Abstract
Background:
For non-small-cell lung cancer (NSCLC) patients who progressed after first-line chemotherapy, immunotherapy targeting programmed cell death (ligand) 1 has shown promising activity. However, the activity is relatively limited in patients harboring epidermal growth factor receptor (EGFR) mutations.
Objectives:
This study aimed to evaluate the efficacy and safety of camrelizumab plus famitinib in previously treated patients with locally advanced and metastatic NSCLC.
Design:
A single-center, single-arm, phase II study.
Methods:
Previously treated patients with locally advanced and metastatic NSCLC were enrolled to receive camrelizumab (200 mg, administered intravenously every 3 weeks) and famitinib (20 mg, administered orally once daily). Patients harboring EGFR mutation genes had received at least one EGFR tyrosine kinase inhibitor and no more than two lines of chemotherapy regimen before the enrollment. The other patients had progressed on first-line chemotherapy with or without immunotherapy before the enrollment. The primary endpoint was the objective response rate (ORR) per RECIST v1.1 by the investigator.
Results:
Our study encompassed 23 NSCLC patients between October 2019 and October 2022. For all patients, the confirmed ORR was 30.4%, and the disease control rate was 95.7%. The median progression-free survival (PFS) was 6.9 months (95% CI: 4.9 months–not reached). The median overall survival (OS) was not reached. 1- and 2-year OS rates were 85.6% (95% CI: 71.8%–100.0%) and 56.8% (95% CI: 37.7%–85.7%). Especially, for the 6 patients with EGFR genetic aberrations, the confirmed ORR was 33.3%, the median PFS was 10.3 months (95% CI: 1.8–18.8 months), and the median OS was 20.3 months (95% CI: 0.8–39.8 months). The most common grade 3 and above treatment-related adverse events were platelet count decreased, white blood cell count decreased, and hypertension. No unexpected adverse events were reported.
Conclusion:
Camrelizumab plus famitinib demonstrated encouraging clinical activity with a manageable safety profile in previously treated patients with locally advanced and metastatic NSCLC. The results warranted further validation.
Trial registration:
Chinese Clinical Trial Registry identifier: ChiCTR1900026641.
Introduction
Immunotherapy has significantly transformed the management of advanced/metastatic non-small-cell lung cancer (NSCLC). Numerous pivotal phase III trials have demonstrated that the incorporation of immune checkpoint inhibitors (ICIs) such as pembrolizumab, nivolumab, atezolizumab, or combinations of ICIs like nivolumab plus ipilimumab, alongside chemotherapy, yields substantial improvements in efficacy compared to chemotherapy alone in this clinical context. 1 ICIs that target the programmed death 1 (PD-1)/PD ligand 1 (PD-L1) pathway have become the established second-line treatment for advanced NSCLC. However, the majority of patients eventually progress during immunotherapy.2–4 Chemo-immunotherapy combinations are associated with improved outcomes, especially in patients with PD-L1 expression less than 50%.5–7 Even with chemo-immunotherapy combinations, a significant proportion of patients do not respond to treatment or respond only for a limited period, with median PFS (mPFS) varying from 4 to 10 months in different trials. 8 Nevertheless, the rapid pace of these advancements has raised a pressing clinical inquiry: with ICIs for NSCLC transitioning effectively from second-line to first-line treatment, what is the optimal course of action for patients who do not respond to ICI-based first-line regimens?
Currently, the identification of oncogenic driver genes also dramatically facilitates the development of targeted therapy for NSCLC. Studies have demonstrated that immunotherapy also significantly improved the survival of NSCLC patients without driver gene mutations, but the benefits for patients with driver gene mutations are limited. 9 EGFR tyrosine kinase inhibitors (TKIs) have revolutionized the management of metastatic EGFR-mutated NSCLC as it has demonstrated a notable response rate in first-line treatment.10–12 However, patients develop disease progression after 9–13 months of treatment with first-generation EGFR TKIs. 13 In a real-world setting, salvage chemotherapy with a platinum-based regimen is the most commonly used regimen, yet the response rate of chemotherapy was low with mPFS of 4–6 months. 14 For these patients, ICIs alone or in combination with chemotherapy did not show better clinical outcomes than standard chemotherapy alone.15–17
Angiogenesis and immunosuppression are intricately interconnected processes with shared regulators.18,19 The effectiveness of ICIs relies on an inflammatory tumor microenvironment (TME) that is characterized by reactivatable T cells, which unleash the pre-existent immune responses. 20 Preclinical evidence suggests that anti-angiogenesis therapies targeting the vascular endothelial growth factor (VEGF) pathway, which can also exert immunosuppressive effects by binding to VEGF receptors present on CD8+ cells, regulatory T-cells, tumor-associated macrophages, and myeloid-derived suppressor cells (MDSCs) within the tumor microenvironment.21,22 In patients with NSCLC, VEGF/VEGFR-targeted therapy increased CD8+ T-cell infiltration and PD-L1 expression in the TME. 23 Thus, combining antiangiogenic TKIs with immunotherapy has been considered an exciting strategy in NSCLC. In advanced NSCLC, bevacizumab was also found to enhance atezolizumab in combination with chemotherapy in patients with metastatic non-squamous NSCLC in the first-line setting. 24 In the ORIENT-31 trial, sintilimab plus bevacizumab biosimilar IBI305 and chemotherapy was generally efficacious and well tolerated in patients with EGFR-mutated NSCLC who had progressed on EGFR TKI. 25 In the HARMONi-A trial, the PD-1/VEGF bispecific antibody ivonescimab (AK112) combined with chemotherapy achieved a significant benefit compared with chemotherapy in mPFS versus chemotherapy alone in patients with advanced NSCLC who progressed on EGFR TKI. 26 In a randomized phase II Lung-MAP S1800A, the combination of pembrolizumab with the VEGF receptor inhibitor ramucirumab demonstrated a survival benefit versus standard of care in patients with advanced NSCLC who had progression on prior ICIs and platinum-based chemotherapy, 27 and the further study PRAGMATICA-Lung is being conducted. 28 Whereas atezolizumab plus cabozantinib compared with docetaxel in patients with metastatic NSCLC previously treated with platinum-based chemotherapy and an anti-PD (L) 1 agent did not meet the primary endpoint of OS. 29 There was no difference in the OS of patients who received sitravatinib with nivolumab compared with docetaxel as a second- or third-line treatment for advanced NSCLC following progression on or after ICI and chemotherapy. 30
Camrelizumab (SHR-1210) is an anti-PD-1 antibody, whose binding sites (CC′ and FG loop of PD-1) with PD-1 are different from pembrolizumab (C′D loop) or nivolumab (BC loop). 31 Camrelizumab combined with chemotherapy has been approved for first-line treatment of NSCLC, esophageal cancer, and nasopharyngeal cancer in China.32–35 Famitinib is a novel multi-targeted TKIs against stem-cell factor receptor (c-kit), VEGFR-2/3, and platelet-derived growth factor receptor β (PDGFR-β), proto-oncogene tyrosine-protein kinase receptor, and FMS-like tyrosine kinase-1/3 receptor. 36 Since these targets are involved in processes such as tumor angiogenesis, proliferation, and immune suppression,37–40 famitinib holds the potential to enhance the antitumor immune response to camrelizumab. 41 Therefore, we conducted this phase II trial to explore the efficacy and safety of camrelizumab plus famitinib in pretreated patients with locally advanced and metastatic NSCLC. The results were presented in accordance with the TREND reporting checklist.
Methods
Study design and patients
The study was a single-arm, phase II clinical trial. Eligible patients were those aged 18–75 years, had histologically or cytologically confirmed unresectable stage IIIB–IV NSCLC, Eastern Cooperative Oncology Group performance status (ECOG PS) 0–2, at least one measurable lesion that met Response Evaluation Criteria in Solid Tumors Version 1.1 (RECIST V1.1), with the adequate function of marrow and major organs and a life expectancy of more than 12 weeks at screening, and with disease progression after at least first-line standard treatment. For patients without driver gene mutation, those who progressed after first-line chemotherapy without or with immunotherapy were eligible. For patients with mutations in EGFR, those who progressed on EGFR-TKIs and no more than two prior lines of systemic therapy were eligible. Exclusion criteria included the following: patients with mutations in EGFR T790M and have not progressed on osimertinib treatment, symptomatic brain metastases, obvious tumor infiltration into the large blood vessels, high risk of fatal bleedings, hypertension and cardiovascular disease that are not well controlled, immunodeficiency and active HBV/HCV infection.
The study protocol was approved by the Ethics Committees of The Fifth Medical Center of PLA General Hospital. All enrolled patients provided written informed consent. The study was carried out in accordance with the principles of the Declaration of Helsinki and the International Conference on Harmonization Good Clinical Practice guidelines. The reporting of this study conforms to the Consolidated Standards of Reporting Trials (CONSORT) guidelines, and the checklist has been provided as Supplemental File 1. 42 The summary of the protocol has been provided as Supplemental File 2.
Study treatment
All patients received camrelizumab (200 mg, administered intravenously on day 1 of each 3-week treatment cycle) and famitinib (20 mg, administered orally once daily) until progressive disease assessed by investigators per RECIST v1.1, unacceptable toxicity, death, up to 2 years, or withdrawal of consent. The patients could continue treatment beyond disease progression based on the investigator’s judgment. Dose modification of camrelizumab was not allowed and dose interruptions of camrelizumab were permitted for up to 12 weeks. Dose interruption, dose reduction, and administration schedule modifications (14 days on, 7 days off) of famitinib were allowed.
Endpoints and assessments
The primary endpoint was ORR per RECIST version 1.1, defined as the proportion of patients achieving a confirmed complete response (CR) or partial response (PR) based on investigators’ assessment. The secondary endpoints included disease control rate (DCR, defined as proportion of patients with CR or PR, or SD (Stable disease)), time to response (TTR, defined as the time from the first dose to first documented objective response), duration of response (DoR, defined as the time from first documented objective response to disease progression or death from any cause), progression-free survival (PFS, defined as time from first dose of study treatment to disease progression per RECIST version 1.1 or death, whichever occurred first), overall survival (OS, defined as time from first dose of study treatment to death due to any cause) and safety. Investigator-assessed tumor response was performed every 2 cycles. After treatment discontinuation, patients were followed every 2 months to collect survival.
Adverse events (AEs) were collected and coded according to the Medical Dictionary for Regulatory Activities Version 23.0, and graded in accordance with the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, and the causality to study drugs was recorded. AEs and serious AEs were monitored until 30 days after the last administration.
Statistical analysis
To minimize the sample size if the study treatment was deemed ineffective, Simon’s two-stage design was used. The null hypothesis was set at 14%, based on historical data from the OAK study, in which the ORR was 14% for previously treated NSCLC with atezolizumab. 15 In the first stage, 12 patients would be accrued. If no response was observed in these 12 patients, the study would be stopped. Otherwise, 11 additional patients would be accrued for a total of 23. The null hypothesis would be rejected if 6 or more responses were observed in 23 patients. This design yields a type I error rate of 0.1 and a power of 80% when the true response rate was 33%. The study did not have pre-specified subgroups, and the sample size and statistical power were based on people with locally advanced and metastatic NSCLC who progressed after standard treatment.
The full analysis set included all patients who received at least one dose of study treatment. The safety analysis set included all patients who received at least one dose of study treatment and underwent safety assessment after treatment. The ORR and DCR and the corresponding 95% CIs were calculated using the Clopper–Pearson method. TTR was summarized descriptively for responding patients only. The Kaplan–Meier method was used to estimate the PFS and OS, and the corresponding 95% CIs were calculated with the Brookmeyer and Crowley method. All statistical analyses were performed with SAS software version 9.4 (SAS Institute Inc. Cary, NC, USA).
Results
Patient characteristics and disposition
Between October 2019 and October 2022, a total of 23 previously treated patients were enrolled and received the combination treatment of camrelizumab (200 mg every 3 weeks) plus famitinib 20 mg once daily. The baseline characteristics of patients are shown in Table 1. The median line of the previous treatment was 1. The median age of enrolled patients was 63 years (range, 41–73). All patients had an ECOG PS of 1. Eighteen (78.3%) patients had stage IV disease and 5 (21.7%) patients had brain metastases. In addition, 8 (34.8%) patients had PD-L1 tumor proportion score (TPS) ⩾1%, and 6 (26.1%) patients had PD-L1 TPS <1%. The majority of patients (n = 17, 73.9%) had received chemotherapy. Seven (30.4%) patients had received immunotherapy and EGFR TKIs, respectively. Six (26.1%) patients were harboring EGFR genetic aberration.
Baseline characteristics.

ALK, anaplastic lymphoma kinase; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; ICI, immune checkpoint inhibitor; PD-L1, programmed death-ligand 1; TKI, tyrosine kinase inhibitor; TPS, tumor proportion score.
At the data cutoff (March 25, 2024), the median follow-up time was 22.8 months (range, 2.8–53.8 months). The reasons for study discontinuation were disease progression (n = 15, 65.2%), AEs (n = 3, 13%), and end of treatment per study design (n = 5, 13%).
Efficacy
At the time of data cutoff, 23 patients were included in the full analysis set. Figure 1(a) presents the best percent of change from baseline in the target lesion size, 19 (82.6%) patients showed decreased total tumor burden. As shown in Table 2, 9 patients (39.1%) achieved an objective response, including 2 unconfirmed PR patients, 13 (56.5%) patients had SD as their best response, and 1 (4.3%) patient had progressive disease (PD). Thus, the confirmed ORR assessed was 30.4% (95% CI: 13.2%–52.9%) and DCR was 95.7% (95% CI: 78.1%–99.9%). In addition, for driver-negative patients, ORR and DCR were 29.4% (95% CI: 10.3%–56.0%) and 94.1% (95% CI: 71.3%–99.9%), respectively; for those with EGFR mutation, ORR and DCR were slightly increased by 33.3% (95% CI: 4.3%–77.7%) and 100.0% (95% CI: 54.1%–100%), respectively. Moreover, response occurred at a median of 1.41 months (range, 1.28–2.93 months; Table 2), and the median duration of response (DoR) was NR (range, 4.34–NR months; Table 2). Treatment duration and tumor response are shown in Figure 1(b), most patients showed improvement in tumor burden during the initial assessment and the decreased tumor burden was sustained over several assessments.

Clinical activity of camrelizumab plus famitinib in previously treated advanced non-small-cell lung cancer. (a) Best percentage changes in the sum of the diameters of target lesion from baseline. (b) Treatment duration and tumor response in all patients.
Investigator-assessed best overall tumor response and PFS.

Among the 15 patients with SD, including the two unconfirmed PR patients.
BOR, best overall response; CI, confidence interval; CR, complete response; DCR, disease control rate; DOR, duration of response; NR, not reached; ORR, objective response rate; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease; TTR, time to response.
A total of 14 patients had PFS events and the median PFS was 6.9 months (95% CI: 5.2 months–NR; Figure 2(a)). For those without or with EGFR mutation, the median PFS rates were 6.9 months (95% CI: 4.9 months–NR) and 10.3 months (95% CI: 4.6 months–NR), respectively (Figure 2(b)). As of the data cutoff, the median OS of all patients remained immature (Figure 3(a)). 1- and 2-year OS rates were 85.6% (95% CI: 71.8%–100.0%) and 56.8% (95% CI: 37.7%–85.7%). Moreover, the median OS was 20.3 months (95% CI: 0.8–39.8 months, Figure 3(b)) in EGFR mutation patients. Longer follow-up on OS data was needed, and currently, the collection of long-term follow-up data for patients is still continuing.
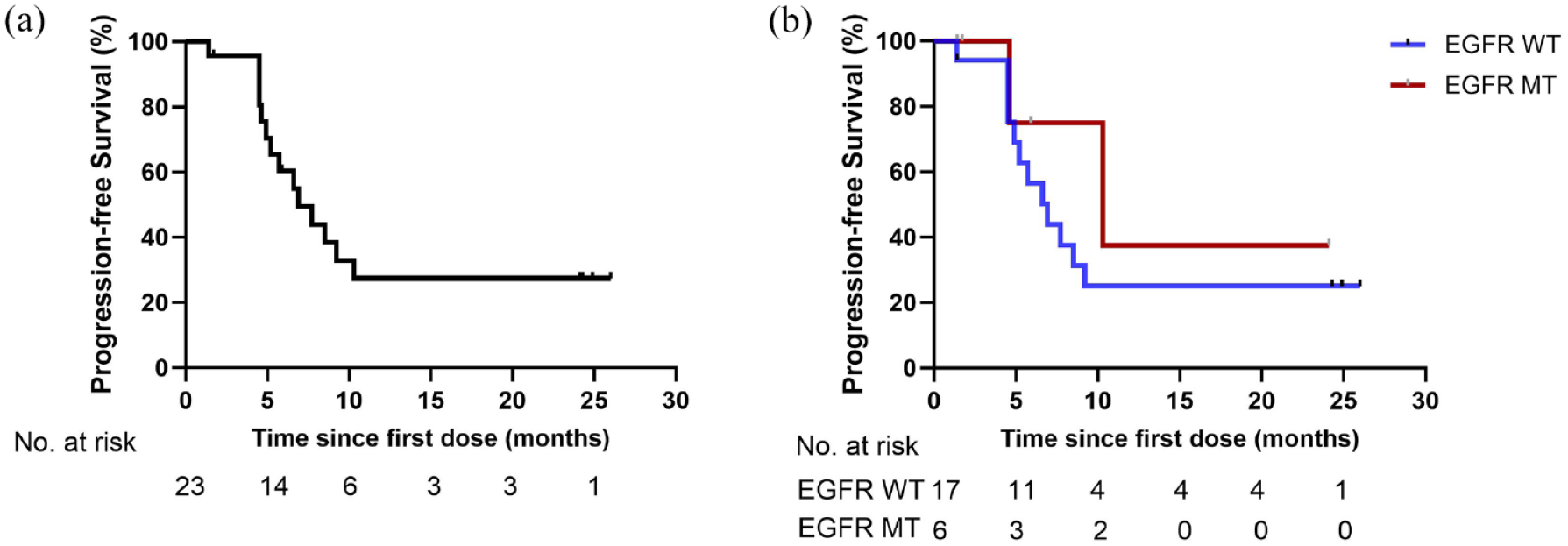
PFS in previously treated advanced non-small-cell lung cancer (n = 23). (a) Kaplan–Meier curves for PFS; (b) Kaplan–Meier curves for PFS of patients without (EGFR WT) or with EGFR mutation (EGFR MT).

OS in previously treated advanced non-small-cell lung cancer (n = 23). (a) Kaplan–Meier curves for OS; (b) Kaplan–Meier curves for OS of patients without (EGFR WT) or with EGFR mutation (EGFR MT).
Exposure and safety
All 23 patients were evaluable for safety analysis. The median duration of camrelizumab exposure was 6.6 months (range, 0.7–27.6 months), and the median duration of famitinib exposure was 6.6 months (range, 0.6–27.6 months). Among the 23 patients, 3 (13%) patients discontinued any study treatment due to treatment-related adverse events (TRAEs), 6 (26.1%) had camrelizumab dose interruption because of TRAEs, and 7 (30.4%) had famitinib dose interruption caused by TRAEs (Table 3).
Summary of TRAEs.

SAE, serious adverse event; TRAE, treatment-related adverse event.
As illustrated in Table 4, 22 (95.7%) patients had at least grade 1 TRAEs, and the most commonly reported TRAEs of any grade were proteinuria (78.3%), hypercholesterolemia (65.2%), electrocardiogram T wave abnormal (60.9%), platelet count decreased (56.5%), and anemia (52.2%). Grade 3 or higher TRAEs occurred in 39.1% (9/23) of the patients, including platelet count decreased (8.7%), white blood cell count decreased (8.7%), and hypertension (8.7%). Treatment-related serious adverse events (SAEs) were reported in 3 (14.3%) patients, which were hemoptysis (n = 1 (4.3%)), myasthenia gravis (n = 1 (4.3%)), and peripheral neuropathy (n = 1 (4.3%)). Grade ⩾3 immune-related AEs (irAEs) occurred in 6 (26.1%) patients, with platelet count decreased (n = 2 (9.5%)) and white blood cell count decreased (n = 2 (9.5%)) as the most common events. No unexpected AEs were observed.
TRAEs that occurred in ⩾10% of patients.

TRAE, treatment-related adverse event.
Discussion
To our knowledge, this is the first phase II study to evaluate the combination therapy of camrelizumab plus famitinib in pretreated patients with locally advanced and metastatic NSCLC. And camrelizumab (200 mg every 3 weeks) plus famitinib (20 mg qd) observed significant clinical efficacy with a confirmed ORR of 30.4%. The median PFS was 6.9 months. Especially, patients with EGFR mutation resulted in a relatively long mPFS of 10.3 months. Concurrently, the TRAEs were also manageable and no new safety signals were observed.
For patients who progressed after first-line chemotherapy, immunotherapy has dramatically changed the landscape of treatment. Compared with docetaxel, nivolumab has significantly improved OS in both squamous NSCLC (9.2 months vs 6.0 months, HR = 0.59, p < 0.001)43,44 and non-squamous NSCLC (12.2 vs 9.4 months, HR = 0.73, p = 0.002).44,45 For advanced NSCLC patients with PD-L1-positive (TPS ⩾1%) tumors, pembrolizumab compared with docetaxel has shown a median OS of 10.4 months versus 8.5 months (HR = 0.71).16,46 The OAK trial revealed that atezolizumab improved OS (13.8 vs 9.6 months, HR = 0.73, p = 0.003) over that with docetaxel in previously treated NSCLC. 15 However, despite the considerable progress made with checkpoint inhibitor-based immunotherapies, including pembrolizumab, the majority of patients with metastatic NSCLC will still die of the disease. Moreover, the efficacy is relatively limited in certain types of patients, such as those progressing after first-line immunotherapy and those progressing after TKI treatment with EGFR mutation.
One explanation for this is the presence of other molecules in the tumor environment, making “immune-cold” tumors resistant to PD-1/PD-L1 antibodies. 22 Preliminary clinical data in NSCLC indicate that the synergistic effects of combining anti-angiogenesis TKIs with PD-1/PD-L1 antibodies may enhance tumor responses by modulating the tumor microenvironment. 47 Thus, the combination therapy of anti-angiogenesis TKIs and ICIs has shown promising results.
Lenvatinib plus pembrolizumab demonstrated a manageable safety profile and promising antitumor activity in many cancers such as advanced renal cell carcinoma, endometrial cancer, and NSCLC. 48 This combination therapy had clinical activity with pretreated NSCLC, and ORR was 33% (7/21; 95% CI: 14.6–57.0), the median PFS was 5.9 months at data cutoff. 49 The COSMIC-021 trial evaluated the efficacy of cabozantinib plus atezolizumab (CA) or cabozantinib (C) alone in advanced NSCLC previously treated with an immune checkpoint inhibitor, the data showed that the median of OS (13.8 vs 9.4 months) and PFS (4.5 vs 3.4 months) were both longer in cohort CA compared to cohort C. In addition, ORR was also improved a lot (19% vs 6%). 50 Similarly, our study also demonstrated significant clinical efficacy, with a confirmed ORR of 30.4% and a median PFS of 6.9 months. Therefore, combining multiple studies, it has been shown that lenvatinib, cabozantinib, and famitinib exhibit antiangiogenic and antiproliferative properties by inhibiting signaling pathways of the VEGF receptor and fibroblast growth factor receptor. This mechanism potentially decreases the presence of intratumoral regulatory T cells and impedes the survival and migration of tumor-associated macrophages within tumors. 48
A meta-analysis involving Keynote-010, CheckMate-057, OAK, and POPLAR trials demonstrated limited benefits in previously treated EGFR-mutant NSCLC patients than in the wild-type population. 51 A phase II trial (NCT02879994) evaluated pembrolizumab in TKI-naive patients with EGFR mutation-positive, advanced NSCLC, and PD-L1-positive (TPS ⩾1%) tumors. Although 73% of patients had PD-L1 expression ⩾50%, no patient had an objective response. 52 IMpower 150 trial showed that the addition of bevacizumab to atezolizumab and chemotherapy as first-line therapy demonstrated efficacy in the subgroup of patients with sensitive EGFR mutations. 53 Furthermore, the Orient-31 study of sintilimab in combination with IBI305 and chemotherapy, as well as the HARMONi-A study of PD-1/VEGF bispecific antibody ivonescimab in combination with chemotherapy, demonstrated an increase in PFS and response rates in EGFR-TKI-resistant populations, especially in T790M-negative patients, emphasizing the potentially critical role of anti-angiogenesis in EGFR-TKI-treated NSCLC.25,26 Recently, amivantamab plus chemotherapy has shown promising results in osimertinib-resistant populations. 54 In our present study, an ORR of 33.3% and a mPFS of 10.3 months were observed in patients with EGFR mutation. Despite potential issues in cross-trial comparisons, the median PFS with our combination regimen achieved a longer value compared with that of amivantamab plus chemotherapy (6.3 months). 54 These results supported that the combination of ICIs and anti-angiogenic inhibitors might improve the tumor immune microenvironment, which confers more antitumor activity than anti-PD-1 monotherapy. Therefore, further trials are warranted to confirm this conclusion with a larger patient population.
Ivonescimab significantly improved PFS when compared to pembrolizumab as first-line therapy in patients with advanced NSCLC and PD-L1 TPS ⩾1% in the phase III HARMONi-2 trial. 55 By contrast, lenvatinib plus pembrolizumab did not have a favorable benefit–risk profile versus placebo plus pembrolizumab as shown in the LEAP-007 trial. 56 In terms of safety, the incidence rate of grade 3 or higher TRAEs was 29.4%, treatment-related SAE was 20.8%, and TRAEs leading to treatment discontinuation was 1.5% in the HARMONi-2 study. 55 But in the Leap-007 study, the observed grade 3 or higher TRAEs was 57.9%, and treatment discontinuation due to TRAEs is 27.5% for lenvatinib and 14.6% for pembrolizumab. 56 The high rate of treatment discontinuation in LEAP-007 compared to HARMONi-2 may be the reason for the limited efficacy. In this phase II study, the adverse effect profile of camrelizumab plus famitinib was similar to that of ivonescimab in the HARMONi-2 study, but the incidence of hematologic toxicity was higher, and no new safety signals were identified in this study. Most of the TRAEs were grade 1–2, the incidence of grade 3 and higher TRAE was 39.1%, and the incidence of severe TRAE was 13%, importantly, the observed treatment discontinuation due to TRAEs is 4.3% for camrelizumab and 4.3% for famitinib. Surprisingly, reactive cutaneous capillary endothelial proliferation (RCCEP), a clinically controllable and self-limiting TRAE induced by camrelizumab, occurred in only one (4.8%) patient, with no grade 3 or higher events reported. This finding suggested that the mechanism of RCCEP might be correlated with the angiogenesis signaling pathway. 57 Our findings indicated that the combination of camrelizumab plus famitinib has an acceptable safety profile.
There were several limitations to our study. First, this trial was a single-arm phase II study without a control arm. Second, the sample size was restricted and we did not have a pre-specified subgroup analysis, leading to challenges in interpreting these findings, especially in pre-treated patients with EGFR mutations. Third, PD-L1 expression status was unknown in more than 1/3 of patients because providing a tissue sample was not a mandatory requirement for enrollment. Therefore, the identification of predictive biomarkers to select suitable candidates for anti-angiogenic drug and immunotherapy combinations still needs to be addressed. Fourth, due to the outbreak of coronavirus disease 2019 (COVID-19), the recruitment of patients was affected. Lastly, the OS was not reached by the data cutoff date, necessitating a longer follow-up period for reporting survival outcomes, particularly for patients receiving immunotherapy.
Conclusion
In conclusion, our phase II trial demonstrated that camrelizumab plus famitinib showed promising antitumor activity and acceptable safety profile in previously treated patients with locally advanced and metastatic NSCLC, especially with EGFR mutations. The results warranted further validation in a randomized trial.
Supplemental Material
sj-doc-2-tam-10.1177_17588359241311058 – Supplemental material for Efficacy and safety of camrelizumab plus famitinib in patients with previously treated non-small-cell lung cancer: a single-arm, phase II trial
Supplemental material, sj-doc-2-tam-10.1177_17588359241311058 for Efficacy and safety of camrelizumab plus famitinib in patients with previously treated non-small-cell lung cancer: a single-arm, phase II trial by Ming Gao, Xia Zhang, Huan Yan, Yan Zhao, Fang Yuan, Decong Sun, Xuejiao Yang, Yanfang Ju, Lijie Wang, Haitao Tao, Luyuan Tian, Changhong Zhao, Junxun Ma, Yi Hu and Zhefeng Liu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-1-tam-10.1177_17588359241311058 – Supplemental material for Efficacy and safety of camrelizumab plus famitinib in patients with previously treated non-small-cell lung cancer: a single-arm, phase II trial
Supplemental material, sj-docx-1-tam-10.1177_17588359241311058 for Efficacy and safety of camrelizumab plus famitinib in patients with previously treated non-small-cell lung cancer: a single-arm, phase II trial by Ming Gao, Xia Zhang, Huan Yan, Yan Zhao, Fang Yuan, Decong Sun, Xuejiao Yang, Yanfang Ju, Lijie Wang, Haitao Tao, Luyuan Tian, Changhong Zhao, Junxun Ma, Yi Hu and Zhefeng Liu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
