Abstract
Background:
Due to the rarity of sinonasal squamous cell carcinoma (SNSCC), the distribution patterns of lymph node metastasis (LNM), the relationship between LNM and prognosis, and the optimal treatment of LNM lack sufficient evidence-based support.
Objectives:
To investigate the patterns of LNM in SNSCC and evaluate the impact of LNM on prognosis.
Design:
This was a retrospective cohort study.
Methods:
The medical records of 441 patients with SNSCC between 2009 and 2022 in one institution were retrospectively reviewed. We assessed the incidence, the distribution of LNM, and the relationship between LNM and long-term survival.
Results:
Seventy-three out of 441 patients (16.6%) presented LNM initially. Among the 73 patients, 34 patients (46.6%) had LNM in the region of ipsilateral level II; 22 patients (30.1%) had positive retropharyngeal lymph nodes; 20 patients (27.4%) had LNM in the region of ipsilateral level Ib; and nine patients (12.3%) had evidence of parotid LNM. Poor differentiation (p = 0.001), nasal cavity (p = 0.018), skin involvement (p = 0.036), and nasopharynx involvement (p = 0.009) were the risk factors for LNM. In the univariate and multivariate analyses, the overall survival (p = 0.25), progression-free survival (p = 0.22), regional failure-free survival (p = 0.20), and distant metastasis-free survival (p = 0.14) rates were not significantly decreased by the LNM. After the propensity score matching, LNM was still not correlated with poor long-term survival.
Conclusions:
The incidence of retropharyngeal and parotid LNM was higher than in previous studies. At initial diagnosis, the risk factors for LNM were identified, and LNM was not associated with poor survival outcomes.
Keywords
Introduction
Sinonasal squamous cell carcinoma (SNSCC) is a complex and relatively rare malignancy, representing only 3% of all head and neck cancers. 1 Nonetheless, squamous cell carcinoma (SCC) is still the most common malignancy of the sinonasal cavity, accounting for 50%–60% of cases. 2 Overall, outcomes for SNSCC have remained poor, with a 5-year overall survival (OS) rate of around 50% in most studies. 3 Due to the rarity of SNSCC, the patterns of lymph node metastasis (LNM) lack sufficient evidence-based support. It has been found that 13.2% of patients (854/6448) had clinical evidence of cervical LNMs in a review of 6448 primary SNSCC cases, and the incidence of bilateral or contralateral cervical LNM was 5.6%. 4 19.3% of LNM occurred in paranasal sinuses (maxillary sinus, ethmoid sinus, sphenoid sinus, frontal sinus). Regarding the site, the maxillary sinus had the highest rate of lymphatic metastasis, reaching 21.0% and approximately 7.9% in the nasal cavity. There is no system analysis on patterns of LNMs, including the incidence of lymph nodes in varied levels, the contralateral neck, and the retrospective space. Knowledge of patterns of cervical LNM is required to determine the need for elective neck treatment.
LNM at initial diagnosis was significantly and independently associated with poor survival (p = 0.0012). 5 The treatment of SNSCC with LNM was achieved in a consensus, including neck dissection and adjuvant radiotherapy. However, for patients with cN0, the treatment of the neck remains controversial. Lee et al. 6 found that elective neck therapy had no benefit on survival or node failure in SNSCC patients regardless of the neck treatment modality. Crawford et al. 7 found no statistically significant difference in OS between patients who underwent elective neck dissection and those who did not after analyzing 1220 patients with SNSCC. No evidence exists to guide the optimal management, and the prophylactic neck treatment remains controversial.
The incidence of LNM in SNSCC, the distribution patterns of LNM, the relationship between LNM and prognosis, and the optimal treatment of LNM are still the main attractive clinical topics. The researchers have been investigating the issues; however, they have not been well established with big data till now. A thorough understanding of the incidence and the patterns of LNM may aid in predicting the prognosis and selecting the optimal treatment modality. To better solve the puzzles, we retrospectively collected data from one institution to analyze the characters and predictors of LNM in SNSCC in a large population treated with curative intent.
Materials and methods
Patients
This retrospective study examined the records of consecutive patients with primary SNSCC who underwent treatment at Eye & ENT Hospital, Fudan University, between January 2009 and January 2022. Patients with other malignant tumors, evidence of distant metastasis, and palliative treatment were excluded. This study received ethical approval from the Ethical Committee of Eye & ENT Hospital, Fudan University.
None of these patients had undergone lymph node biopsy. The cervical LNM was confirmed at our institution by two head and neck radiologists. Patient characteristics, including age, sex, primary tumor site, and clinical stage, were collected retrospectively from patients’ records. Pathological findings and therapeutic intervention were identified and recorded. The location of LNM was categorized according to the cervical regions, as described by the multi-center consensus guidelines. 8
Assessment of lymph node involvement
Clinical lymph node assessment was based on the evaluation of pretreatment CT/MRI examinations and/or PET-CT scans. No histopathological data were obtained during patients’ treatment, as no dissection of the retropharyngeal space was performed. The criteria of clinical LNM were as follows: Short-axis diameter in axial plane ⩾10 mm, ⩾5 mm in lateral retropharyngeal group and parotid lymph node; evidence of necrosis/cystic areas or extracapsular invasion; a cluster of three or more borderline nodes, each ⩾8 mm in short-axis diameter; long-to-short axis ratio <2; and intense 18F-FDG uptake in PET-CT. 9
Treatment
All patients with lymph node metastases were preferred to be treated with chemoradiotherapy or radiotherapy. Neck dissections were performed in patients with residual lymph nodes or disease recurrence. Radiotherapy was administered as intensity-modulated radiotherapy in 256 patients, 3D-conformal radiotherapy in 168 patients, and volumetric intensity-modulated arc therapy in 17 patients. The average prescribed dose for lymph node was 64–70 Gy with 2.0–2.2 Gy/fractions, and the elective nodal irradiation of the neck was delivered with a dose of 50–60 Gy given with 1.8–2.0 Gy/fractions.
Induction chemotherapy (IC), concurrent chemotherapy (CCRT), and their combination were the main chemotherapy modes. The protocols of IC, including TPF (docetaxel + cisplatin + 5-FU) and TP (docetaxel + cisplatin), PF (cisplatin + 5-FU), and GP (gemcitabine + cisplatin), were also commonly used. Cisplatin was administered as concurrent therapy at 75 mg/m2 every 3 weeks. Alternatively, other regimes were used, including nedaplatin or cisplatin and fluorouracil in a minority of cases.
Statistics
Statistical analyses were performed using IBM SPSS version 26.0 (IBM, Armonk, NY, USA), GraphPad VR Prism 8.0 (GraphPad Software Inc., La Jolla, CA, USA), and R software (version 3.5.0 for Windows; Bell Laboratories, New York, United States). Kaplan–Meier analysis was used to determine OS, progression-free survival (PFS), regional-failure-free survival (RFS), and distant metastasis-free survival (DMFS). Categorical variables were compared using the chi-square test or Fisher’s exact test. Logistic regression analysis was used to investigate the risk factors causing LNM. Univariate analysis for OS and RFS was done using the log-rank test, and we fitted a multivariable model based on the selected risk factors on univariate analysis. Cox proportional hazards regression analysis was used to calculate the hazard ratio (HR), confidence intervals (CI), and p-values. Two-tailed p < 0.05 was statistically significant for tests.
Results
Demographic data
Table 1 summarizes the disease characteristics of patients with and without lymph node metastases. Patients with cervical lymph nodes were identified after reviewing radiology images and radiotherapy plans. The typical presentations of lymph node metastases are shown in Supplemental Figure 1.
Patients’ clinical characteristics (N = 441).

LN, lymph node.
At the initial presentation, 73 out of 441 patients (16.6%) presented LNM. Among the 73 patients with LNM, the primary tumor site was the nasal cavity in 38 patients (8.6%, 38/441), maxillary sinus in 24 patients (5.4%, 24/441), ethmoid sinus in 9 patients (2.0%, 9/441), and other sites in 2 patients (0.45%, 2/441). There was no difference in incidence among the primary tumor sites (χ2 = 4.48, p = 0.21). Of these patients, 41 were with cN1 disease, 16 had cN2b disease, 15 had cN2c disease, and 1 had cN2a disease. Anecdotally, LNM was more common in patients with ⩾60 years (10.9% vs 5.7%; χ2 = 4.01, p = 0.045). Compared with the T1–3 stage (11.2%, 15/134), a higher rate of cervical node metastasis (18.9%, 58/307) occurred in the T4 stage (χ2 = 4.00, p = 0.045). Lymph node metastases were more common in poorly differentiated cases (p = 0.02), and there was no correlation between LNM and other pathological features.
During the follow-up, 24 out of 441 patients (5.4%) were affected with regional recurrence. Among the 73 patients with LNM at diagnosis, 8 patients (11.0%) had regional disease recurrence; while 16 patients (4.3%) out of the 368 patients with cN0 disease experienced regional recurrence (p = 0.042). A total of 65 patients developed distant metastasis, 18 (24.7%) of the 73 patients with LNM at diagnosis, and 47 (12.8%) of the 368 patients with N0 disease (χ2 = 6.85, p = 0.009). Hence, patients with initial cervical LNM were more likely to develop distant metastases.
Regional patterns of spread
Table 2 shows the level distribution of lymph node metastases. The ipsilateral parotid lymph nodes were involved in nine patients (12.3%, 9/73), two patients (2.7%, 2/73) had ipsilateral level III disease, and one patient (1.4%, 1/73) had ipsilateral level IV disease. Metastasis to the contralateral lymph nodes was present in three patients (4.1%, 3/73; two with level II disease and one with level Ib disease), and all the primary tumors crossed the midline. LNM in bilateral level II region was found in 11 patients (15.1%, 11/73), a bilateral retropharyngeal region in five patients (6.8%, 5/73), a bilateral level Ib region in two patients (2.7%, 2/73), a bilateral level III region in two patents (2.7%, 2/73), and a bilateral level IV region in one patient (1.4%, 1/73).
Distribution of lymph node metastases (N = 73).

According to the primary tumor sites, the percentage of cervical LNM in the regions of levels II, VIIA, Ib, and VIII were 27.4%, 21.9%, 16.4%, and 2.7%, respectively, in the nasal cavity; 6.8%, 5.5%, 4.1%, and 2.7% in the ethmoid sinus; 20.5%, 9.6%, 11.0%, and 5.5% in the maxillary sinus (Supplemental Table 1). The relative incidence of LNM in various cervical regions in patients with different primary tumor sites is presented in Figure 1. Nine patients (12.3%) showed level VIII LNMs, and all the cases had ipsilateral cervical LNM. Seven out of 23 patients (30.4%, 7/23) with facial skin invasion were found to have parotid lymph node metastases, while only 2 of 50 patients (4.0%, 2/50) without facial skin invasion had parotid lymph node metastases. Fisher’s exact test showed that there was a strong correlation between LNM in the region of level VIII and facial skin invasion (30.4% vs 4.0%, p = 0.003).

The relative incidence of lymph node regions at different primary tumor sites.
Risk factors of initial node metastasis
We performed a logistic regression to evaluate the strength of seven variables on cervical LNM. The multivariate analyses revealed that LNM was independently associated with poor differentiation (odd ratio (OR) = 0.61, 95% CI: 0.45–0.83; p = 0.001), nasal cavity (OR = 0.65, 95% CI: 0.46–0.93; p = 0.018), skin involvement (OR = 1.95, 95% CI: 1.05–3.64; p = 0.036), and nasopharynx involvement (OR = 2.72, 95% CI: 1.28–5.72; p = 0.009). Patients with pterygopalatine fossa involvement at diagnosis tend to develop LNM (OR = 1.82, 95% CI: 0.97–3.41; p = 0.063). However, the involvement of hard plate (OR = 0.82, 95% CI: 0.39–1.71; p = 0.593) and orbit involvement (OR = 1.19, 95% CI: 0.67–2.10; p = 0.55) has no risk for cervical LNM (Figure 2).

Multivariate logistic analysis of risk factors for initial lymph node metastasis.
Survival analysis
The median follow-up time of the entire cohort was 40 months. The 5-year OS rate was 49.6% in patients with clinical LNM (cN+) compared with 59.3% in patients without clinical LNM (cN0; HR = 1.24, 95% CI: 0.83–1.85, p = 0.25), and the 5-year PFS rates were 54.0% and 42.2% respectively (HR = 1.25, 95% CI: 0.86–1.81, p = 0.22). The 5-year RFS rate in patients with cN+ was 57.6%, compared with 48.5% in those with cN0 (HR = 1.27, 95% CI: 0.85-1.88, p = 0.20). The 5-year distant metastasis failure rates were 58.8% and 46.6%, respectively, in patients with and without LNM (HR = 1.31, 95% CI: 0.88–1.96, p = 0.14; Figure 3).
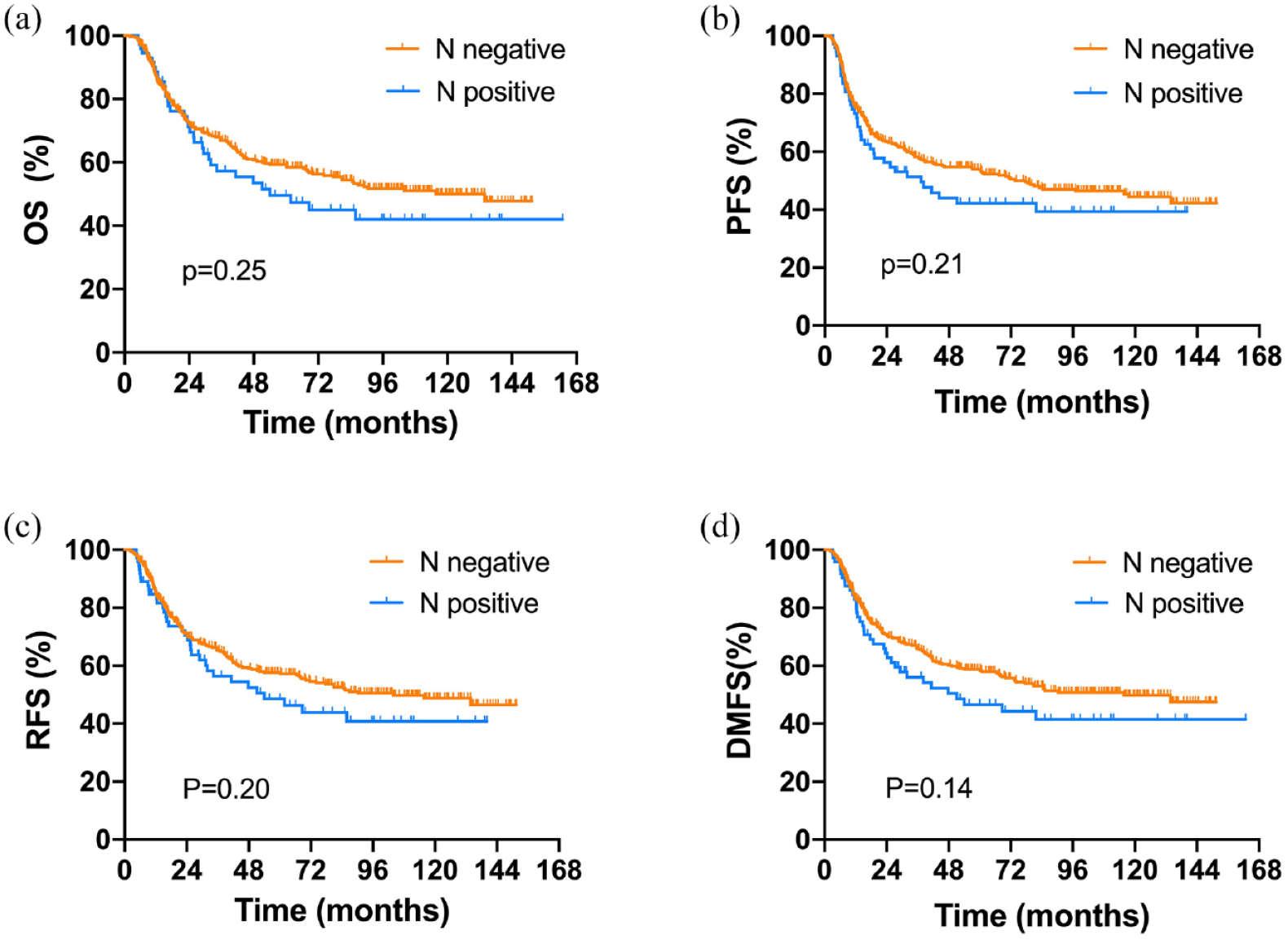
Kaplan–Meier curves showed a comparison of (a) overall survival, (b) progression-free survival, (c) regional-failure-free survival, and (d) distant metastasis-free survival for cases with or without lymph node metastases, and there were no significant differences between the two groups.
A multivariate analysis was used to evaluate the effect of clinicopathological variables on the survival outcome, including T stage, N stage, tumor differentiation, chemotherapy, and surgery. LNM was not found to be associated with OS (HR = 1.05, 95% CI: 0.70–1.56, p = 0.826) and RFS (HR = 1.09, 95% CI: 0.73–1.60, p = 0.826; Supplemental Table 2).
Propensity scoring match (PSM) analysis was performed in this study with the matching package in R software and conducted with the 1:2 nearest neighbor matching method. The covariates included age, sex, T stage, differentiation, surgery, and chemotherapy. After the PSM, 71 patients in the cN+ group and 121 in the cN0 group were enrolled for analysis, and the patient’s clinical characteristics are shown in Supplemental Table 3. The 5-year OS rates were 49.5% in the cN+ group and 57.2% in the cN0 group (HR = 1.11, 95% CI: 0.71–1.73, p = 0.64), 5-year PFS rates were 41.8% and 53.3% (HR = 1.12, 95% CI: 0.74–1.68, p = 0.58), 5-year RFS rates were 48.4% and 56.3% (HR = 1.17, 95% CI: 0.75–1.81, p = 0.48), and 5-year DMFS was 46.4% and 56.6% (HR = 1.15, 95%CI: 0.75–1.78, p = 0.52; Figure 4).
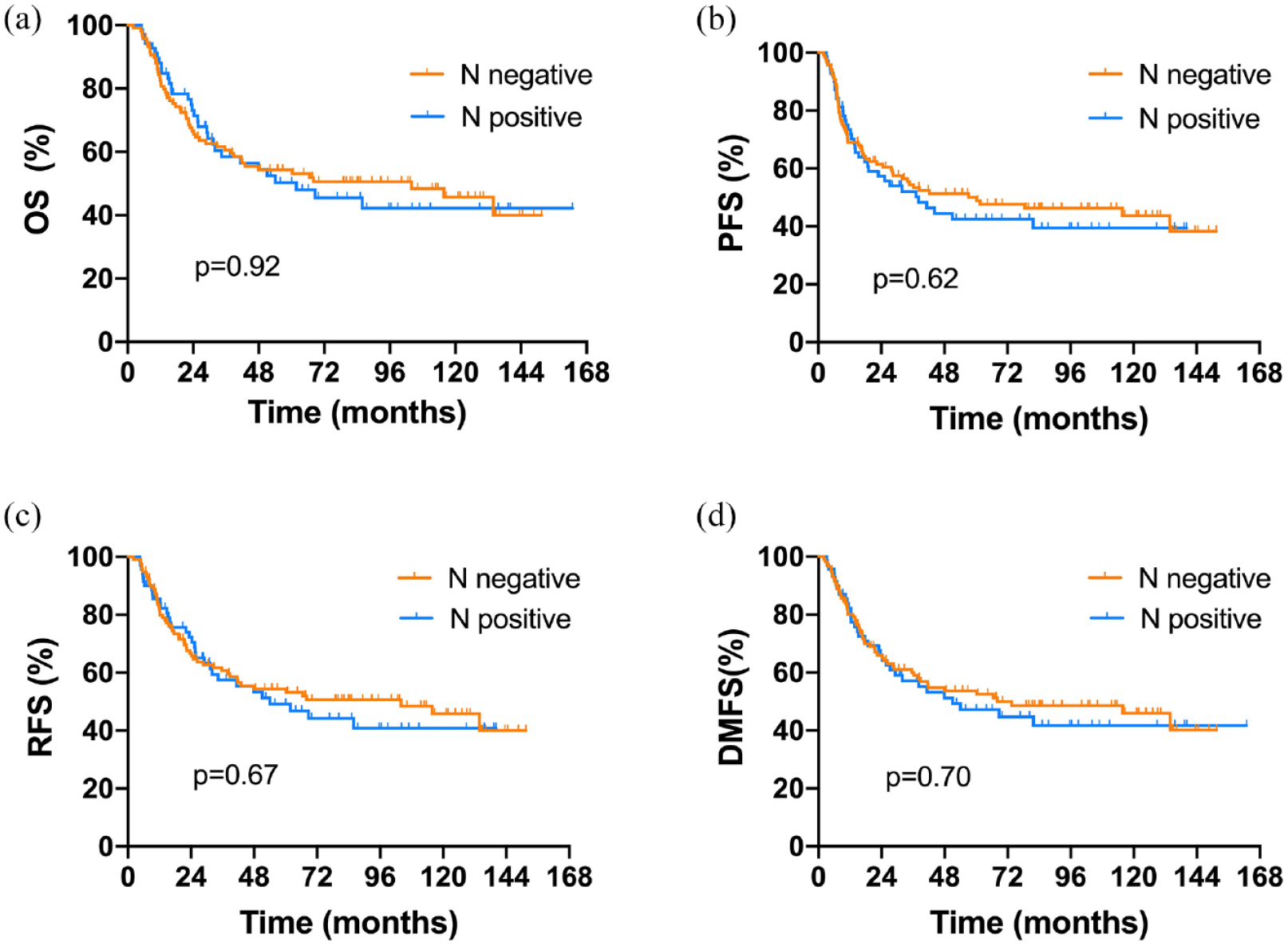
After 1:2 propensity score matching, 188 patients were analyzed. Kaplan–Meier curves showed a comparison of (a) overall survival, (b) progression-free survival, (c) regional-failure-free survival, and (d) distant metastasis-free survival for cases with or without lymph node metastases, and there were no significant differences between the two groups.
Discussion
Some studies on the incidence of LNM in SNSCC were based on National Cancer Database (NCDB) or SEER database analysis. The incidence of primary nodal metastasis at presentation ranged from 3.3% to 33%. 10 The disparity in different studies was almost 10 times, and the evaluation methods and tools might be the primary reason for this difference. The diagnostic accuracy has been dramatically improved with the application of high-resolution CT and MRI. In the previous literature, no parotid LNM was reported in SNSCC. However, in this study, nine patients had evidence of LNM in the parotid, accounting for 12.3% (9/73) of the cases with LNM. MRI, especially, is critical to screen the small lymph nodes in the parotid. We recommend that all the patients undergo MRI examinations, if possible. Not only parotid LNM but also retropharyngeal LNM is rarely reported before 2000. With the application of MRI, in recent years, there have been increasing reports of LNM in sinonasal tumors. 11 Guan et al. 12 first investigated the retropharyngeal LNM using MRI in 59 patients, and found LNM in 18 patients, with two cases of retropharyngeal LNM. Later, Sai et al. 13 identified 6 of 41 SNSCC (15%) as having LNM at initial diagnosis. Gangl et al. 14 reported that the incidence of metastatic retropharyngeal lymph nodes was as high as 43.5% in SNSCC patients (10/23). In our cohort, retropharyngeal LNM accounts for 30.1% of the SNSCC patients with cervical lymph nodes, five of whom had bilateral lesions. The high incidence of parotid and retropharyngeal LNM indicates the different lymph basins in SNSCC.
Several researchers have investigated the patterns of lymph node distribution in SNSCC. 15 Liu et al. 16 showed that the most frequently involved site in SNSCC was level II LNs (16.1%), followed by level Ib LNs (10.2%) and RPLNs (4.7%). In our cohort, level Ib, level VIIa, and level II were the most involved sites regardless of whether the primary tumor was located in the nasal cavity, ethmoid, or maxillary sinus. In contrast to previous findings, parotid LNM was detected. Furthermore, all the parotoid LNM occurred in the patients with facial skin involvements, and the facial skin and lymph node metastases were in the same laterals. We proposed three main lymph node drainage routes: the primary site to level VIIa to level II; the primary site to level Ib to level II; and the primary site to level VIII to level Ib/level II (for patients with facial skin involvement). Except for several bilateral LNMs, the spread of the LNM almost occurred in the ipsilateral. The patients with contralateral LNM had evidence of primary lesions crossing the midline, consistent with other studies. Ahn et al. 17 reported 9.3% and 20.7% of initial nodal involvement rates in patients with nasal cavity SCC and maxillary sinus SCC, and only 3.3% of bilateral or contralateral neck involvement was present.
LNM in SNSCC differs from that in other head-neck tumors, and a huge cervical mass was rarely found. In our cohort, only two patients presented a massive mass in the neck at diagnosis. Therefore, identifying the LNM accurately by radiology is a challenge. We investigated the relationship between the common risk factors and LNM. The multivariate analyses revealed that poor differentiation, skin involvement, and nasopharynx involvement indicated a risk of LNM. In contrast to other findings, however, no evidence of the sinus having a significantly increased risk of nodal metastasis compared to the nasal cavity. 4 Miyaguchi’s study presented a different conclusion that LNM was unrelated to the degree of differentiation. 18 However, the LNM in this study represents the regional failure after treatment other than the lymphatic status at diagnosis. Liu et al. 16 evaluated the common risk factors, including nasopharynx invasion, orbit invasion, pterygopalatine fossa invasion, infratemporal fossa invasion, dura invasion, and hard palate invasion, and found only nasopharynx involvement was associated with a significantly higher initial LNM rate. There is a study indicating that EBV-positive sinonasal tumors are more likely to develop regional LNM. 19 In practice, those hint at LNM possibility, especially those with retropharyngeal LNM and parotid LNM. The patterns of LNM guide the decision on the treatment modality of the neck. For cN0 patients, whether neck dissection or prophylactic neck irradiation remains controversial.20–22 The regions of the neck dissection and prophylactic neck irradiation are less discussed. Jeon et al. 23 suggested that elective neck treatment involving ipsilateral levels I and II is recommended for SCC of the maxillary sinus with posterior wall invasion. Le et al. 24 recommended elective ipsilateral neck irradiation for patients with T3–4 SCC of the maxillary sinus. In this study, a high incidence of retropharyngeal LNM indicates the possibility of prophylactic irradiation coverage of retropharyngeal space. Treating the ipsilateral parotid region is necessary for those with facial skin involvement.
A study based on SEER demonstrated that cN0 patients had significantly higher disease-specific survival than cN+ patients. 25 Another study data from NCDB showed that cN+ patients presented a risk of poor OS compared to cN0 patients. 4 Apart from such analyses based on databases, several studies investigated the LNM’s impact on survival in SNSCC. However, all the studies had limitations, either from small-size samples or without pathological sorts. Even though some studies reported that LNM was an independent factor for unfavored prognosis, some disagree with this conclusion. Gangl et al. 14 reported that retropharyngeal lymph node involvement was associated with statistically significant decreased OS (p = 0.0046) and PFS (p = 0.0065) in SNSCC. Liu et al. 16 reported a correlation between regional LNM and poor survival in SNSCC. However, they defined the initial and recurrent lymph nodes as regional LNM and performed the analysis. Mirghani et al. 5 reported that lymph nodes at diagnosis time were significantly and independently associated with poor survival, but in this study, they did not analyze SNSCC separately. Miyaguchi et al. 18 presented that the 5-year survival rate was 63% in the non-LNM group and 57% in the LNM group (p = 0.77). Although no correlation was observed between LNM and long-term survival, patients diagnosed with LNM were more likely to develop regional failure and distant metastasis.
To the best of our knowledge, this is the sample size study in a single institute; we confirmed that cervical LNM was not a poor prognostic factor for survival for SNSCC in univariate and multivariate analyses. We still could not find the survival difference between patients with or without LNM after propensity score matching. However, the LNM at diagnosis is associated with distant metastasis during the follow-up. The reasons we could achieve a correlation between the LNM and prognosis might be as follows: the majority of the lymph nodes were not huge cervical masses or extra-nodal extension or necrosis; all the lymph nodes were irradiated with radical dose.
Various limitations to this study must be considered when interpreting the results. First, it is a retrospective study; second, all the lymph nodes have no histological confirmation; in addition, no analysis on the relationship between LNM and tumor variants (e.g., keratinizing, non-keratinizing, spindle-cells, basaloid SCC) was performed; finally, the treatment modality was not completely uniform. We believe that the findings from this study bring a new perspective to LNM metastases in SNSCC.
Conclusion
In primary SNSCC, the incidences of retropharyngeal LNM and parotid were higher than in previous studies. The distribution levels of LNM demonstrate three spread routes in SNSCC. The risk factors for LNM at initial diagnosis could help us to decide on neck treatment. Especially this work showed us that LNM was not the independent factor in prognosis.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241299331 – Supplemental material for Patterns of lymph node metastasis in 441 patients with sinonasal squamous cell carcinoma
Supplemental material, sj-docx-1-tam-10.1177_17588359241299331 for Patterns of lymph node metastasis in 441 patients with sinonasal squamous cell carcinoma by Li Wang, Jie Wang, Tian Wang, Yi Li and Xinmao Song in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241299331 – Supplemental material for Patterns of lymph node metastasis in 441 patients with sinonasal squamous cell carcinoma
Supplemental material, sj-docx-2-tam-10.1177_17588359241299331 for Patterns of lymph node metastasis in 441 patients with sinonasal squamous cell carcinoma by Li Wang, Jie Wang, Tian Wang, Yi Li and Xinmao Song in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359241299331 – Supplemental material for Patterns of lymph node metastasis in 441 patients with sinonasal squamous cell carcinoma
Supplemental material, sj-docx-3-tam-10.1177_17588359241299331 for Patterns of lymph node metastasis in 441 patients with sinonasal squamous cell carcinoma by Li Wang, Jie Wang, Tian Wang, Yi Li and Xinmao Song in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359241299331 – Supplemental material for Patterns of lymph node metastasis in 441 patients with sinonasal squamous cell carcinoma
Supplemental material, sj-docx-4-tam-10.1177_17588359241299331 for Patterns of lymph node metastasis in 441 patients with sinonasal squamous cell carcinoma by Li Wang, Jie Wang, Tian Wang, Yi Li and Xinmao Song in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
