Abstract
Background:
Depth of tumor is a risk factor for lymph node metastasis in rectal cancer, but impact of yield pathologic T (ypT) stage on lymph node involvement in rectal cancer remains unclear. The aim of this study was to evaluate the correlation between ypT stage and lymph node metastasis.
Methods:
From January 2010 to December 2015, 602 patients who were diagnosed with rectal cancer and treated with neoadjuvant chemoradiotherapy (CRT) followed by radical operation were reviewed retrospectively. The correlations between ypT stage and lymph node status and survival were evaluated.
Results:
On pathology, 179 (29.7%) patients exhibited regional lymph node metastasis. Lymph node metastasis was seen in 8.5% of ypT0 patients, 20% of ypT1, 18.4% of ypT2, 47.5% of ypT3, and 27.3% of ypT4. Positive lymph node metastasis was correlated with ypT stage. In addition, the difference of lymph node metastasis in ypT stage subgroups was statistically significant (p < 0.001). Five-year disease-free survival was significantly different in the ypT stage subgroups (88.7% versus 86.7% versus 82.6% versus 64.7% versus 72.7%, p < 0.001), as was 5-year overall survival (96.2% versus 90.0% versus 95.8% versus 80.0% versus 90.9%, p < 0.001).
Conclusion:
YpT stage is associated with lymph node metastasis in rectal cancer treated with neoadjuvant CRT and radical operation, and ypT0 patients exhibited an 8.5% lymph node metastasis rate. Therefore, the decision for local excision or the watch-and-wait strategy for rectal cancer treated with neoadjuvant CRT and predicted to show a pathologic complete response should be considered with caution.
Introduction
Rectal cancer is one of the most common cancers worldwide, and standard treatment for locally advanced rectal cancer is neoadjuvant chemoradiotherapy (CRT) and proctectomy with total mesorectal excision. The advantages of neoadjuvant CRT are a reduction in size and downstaging of the primary cancer, resulting in increasing sphincter preservation and reduced toxicity.1–3 In addition, prognosis of rectal cancer between neoadjuvant CRT and adjuvant CRT has been reported, and neoadjuvant CRT exhibited better local control and survival compared to adjuvant CRT.4–6Also, several studies were conducted to evaluate the prognostic factors for survival after neoadjuvant CRT with radical operation in rectal cancer, and nodal status was a prognostic factor for recurrence and survival.7-9
Pathologic T stage is a risk factor for lymph node metastasis in rectal cancer.10,11 Furthermore, post-chemoradiation yield pathologic T (ypT) stage is a well-known risk factor for nodal status in rectal cancer treated with neoadjuvant CRT.12,13
Patients diagnosed with pathologic complete response after neoadjuvant CRT were considered for treatment with local excision because nodal metastases are rare in such patients with mural complete pathologic response after neoadjuvant CRT, and the oncologic outcome was not poor compared to that of a radical operation group.14,15 In addition, the watch-and-wait strategy was introduced for clinical complete response patients, and several studies showed comparable oncologic outcomes with those of a surgical group.16-18 However, lymph node metastasis in pathologic Complete response (CR) patients ranged in occurrence from 0% to 17.7%, and treatment by local excision or the watch-and-wait strategy could result in a poor oncologic outcome.19,20
The main purpose of this study was to evaluate whether ypT stage could predict lymph node status. The secondary purpose was to determine if ypT0 stage equated to ypN0, which would be treated with local excision or the watch-and-wait strategy.
Method
Flowchart of patient selection is shown in Figure 1. Patients, who underwent neoadjuvant CRT followed by radical operation with total mesorectal excision for locally advanced rectal cancer at our institution between January 2010 and December 2015, were reviewed retrospectively. Patients diagnosed with clinical stage II or III rectal cancer by abdominopelvic and chest computed tomography (CT) and rectum magnetic resonance imaging (MRI) were selected to administer neoadjuvant CRT. Patients with history of recurrent disease, familial cancer, palliative operation, or local excision were excluded. Also, patients who did not undergo adjuvant chemotherapy after the operation were excluded. Finally, a total of 602 patients was enrolled after 89 patients were excluded because of insufficient follow-up data. The study was approved by the Institutional Review Board of Samsung Medical Center.

Flowchart showing study population selection.
Patients were treated with preoperative radiotherapy of 44–50.4 grays administered to the entire pelvis and a regimen of preoperative chemotherapy of 5-fluorouracil (5-FU)-based therapy in 307 patients, capecitabine in 334 patients, oxaliplatin-based therapy in 6 patients, and others in 16 patients. After completion of neoadjuvant CRT, all patients underwent radical proctectomy with total mesorectal excision. The median time interval between preoperative CRT and operation was 7 weeks (range, 5–46 weeks).
Postoperative chemotherapy of 5-FU-based therapy was initiated 4–6 weeks after radical operation regardless of pathologic result. Postoperative chemotherapy was not performed in 61 patients because of poor general condition, patient refusal, or postoperative complications.
During the evaluation of primary cancer, all patients underwent digital rectal examination, colonoscopy, chest and abdominopelvic CT, and rectum MRI. After the operation, cancer stage was determined according to the tumor-node-metastasis classification (AJCC 7th classification). 21 Pathologic therapeutic regression by neoadjuvant concurrent CRT was assessed using the Dworak tumor regression grade.
Statistical analyses were performed using SPSS version 27 (SPSS Inc., Chicago, IL, USA). Sex, pre-CRT carcinoembryonic antigen (CEA), post-CRT CEA, interval between CRT and operation, route of access, diverting stoma, cancer perforation, gross type, differentiation, circumferential resection margin (CRM), number of harvested lymph nodes, lymphovascular invasion, perineural invasion, tumor budding, and tumor regression grade were considered binary variables. Type of surgery, ypT stage, and ypN stage were considered categorical variables. Age, body mass index (BMI), hemoglobin level, and tumor size were continuous variables. Categorical variables were analyzed using the Chi-square and Fisher’s exact test, and continuous variables were analyzed using the independent samples t-test. One-way analysis of variance was used to determine the quantitative differences between the groups. Correlation between ypT stage and lymph node metastasis was analyzed using the Spearman correlation coefficient test. The Cox proportional hazards regression model was used to analyze the variables that independently influenced survival and risk grouping. Factors with a p value ⩽0.05 in univariable analysis were entered into a multivariable analysis. To analyze the factors associated with lymph node metastasis, logistic regression was used. Survival curves were analyzed using the Kaplan–Meier method. A p value <0.05 was considered statistically significant.
Results
A total of 602 patients diagnosed with rectal cancer, treated with neoadjuvant CRT, and who underwent radical operation was analyzed. The median age of the study population was 56 years (range 28–83 years), and the number of male patients was 408 (67.8%). Median BMI was 23.9 kg/m2 (range, 16–38 kg/m2). Baseline characteristics of the study population are described in Table 1.
Baseline characteristics of the patients (n = 602).
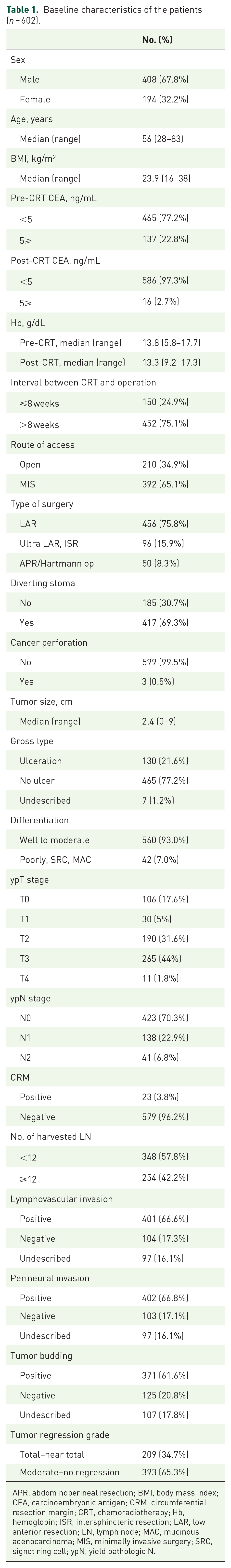
APR, abdominoperineal resection; BMI, body mass index; CEA, carcinoembryonic antigen; CRM, circumferential resection margin; CRT, chemoradiotherapy; Hb, hemoglobin; ISR, intersphincteric resection; LAR, low anterior resection; LN, lymph node; MAC, mucinous adenocarcinoma; MIS, minimally invasive surgery; SRC, signet ring cell; ypN, yield pathologic N.
The number of stage 0 patients was 92 (15.3%), stage I was 183 (30.4%), stage II was 148 (24.6%), and stage III was 179 (29.7%). The 5-year disease-free survival and overall survival were significantly different by stage (p < 0.001, p < 0.00).
The distribution of pathologic stages after neoadjuvant CRT is summarized in Table 2. Pathology showed 179 (29.7%) patients with regional lymph node metastasis. Of the 106 patients with no residual tumor after neoadjuvant CRT, 9 (8.5%) experienced regional lymph node metastasis. Positive lymph node metastasis was observed in 6 (20%) of 30 ypT1 patients, 35 (18.4%) of 190 ypT2 patients, 126 (47.5%) of 265 ypT3 patients, and 3 (27.3%) of 11 ypT4 patients (Table 2). Positive lymph node metastasis was confirmed to be correlated with ypT stage [correlation coefficient (r) = 0.340, p < 0.001). In addition, the difference between ypT stage subgroups by lymph node metastasis was statistically significant (p < 0.001). In univariable analysis, differentiation, ypT stage, number of harvested lymph nodes, lymphovascular invasion, perineural invasion, tumor budding, and tumor regression grade were predictive factors for lymph node metastasis. In multivariable analysis, differentiation, ypT stage, number of harvested lymph nodes, and lymphovascular invasion were predictive factors for lymph node metastasis (p = 0.007, p = 0.024, p < 0.001, p < 0.001; Table 3).
Number of positive lymph node patients according to ypT stage.

ypN, yield pathologic N; ypT, yield pathologic T.
Univariable and multivariable analyses of factors associated with lymph node metastasis.
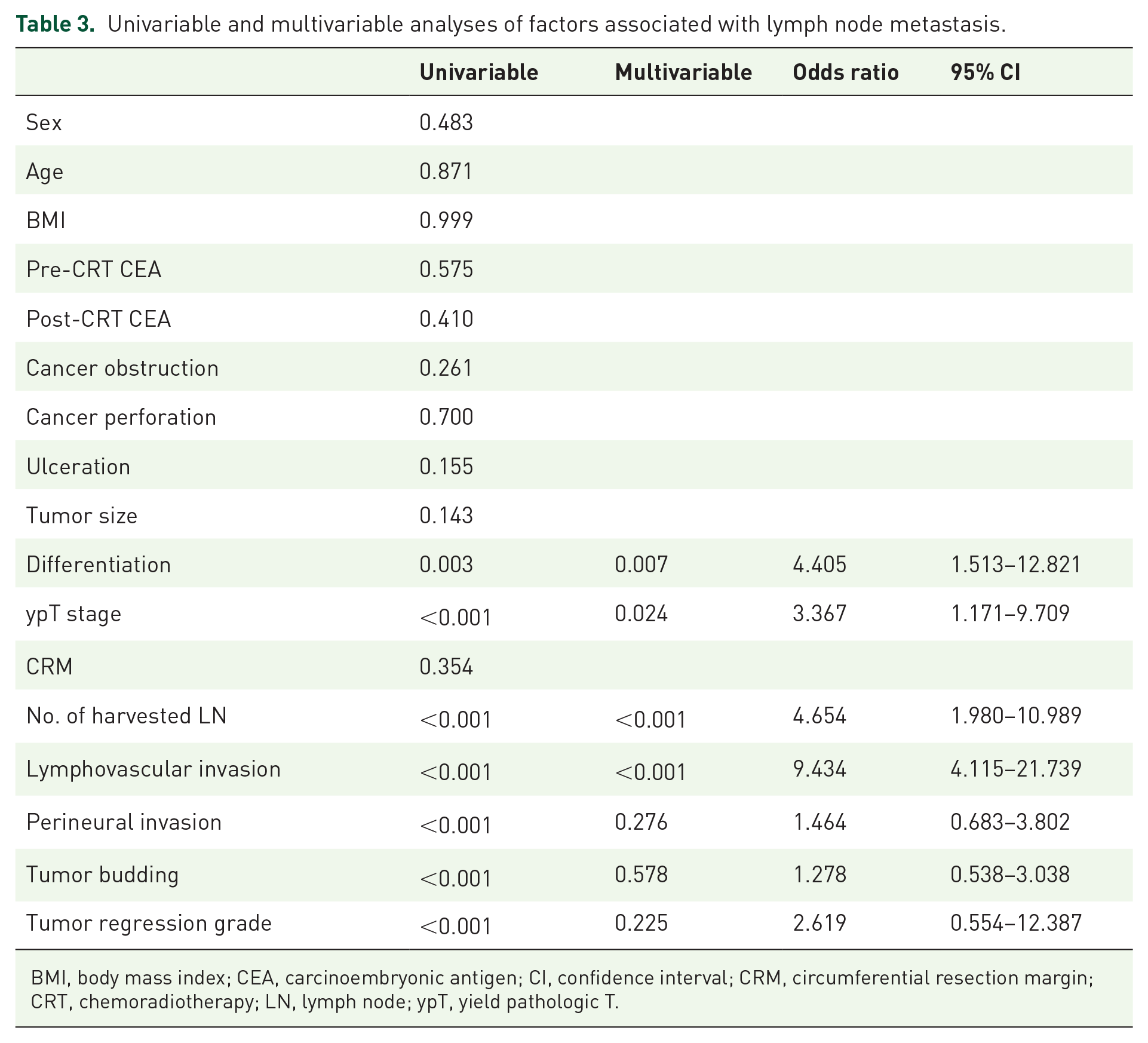
BMI, body mass index; CEA, carcinoembryonic antigen; CI, confidence interval; CRM, circumferential resection margin; CRT, chemoradiotherapy; LN, lymph node; ypT, yield pathologic T.
The median follow-up was 79 months. The 5-year disease-free survival was significantly different in ypT stage subgroups (88.7% versus 86.7% versus 82.6% versus 64.7% versus 72.7%, p < 0.001). In univariable analysis, pre-CRT CEA, ypN stage, CRM, lymphovascular invasion, perineural invasion, tumor budding, and tumor regression grade were prognostic factors for recurrence. In multivariable analysis, ypN stage, CRM, lymphovascular invasion, and perineural invasion were prognostic factors for recurrence (p < 0.001, p = 0.036, p = 0.008, p = 0.023, p = 0.022; Table 4). The 5-year overall survival was significantly different by ypT stage subgroup (96.2% versus 90.0% versus 95.8% versus 80.0% versus 90.9%, p < 0.001). In multivariable analysis, ypN stage, CRM, and tumor budding were prognostic factors for death (p < 0.001, p = 0.001, p = 0.044; Table 5). In addition, local recurrence-free survival was significantly different in ypT stage subgroups (96.2% versus 96.7% versus 95.3% versus 88.6 versus 90.9%, p = 0.007).
Univariable and multivariable analyses of prognostic factors for recurrence.

BMI, body mass index; CEA, carcinoembryonic antigen; CI, confidence interval; CRM, circumferential resection margin; CRT, chemoradiotherapy; LN, lymph node; ypN, yield potential N; ypT, yield potential T.
Univariable and multivariable analyses of prognostic factors for overall survival.
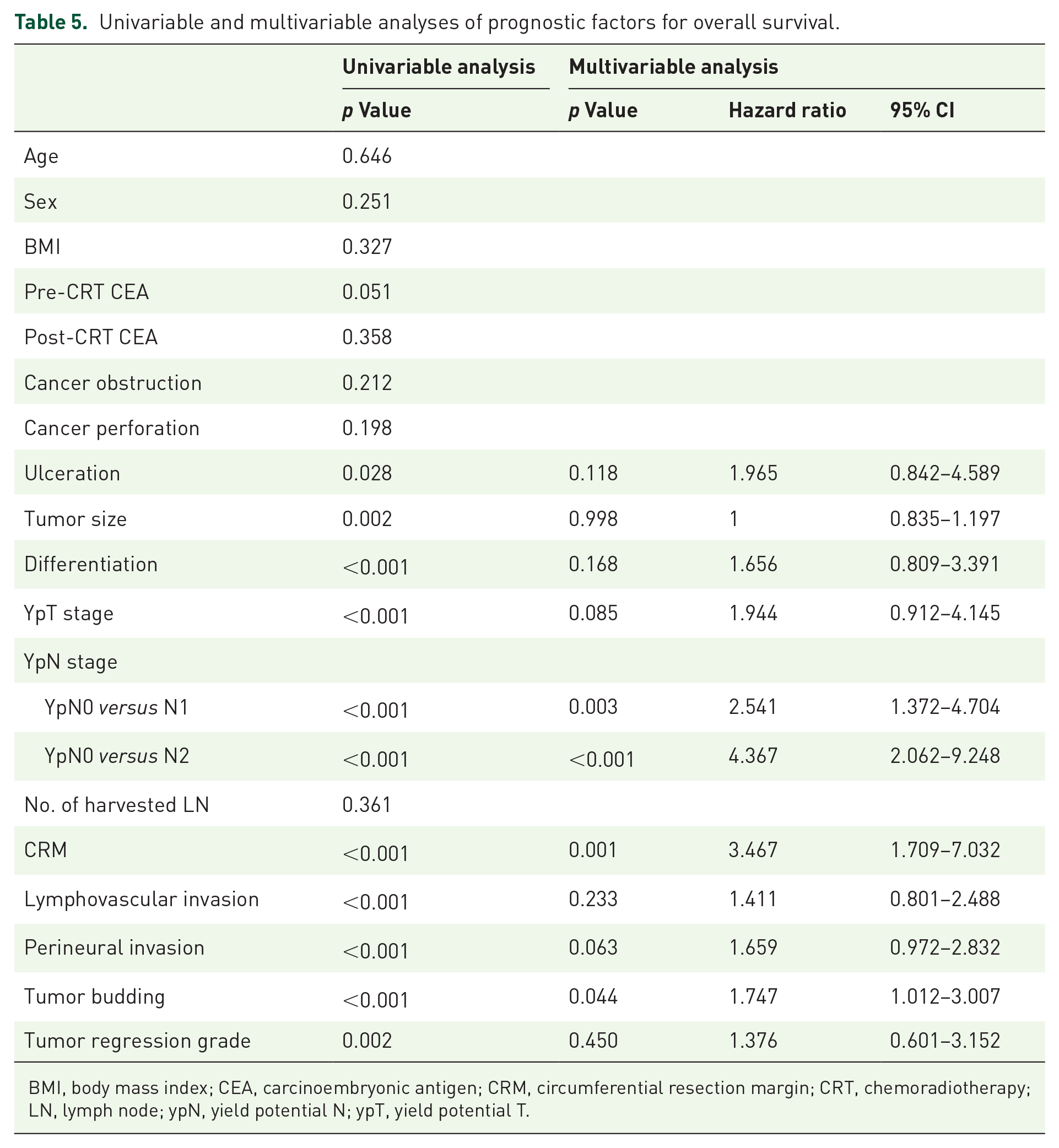
BMI, body mass index; CEA, carcinoembryonic antigen; CRM, circumferential resection margin; CRT, chemoradiotherapy; LN, lymph node; ypN, yield potential N; ypT, yield potential T.
Discussion
Standard treatment for mid-to-low advanced rectal cancer is neoadjuvant CRT following radical operation including total mesorectal excision. Neoadjuvant CRT has the potential advantage of reducing the size and downstaging the primary tumor before surgery and exhibiting favorable oncologic outcomes with increasing sphincter preservation, leading to a better quality of life.1,22 Of such patients, 10–30% are diagnosed with pathologic complete response, characterized by eradication of all tumor cells; they show favorable oncologic outcome compared to non-responsive tumor, and local excision and watch-and-wait strategy are alternative treatment options.23,24 Despite resolution of the primary tumor after neoadjuvant CRT, the risk of lymph node metastasis remains and could be a cause of recurrence.
Nodal status is a strong prognostic factor for survival in rectal cancer.25,26 The prognostic impact of lymph node metastasis after neoadjuvant therapy is evaluated in many studies.27,28 In our study, the proportion of nodal metastasis in pathologic complete response after neoadjuvant CRT was 8.3% with a 5-year disease-free survival of 90.1%. Bosch et al. 29 reported lymph node metastasis status in ypT0 patients at a rate of 17.4%, ypT1 patients at a 14.8% rate, ypT2 patients at a 25.8% rate, and ypT stage did not predict residual nodal status after neoadjuvant CRT. However, Pucciarelli et al. 13 reported the percentage of lymph node metastasis in ypT0 at 1.8%, in ypT1 at 16.9%, in ypT2 at 37.8%, and in ypT3 at 37.8%, and ypT stage was a predictive factor for lymph node involvement. In our study, there was a rapid increase in nodal involvement between ypT0 and ypT1 (8.3%–17.6%) and between ypT1-2 and ypT3-4 (17.6%–46.5%).
Differentiation, ypT stage, lymphovascular invasion, and number of harvested lymph nodes were predictive factors for lymph node metastasis. Kim et al. 12 reported ypT stage and tumor regression grade as predictive factors for lymph node metastasis. Huh et al. 30 reported lymphovascular invasion as a predictive factor for lymph node metastasis in T1 and T2 colorectal cancers. In addition, Chang et al. 31 reported lymphovascular invasion, poor differentiation, and T stage to be significantly related to nodal involvement.
ypN stage, CRM, lymphovascular invasion, and perineural invasion were significant prognostic factors for recurrence, and ypN stage, CRM, and tumor budding were significant prognostic factors for death in rectal cancer treated with neoadjuvant CRT and radical operation. Hwang et al. 32 described CRM and yp stage as risk factors for local recurrence and disease-free survival. Huh et al. 33 reported differentiation, perineural invasion, postoperative chemotherapy, tumor regression grade classification, and post-CRT CEA level as prognostic factors for overall survival. Kim et al. 28 described ypN stage, CRM, lymphovascular invasion, and perineural invasion as prognostic factors for recurrence, as in our study.
For ypT0 patients with rectal cancer, there are many studies reporting local excision or the watch-and-wait strategy as treatment of choice in those treated with neoadjuvant CRT, resulting in a local recurrence rate of 1.9%-6.9%.34,35 Based on our study results, 8.3% of patients are diagnosed with ypT0 lymph node metastasis; the recurrence rate was 9.9% with radical operation, with a local recurrence rate of 3.3%. According to our study results, local excision after neoadjuvant CRT with rectal cancer with a pathologic result of ypT0 would be insufficient for controlling recurrence.
This study has several limitations. First, it is a single-center study with retrospective data collection that is subject to various biases. Second, the numbers of ypT1 and ypT4 patients was small, and lymph node metastasis was lower in ypT2 than in ypT1 and in ypT4 than in ypT3. The obtained data were insufficient to perform a proper statistical analysis. Further studies with larger sample size are needed to reevaluate and confirm these observations. Third, there is no mention of radiologic evaluation after neoadjuvant CRT, which could better identify clinical complete response even though the accuracy is low.
YpT stage is associated with lymph node metastasis in rectal cancer treated with neoadjuvant CRT and radical operation, and ypT0 exhibited 8.5% positive lymph node metastasis. Therefore, the decision for local excision or the watch-and-wait strategy for rectal cancer treated with neoadjuvant CRT and predicted as clinical complete response should be considered with caution.
