proportion scores were categorized as follows: 0%–1% (negative expression), 1%–49% (low expression), and ⩾50% (high expression).
EBER and EBV-DNA detection
EBER positivity was evaluated using fluorescence in situ hybridization (FISH). EBV-DNA in peripheral blood was detected via polymerase chain reaction (PCR), with clearance defined as the absence of EBV-DNA post-treatment.
Epidermal growth factor receptor mutation and anaplastic lymphoma kinase rearrangement
At baseline, epidermal growth factor receptor (EGFR) mutations (EGFR L858R and EGFR 19del) were detected in tumor tissue specimens using the Amplification Refractory Mutation System-PCR. Anaplastic lymphoma kinase (ALK) rearrangements were identified via FISH. Due to less than 10% of patients undergoing second-generation sequencing, the data are not included in the paper.
Treatment regimen
All patients received at least three treatment cycles. The treatment regimens included the following: (1) platinum complexes combined with non-platinum anticancer agents; (2) single-agent non-platinum anticancer agents; (3) platinum complexes combined with non-platinum anticancer agents and PD-1/PD-L1 monoclonal antibodies; (4) non-platinum anticancer agents combined with PD-1/PD-L1 monoclonal antibodies; and (5) single-agent PD-1/PD-L1 monoclonal antibodies.
In our cohort, only one patient received single-agent chemotherapy (pemetrexed), and this decision was based on the clinical judgment of the treating physician. This patient had comorbidities that contraindicated combination therapy, but they met the overall inclusion criteria for ECOG PS and treatment history. Due to the limited number of patients receiving single-agent chemotherapy, we do not expect this to significantly impact the overall statistical results. We conducted a sensitivity analysis, and the outcomes remained consistent with the overall findings; however, the results of this analysis are not included in the main text.
Given the progressive nature of advanced pLELC, the inclusion of second-line treatment data enables a more nuanced analysis of immunotherapy’s potential benefits across different stages of treatment. Patients were grouped by treatment line to mitigate the influence of prior therapies on response rates and outcomes.
Although pLELC is reported to be chemo-sensitive and radio-sensitive, radiotherapy was not included in the comprehensive treatment regimen for patients with stage IIIB/IIIC disease in this study. Including this subgroup, which demonstrated significantly different survival outcomes, would not allow for a uniform assessment of immunotherapy efficacy in non-radiotherapy patients. We focused on patients who did not receive radiotherapy to ensure consistency in treatment exposure and to directly assess the impact of immunotherapy in a more homogeneous population.
Patients were classified into the immunotherapy group and the non-immunotherapy group based on whether they received PD-1/PD-L1 monoclonal antibodies in their treatment regimen. Immunotherapy was defined as receiving at least three consecutive treatment cycles containing PD-1/PD-L1 monoclonal antibodies. Both first-line and second-line immunotherapies were included in the analysis.
Treatment evaluation
OS was defined as the time from the start of treatment to death. PFS was defined as the time from the initiation of treatment to the first occurrence of disease progression or death from any cause. Tumor burden was assessed by CT examination in accordance with the Response Evaluation Criteria in Solid Tumors (RECIST, version 1.1). To avoid interference from tumor pseudoprogression, two consecutive CT examinations were performed following the administration of PD-1/PD-L1 monoclonal antibodies. All CT examinations were independently evaluated by two radiologists to ensure accuracy.
STMs assay
STMs were collected before immunotherapy and 6–8 weeks after treatment. For this cohort, STM analysis was conducted using the cobas e801 immunoassay module (Roche Diagnostics, Rotkreuz, Switzerland) and corresponding electrochemiluminescence immunoassay kits from Roche. According to the manufacturer’s instructions, the reference ranges for CEA, NSE, CYFRA 21-1, CA19-9, and CA125 were 0.00–5.00, 0.00–16.3, 0.00–3.30, 0.00–27.0, and 0.00–35.0 ng/ml, respectively. Based on previous studies involving advanced NSCLC patients receiving standard first-line and second-line immunotherapies, we used the upper limit of normal values for baseline STMs as cutoffs to explore their impact on immunotherapy efficacy. Patients were subsequently grouped based on significant improvement in at least two serum biomarkers. For dynamic STMs, a post-treatment reduction of ⩾20% from baseline was considered a significant improvement. Similarly, patients were grouped according to significant improvement in at least two serum biomarkers.
Statistical analysis
In this study, patients’ ages were categorized as a discrete variable: >65 years old as “High” and ⩽65 years old as “Low.” Chi-square tests and Fisher’s exact tests were used to compare differences in discrete variables between groups. PFS and OS curves were generated using the Kaplan–Meier method and validated with the log-rank test. HR and 95% CIs were assessed via univariate regression analysis. A p value of <0.05 was considered statistically significant, and all p values were two-sided.
Results
Patients characteristics
A total of 252 patients were included in this study. For first-line treatment, 133 patients received chemotherapy, 36 received immunotherapy monotherapy, and 83 received chemoimmunotherapy. Of the 248 patients who received second-line treatment, 105 had previously undergone first-line chemotherapy, 22 had received first-line immunotherapy, and 67 had been treated with first-line chemoimmunotherapy. Among those in the second-line immunotherapy group (N = 26), six patients had also received immunotherapy during their first-line treatment. Similarly, in the second-line chemoimmunotherapy group (N = 28), six patients had previously been treated with first-line immunotherapy. The demographic and clinicopathological characteristics of the patients in each group are summarized in Tables 1 and 2.
General clinicopathological features of patients with different first-line treatments.
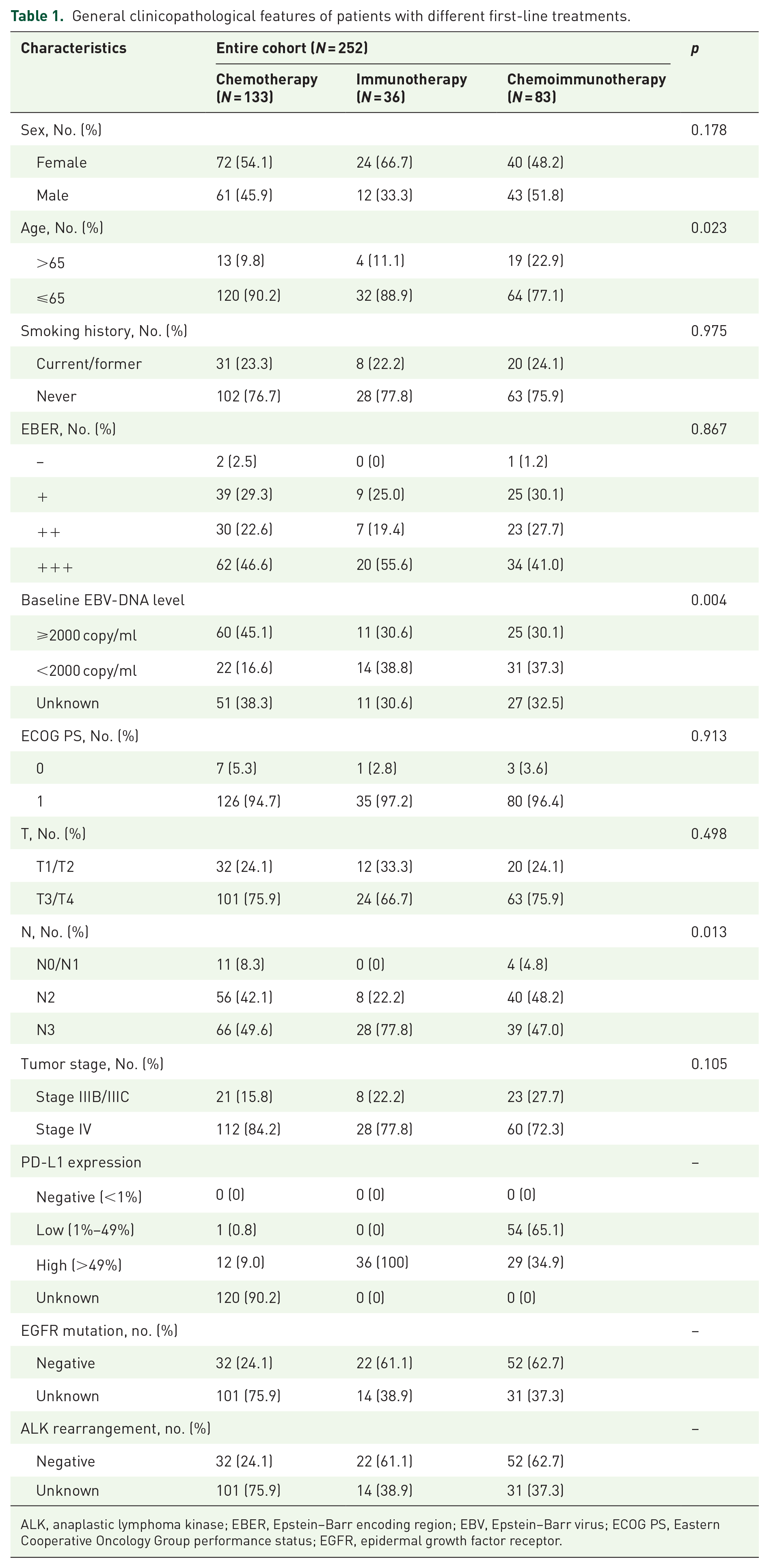
ALK, anaplastic lymphoma kinase; EBER, Epstein–Barr encoding region; EBV, Epstein–Barr virus; ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor.
General clinicopathological features of patients with different second-line treatments.

EBER, Epstein–Barr encoding region; EBV, Epstein–Barr virus; ECOG PS, Eastern Cooperative Oncology Group performance status; PD-L1, programmed death ligand 1.
The majority of patients included in the study were non-smoking females under the age of 65. Most patients were positive for the EBER. Among the 106 patients who were tested for EGFR mutations and ALK rearrangements, no mutations or rearrangements were detected.
The mean follow-up time was 29.1 months for first-line treatment and 11.1 months for second-line treatment. Follow-up data were updated prior to manuscript submission, with the last follow-up date being July 31, 2024.
Efficacy in first-line treatment
In first-line treatment, the objective response rate (ORR) was 51.13% for chemotherapy, 33.33% for immunotherapy, and 67.47% for chemoimmunotherapy. The disease control rate (DCR) was 81.95% for chemotherapy, 94.44% for immunotherapy, and 100% for chemoimmunotherapy. No patients achieved complete response (CR). The median PFS was 8.7 months for chemotherapy, 14.5 months for immunotherapy, and 17.6 months for chemoimmunotherapy. The median OS was 19.2 months for chemotherapy, 23.1 months for immunotherapy, and 26.1 months for chemoimmunotherapy. The log-rank test results indicated that (Figure 2), compared to the chemotherapy group, the immunotherapy group was associated with significantly improved PFS and OS (HR = 0.38, 95% CI: 0.26–0.56, p < 0.001; HR = 0.60, 95% CI: 0.41–0.88, p < 0.001, respectively). In addition, chemoimmunotherapy was significantly associated with improved PFS and OS compared to chemotherapy alone (HR = 0.27, 95% CI: 0.20–0.36, p < 0.001; HR = 0.41, 95% CI: 0.31–0.55, p < 0.001, respectively).

Progression-free survival (a), overall survival (b), and optimum change (c) of different groups in first-line treatment.
Efficacy in second-line treatment
In second-line treatment, the ORR was 10.82% for chemotherapy, 11.54% for immunotherapy, and 17.86% for chemoimmunotherapy. The DCR was 50.52% for chemotherapy, 46.15% for immunotherapy, and 57.14% for chemoimmunotherapy. No patients achieved CR. The median PFS was 3.3 months for second-line chemotherapy, 4.3 months for immunotherapy, and 5.1 months for chemoimmunotherapy. The median OS was 8.9 months for second-line chemotherapy, 10.7 months for immunotherapy, and 13.5 months for chemoimmunotherapy. Log-rank test results demonstrated that (Figure 3), compared to the chemotherapy group, the immunotherapy group was associated with significantly better PFS and OS (HR = 0.30, 95% CI: 0.19–0.47, p < 0.001; HR = 0.61, 95% CI: 0.40–0.94, p < 0.001, respectively). Furthermore, chemoimmunotherapy was significantly associated with improved PFS and OS compared to chemotherapy alone (HR = 0.17, 95% CI: 0.11–0.27, p < 0.001; HR = 0.37, 95% CI: 0.24–0.57, p < 0.001, respectively).
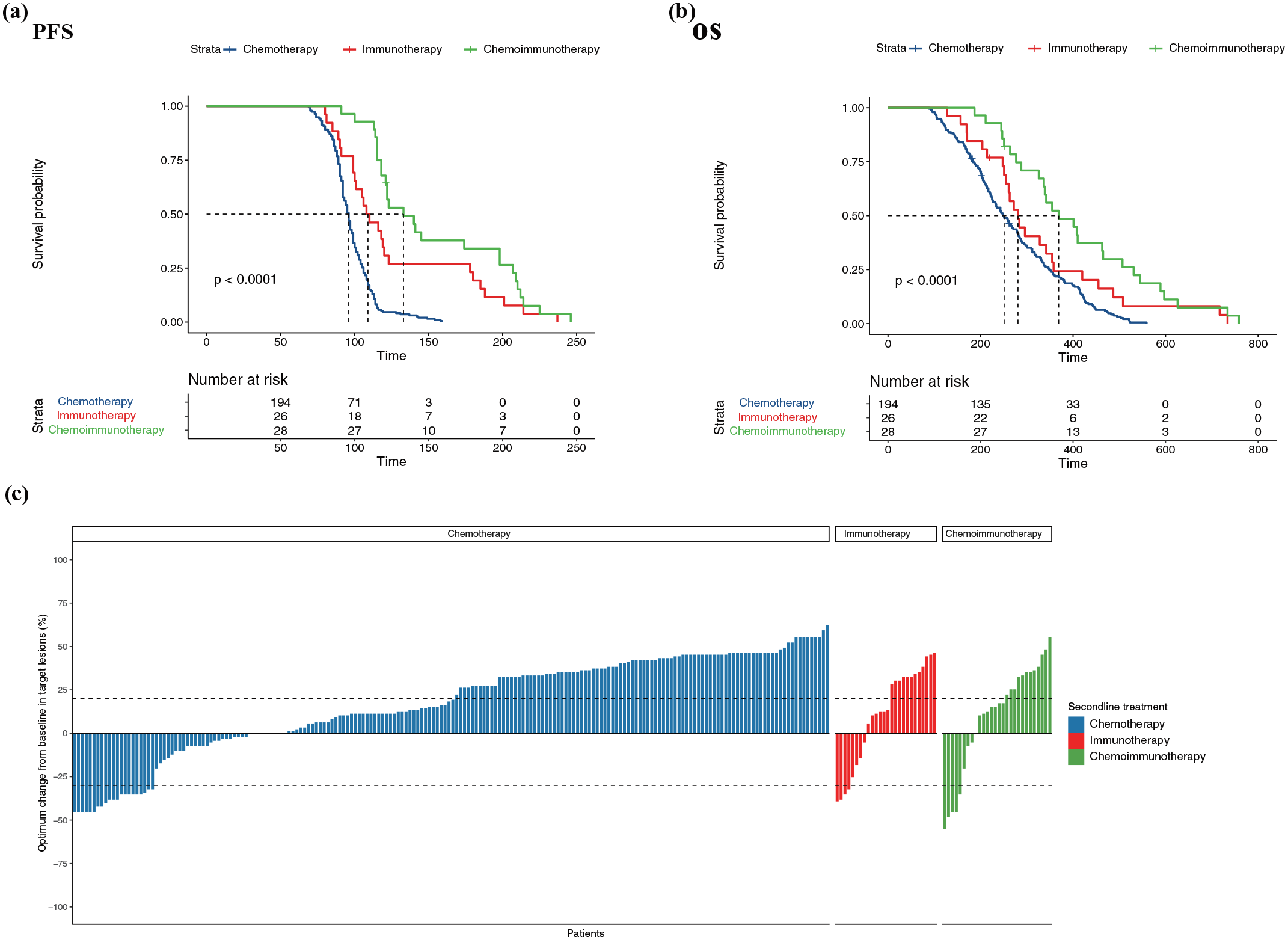
Progression-free survival (a), overall survival (b), and optimum change (c) of different groups in second-line treatment.
Safety and toxicity
No Grade 5 adverse events were observed in this study. In the chemotherapy group, the incidence of leukopenia was higher. In the immunotherapy group, liver damage was more prevalent. In the chemoimmunotherapy group, leukopenia was the most observed adverse event. Overall, myelosuppression was the most frequently reported adverse event in both the chemotherapy and chemoimmunotherapy groups (Table 3).
Treatment-related grade 3/4 adverse events of three groups of first-line and second-line treatment.

EBV-DNA levels in pLELC
Among patients in the first-line immunotherapy and chemoimmunotherapy groups, a total of 81 (68.1%) had baseline EBV-DNA-level data available. We divided these patients into a high EBV-DNA expression group and a low EBV-DNA expression group using a baseline EBV-DNA level of 2000 copies/ml as the cutoff. The median PFS and OS in the high EBV-DNA expression group were 13.0 and 20.9 months, respectively, compared to 19.5 and 28.4 months in the low EBV-DNA expression group. The log-rank test results indicated that (Figure 4), compared to the low EBV-DNA expression group, the high EBV-DNA expression group was associated with significantly worse PFS and OS (HR = 1.97, 95% CI: 1.25–3.11, p = 0.003; HR = 2.95, 95% CI: 1.82–4.79, p < 0.001).

Progression-free survival (a) and overall survival (b) according to the level of baseline EBV-DNA for immunotherapy and chemoimmunotherapy.
PD-L1 expression in pLELC
In our study, PD-L1 expression levels were assessed in both first-line and second-line treatment cohorts, as detailed in Tables 1 and 2.
In the first-line treatment cohort (Table 1), none of the patients had negative PD-L1 expression (<1%). Among the chemotherapy group (N = 133), 0.8% of patients had low PD-L1 expression (1%–49%), 9.0% had high PD-L1 expression (>49%), and 90.2% had unknown PD-L1 expression. The high percentage of unknown PD-L1 status in the chemotherapy group is primarily due to the fact that many patients were treated in earlier years before PD-L1 testing became standard practice. In the immunotherapy group (N = 36), all patients (100%) demonstrated high PD-L1 expression (>49%). In the chemoimmunotherapy group (N = 83), 65.1% of patients had low PD-L1 expression, while 34.9% showed high expression. Based on PD-L1 expression levels, patients were divided into a high-expression group (⩾50%) and a low-expression group (<50%). The median PFS and OS in the high PD-L1 expression group were 20.1 and 28.8 months, respectively, compared to 16.2 and 24.2 months in the low PD-L1 expression group. Log-rank test results indicated that (Figure 5), compared to the high PD-L1 expression group, the low PD-L1 expression group was associated with worse PFS and OS (HR = 1.62, 95% CI: 1.01–2.58, p = 0.043; HR = 1.67, 95% CI: 1.02–2.74, p = 0.040, respectively).
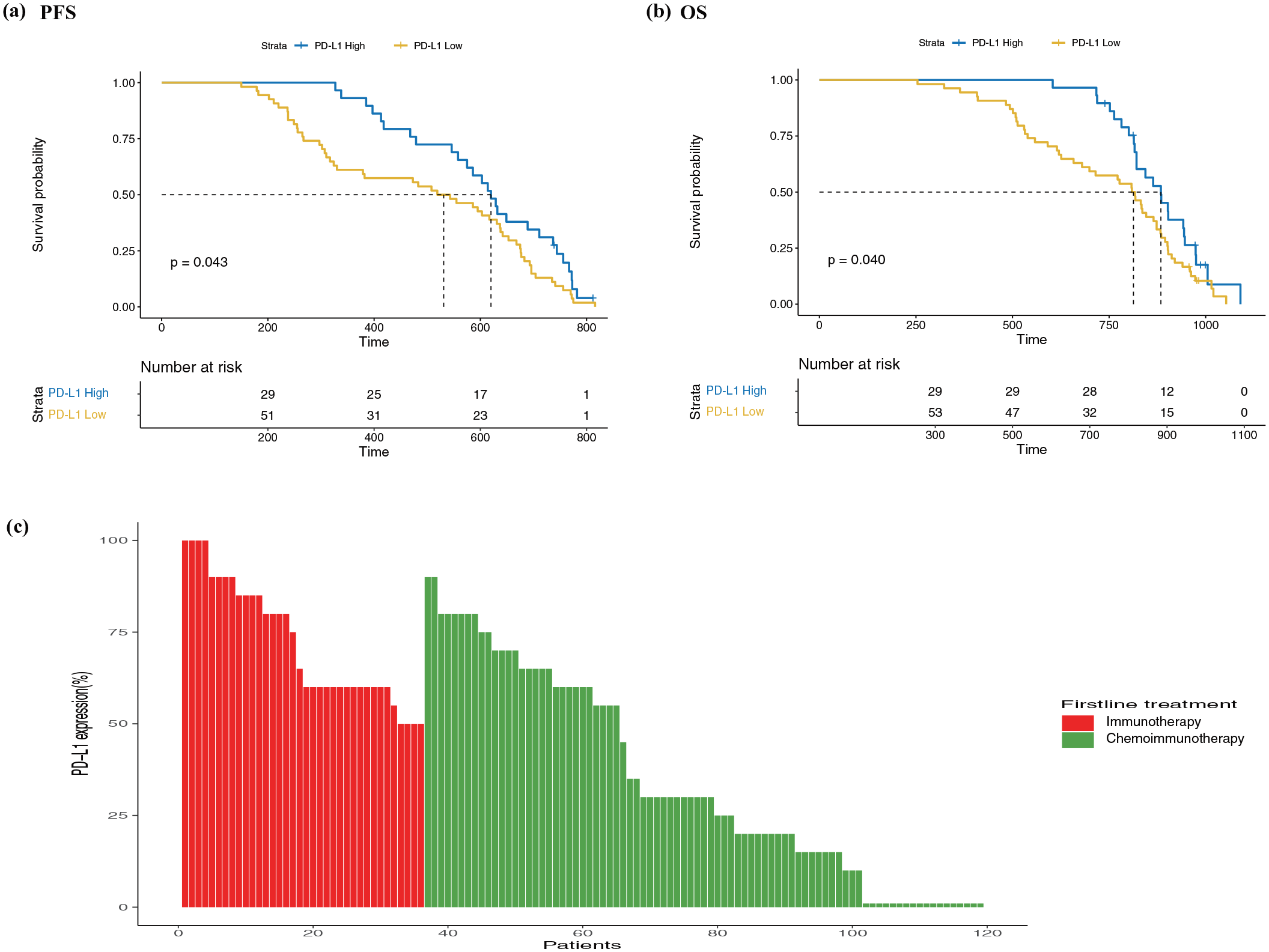
Progression-free survival (a) and overall survival (b) according to the expression of PD-L1 for first-line chemoimmunotherapy. PD-L1 expression levels (c) in first-line immunotherapy and chemoimmunotherapy.
In the second-line treatment cohort (Table 2), none of the patients had negative PD-L1 expression. Among the chemotherapy group (N = 194), 26.3% had low PD-L1 expression, 21.1% had high expression, and 52.6% had unknown PD-L1 status. Similarly, the high percentage of unknown PD-L1 expression in the chemotherapy group is attributed to the fact that many of these patients were treated in earlier years. In the immunotherapy group (N = 26), all patients (100%) exhibited high PD-L1 expression. In the chemoimmunotherapy group (N = 28), 14.3% of patients had low PD-L1 expression, 21.4% had high expression, and 64.3% had unknown PD-L1 status.
Baseline STMs and immunotherapy efficacy
Previous studies have confirmed that both baseline and dynamic STMs can predict the efficacy of immunotherapy in patients with NSCLC.25,26
In this study, we combined patients who received first-line immunotherapy and chemoimmunotherapy (Figure 6). Based on the upper limit of normal values for baseline STMs as the cutoff, patients were grouped, revealing that the median PFS and OS were 11.7 and 20.1 months, respectively, in the high baseline STMs group, compared to 18.9 and 27.5 months, respectively, in the low baseline STMs group. Log-rank test results showed that the high baseline STMs group was associated with significantly worse PFS and OS compared to the low baseline STMs group (HR = 4.21, 95% CI: 2.67–6.62, p < 0.001; HR = 3.98, 95% CI: 2.56–6.19, p < 0.001, respectively).
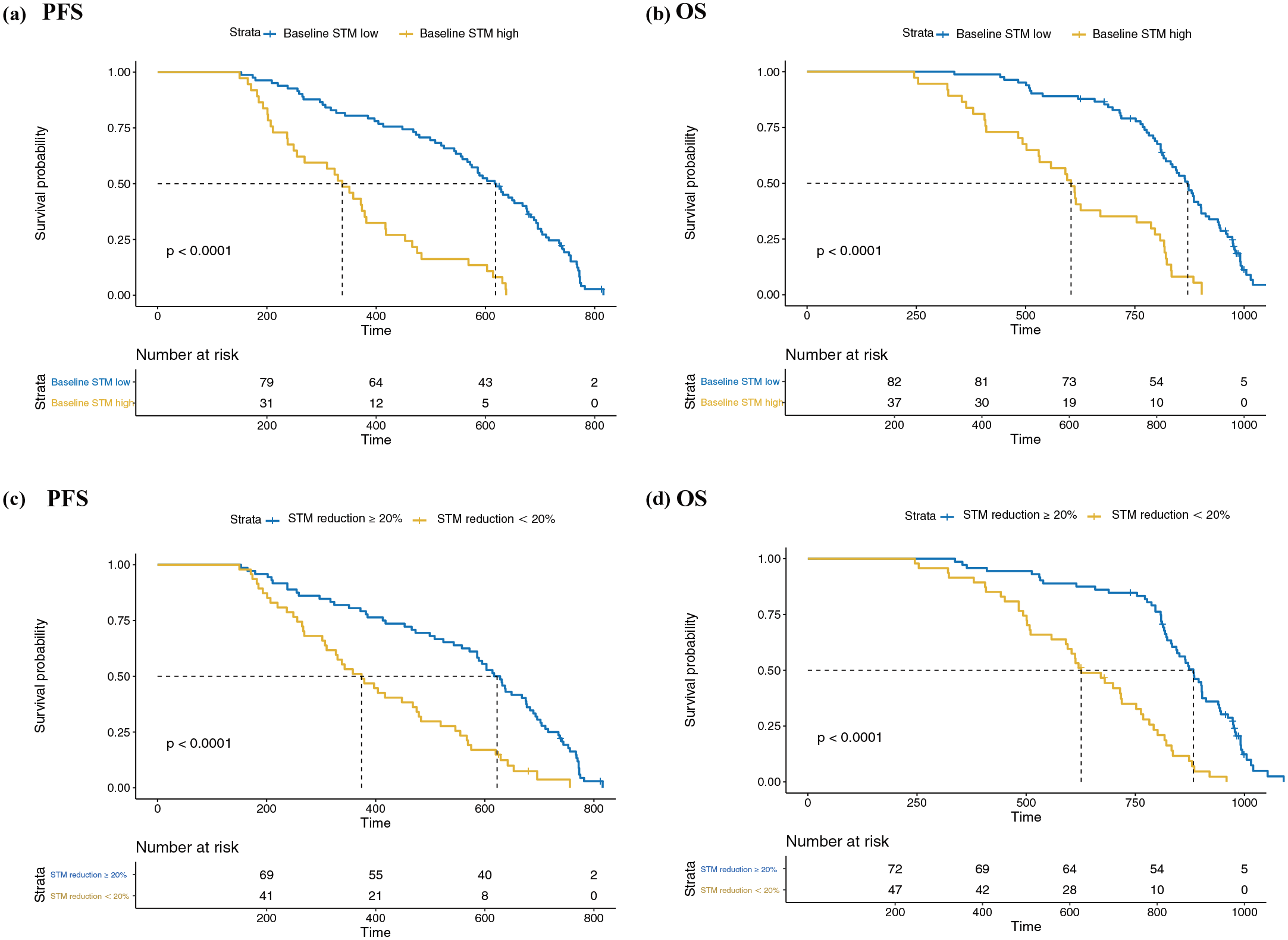
Kaplan–Meier curves of progression-free survival and overall survival for first-line immunotherapy and chemoimmunotherapy. Kaplan–Meier curves were based on baseline STMs (a and b). Kaplan–Meier curves were based on dynamic changes in STMs (c and d).
Similarly, we combined patients who received second-line immunotherapy and second-line chemoimmunotherapy. In this cohort, the median PFS and OS were 3.9 and 8.7 months, respectively, in the high baseline STM group, compared to 5.4 and 15.4 months in the low baseline STM group. Log-rank test results showed that the high baseline STM group was associated with significantly worse PFS and OS compared to the low baseline STM group (HR = 2.61, 95% CI: 1.48–4.59, p < 0.001; HR = 6.45, 95% CI: 3.27–12.73, p < 0.001, respectively).
Dynamics of STMs and immunotherapy efficacy
Similarly, dynamic STMs were analyzed in patients receiving first-line immunotherapy and chemoimmunotherapy (Figure 7). Using a 20% reduction in dynamic STMs as the cutoff, we found that the PFS and OS in the group with a reduction in STMs of ⩽20% were 13.3 and 20.9 months, respectively, compared to 18.8 and 28.0 months in the group with a reduction in STMs of >20%. The log-rank test results indicated that the PFS and OS in the group with an STM reduction of ⩽20% were significantly worse than those in the group with an STM reduction of >20% (HR = 2.70, 95% CI: 1.79–4.08, p < 0.001; HR = 4.08, 95% CI: 2.65–6.28, p < 0.001, respectively).

Kaplan–Meier curves of progression-free survival and overall survival for second-line immunotherapy and chemoimmunotherapy. Kaplan–Meier curves were based on baseline STMs (a and b). Kaplan–Meier curves were based on dynamic changes in STMs (c and d).
A similar pattern was observed in second-line treatment, where the median PFS and OS were 3.9 and 8.7 months, respectively, in the group with a ⩽20% reduction in dynamic STMs, compared to 5.6 and 16.2 months in the group with a >20% reduction. The log-rank test results showed that the PFS and OS in the group with a ⩽20% reduction in dynamic STMs were significantly worse than those in the group with a >20% reduction (HR = 2.64, 95% CI: 1.48–4.70, p < 0.001; HR = 6.45, 95% CI: 3.27–12.73, p < 0.001, respectively).
Multivariate analysis for first-line and second-line treatments
We performed multivariate analyses to identify prognostic factors for both PFS and OS in patients receiving different first-line and second-line treatments. Tables 4 and 5 summarize the results of the univariate and multivariate analyses for first-line and second-line treatments, respectively.
Prognostic factors for PFS and OS in patients with different first-line treatments.

CI, confidence interval; EBV, Epstein–Barr virus; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; OS, overall survival; PD-L1, programmed death ligand 1; PFS, progression-free survival.
Prognostic factors for PFS and OS in patients with different second-line treatments.
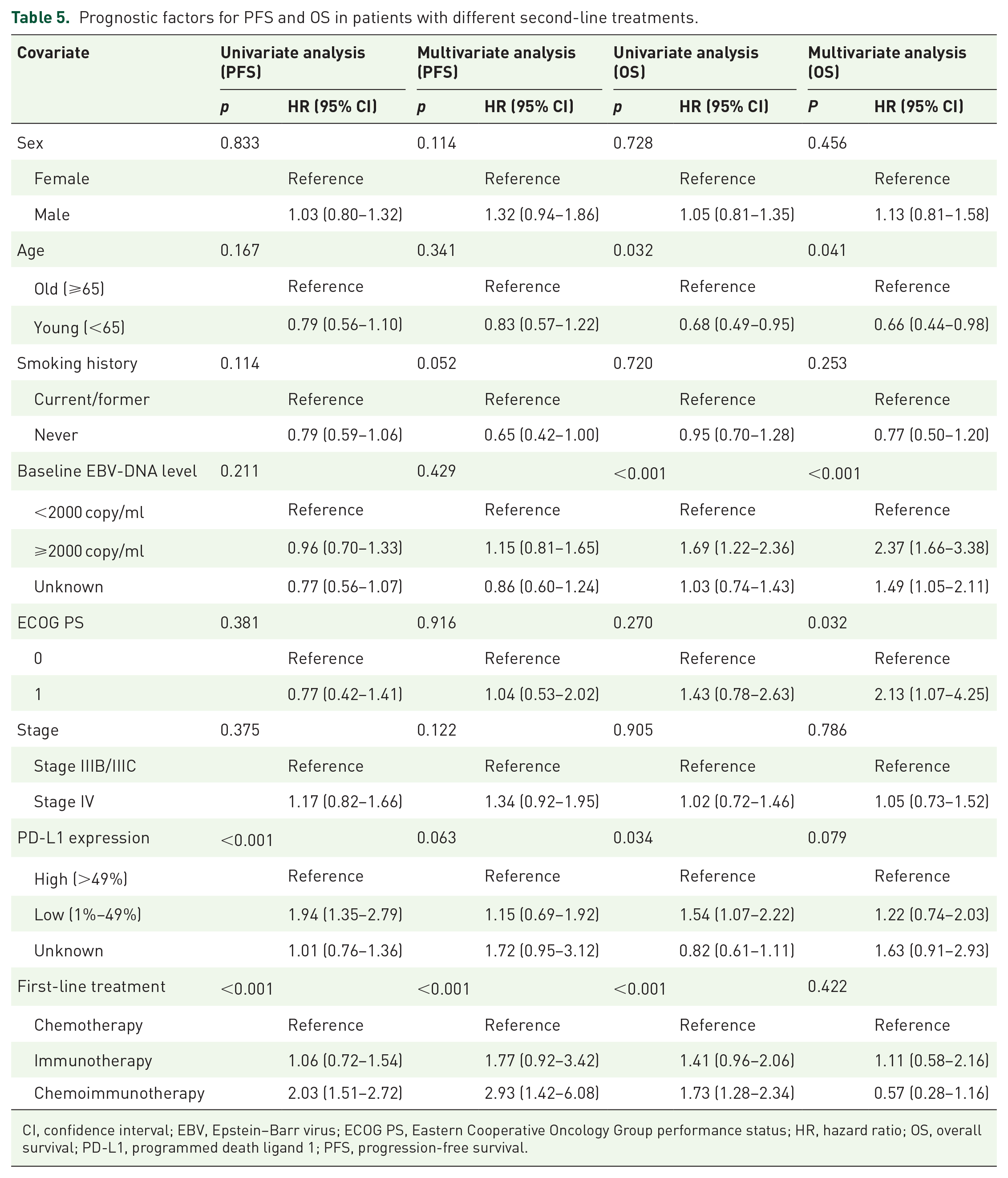
CI, confidence interval; EBV, Epstein–Barr virus; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; OS, overall survival; PD-L1, programmed death ligand 1; PFS, progression-free survival.
For first-line treatment (Table 4), baseline EBV-DNA levels and PD-L1 expression were significant prognostic factors for both PFS and OS. Patients with baseline EBV-DNA levels ⩾2000 copies/ml had significantly worse PFS and OS compared to those with lower levels (multivariate analysis, p < 0.001, HR = 2.14 for PFS; HR = 2.50 for OS). Similarly, patients with unknown baseline EBV-DNA levels also had worse outcomes (multivariate analysis, p < 0.001, HR = 1.87 for PFS; HR = 1.61 for OS). In addition, patients with unknown PD-L1 expression levels had worse PFS and OS compared to those with high PD-L1 expression (>49%; multivariate analysis, p < 0.001, HR = 2.77 for PFS; HR = 1.92 for OS).
For second-line treatment (Table 5), baseline EBV-DNA levels and first-line treatment were identified as significant prognostic factors for OS. Patients with baseline EBV-DNA levels ⩾2000 copies/ml had significantly worse OS compared to those with lower levels (multivariate analysis, p < 0.001, HR = 2.37). In addition, patients with unknown baseline EBV-DNA levels also showed worse OS (multivariate analysis, p < 0.001, HR = 1.49). In terms of PFS, patients who had received chemoimmunotherapy in the first-line treatment had significantly worse PFS compared to those who had received chemotherapy alone (multivariate analysis, p < 0.001, HR = 2.93).
Other significant factors for OS included ECOG PS and age in both first-line and second-line analyses, where younger patients (<65 years) and those with an ECOG PS of 0 had better survival outcomes.
Survival outcomes of immunotherapy versus chemotherapy
To further explore the role of immunotherapy in advanced lymphoepithelioma-like carcinoma (pLELC), we divided patients who received immunotherapy (monotherapy or combination therapy) in either first-line or second-line treatment into the WI (With Immunotherapy) group. Patients who received only chemotherapy in both first-line and second-line treatments were classified into the CO (Chemotherapy Only) group (Figure 8). The results indicated that the WI group showed greater benefits in both PFS and OS compared to the CO group (HR = 0.24, 95% CI: 0.18–0.32, p < 0.001; HR = 0.30, 95% CI: 0.22–0.40, p < 0.001, respectively).
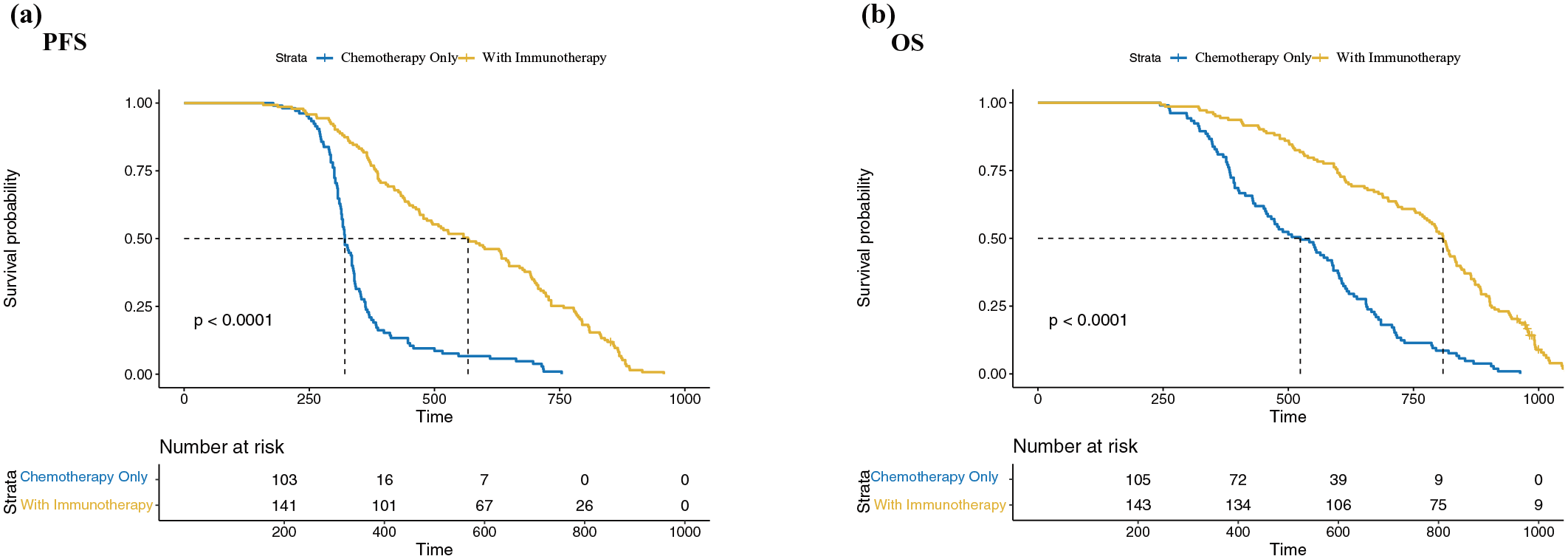
Comparison of progression-free survival (a) and overall survival (b) between the WI group and CO group in advanced pLELC patients.
Discussion
So far, due to the rarity of this disease, there is no standard treatment for advanced pLELC. Our previous studies were the first to demonstrate that the 2-year PFS of chemoimmunotherapy was significantly better than that of chemotherapy, with a median PFS that was 5 months longer. 19 In addition, through a propensity score-matched study, we confirmed that patients with advanced pLELC benefit more from immunotherapy compared to those with advanced squamous cell carcinoma of the lung. 20 Recent studies have also confirmed the efficacy of first-line immunotherapy in advanced pLELC.21,22 Furthermore, Pang et al. 22 included 46 patients who received second or more line immunotherapy, but they did not analyze this population separately. While the role of second-line immunotherapy and chemoimmunotherapy in advanced NSCLC has been established in multiple clinical studies,27–32 its efficacy and safety in advanced pLELC have not yet been studied.
This study’s retrospective design introduces inherent limitations, as treatment decisions were not randomized but rather influenced by various clinical factors, potentially leading to selection bias. Notably, radiotherapy was not included as a standard component of the treatment regimen for patients with stage IIIB/IIIC pLELC. Although some studies have suggested that pLELC is radiosensitive, particularly in early-stage disease, we excluded patients who received curative radiotherapy to avoid confounding the assessment of immunotherapy outcomes in a more homogeneous cohort, as these patients often have different prognoses.33,34 We recognize that this exclusion may narrow the scope of our conclusions. However, this choice reflects the limitations typical of retrospective studies, where treatment paths are often individualized based on clinical judgment rather than randomized assignment, introducing possible selection bias. Currently, we are analyzing data on locally advanced pLELC patients who received curative radiotherapy, with or without concurrent immunotherapy, aiming to offer more comprehensive insights into optimal treatment strategies for this distinct patient population.
Our study updates the data by including a larger population from more centers, further confirming the efficacy and safety of first-line immunotherapy and chemoimmunotherapy in advanced pLELC. In addition, we analyzed patients who failed first-line treatment and found that second-line immunotherapy and chemoimmunotherapy still provide significant benefits in advanced pLELC.
Our results showed that immunotherapy and chemoimmunotherapy significantly prolonged both PFS and OS in patients with pLELC, whether in first-line or second-line treatment. Specifically, in first-line treatment, the median PFS and OS for chemoimmunotherapy were 17.6 and 26.1 months, respectively, outperforming chemotherapy (median PFS of 8.7 months and OS of 19.2 months). These findings are consistent with previous studies and further support the application of immunotherapy and chemoimmunotherapy in pLELC. In second-line treatment, although the ORR was lower than in first-line treatment, chemoimmunotherapy still outperformed chemotherapy alone. The median PFS and OS rates in the chemoimmunotherapy group were 5.1 and 13.5 months, respectively, compared to 3.3 and 8.9 months in the chemotherapy group. These findings suggest that even after initial treatment failure, incorporating immunotherapy or chemoimmunotherapy into subsequent treatment regimens may still provide survival benefits for pLELC patients.
pLELC primarily affects younger, non-smoking individuals living in regions with a high prevalence of EBV.10,35 In our cohort, the median age of patients was 52 years (interquartile range, 46–59), and they were predominantly non-smokers, which aligns with epidemiological data on pLELC. In addition, it is widely recognized that pLELC has a strong association with EBV infection and shares histological characteristics with NPC.11,12 In this study, almost all patients tested positive for EBER (249/252 (98.8%)). These results further confirm the unique role of EBV in the tumorigenesis of pLELC. However, EBV positivity may vary by race and region; Asian patients predominantly test positive for EBV, whereas most non-Asian patients do not.36,37 In addition, some patients in our cohort were tested for EGFR and ALK mutations, all of which were negative, suggesting that common driver mutations may not play a crucial role in pLELC. 12 Furthermore, PD-L1 expression in pLELC patients is higher than in other types of lung cancer patients. 17 These epidemiological and biological characteristics provide evidence for the potential benefits of immunotherapy. Moreover, our findings strongly support the application of immunotherapy and chemoimmunotherapy in this unique cancer subtype, significantly improving patient prognosis.
Previous studies have shown that baseline EBV-DNA levels are associated with the efficacy of chemotherapy or immunotherapy.38–40 Our study indicates that among patients receiving first-line immunotherapy and chemoimmunotherapy, those with baseline EBV-DNA <2000 copies/ml experienced greater PFS and OS benefits, suggesting that antiviral therapy before antitumor treatment may improve the quality of life and survival of these patients.
Several studies have confirmed the correlation between PD-L1 expression levels and the efficacy of immunotherapy in NSCLC. 41 Possibly due to the sample size, previous studies on advanced pLELC did not find a similar phenomenon. Our data indicate that in the chemoimmunotherapy group, patients with high PD-L1 expression had better PFS and OS benefits, suggesting that PD-L1 could be a potential predictor of the benefits of immunotherapy in advanced pLELC patients.
Many studies have previously demonstrated that baseline STMs and dynamic STMs can predict the efficacy of immunotherapy.25,26,42,43 However, no studies have examined the role of STMs in pLELC. Our study suggests that both baseline and dynamic STMs can predict the efficacy of immunotherapy and chemoimmunotherapy. However, unlike our previously published studies,25,26 we did not analyze individual STMs but instead conducted a pooled analysis of the five common STMs in lung cancer, which provides a more comprehensive understanding of the role of tumor markers in pLELC.
There were no cases of Grade 5 Treatment-Related Adverse Event (TRAE) during systematic treatment in our study. Notably, myelosuppression, particularly leukopenia, was the most common grade 3/4 TRAE in patients with advanced pLELC receiving chemoimmunotherapy. Therefore, it is recommended that blood counts be closely monitored when using chemoimmunotherapy in patients with advanced pLELC.
This study has several limitations. First, as a retrospective study, there is an inherent risk of selection bias and information bias. In addition, retrospective studies cannot control for potential confounding factors, which may affect the interpretation of results. Future prospective, randomized controlled trials are needed to validate these findings, thereby improving the quality and credibility of the evidence. Second, this study included various treatment regimens, and the effectiveness of different treatment regimens may vary. Due to the limited sample size, subgroup analyses of the various treatment regimens were not feasible. Third, this study evaluated biomarkers such as PD-L1 expression, EBV-DNA levels, and STMs. However, the detection methods, cutoff selection, and evaluation time points for these biomarkers may impact the results. For example, heterogeneity in PD-L1 expression and differences in detection methods may lead to variability in the results. In addition, the dynamic changes in STMs may be influenced by multiple factors, requiring further investigation into their clinical applicability. Finally, we only detailed the recording of grade 3–4 adverse events; due to incomplete records, mild adverse events were not reported. Therefore, we do not know whether there are specific adverse events related to pLELC.
Conclusion
Our study demonstrates that immunotherapy, particularly when combined with chemotherapy, significantly improves the survival outcomes of patients with advanced pLELC. PD-L1 expression and STMs are important factors in predicting treatment response, providing a foundation for more personalized therapeutic approaches. Our analysis of second-line treatment outcomes provides important insights into the extended utility of immunotherapy in advanced pLELC. Despite potential influences from prior therapies, the second-line data indicate that some patients continue to derive significant PFS and OS benefits. This highlights the need for further research to refine treatment strategies for pLELC in later lines. As our understanding of pLELC continues to deepen, integrating these findings into clinical practice may improve the prognosis of patients with this rare and challenging form of lung cancer.
