Abstract
Background:
Chemoradiotherapy (CRT) is the main treatment for elderly patients with non-metastatic rectal cancer who are ineligible for or decline surgery, but the optimal modality remains unclear.
Objectives:
This study was to validate the safety and efficacy of comprehensive geriatric assessment (CGA) guided radiotherapy in older patients.
Design:
An exploratory analysis of a single-arm, multicenter, Phase II trial.
Methods:
Patients aged over 70 and diagnosed with rectal cancer were enrolled and evaluated by CGA. CGA-guided radiotherapy was individually conducted in a multidisciplinary setting. Patients in fit, intermediate, and frail groups were scheduled to receive CRT, long-course radiotherapy, and short-course radiotherapy (SCRT) alone respectively. Patients who were unfit for or refused surgery were analyzed for acute toxicities and survival outcomes.
Results:
In a total of 109 enrolled patients, 47 individuals who did not undergo surgery were included, with 26, 9, and 12 categorized into fit, intermediate, and frail groups. Only 11 (23.4%) grade 3 or above toxicities were observed overall. Within a median follow-up of 69.0 months, the 3-year overall survival (OS), progression-free survival (PFS), and cancer-specific survival (CSS) rates were 44.3% (95% CI: 32.1%–61.2%), 25.5% (95% CI: 15.7%–41.6%) and 61.0% (95% CI: 47.8%–77.6%) in total. The 5-year OS, PFS, and CSS reached 15.0% (95% CI: 7.4%–30.3%), 14.6% (95% CI: 7.3%–29.4%), and 36.2% (95% CI: 22.0%–59.4%), with no significant difference among the three subgroups. SCRT (p < 0.001) and dose boost (p = 0.045) contributed to lower tumor-related death rates in multiple competing risk regressions.
Conclusion:
Radiotherapy guided by CGA was effective and well-tolerated in non-surgical elderly patients. SCRT alone seemed to achieve similar clinical outcomes as CRT in corresponding subgroups. However, given the limited size of this study, further investigation in a larger population is still needed for this strategy.
Keywords
Introduction
More than half of advanced rectal cancer (LARC) patients age over 70 when diagnosed, but evidence in standard treatment modality is lacking. During the past decades, the standard of care for aging patients with LARC has shifted from surgery alone or surgery with adjuvant therapies to preoperative treatments followed by surgery or the “Watch and Wait (W&W)” strategy in patients who respond well. While long-course chemoradiotherapy (CRT) is perceived as the cornerstone of treatment, the ideal strategy for vulnerable and frail patients is still under investigation, since fewer data are derived from prospective datasets.
In fact, the mortality related to surgery varies from 11.3% to higher,1,2 especially in frail and functionally declined individuals. In consequence, non-surgical options, including concurrent CRT or radiotherapy (RT), have evolved into promising alternatives for those who need a balanced between tumor treatments and general care. 3 Recent studies have witnessed a growing proportion of nonoperative treatments in older populations, rising from 4% in those aged under 64 to 21% in their counterparts over 85. The fluctuation is mainly due to various comorbidities, disease stages, anesthesia intolerance, and patient concerns for recovery after surgery. 1 Additionally, different from the most commonly used strategy established on data from younger patients, elderly patients show low adherence to concurrent CRT. After all, the regimen is at high risk of adverse effects. 4 Therefore, there is an urgent need in exploring a favorable modality for older and frail patients.
Attempts on screening better modality and tailoring interventions have been made. 5 It was revealed in the PRODIGE 42 study that the R0 resection rate in preoperative short-course radiotherapy (SCRT) and traditional CRT was close (84.3% vs 88%, though non-inferiority p = 0.28). Instrumental functionality was better preserved in the SCRT group. 6 Haak et al. carried out a pooled analysis of older patients who achieved (near) clinical complete response in two studies, in which either long-course CRT (based on capecitabine) or SCRT were adopted. The 3-year overall survival (OS) and local regrowth-free rate were reported to be 97% and 88%, respectively. 7 Another randomized controlled trial did not find any disparity in rates of complications between patients receiving personalized treatments based on preoperative geriatric evaluations and usual care. However, only Vulnerable Elderly Survey-13 and 4 clinical criteria were applied for frailty screening in the study. 8 Overall, the existing evidence hardly shows a divergence between SCRT and long-course CRT in benefits and risks, possibly attributed to the fact that frailty is mainly introduced as a dichotomous or categorical factor. Thus, prospective researches based on quantitative geriatric assessments are needed to shed light on the optimal choices for the elderly.
While oncological endpoints might rank first in their younger counterparts, functional independence is valued as vital as the same in elderly and frail individuals. Consequently, frailty should be assessed and integrated into decision-making procedures of elderly patients. 1 The definition of comprehensive geriatric assessment (CGA) and its latent beneficiaries have been determined. 9 Strong recommendation is given to CGA assessments, multidisciplinary evaluation, and prehabilitation implementation according to current guidelines on nonoperative management of elderly and frail patients.4,10 Though CGA-driven methods may help in prescribing appropriate therapies in selected patients to avoid under-treatment or over-treatment, the majority of studies focus on the role of RT, particularly brachytherapy, rather than CGA-stratified treatment. 11 Therefore, the feasibility of CGA-motivated approaches yet needs to be evaluated.
Our previous study verified the oncological outcomes of neoadjuvant chemoradiotherapy followed by total mesorectal excision in elderly patients who were defined as “Fit” and eligible for operation. 12 However, surgery was omitted in a considerably large portion of enrolled patients, and those who were “Vulnerable,” “Frail” or refused operation were underrepresented. This study aimed to probe into the safety and efficacy of CGA-based radiotherapy in inoperable patients.
Methods
Trial design and patient selection
A prospective nonoperative cohort from a single-arm, multicenter, Phase II trial was included in this post-hoc study. Details of the Phase II trial have been previously reported. 13 The study was approved by the ethics committee and written informed consents were obtained from all candidates. In brief, patients aged over 70 and diagnosed with stage II/III intermediate or locally advanced (according to European Society for Medical Oncology Clinical Practice Guidelines) rectal adenocarcinoma were enrolled and assessed with CGA by an experienced geriatric oncologist.
The assessment procedure followed the criteria developed by the International Society of Geriatric Oncology (SIOG), taking comorbidities, functional independence, and nutritional status into consideration comprehensively. Evaluation tools applied are listed in Table 1. Generally, patients scored 0–2 according to Cumulative Illness Rating Scale for Geriatric, independent in instrumental activities of daily living (IADL) assessments, and free of nutritional risks were classified into the “Fit” group. Patients who scored at least one Grade 3 in Cumulative Illness Rating Scale for Geriatric with reversible impairment in independence were deemed as “Vulnerable,” while those with irreversible impairment were defined as “Frail”. The main difference between “Vulnerable” and “Frail” was the severity of comorbidities, dependence, and malnutrition. 14 Individuals who were ineligible for or refused surgery were selected for safety and survival analyses. The reporting of this research adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist 15 (Supplemental Table S1).
Geriatric assessment at baseline.

ADL, activities of daily living; CCI, Charlson Comorbidity Index; CGA, comprehensive geriatric assessment; CIRS-G, Cumulative Illness Rating Scale for Geriatrics; GDS15, Geriatric Depression Scale 15; IADL, instrumental activities of daily living; KPS, Karnofsky Performance Scale; MMSE, Mini-Mental State Examination; MNA, Mini Nutritional Assessment; TUG, Timed “Up & Go.”
Treatment delivery
Radiotherapy and concurrent chemotherapy were individually conducted based on clinical stages, physical status, and personal preference in a multidisciplinary setting. Specifically, patients recognized as being “Fit” were scheduled to receive standard concurrent CRT (Raltitrexed, intravenous infusion, 3 mg/m2 on days 1 and 22), with long-course radiotherapy (LCRT) delivered to planning target volume at a dose of 50 Gy (2.0 Gy per fraction, 35 days in total). Capecitabine (oral administration, 1650 mg/m2, on radiotherapy days) was prescribed to those who were excluded from the Phase II study because of refusal of operation. LCRT was also recommended for “Vulnerable” patients, but concurrent chemotherapy was planned for those without multisystem comorbidities. SCRT alone, which referred to 25 Gy (5.0 Gy per fraction, 5 days in total), was prescribed to the “Frail” subgroup. Sequential dose boost to the primary tumor or metastatic lymph nodes (10 Gy, 2.0 Gy per fraction, 5 days in total) was performed once the surgery was declined unless patients suffered from severe comorbidities or refused dosage boost. Simultaneous dose boost was optional. The dosage establishment was based on the guidelines for rectal cancer published by the European Society for Medical Oncology. 16 All the prescribed RT were executed with 6-MV X-ray by UNIQUETM (Varian, Palo Alto, CA, USA), EdgeTM (Varian) or SynergyTM (Elekta, Stockholm, Sweden). Short-term response evaluation was done by Response Evaluation Criteria in Solid Tumours (RECIST) 1.1. Radiotherapy was carried out with intensity-modulated or volumetric-modulated arc techniques, and image-guided RT with cone beam computed tomography performed in the first 5 days and once per week subsequently was adopted in all patients.
Clinical endpoints
The primary endpoint was defined as progression-free survival (PFS), the period from the completion of CRT or RT to objective disease progression or death from any cause. Additionally, OS, cancer-specific survival (CSS), and loco-regional free survival (LRFS) were included as secondary endpoints. OS was identified as the interval between the end of CRT or RT and death from any cause, while CSS was defined as the time from the end of CRT or RT to death from cancer. Participants who were lost in follow-ups were censored at the date of the last record of vital events. Short-term responses were assessed by objective response rate (ORR) within 3 months after RT. Furthermore, toxicities were graded by the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.0. Acute grade 3 or higher adverse events, which occurred during CRT or RT and the following 180 days, were collected and assessed.
Statistical analysis
A nonoperative registry of the previous Phase II study 12 was included and analyzed to capture oncological outcomes and toxicities. Analysis was carried out in the intention-to-treat (ITT) population. The distribution of PFS, OS, and CSS were summarized with the Kaplan-Meier method and compared with log-rank tests. Additionally, OS was analyzed with competing risk methods with death from other causes rather than tumors as the competing factor. Univariate and multivariate competing risk analyses by Gray’s test were applied to calculate the mortality of tumors and other death reasons. Baseline characteristics, CGA assessments, treatment modality, and compliance, as well as toxicities, were described by frequencies and tested with Chi-square or Fisher’s exact tests. In terms of numerical variables, the Kruskal-Wallis rank sum test was performed. Statistical analyses were tested two-sided using a significance level of 5%. These analyses were performed using R software (v 4.3.2).
Results
Patient characteristics
From September 2016 to October 2019, a total of 109 patients were recruited from 2 clinical centers and assessed by CGA, among whom 86 were recognized as “Fit” patients, 11 as “Vulnerable” and 12 as “Frail” individuals. In the ITT population, two patients in the “Fit” group exhibited oligometastasis at the initiation of radiotherapy, and surgery was subsequently omitted. Patients in the “Vulnerable” and “Frail” groups, and those who did not undergo surgery due to personal preference, physical status, or unresectable diseases were included in the current study. Specifically, except for 2 patients who withdrew informed consent, 47 candidates were selected, with 26, 9, and 12 categorized into “Fit”, “Vulnerable,” and “Frail” groups (Figure 1).

CONSORT diagram. The enrollment and selection procedures of the analyzed cohort.
In this cohort, the median age was 79 years old (range 71–89 years), with 6 (23.1%), 4 (44.4%), and 10 (83.3%) patients in the “Fit”, “Vulnerable,” and “Frail” subgroup aged over 80 (p = 0.011). The median distance from the anal verge was 4.0 cm (range 1.0–12.0 cm), and 33 (70.2%) individuals were diagnosed with lower rectal cancer (⩽5.0 cm). In terms of CGA assessments, worse daily living status, impaired mental health, and physical functions were more frequently observed in patients defined as “Frail” (p < 0.05). There was no significant difference observed in other tumor or geriatric features. The tumor characteristics and CGA evaluations are listed in Tables 1 and 2, respectively.
Baseline characteristics.
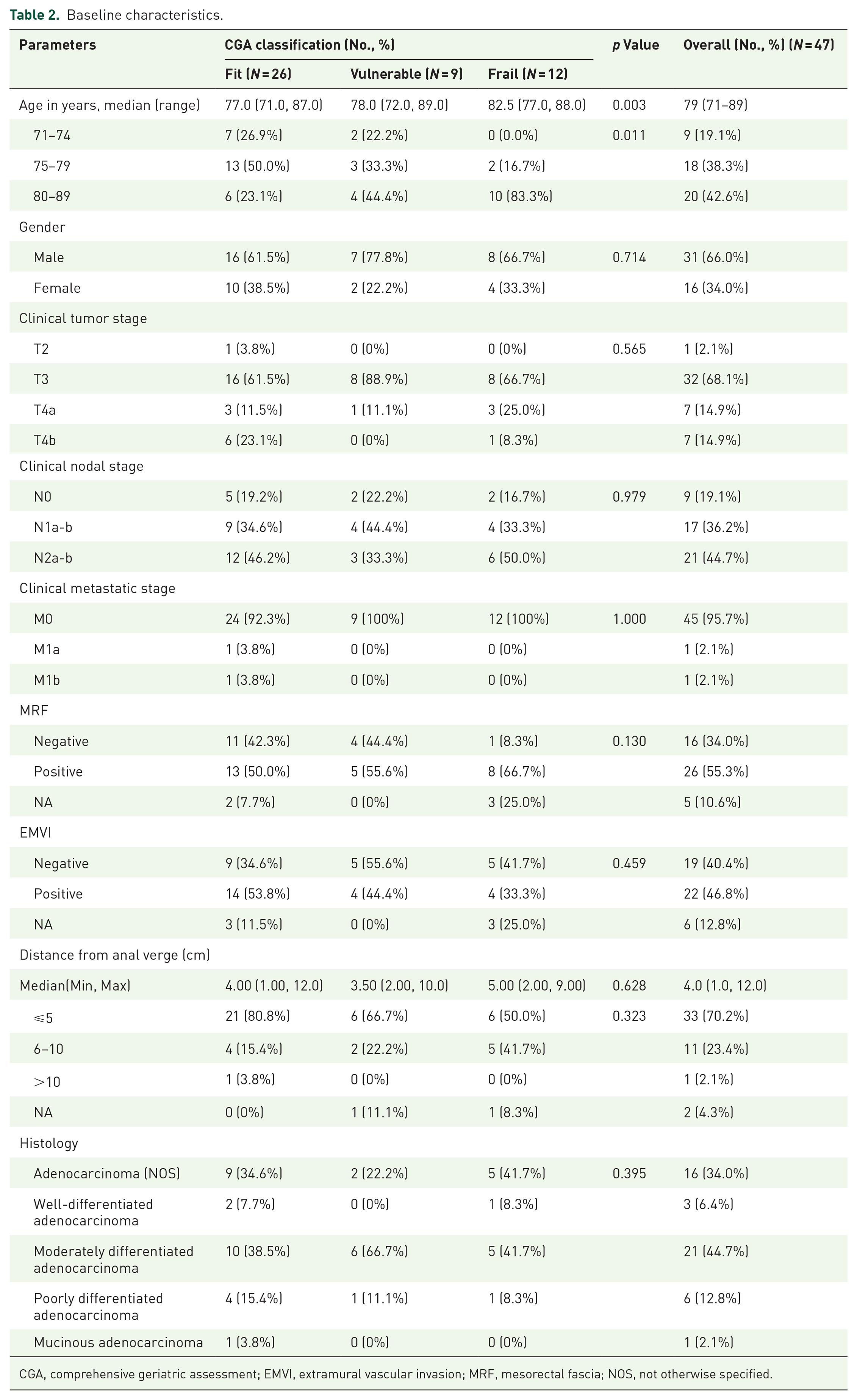
CGA, comprehensive geriatric assessment; EMVI, extramural vascular invasion; MRF, mesorectal fascia; NOS, not otherwise specified.
Treatment modality and compliance
Discussion involving multidisciplinary treatment (MDT) was required for each participant before treatment delivery. CRT was prescribed to the “Fit” and “Vulnerable” groups. Instead, all the patients in the “Frail” group were scheduled to receive RT alone. Totally, 39 (83.0%) patients underwent radiotherapy as planned. LCRT was delivered to patients in the “Fit” and “Vulnerable” groups, along with concurrent chemotherapy based on raltitrexed. Five (19.2%) and 2 (22.2%) patients in the “Fit” and “Vulnerable” groups received capecitabine. Only 8 (30.8%) and 1 (11.1%) patients finished prescribed chemotherapy in the corresponding groups respectively without significant difference (p = 0.785). In the “Frail” group, all patients underwent SCRT without concurrent chemotherapy. There were 11 (42.3%), 5 (55.6%), and 6 (50.0%) patients who had a dose boost to tumors in the 3 groups, respectively, of whom 11 were scheduled to have a dose boost up to 60 Gy, and 9 finally completed. No significant difference was observed in dose boost or reduction proportions among subgroups. Break or cessation was rare in the whole cohort. The treatment delivery and compliance profiles are shown in Table 3.
Treatment modality and compliance profile.
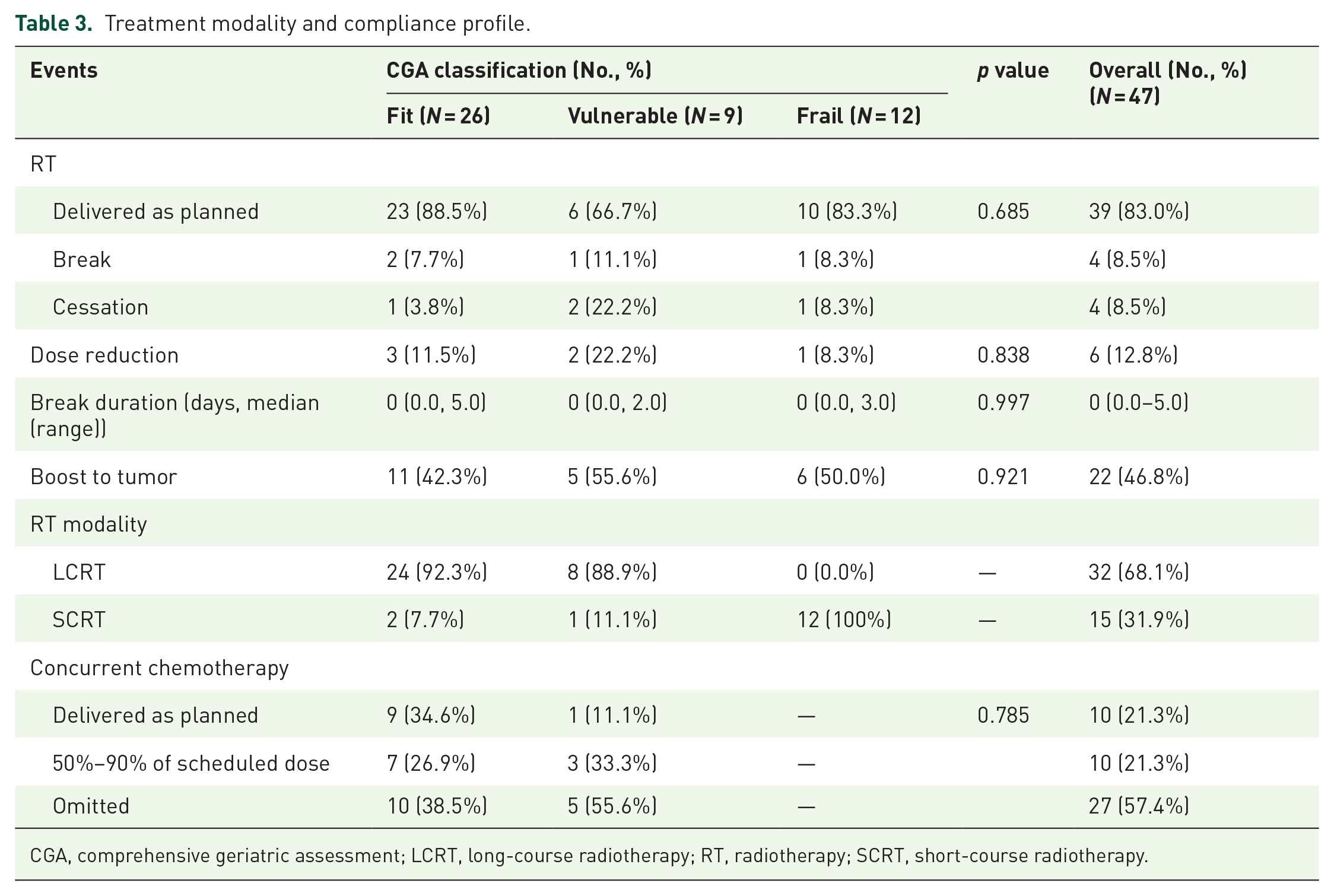
CGA, comprehensive geriatric assessment; LCRT, long-course radiotherapy; RT, radiotherapy; SCRT, short-course radiotherapy.
Oncological and survival outcomes
In total, 3 (6.4%) patients achieved clinical complete response, with 25 (53.2%) defined as partial response. Participants in the “Fit” group exhibited a numerically higher ORR rate of 69.2% than the “Vulnerable” or “Frail” group (p = 0.544). During a median follow-up of 69.0 months (range 1.0–83.0 months), the median OS of the whole cohort reached 33 months (95% CI: 30–41 months). The 3-year OS, PFS, CSS, and LRFS rates were 44.3% (95% CI: 32.1%–61.2%), 25.5% (95% CI: 15.7%–41.6%), 61.0% (95% CI: 47.8%–77.6%) and 58.3% (95% CI: 44.5%–76.4%), respectively. The 5-year OS, PFS, CSS, and LRFS rates were 15.0% (95% CI: 7.4%–30.3%), 14.6% (95% CI: 7.3%–29.4%), 36.2% (95% CI: 22.0%–59.4%) and 34.6% (95% CI: 17.7%–67.4%) (Figure 2). Though the “Fit” group seemed to perform better in 3-year PFS numerically (“Fit” vs “Vulnerable” vs “Frail”: 26.9% (95% CI: 9.4%–66.6%) versus 22.2% (95% CI: 6.6%–75.4%) versus 25.0% (95% CI: 14.3%–50.7%), p = 0.960), no significant difference was found among the 3 groups (Figure 3).

Survival outcomes of the whole cohort presented by the Kaplan-Meier method. (a) Overall survival curve; (b) progression-free survival curve; (c) cancer-specific survival curve; (d) local-regional free survival curve.
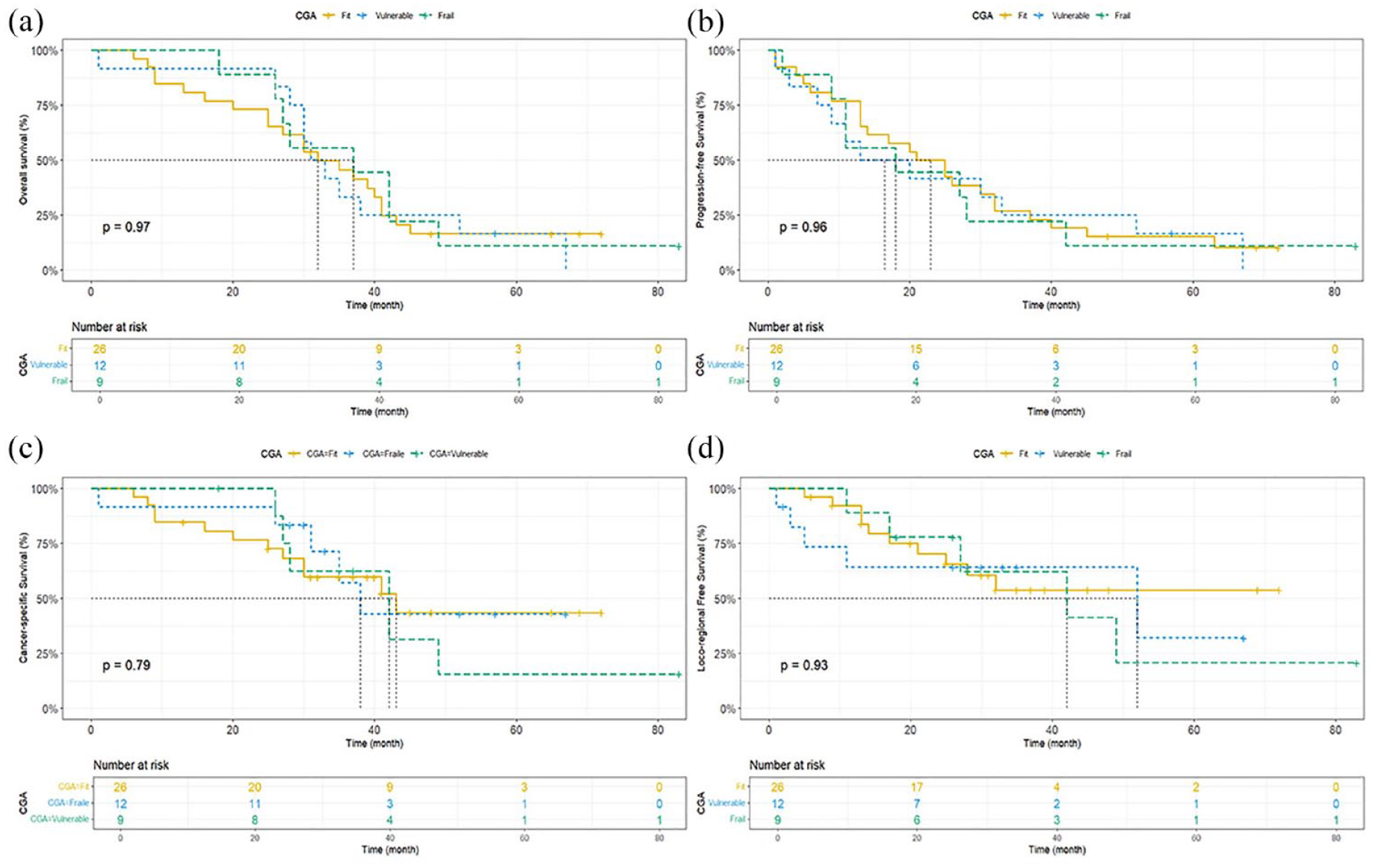
Survival outcomes of separate CGA subgroups presented by the Kaplan-Meier method and compared with log-rank tests. (a) Overall survival curve; (b) progression-free survival curve; (c) cancer-specific survival curve; (d) local-regional free survival curve (p > 0.05).
According to the univariate competing risk regression, no significant correlation was found between the cumulative mortality and CGA assessments in months 24, 48, and 60, either due to tumor or other reasons (Supplemental Table S2). However, in multivariate analysis, SCRT (HR = 0.00, 95% CI: 0.00–0.04, p < 0.001) and dose boost to tumor (HR = 0.10, 95% CI: 0.01–0.95, p = 0.045) contributed to lower tumor-related death rates in multiple competing risk regression. Compared to patients recognized as “Frail”, “Fit” (HR = 0.02, 95% CI: 0.00–0.54, p = 0.018), and “Vulnerable” (HR = 0.03, 95% CI: 0.00–0.43, p = 0.012) patients had decreased mortality regarding tumor. Higher KPS scores and better treatment response tended to have lower death rates in relation to tumors as well (p < 0.001). Age, sex, clinical stage, mesorectal fascia status, extramural vascular invasion status, or concurrent chemotherapy delivery had no significant impact on tumor mortality (Table 4). Similarly, “Fit” and “Vulnerable” patients were less likely to die from other causes, with enhanced mortality rates of other causes demonstrated in individuals who scored higher in the Charlson Comorbidity Index or IADL (p < 0.05). (Table 5). Only one patient exhibited disease progression with ileus during the follow-up, and a stoma was planned for him as a palliative strategy.
Competing risks regression analyses (tumor as the failure reason).

CGA, comprehensive geriatric assessment; EMVI, extramural vascular invasion; KPS, Karnofsky Performance Scale; LCRT, long-course radiotherapy; MRF, mesorectal fascia; SCRT, short-course radiotherapy.
Competing risks regression analyses (other failure reasons).

CCI, Charlson Comorbidity Index; CGA, comprehensive geriatric assessment; GDS15, Geriatric Depression Scale 15; IADL, instrumental activities of daily living; KPS, Karnofsky Performance Scale; MMSE, Mini-Mental State Examination.
Toxicities
Forty-five (95.7%) patients reported acute toxicities during CRT or RT overall. The most common adverse effects were lymphocytopenia (35, 74.5%), leukopenia (24, 51.1%), and diarrhea (24, 51.1%). Radiotherapy-induced side effects happened more frequently in the “Fit” and “Vulnerable” groups, including urinary frequency (p = 0.018) and radiation dermatitis (p = 0.005) (Supplemental Table S3). Only 11 (23.4%) grade 3 or above toxicities were reported in total, while lymphocytopenia occurred more often in the “Fit” and “Vulnerable” group (p = 0.029) (Table 6).
Grade 3–4 acute toxicity during CRT or RT.

ALT, alanine transaminase; CGA, comprehensive geriatric assessment; CRT, chemoradiotherapy.
Discussion
Evidence-based recommendations for elderly patients with LARC were limited since this age category was usually excluded in previous clinical trials. Though postoperative mortality has improved over the last decade, risks have been on the rise with aging. Decision-analytic models illustrated that patients over 80 could benefit more from nonoperative interventions. 17 This study provided evidence in tailoring treatments for the elderly based on exploratory results from the nonoperative cohort of a multicenter prospective study, highlighting the feasibility of CGA-based individualized strategies and the efficacy of external beam radiotherapy (EBRT) in different geriatric subgroups.
Existing guidelines for older and frail patients with LARC were mainly derived from data from observational studies or subgroup analyses. According to a consensus established by an SIOG task force, preoperative LCRT might be an alternative to CRT concerning chemotherapy tolerance. Additionally, SCRT followed by a delayed surgery (commonly 6–8 weeks or longer) was optional in frail patients, consistent with multi-society opinions from Italy. 18 While EBRT was recommended in inoperable low rectal cancer of all stages, high-dose-rate brachytherapy (HDR-BT) and contact X-ray therapy (CXB) also showed promising effects in older adults. 19 Besides, a recently released guidance from the Fox Chase Cancer Center highlighted the attempt on total neoadjuvant therapy. 10 The idea was supported by several breaking studies, including the OPRA and RAPIDO trials, where tolerability and disease control were improved but elderly patients were not excluded.20,21 To plan management properly in older cohorts, patients were classified into four categories based on their physical performance, CGA, and treatment goals by Myint et al. 3 The concept partially overlapped with our previous study that confirmed the efficacy and safety of preoperative CRT in fit older patients. The study also pointed out the necessity of specifying individualized therapies within different CGA categories. 12 However, several questions remained by far since relevant trials were mostly conducted in selected cases. Furthermore, the application of brachytherapy was challenged with technical availability and inability to dose boost to metastatic lymph nodes, leading to uncertainty in its long-term impact.1,3
Though LCRT showed superior pathological complete response (pCR) rates in an early meta-analysis, the advantage did not turn into organ preservation or long-term survival benefits. 22 Furthermore, Cai et al found in a single-institutional retrospective study that only age and Charlson index were prognostic of 3-year OS rate in rectal cancer patients over 70, with no statistically significance found in treatment modality (RT vs CRT) or KPS score. Notably, the results might be confounded by patients with local recurrence and synchronous distant metastases in the cohort, as well as the poorly completed scheduled RT dosage (69%). 23 A population-based study in the Netherlands indicated that 20% of rectal cancer patients aged ⩾70 did not receive surgery, among who nearly a half underwent RT alone. The 3-year relative survival rate dropped from 93% to 37% in favor of surgical patients, possibly resulting from advanced age and disease stages in the inoperable group. CRT and RT appeared to improve both overall and relative survival rates compared to the no-treatment group. 24 Dose escalation to the tumor with EBRT was important 25 but rarely mentioned in previous studies for the elderly. Nearly half patients in our cohort received a dose boost to tumor, and the proportion was analogous among subgroups. Overall, the intensification might guarantee a median OS of 33 months and comparable 2-year OS and CSS rates to our previous study. 12 Survival data of EBRT plus brachytherapy was limited, as the primary endpoint was often defined as local control. Compared to our results, a 10% increase was demonstrated in 1-year PFS among elderly patients treated with CXB (68% vs 78%). 26
As is shown in the current study, the “Frail” cohort treated with SCRT alone seemed to have similar survival outcomes as the “Fit” or “Vulnerable” group who received CRT or LCRT. The results coincided with a large retrospective analysis that claimed similar OS, disease-free survival (DFS), and CSS between patients aged over and below 70. 27 Intriguingly, the older group was less likely to complete the scheduled CRT, but age was proved to be irrelevant to DFS. 27 One potential reason for the favorable results of SCRT in our study might be its high completion rate, which was 83.3% in the “Frail” group, much closer to the average level of the whole cohort (83.0%). In fact, compared with CRT or LCRT, SCRT has garnered more attention in the past decades since several types of research manifested the non-inferiority or even superiority of SCRT. 6 SCRT followed by delayed surgery showed a significantly higher pCR rate than LCRT (10.4% vs 2.2%) in Stockholm III trial. 28 Another randomized study in frail older patients with colorectal cancer suggested reduced total number of complications after preoperative geriatric assessments and adapted treatments. 8 Notably, surgery was adopted in the studies mentioned above, whose extrapolation could indirectly confirmed the rationality of our results. SCRT was reported to be evolved in the activation or polarization of immune cells in rectal cancer, thus potentiating enhanced immunological effects and possibly linked with better clinical responses. 29
With regard to toxicities, quality of life (QoL) and patient desire need to be prioritized because of the significantly worsened performance of the “Frail” group in functional independence and mental health in our CGA evaluations. In the PRODIGE 42 study, 103 patients ⩾75 were randomized to either CRT or SCRT followed by delayed surgery. Despite that the study failed to prove the non-inferiority of SCRT in R0 resection rate, the IADL score was preserved after SCRT at 3 months (IADL deterioration rate: 14.8% vs 44.8%, p = 0.032). This was consistent with the recovery of QoL in non-randomized registry of older patients of the TREC study. 30 Moreover, SCRT exhibited better compliance than CRT (failure of receiving planned therapies: 0% vs 14%). However, the finite methods used in geriatric assessments of PRODIGE 42 might masked the heterogeneity. Given the absence of subgroup analysis based on age stratification, the results should be interpreted with caution while selecting preferred therapies for the elderly and frail in practice. 6 Adverse effects in this study were manageable, with grade 3–4 hematologic toxicities remained 23.4% and non-hematologic ones kept under 5%. Grade 3 or above hematologic adverse effects were observed more frequently in the “Fit” group, probably attributed to the systemic chemotherapy.
In order to further enhance oncological outcomes without sacrificing functional independence, additional interventions are available at present. Local excision and brachytherapy (including HDR-BT and CXB) preceded by EBRT were elective methods as shown by several tentative types of research, including the GRECCAR 2 and HERBERT.1,26,31,32 Unfortunately, geriatric assessments were eliminated from these studies. Therefore, the specific population who might benefit more from local intensification treatments remained unclear. Additionally, immunotherapy might act as a sensitizer to RT and was described as well-tolerated in most frail and elderly patients.25,33 The combination of SCRT and immunotherapy has succeeded in achieving a higher pCR rate even in patients with microsatellite stable tumors. 34 Thus, given that immunotherapy was refrained from technical barriers and CGA-driven EBRT showed a promising median OS in our study, the addition of immunotherapy to personalized RT deserved further exploration.
A strength of this study was the usage of CGA assessments in frailty scaling and the implementation of CGA-driven and MDT-based strategies, which was strongly advocated by recent consensus on personalized treatments for the elderly.10,18 There were also a few limitations of this study. First, brachytherapy was absent from the planned schedule, which might result in reduced local control and survival benefits. Further investigation into a combined therapy of EBRT and brachytherapy was needed to manifest the role of RT in age-stratified cohorts. Moreover, the relatively small sample size would compromise the statistical efficacy of the study. The risk of colinearity of the regression model, probably generated from CGA and response profile, was another statistical concern. These could hamper the extrapolation of the results to external datasets. Finally, the loss of consultation during follow-up due to poor physical status could not be neglected.
Conclusion
Radiotherapy guided by CGA was effective and well-tolerated in non-surgical elderly patients with LARC, and CRT and RT seemed to be feasible alternatives to surgery in frail patients. Considering the time-consuming feature and toxicities, patients who scored higher in frailty evaluations should be treated less aggressively and SCRT might be a preferable option. Still, the application of SCRT in older and frail patients warranted further investigation in a larger population.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241296386 – Supplemental material for Comprehensive geriatric assessment guided radiotherapy in elderly patients with locally advanced rectal cancer—exploratory results on nonoperative cohort of a multicenter prospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359241296386 for Comprehensive geriatric assessment guided radiotherapy in elderly patients with locally advanced rectal cancer—exploratory results on nonoperative cohort of a multicenter prospective study by Yue-Xin Yang, Yuan Tang, Ning Li, Yu Tang, Yun-Jie Cheng, Lin Yang, Hui Fang, Ning-Ning Lu, Shu-Nan Qi, Bo Chen, Shu-Lian Wang, Yong-Wen Song, Yue-Ping Liu, Ye-Xiong Li, Zheng Liu, Jian-Wei Liang, Hai-Zeng Zhang, Hai-Tao Zhou, Jun Wang, Wen-Yang Liu and Jing Jin in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
