Abstract
Background:
The NOVA study (NCT01847274) compared niraparib with placebo as a maintenance treatment for patients with recurrent ovarian cancer (OC) but was not powered to detect an overall survival (OS) improvement.
Objective:
To compare OS in a real-world population of patients with
Design:
A retrospective study using a US-based nationwide deidentified electronic health record-derived database.
Methods:
Patients diagnosed with epithelial OC (January 1, 2011–May 31, 2021) who completed second-line (2L) therapy (January 1, 2017–March 2, 2022) and were
Results:
The overall population comprised 199 patients in the 2LM niraparib monotherapy cohort and 707 patients in the AS cohort; the NOVA study-like subpopulation included 123 patients in the 2LM niraparib monotherapy cohort and 143 in the AS cohort. Demographic and clinical characteristics were similar in both populations. Overall, adjusted mOS was 24.1 months for the 2LM niraparib monotherapy cohort versus 18.4 months for the AS cohort (hazard ratio, 0.8; 95% confidence interval [CI]: 0.7–0.9). In the NOVA study-like subpopulation, adjusted mOS was 28.1 months for the 2LM niraparib monotherapy cohort versus 21.5 months for the AS cohort (hazard ratio, 0.6; 95% CI: 0.5–0.9).
Conclusion:
These results provide important real-world OS data for patients with recurrent
Plain language summary
This study examined the real-life survival of patients with ovarian cancer (OC) who received two lines of chemotherapy for OC, known as recurrent OC. We compared two groups: one patient group received a subsequent oral medication called niraparib (a type of maintenance therapy) to delay recurrence, whereas the other group was monitored by their physicians without receiving any medication (also known as active surveillance). This study analyzed health records from across the United States, focusing on patients who were diagnosed with OC between January 2011 and May 2021. Importantly, these patients completed their second line of therapy between January 2017 and March 2022 and had
Keywords
Introduction
Ovarian cancer (OC) is the eighth most common cancer among women worldwide. 1 In 2023, there were an estimated 19,710 new OC cases and 13,270 OC-related deaths projected in the United States. 2 Treatment for OC typically involves primary cytoreductive surgery with adjuvant chemotherapy or neoadjuvant chemotherapy with subsequent interval cytoreductive surgery and additional chemotherapy.3,4 Although most patients respond to the initial treatment, approximately 70% of patients with advanced disease experience disease progression 5 and frequently require subsequent therapies. 6 Nevertheless, advances in supportive care and therapeutic options have had a measurable impact on increasing patient survival and consequently the disease prevalence in the United States, which has steadily risen over the past 20 years and accounts for approximately 236,000 patients alive with this diagnosis. 7
After response to chemotherapy, treatment options include active surveillance (AS) or maintenance therapy. In the maintenance setting, poly(ADP-ribose) polymerase (PARP) inhibitors are often prescribed. 6 Niraparib, an oral PARP-1 and PARP-2 inhibitor, has been shown to improve progression-free survival (PFS) in patients with OC while demonstrating a consistent safety profile in first-line (1L) maintenance and recurrent settings.8,9
In NOVA, a randomized, placebo-controlled phase III trial, patients who received niraparib monotherapy in the second-line maintenance (2LM) or later setting had significantly longer median PFS than patients who received placebo, regardless of biomarker status.
8
Specifically, the hazard ratio comparing median PFS for niraparib versus placebo in a germline
The objective of this retrospective observational study was to compare OS in a US-based, real-world population of patients with
Methods
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. 10 The order of the STROBE checklist items has been modified to group similar items together. This file is found in the Supplemental Materials.
Data source
This study was conducted using data from the Flatiron Health database, a US-based nationwide database of deidentified electronic health record (EHR)-derived data from approximately 280 cancer clinics representing an estimated 800 sites of care.11,12 The database contains patient-level structured and unstructured data, curated via technology-enabled abstraction from physician notes and other unstructured documents.11,12 Data consisted of a random sample of the broad OC population in the real-world database, plus a custom-curated dataset of all patients diagnosed with OC who received niraparib but were not included in the deidentified database. The deidentified data were subject to obligations to prevent reidentification and protect patient confidentiality.
Study population
Female patients were included if they had a diagnosis of the ovarian, fallopian tube, or peritoneal cancer (collectively referred to as OC) as defined by International Classification of Diseases (ICD) codes 183x and 158x (ICD 9) and C56x, C57.0x, C48x (ICD-10) and at least two documented clinical visits between January 1, 2011 and May 31, 2022 (hereafter referred to as the study period). All patients were aged 18 years or older at index, were diagnosed with epithelial OC during the patient selection period (January 1, 2011–May 31, 2021), had received two prior lines of therapy, and were
A NOVA study-like subpopulation was created from the overall study population by applying additional inclusion criteria of an Eastern Cooperative Oncology Group (ECOG) performance status score of 0–1, known histology, and platinum-sensitive disease, defined as 6 months or more between the end of 1L platinum-based treatment and the start of 2L platinum-based treatment. Platinum-based treatment was defined as chemotherapy treatment lines that include carboplatin, cisplatin, and oxaliplatin. These criteria were based on similar inclusion criteria in the NOVA clinical trial. 8
Patients who met the inclusion criteria from both the overall population and the NOVA study-like subpopulation were assigned to either the 2LM niraparib monotherapy cohort or the AS cohort. AS was not recorded in the database but rather was derived from the line of therapy available in the database as patients who did not initiate 2LM during a grace period after the end of 2L non-maintenance therapy. Patients were followed from index date to date of death, last clinical activity, or study period end (May 31, 2022), whichever occurred first.
In contrast to the NOVA study
8
that classified patients as either g
Study outcome
The study outcome was OS, defined as the time from index date to death from any cause. Patients who did not have a death event during the study period were censored at the earliest occurrence of the last clinical activity or the end of the study period.
Statistical methods
To adjust for immortal time bias associated with a potential artificial increase in the follow-up time for the maintenance cohort (e.g., when the start of follow-up and treatment initiation do not coincide), a target trial emulation with a cloning-weighting-censoring approach was selected a priori. 13 Inverse-probability-of-censoring weighting (IPCW) was used to control for informative censoring bias arising from the cloning approach, and weights were stabilized to reduce the size of extreme weights. The methods used in this study have been previously described in detail in a prior report. 14
Patient demographic and clinical characteristics were summarized descriptively for the overall study and the NOVA study-like subpopulation before cloning. The balance of key baseline covariates between both cohorts was assessed using standardized mean differences with a threshold of less than 15% chosen as the midpoint between acceptable ranges.15,16 These variables were defined a priori because of their potential to affect treatment decisions and were as follows: age at index, race, region of residence, practice type (academic and community), epithelial histology, stage at initial diagnosis, ECOG performance status score,
Survival curves for the 2LM niraparib monotherapy cohort and the AS cohort were estimated using an inverse-probability-of-censoring (IPC)-weighted nonparametric Kaplan–Meier estimator. Median OS (mOS) in months, survival rate at 24 months, and associated 95% CIs were generated. IPC-weighted Cox proportional hazards regression models with a robust variance estimator to account for within-person correlation were used to compare OS between both cohorts. Hazard ratios and 95% CIs were generated. All survival analyses were conducted for the overall study and the NOVA study-like subpopulation. All analyses were performed using SAS® 9.4 (SAS Institute, Cary, NC, USA).
Results
Participants
In this real-world database, 10,394 patients were diagnosed with OC during the study period. Of these, 906 and 266 patients met eligibility criteria for the overall population and the NOVA study-like subpopulation, respectively, and were included in analyses (Figure 1). In the overall population, 199 patients were assigned to the 2LM niraparib monotherapy cohort and 707 patients to the AS cohort. In the NOVA study-like subpopulation, 123 patients were assigned to the 2LM niraparib monotherapy and 143 to the AS cohort.
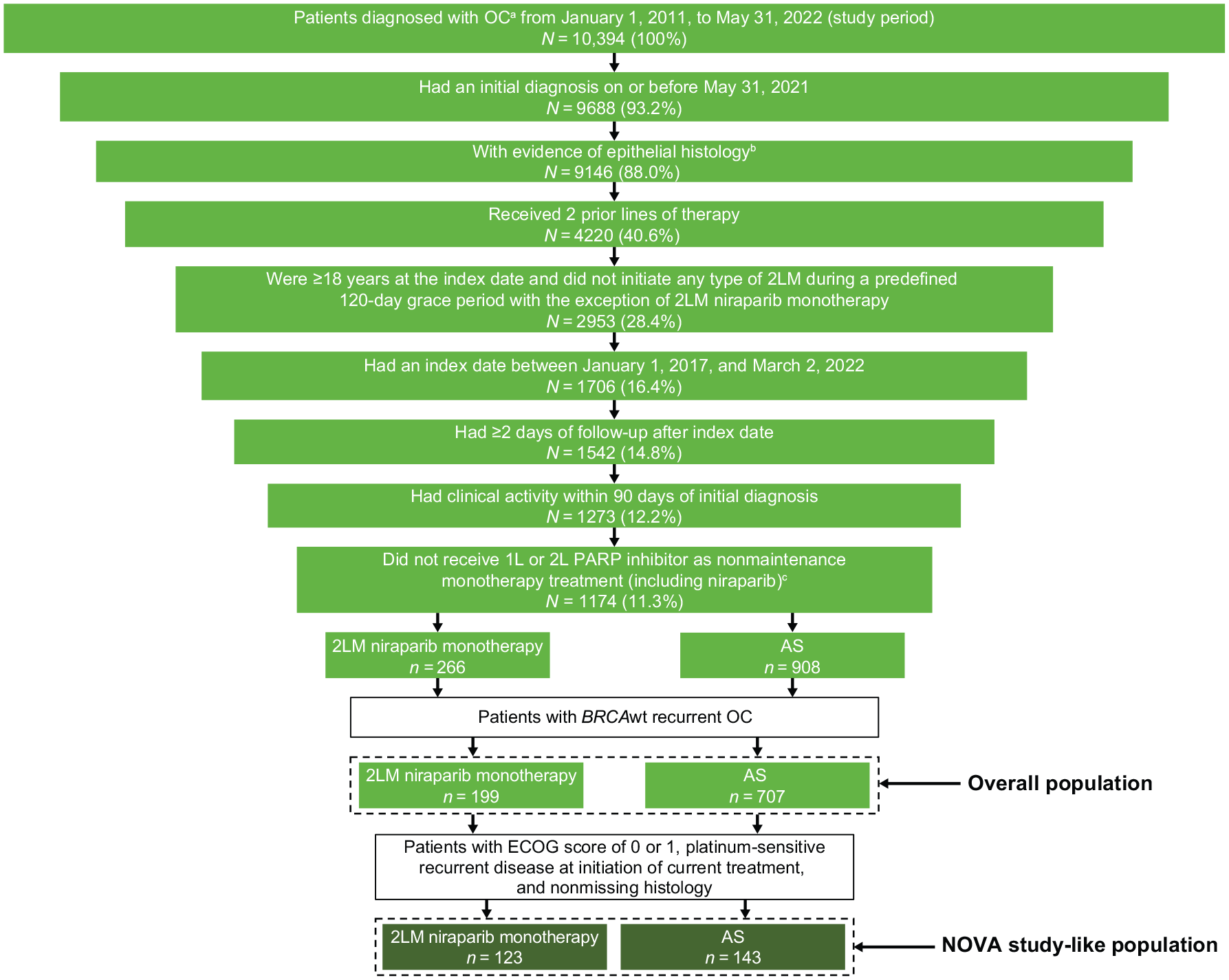
Study population attrition.
Patient demographic and clinical characteristics before adjustment are shown in Table 1. In the overall population, the median age at index was 68 years (interquartile range [IQR], 61–75 years) for the 2LM niraparib monotherapy cohort and 69 years (IQR, 60–76 years) for the AS cohort. Most patients were White (66.3% in the 2LM niraparib monotherapy cohort and 71.9% in the AS cohort) and from a community setting (84.4% in the 2LM niraparib monotherapy cohort and 81.2% in the AS cohort). In the 2LM niraparib monotherapy and AS cohorts, 83.9% and 71.4% of patients had an ECOG performance status score of 0 or 1, respectively. The median duration between the last treatment date of 1L and the start of 2L was 13.6 months (IQR, 7.6–20.9 months) for the 2LM niraparib monotherapy cohort and 6.2 months (IQR, 2.1–12.5 months) for the AS cohort. Demographic and clinical characteristics for the NOVA study-like subpopulation were similar to the overall population in most instances (Table 1). Two exceptions were by study design and involved all patients in both cohorts having an ECOG performance status score of 0 or 1 and epithelial histology not otherwise specified or unknown. Key characteristics that may affect treatment decisions were balanced after cloning and stabilized IPCW (Table 2).
Baseline demographic, clinical, and tumor characteristics before adjustment.
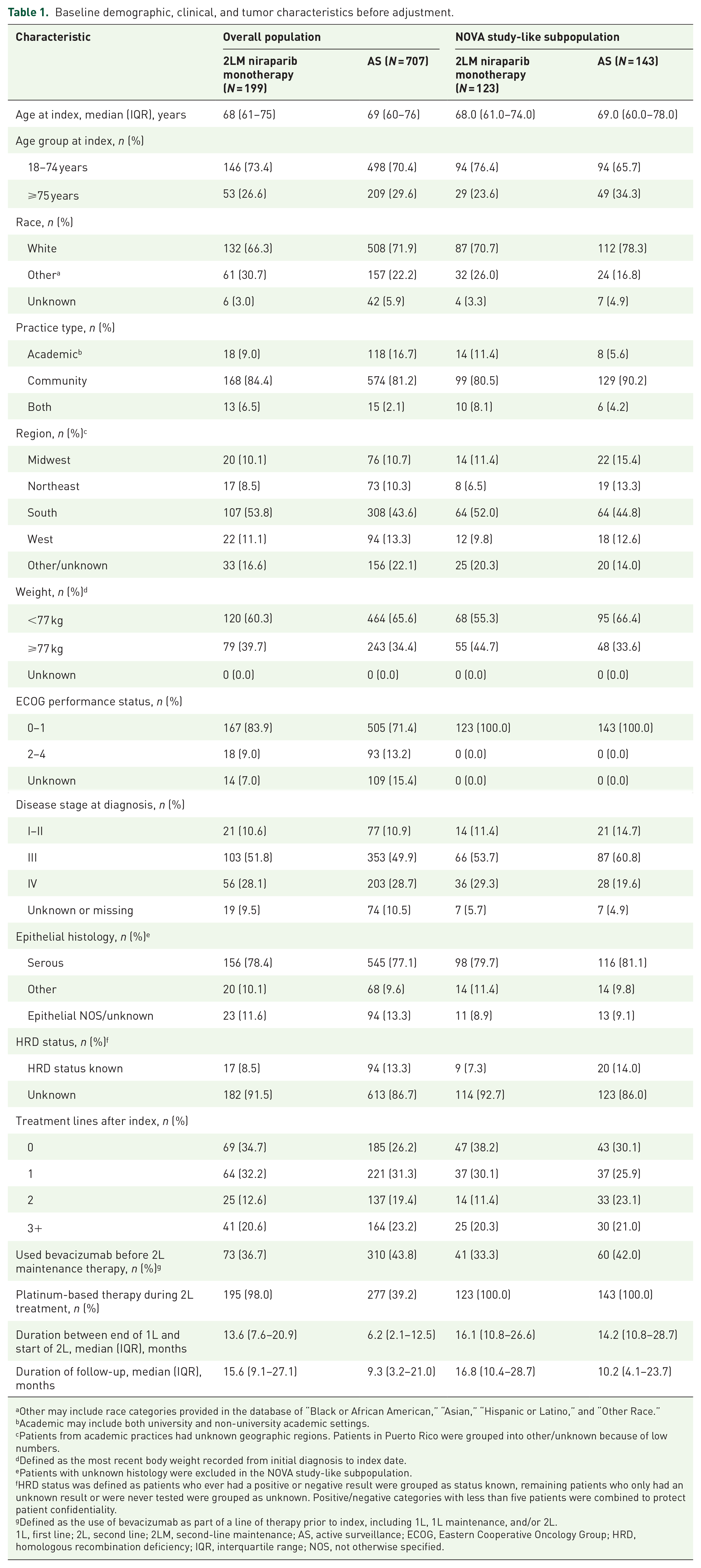
Other may include race categories provided in the database of “Black or African American,” “Asian,” “Hispanic or Latino,” and “Other Race.”
Academic may include both university and non-university academic settings.
Patients from academic practices had unknown geographic regions. Patients in Puerto Rico were grouped into other/unknown because of low numbers.
Defined as the most recent body weight recorded from initial diagnosis to index date.
Patients with unknown histology were excluded in the NOVA study-like subpopulation.
HRD status was defined as patients who ever had a positive or negative result were grouped as status known, remaining patients who only had an unknown result or were never tested were grouped as unknown. Positive/negative categories with less than five patients were combined to protect patient confidentiality.
Defined as the use of bevacizumab as part of a line of therapy prior to index, including 1L, 1L maintenance, and/or 2L.
1L, first line; 2L, second line; 2LM, second-line maintenance; AS, active surveillance; ECOG, Eastern Cooperative Oncology Group; HRD, homologous recombination deficiency; IQR, interquartile range; NOS, not otherwise specified.
Balance of patient characteristics before and after IPCW, using a 15% threshold.

Standardized mean difference is calculated as the absolute difference in means, mean ranks, or proportions divided by the pooled standard deviation. For categorical variables, the overall standardized difference was calculated using Mahalanobis distance. 17
Practice type had a standardized mean difference of 15.3% in the overall population after stabilized IPCW, but the balance was achieved at the categorical level.
1L, first-line; 2L, second-line; ECOG, Eastern Cooperative Oncology Group; IPCW, inverse-probability-of-censoring weights.
IPCW overall survival
For the overall population, mOS was significantly longer for the 2LM niraparib monotherapy cohort (24.1 months; 95% CI: 20.9–29.5 months) than the AS cohort (18.4 months; 95% CI: 15.1–22.8 months; hazard ratio, 0.8; 95% CI: 0.7–0.9; Figure 2(a)) after adjustment. Survival rates at 24 months were 50.6% (95% CI: 42.5%–58.1%) for the 2LM niraparib monotherapy cohort and 41.6% (95% CI: 35.4%–47.6%) for the AS cohort.
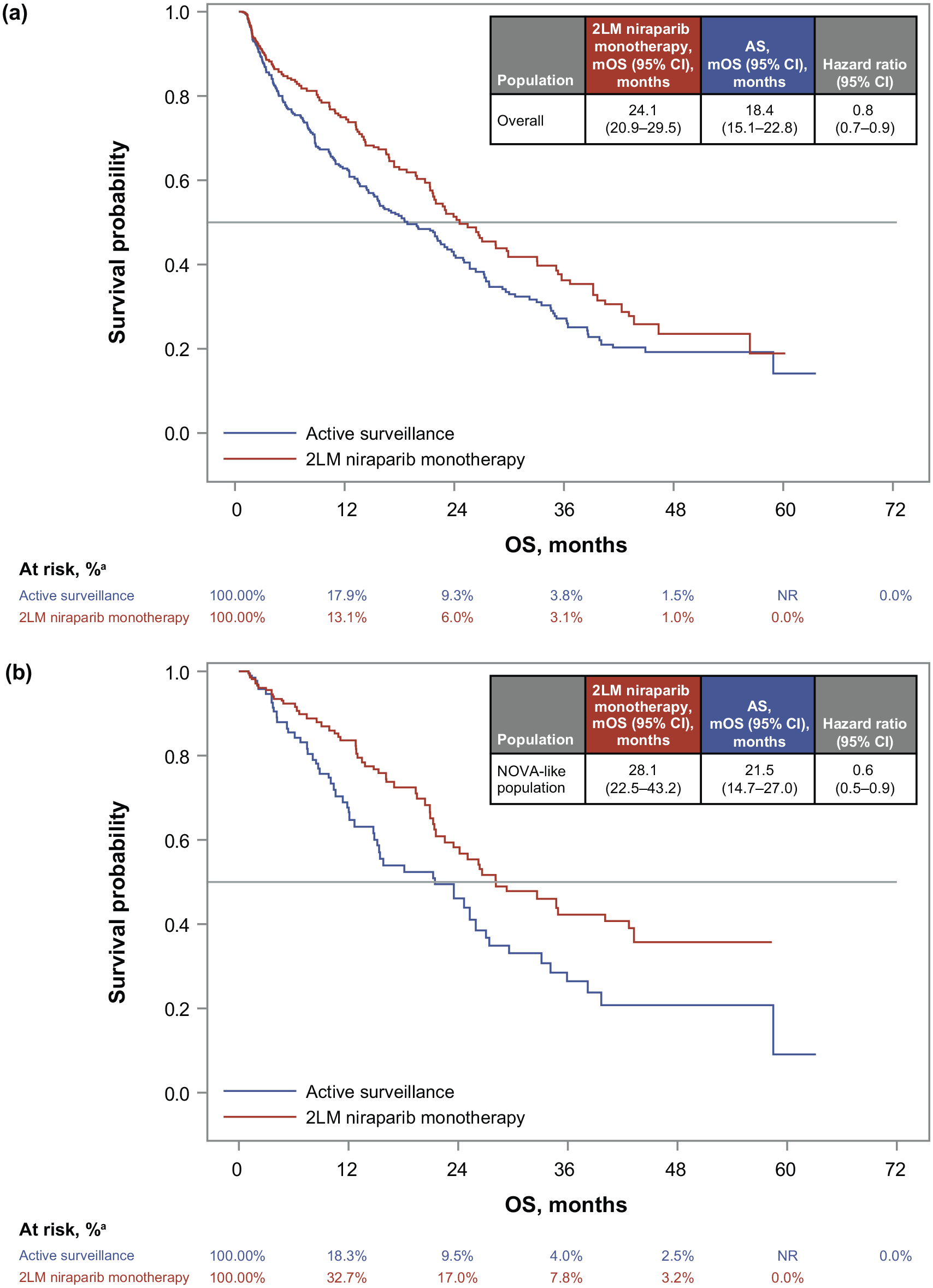
Adjusted Kaplan–Meier curves and adjusted hazard ratios for the (a) overall population and (b) the NOVA study-like subpopulation.
OS results were similar for the NOVA study-like subpopulation after adjustment (Figure 2(b)). The mOS was 28.1 months (95% CI: 22.5–43.2 months) and 21.5 months (95% CI: 14.7–27.0 months) for the 2LM niraparib monotherapy and AS cohorts, respectively (hazard ratio, 0.6; 95% CI: 0.5–0.9). The survival rate at 24 months was 58.2% (95% CI: 47.5%–67.6%) for the 2LM niraparib monotherapy cohort and 46.1% (95% CI: 33.6%–57.7%) for the AS cohort.
Discussion
Given a lack of real-world data reporting outcomes for patients with OC who receive niraparib maintenance monotherapy in the recurrent setting, this retrospective observational study provides an informative real-world comparison of OS for patients with
These real-world results support niraparib’s clinical benefit in treating recurrent OC.
8
These results also support preplanned exploratory NOVA analyses that demonstrated a continued clinical benefit of niraparib in the maintenance setting beyond the first progression. After a median follow-up of more than 75 months, the mOS in a g
The results from this study should be interpreted within the context of some potential limitations. Real-world, retrospective, EHR database analyses are limited by the type of data collected and the quality control of data contained within the database.19,20 However, the database used in this study is considered to have a high degree of accuracy in the essential dataset required when evaluating real-world OS, namely all-cause mortality data.
21
The study population is limited to data from the real-world database, collected primarily from community practices, and may not represent the entire OC population in the United States. The study included a broad patient cohort to be reflective of the real-world OC population, which included patients who were not treated with platinum-based 2L therapy or patients who may have had platinum-resistant or -refractory disease (which is inconsistent with the niraparib label), who were therefore at a higher risk of having poorer outcomes. However, in the NOVA study-like subpopulation, all patients received a platinum-based 2L therapy and were required to have at least 6 months between the 1L and 2L, to exclude patients with platinum-resistant or -refractory disease. For both cohorts, patients who were treated with 2LM had longer OS than patients who were under AS. Relative to the NOVA study, this study excluded s
Conclusion
The findings of this study provide important real-world OS data for patients with recurrent OC who received niraparib monotherapy in the 2LM setting. In this real-world study from a clinical practice population in the United States, results provide evidence of an OS benefit from using 2LM niraparib monotherapy over AS that supports data generated in the NOVA clinical trial, specifically in patients with
Supplemental Material
sj-docx-1-tam-10.1177_17588359241292272 – Supplemental material for Real-world overall survival in second-line maintenance niraparib monotherapy versus active surveillance in patients with BRCA wild-type recurrent ovarian cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359241292272 for Real-world overall survival in second-line maintenance niraparib monotherapy versus active surveillance in patients with BRCA wild-type recurrent ovarian cancer by Robert L. Coleman, Jessica A. Perhanidis, Linda Kalilani, Nicole M. Zimmerman, Amanda Golembesky and Kathleen N. Moore in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Statistical programming support was provided by Pratyk Gomez, BA, and Warsha Kumari Singh, BTech, both of GSK. Medical writing and editorial assistance, funded by GSK (Waltham, MA, USA) and coordinated by Chun Zhou, PhD, CMPP, of GSK, were provided by Tafara T.R. Kunota, PhD, and Kathleen Blake, PhD, of Ashfield MedComms, an Inizio Company.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
